Abstract
OBJECTIVE:
This study aims to determine the correlation between p-cofilin expression and cisplatin resistance in patients with ovarian cancer, and also to investigate the role of p-cofilin in prognosis.
PATIENTS AND METHODS:
The ovarian cancer cell line A2780/DDP resistant to cisplatin was prepared. The cell resistance to cisplatin was measured via MTT assay. The cell invasion capacity was identified via Transwell assay. The mRNA expression and protein level was evaluated via semi-quantitative PCR and Western blot, respectively. The tumor tissues of patients with cisplatin-resistant ovarian cancer were collected. The relationship between prognosis and p-cofilin expression was analyzed.
RESULTS:
The growth rate of A2780 was similar to that of A2780/DDP. The sensitivity of A2780 to cisplatin was significantly higher than that of A2780/DDP (
CONCLUSION:
The cisplatin resistance of ovarian cancer is closely related to the expression level of p-cofilin, which affects the prognosis of patients with ovarian cancer.
Introduction
The incidence of ovarian cancer ranks the third in the gynecological malignancy at present, which causes high mortality. Cisplatin is extensively used for the treatment of tumors as a kind of chemotherapeutic agent, inefficacy due to the drug resistance remains impede its further application in clinic [1, 2, 3]. Cofilin is a member of actin-binding protein family encoded by CFL1 gene, and it can affect the recombination of actin skeleton via promoting the circulation of actin microfilaments in mammals, which is one of the important proteins of cytoskeleton shaping [4, 5, 6]. Several studies have demonstrated the functional roles of cofilin phospho-regulation in cell migration, tumor invasion, mitosis, neuronal development, and synaptic plasticity [7]. Accumulating evidence demonstrates that the phospho-regulation of cofilin is a key convergence point of cell signaling networks that link extracellular stimuli to actin cytoskeletal dynamics and perturbations in the normal control of cofilin activity underlie many pathological conditions, including cancer metastasis and neurological and cardiovascular disorders [7]. Goyal et al. [8] studied and showed that cofilin can affect the direction of cell movement via inducing the formation of lamellar pseudopodia. Li et al. [9] found that taxo-resistance easily occurs in patients with high expression of p-cofilin, suggesting that it may be related to tumor resistance. But there is no report on the exact relation of p-cofilin expression with cisplatin resistance. This study aims to investigate the relationship between p-cofilin level and cisplatin resistance in patients with ovarian cancer and its effect on prognosis of patients.
Patients and methods
Objects
A total of 70 paraffin-embedded sections of patients with ovarian cancer admitted and diagnosed via pathological examination in Weihai maternal and child health hospital after surgical resection from September 2013 to September 2015 were collected in this study. The above patients were aged from 38–87 years. Patients with other types of diseases were excluded. All patients had the complete clinical and pathological data. Patients enrolled had the 1-year follow-up records and complete treatment regimens. All cancer tissue samples were obtained and stored in liquid nitrogen. The study was approved by the ethics committee of Weihai maternal and child health hospital and all the participants have signed the informed consent.
Instruments and materials
Human ovarian adenocarcinoma cell A2780 was purchased from Shanghai Cell Bank (Chinese Academy of Sciences, Shanghai, China). Methyl thiazolyl tetrazolium (MTT) and dimethylsulfoxide (DMSO) was from Sigma (Temecula, CA, USA). Cisplatin was provided from Shanghai Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). TRIzol kit, SSIII reverse transcription kit and enhanced chemiluminescence (ECI) solution were offered by Invitrogen (Carlsbad, CA, USA). Rabbit anti-p-cofilin and glyceraldehyde-phosphate dehydrogenase (GAPDH) were purchased from Cell Signaling Technology (Beverly, MA, USA). DreamTaq PCR Master Mix (2X) and inversed fluorescent microscope was made from Thermo Fisher Scientific (Waltham, MA, USA). Cell culture flask was obtained from Corning (Waltham, MA, USA). The pipettor and transwell chamber were from Eppendorf (Hamburg, Germany). PCR instrument was provided by ABI (Waltham, MA, USA). UV imaging system Biometra (Berlin, Germany), electronic balance (BP121S) was purchased from Sartorious (Berlin, Germany).
Construction of ovarian cancer-resistant cell lines
After human ovarian adenocarcinoma cell A2780 was resuscitated, it was cultured in the incubator, and the in vitro cisplatin-resistant cell line was constructed by stepwise progressive concentration method. The cells in the exponential growth phase were placed into the culture flask, and then cisplatin with concentrations of 0.1, 0.2, 0.5, 1 and 2
Determination of cell growth cycle and drug sensitive test
Determination of growth cycle: The cisplatin- resistant ovarian cancer cell lines in the exponential growth phase were prepared into the suspension and placed onto the culture dish at the concentration of 1
Determination of cell invasion capacity
The cell invasion capacity was detected via Transwell assay. After the cisplatin-resistant ovarian cancer cell line A2780/DDP in good growth status was selected, the cell concentration was adjusted to 5
Determination of CFL1 gene expression level via semi-quantitative PCR
The total RNA in cisplatin-resistant ovarian cancer cell line A2780/DDP treated with cisplatin (10
PCR primers
PCR primers
The proteins in cisplatin-resistant ovarian cancer cell line A2780/DDP and cell line A2780 were extracted. After centrifugation, all the supernatant was removed and the total protein was obtained. For tissues, the cancer tissue samples of patients with cisplatin-resistant ovarian cancer and patients with cisplatin-sensitive ovarian cancer were taken from the liquid nitrogen. The tissues were cut up with scissors and then the lysis buffer (1:20) was added for homogenate. After centrifugation, all the supernatant was removed and the total protein was obtained. The protein loading samples were prepared via protein quantification into the loading sample system with the same concentration. Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was performed for loading, and the target band was cut off after sealing and cleaning. The p-cofilin (ab100836, Abcam, Cambridge, MA, USA) or total cofilin (ab42824, Abcam) primary antibody was incubated at 4
Statistical analysis
The data in this study were presented as mean
Results
Cell growth cycle and doubling time
The cisplatin-resistant ovarian cancer cell line A2780/DDP was constructed and the cell line A2780 was used as the control group. MTT data showed that the growth rate of A2780 was similar to that of A2780/DDP (
Growth curves of A2780/DDP and A2780. The growth rate and doubling time between A2780/DDP and A2780 are similar (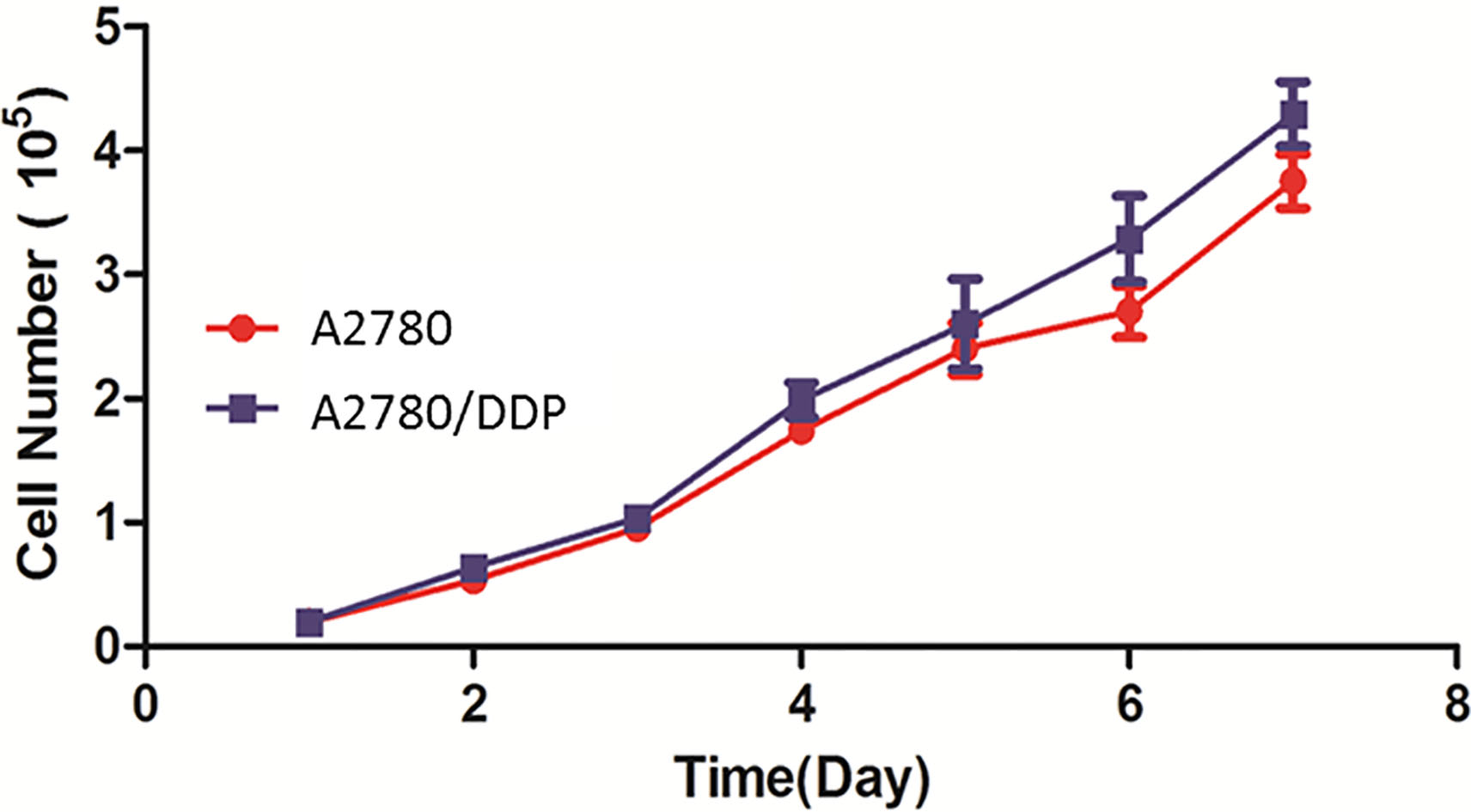
Sensitivity of A2780/DDP and A2780 to cisplatin
Eight different concentration gradients of DDP were added into the cisplatin-resistant ovarian cancer cell line A2780/DDP, and A2780 was used as the control group. The sensitivity of A2780/DDP to the above drug was detected via MTT and the IC
Determination of cell migration capacity
The migration capacity of drug-resistant cell line A2780/DDP was detected via Transwell assay. Of note, the migration capacity of A2780/DDP was significantly increased compared with that of A2780 (
Migration capacity of A2780/DDP via Transwell assay. The migration capacity of A2780/DDP was significantly increased compared with that of A2780 (
The expression levels of p-cofilin gene CFL1 in A2780/DDP and A2780 cells were detected via semi-quantitative PCR. We observed that the mRNA level of CFL1 in A2780/DDP was significantly higher than that of A2780 cells (
CFL1 gene expression via semi-quantitative PCR. A, Diagram of agarose gel electrophoresis. B, The relative expression level of CFL1. The results showed that the CFL1 mRNA expression level in A2780/DDP was not significantly changed, but that in A2780 was significantly decreased (
The p-cofilin protein expression in A2780/DDP and A2780 were detected via Western-blot. In similar scenario, the results indicated that the p-cofilin protein expression in A2780/DDP was significantly increased compared to that of A2780 (
p-cofilin protein level. A, Western blot. B, Statistical chart of p-cofilin protein expression level. The results indicated that the p-cofilin protein level in A2780/DDP was significant higher than that in A2780 (
Also, we detected the p-cofilin expression levels of cancer tissues from patients with cisplatin-resistant ovarian cancer and cisplatin-sensitive ovarian cancer. We found the level in cancer tissues in patients with cisplatin-resistant ovarian cancer was significantly higher than that in patients with cisplatin-sensitive ovarian cancer (
p-cofilin level in cancer tissues in patients with ovarian cancer. A, Western blot. B, Statistical chart of p-cofilin protein expression level. The p-cofilin protein expression level in cancer tissues in patients with cisplatin-resistant ovarian cancer was significantly higher than that in patients with cisplatin-sensitive ovarian cancer (
The severity of cisplatin resistance in 78 patients with ovarian cancer was identified and divided into Grade 0–3 according to the clinical resistance criteria. The p-cofilin protein level was detected by western blot, and the correlation analysis of the p-cofilin protein expression and cisplatin resistance was performed. Pearson correlation analysis showed that the cisplatin resistance in patients with ovarian cancer was positively correlated with the p-cofilin protein expression level (
Correlation analysis between p-cofilin protein expression and cisplatin resistance in patients with ovarian cancer. Cisplatin resistance in patients with ovarian cancer was positively correlated with the p-cofilin protein expression level (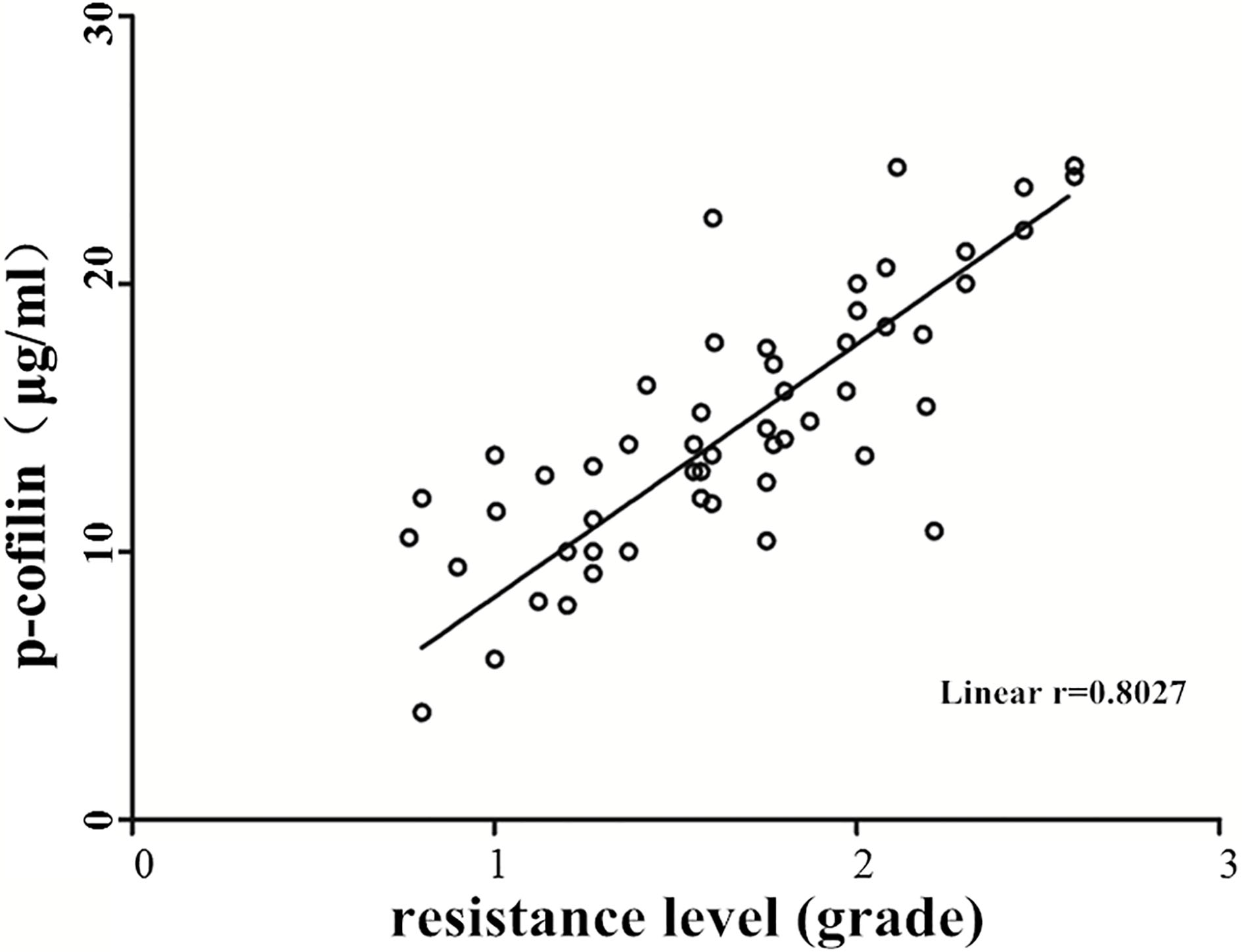
The patients were followed up for 1 year. The clinical data and treatment regimens were recorded in details and the p-cofilin expression level in ovarian cancer tissues was detected. The patients were categorized into high-expression group and normal- or low-expression group according to the p-cofilin expression level. The statistical analysis of survival time among the groups showed that the survival time of patients in p-cofilin high-expression group was significantly lower than that in normal- or low-expression group (
Relationship between p-cofilin protein expression and prognosis of patients with ovarian cancer. The survival time of patients in p-cofilin high-expression group was significantly lower than that in normal-or low-expression group (
Ovarian cancer seriously threatens women’s health. Ovarian adenocarcinoma is a kind of ovarian cancer with the highest mortality rate clinically, and the 5-year survival rate of epithelial ovarian cancer in the world is less than 30%. No obvious pathological features are presented regarding ovarian cancer in the early phase of pathogenesis and it often developed to the advanced stage when diagnosed [10, 11, 12]. With the continuous development and progress of medical technology and anti-tumor drugs, the cure rate of tumors has been significantly improved through surgical resection combined with chemotherapy and radiotherapy, etc., but the 5-year survival rate of patients with advanced ovarian cancer is still around 15–30%. Drug resistance caused by long-term use of chemotherapy drugs in the treatment of tumors adversely affects the efficacy of therapeutic regimens against ovarian cancer [13, 14]. The drug resistance of tumor cells can be induced by many chemicals, and cofilin is one of the important factors resulting in the drug resistance of tumor cells [15]. Castro et al. [16] found that paclitaxel treatment can reduce the p-cofilin expression level in ovarian cancer cells. However, the p-cofilin expression level in drug-resistant cell lines treated with paclitaxel in the same concentration exhibited no significant change, suggesting that the p-cofilin expression level may be closely related to the paclitaxel resistance [17, 18].
In this study, the cisplatin-resistant ovarian cancer cell line A2780/DDP was constructed by inducing human ovarian adenocarcinoma cell line A2780 via cisplatin gradient. The growth rates of A2780/DDP and A2780 were detected via MTT and no significant difference was found. It suggests that the cell metabolic activity was not dramatically affected by DDP. Nevertheless, the sensitivity of A2780/DDP to cisplatin was significantly reduced, and the migration capacity of drug-resistant cell lines was significantly higher than that of A2780. P-cofilin is a major factor in the cytoskeleton and it is closely related to cell migration. Both in vitro and in vivo tests showed that p-cofilin expressions in patients with cisplatin-resistant ovarian cancer and A2780/DDP cells were significantly induced. The above results indicated indirectly that the p-cofilin expression level may be related to the mechanism of cisplatin resistance. Delorme-Walker et al. [19] found that the maintenance of cell morphology required actin, and dephosphorylation of cofilin activated actin. Nadkarni and Brieher [20] demonstrated in pancreatic cancer cell lines that the p-cofilin level in paclitaxel-sensitive cell lines was significantly lower than that in drug-resistant cell lines. The above results also revealed that the p-cofilin expression level may be associated with cell resistance. In the context, pearson correlation analysis further revealed that cisplatin resistance was positively correlated with p-cofilin expression. The therapeutic regimen and general pathological data of patients with ovarian cancer were analyzed and the relationship between the p-cofilin expression level and the prognosis of patients was investigated. The survival data showed that the survival time of patients with low- or normal-expression p-cofilin was significantly longer than that of patients with high-expression p-cofilin (
Conclusion
Our data demonstrate that p-cofilin expression in patients with ovarian cancer is positively correlated with cisplatin resistance, which affects the treatment effect and prognosis of patients. Our results provide new ideas for the clinical treatment of ovarian cancer as well as clues of the mechanism of chemotherapy drug resistance.
Footnotes
Conflict of interest
All author declare that they have no conflict of inter- est.
