Abstract
The objective of this study is to evaluate the relationship between discoidin domain receptor 2 (DDR2) and miR-182 expression with response to platinum-based chemotherapy and survival in women with high-grade serous ovarian cancer (HGSOC). We evaluated 78 women with HGSOC stages I-IV, diagnosed between 1996 and 2013, and followed up until 2016. DDR2 expression was assessed using immunohistochemistry on tissue microarray slides. The microRNAs were evaluated by qRT-PCR. DDR2 expression was high in 11 (14.1%) women. PFS was significantly lower in women with FIGO stage I/II – versus III/IV, post-surgery residual disease and high expression of DDR2. Women with postsurgery residual disease, FIGO stage I/II – versus III/IV and DDR2 expression had worse OS, but only post-surgery residual disease remained an independent prognostic factor for worse OS in multivariable analysis. miR-182 expression levels were significantly lower in patients harboring tumors with higher expression of DDR2 (p < 0.001). In this relatively large cohort of women with HSGOC, higher DDR2 expression was associated with lower miR-182 levels and worse PFS, suggesting that these molecules may be associated with mechanisms of HGSOC progression.
Introduction
Ovarian carcinoma is a common cause of death among women with gynecological cancer, and high-grade serous ovarian carcinoma (HGSOC) is responsible for the majority of deaths. 1 More than 70% of HGSOC are diagnosed at stages III and IV and harbor a dismal prognosis, with meager 30% 5-year survival. 2 Since most cases present with disseminated disease in the peritoneal cavity at diagnosis, current standard treatment protocols generally mandate either cytoreductive surgery, if feasible, followed by adjuvant platinum-based chemotherapy, or neoadjuvant chemotherapy followed by surgery.3,4 Despite high response rates to first-line treatment with platinum-based chemotherapy, most patients with HGSOC will relapse or progress. 5 Therefore, the identification of prognostic and predictive biomarkers in HGSOC may help tailor pharmacological treatment.
Recently, some studies demonstrated that protein components, such as discoidin domain receptor 2 (DDR2),6–8 and specific microRNAs (miRNAs), such as miR-182 and miR-9,9–11 are involved in well-recognized oncogenic pathways. DDR2 belongs to the tyrosine kinase receptor family, which mediates cell interactions with structural components of the extracellular matrix (ECM). 12 DDR2 is activated through fibrillar collagen type I, which is one of the most abundant components of the ECM. 13 The ECM contributes to cancer cell migration, acts as an immunologic barrier, forms a niche for new metastatic cells, and provides survival and proliferation signals. 14 Aberrant DDR2 cytoplasmic expression patterns have been studied in different sets of tumor types. In head and neck squamous carcinoma, DDR2 expression was associated with high-grade tumors, but no prognostic value was demonstrated. 15 In hepatocellular carcinoma (HCC), DDR2 expression was found to be an independent prognostic factor, since patients whose tumors present higher DDR2 expression had a lower 5-year disease-free and overall survival (OS). 7 In breast cancer, high expression of DDR2 was associated with high tumor grade and triple-negative subtype. 6 DDR2 was also associated with breast cancer proliferation, migration, and metastasis. 14 To our knowledge, there is less than a handful of studies addressing DDR2 expression in ovarian cancer.8,16 Using quantitative real-time polymerase chain reaction (qRT-PCR), Fan et al. found an association between higher DDR2 expression levels in ovarian carcinoma compared to normal ovarian tissue. They also found a significant association between higher cytoplasmic DDR2 immunohistochemical expression with advanced stage and poor OS. In addition, Grither et al. detected a relationship between DDR2 expression in ovarian cancer specimens with advanced stage and metastasis. In vivo studies in breast cancer and HCC showed that DDR2 stabilizes SNAIL1, inducing cell invasion and migration.7,14,17 In ovarian cancer, DDR2 plays a critical role in invasion and cell migration by stabilizing SNAIL1, sanctioning for a sustained mesenchymal phenotype. 16 MiR-182 expression in HGSOC is associated with tumor growth and invasion, largely by its negative regulation of multiple tumor suppressor genes involved in metastasis and DNA instability. 18 In breast cancer, one study showed that miR-182 expression was also influenced by SNAIL1, and its overexpression was associated with lung metastases. 19
This study was designed to evaluate the prognostic value of DDR2 as immunohistochemical marker and its association with miR-182 expression in women with HGSOC.
Materials and methods
Patients and tissue specimens
For this retrospective cohort study, the medical records of 114 women with HGSOC were retrieved. These patients had been diagnosed and treated at the Women’s Hospital Prof. Dr. José Aristodemo Pinotti, State University of Campinas, Campinas, Brazil, from 1996 to 2013 and followed up until 2016. This study was approved by the local institutional ethics committee (CEP 1086/2009 and CEP 710.452/2014). All pathological specimens, collected during primary surgery or percutaneous biopsy before chemotherapy, were analyzed by an expert gynecological pathologist according to the guidelines of the World Health Organization 20 and staged according to Federation of Gynecology and Obstetrics (FIGO) classification. 21 Exclusion criteria were as follows: second primary cancer (2 women), no available formalin-fixed paraffin-embedded (FFPE) tissue sample (17 women), and incomplete files (6 women). Due to technical difficulties, adequate immunohistochemistry reading of DDR2 was not possible for 11 women. FFPE tissue samples from the remaining 78 HGSOC women with complete data were included. MiRNA expression was evaluated for the 63 women who underwent at least three cycles of platinum-based chemotherapy.
Age, disease stage, CA125, post-surgical residual disease, number of cycles of platinum-based chemotherapy, platinum response, progression-free survival (PFS), and OS data were obtained from the clinical files. For PFS and OS, time was estimated in months, from the date of diagnosis to the last follow-up visit, recurrence, or death. For platinum response, time was estimated in months, from the end of primary platinum-based chemotherapy treatment to the date of progression. Platinum response was classified as recommended by Patch et al. 22 Women with less than three cycles of platinum-based chemotherapy were not included in platinum response analysis.
Immunohistochemistry
Slides stained with hematoxylin and eosin from the original FFPE blocks were analyzed for the selection of representative tumor regions. Tissue microarray (TMA; Beecher Instruments Microarray Technology, Silver Spring, CA, USA) was built and TMA sections were placed on electrically charged slides for immunohistochemical procedures. Sections were deparaffinized with xylol and dehydrated in alcohol series. Washes in hydrogen peroxide were performed, followed by distilled water washes. For antigen retrieval, we used a commercially available pressure cooker (T-fal®), in which slides were immersed in citrate buffer (pH: 6.0) for 30 min. The slides were dried at room temperature and washed in distilled water. After that, the sections were incubated in a moist chamber, with monoclonal mouse antibody against DDR2 (1:1500; Abcam, Cambridge, MA, USA) at 4°C, overnight. The slides were then washed in phosphate-buffered saline (PBS), pH 7.4. Slides were then incubated in ADVANCE™ HRP Detection System (Dako, Santa Clara, CA, USA) at 37°C for 1 h and washed in PBS. After that, DAB chromogenic substrate (3′-diaminobenzidine; Sigma, St Louis, MA, USA) was applied at a proportion of 0.06 g to 100 mL of PBS, 500 μL of hydrogen 3% peroxide, and 1 mL dimethylsulfoxide (DMSO) at 37°C for 5 min. Finally, the slides were washed in tap water and counterstained with Harris hematoxylin for 30–60 s. After being dehydrated, the slides were mounted in resin (Entellan®; Merck, Darmstadt, Germany). TMA analysis was performed according to protocols fully validated in ovarian carcinoma. 23 Internal/external and positive/negative controls were used for validation of the reactions. Positive control was done in myocardial (heart) tissue. Immunostaining was separately reviewed and scored by an expert gynecologic pathologist.
Image analysis
DDR2 protein expression was analyzed in the cytoplasm. The proportion and intensity of positively stained cancer cells were assessed. Scores were rendered as follows: Extension (1): number of positive stained cells ≤5%, score = 0; 6%–25%, score = 1; 25%–50%, score = 2; 51%–75%, score = 3; and >75%, score = 4; and intensity of stain and (2): colorless, score = 0; pallideflavens, score = 1; yellow, score = 2; and brown, score = 3. Scores (1) and (2) were multiplied, resulting in a final score ranging 0 to 12, which was further stratified as absent (score 0), weak (scores 1–4), moderate (scores 5–8), or strong (scores 9–12).8,15 Figure 1(a)–(d) shows examples of cases and their scores. For statistical purposes (simplification), the scoring system was further clustered in low/medium expression for scores of 0–8 and as high expression for scores of 9–12.

Representative images of DDR2 expression through immunohistochemistry assays in high-grade serous ovarian carcinoma: (a) DDR2 absent score 0, (b) DDR2 weak scores 1–4, (c) DDR2 moderate scores 5–8, and (d) DDR2 strong scores 9–12.
RNA extraction
Four 20-µm-thick FFPE sections from each sample were submitted to deparaffinization with xylene at 55°C and then washed in ethanol. Total RNA was extracted using the RecoverAll™ Total Nucleic Acid Isolation Kit (Ambion, Kruss, Denmark). RNA extraction was performed according to the manufacturer’s instructions, with the following modifications: digestion with lyse solution was performed with the addition of 8 µL proteinase K at 100 µg/mL (Invitrogen, Carlsbad, CA, USA) at 55°C overnight. Quantity and quality of total RNA were assessed with Epoch Spectrophotometer (BioTek Instruments, Winooski, VT, USA) using 260/280 and 260/230 ratios as quality controls. All samples had adequate RNA quantity and quality.
Reverse transcription and qRT-PCR analysis
Reverse transcription of 10 µg of total RNA (5 µL of total RNA at 2 µg/µL) to complementary DNA (cDNA) was done using TaqMan® MicroRNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) as per the manufacturer’s instructions. The primer sets for miR-182 and for the endogenous control (U6 snRNA) for specific reverse transcription were purchased from Applied Biosystems. Reverse transcription was carried out using Mastercycler Ep Gradient S (Eppendorf, Hamburg, Germany). TaqMan Fast Advanced Master Mix (Applied Biosystems) containing 10 µL of TaqMan Fast Advanced Master Mix, 1 µL of 20× Gene Expression Assay, 1 µL of cDNA and 8 µL of RNase-free water, total of 20 μL volume were processed in a 96-well optical plate at 50°C for 2 min, followed by 95°C for 30 s and then 40 cycles of 95°C for 10 s and 60°C for 30 s performed in 7500 System (Roche, Germany). The signal was collected at the finalization of every cycle. All reactions were run in triplicate for each case and the relative expression miR-182 was normalized using the 2−ΔΔCt method. 24
Statistical analysis
Data normality was assessed with the Shapiro–Wilk test. The relationship of DDR2 with stage, post-surgical residual disease, cycle of platinum, and platinum response was assessed using χ2 or Fisher tests; for continuous data, the Mann–Whitney test was applied. Hazard ratios (HR) and Cox proportional HR with 95%confidence interval (CI) were used to analyze PFS and OS. MiR-182 relative expression was normalized to U6 snRNA. The comparison between miRNA expression and DDR2 expression was performed using T tests. A p value lower than 0.05 was considered significant. All statistical analyses were performed using the R Project for Statistical Computing. 25
Results
Among the 78 women with HGSOC, high expression of DDR2 was found in 11 (14.1%). Older age was significantly associated with high DDR2 expression (p = 0.004). DDR2 expression was associated with FIGO stage, CA125 level, post-surgery residual disease, and response to platinum-based chemotherapy (Table 1).
High-grade serous ovarian carcinoma (HGSOC) patient distribution according to clinical features and discoidin domain receptor 2 (DDR2).
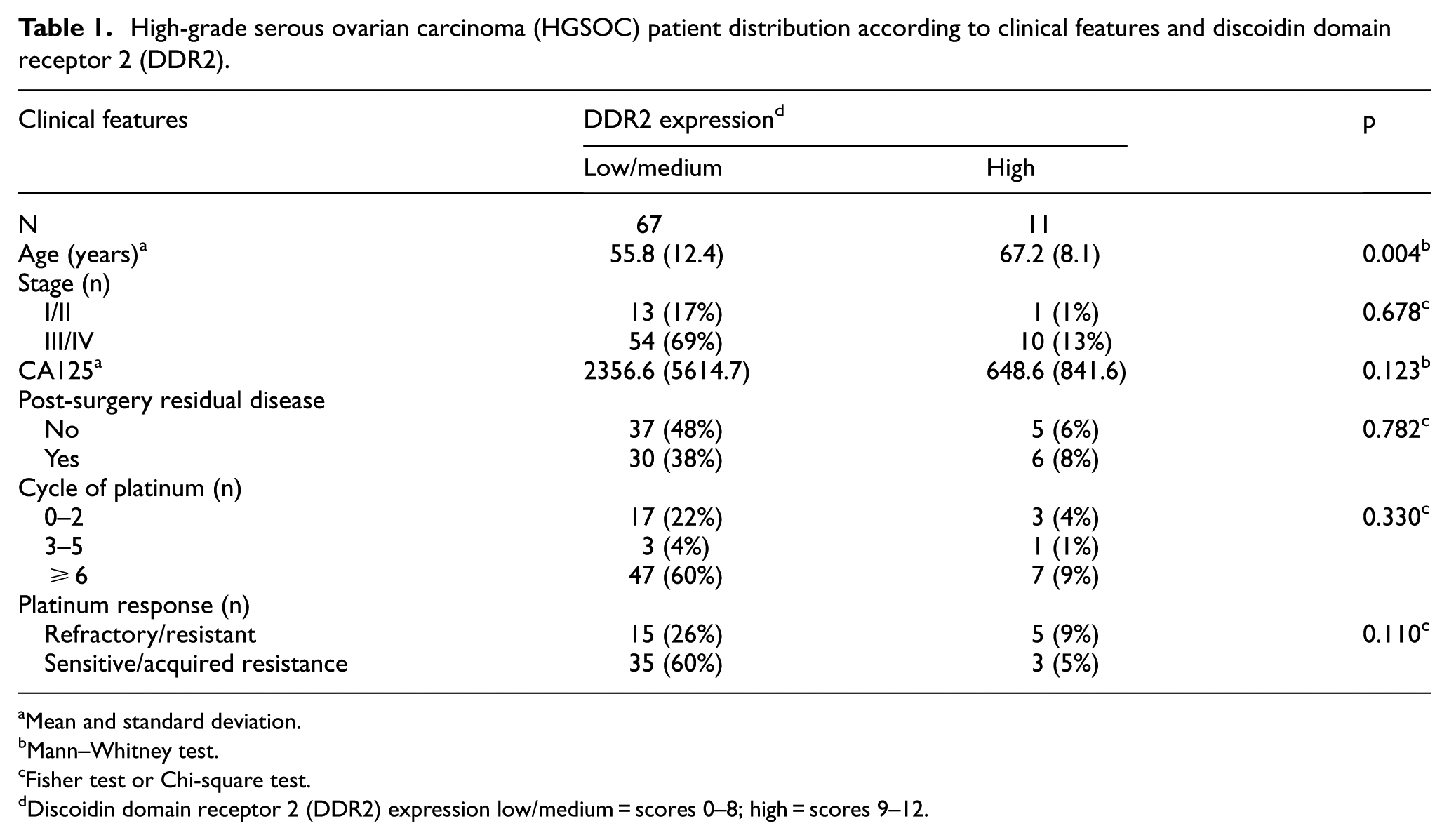
Mean and standard deviation.
Mann–Whitney test.
Fisher test or Chi-square test.
Discoidin domain receptor 2 (DDR2) expression low/medium = scores 0–8; high = scores 9–12.
In the univariate analysis, PFS was significantly lower in women with stage III/IV disease compared to that in women with stage I/II disease (HR = 5.40; 95% CI = 1.95–14.9; p = 0.001), post-surgery residual disease (HR = 2.29; 95% CI = 1.36–3.85; p = 0.001), and high expression of DDR2 (HR = 3.41; 95% CI = 1.58–7.34; p < 0.001). Women with III/IV FIGO stage (HR = 3.92; 95% CI = 1.13–13.60; p = 0.031), post-surgery residual disease (HR = 2.12; 95% CI = 1.10–4.06; p = 0.023), and high expression of DDR2 (HR = 2.60; 95% CI = 1.06–6.35; p = 0.035) had worse PFS after multivariate analyses. III/IV FIGO stage (HR = 4.09; 95% CI = 1.46–11.46; p = 0.007), post-surgery residual disease (HR = 2.05; 95% CI = 1.17–3.59; p = 0.012), and DDR2 expression (HR = 2.52; 95% CI = 1.01–6.25; p = 0.047) were associated with worse OS. However, in the adjusted analysis, only post-surgery residual disease was still associated with worse OS (HR 2.28; 95% CI = 1.04–4.95; p = 0.037) (Table 2).
Survival analysis considering key clinical features, discoidin domain receptor 2 (DDR2).

Cox proportional hazard ratio: HR (95% confidence interval).
Adjusted by stage, CA 125 level, post-surgery residual disease, and platinum response.
As shown in Figure 2, miR-182 expression levels were significantly lower in patients harboring tumors with higher expression of DDR2 (p < 0.001).

Representative images of miR-182 levels according to DDR2 expression.
Discussion
In our study, we investigated the association of DDR2 and miR-182 expression levels in a large set of women with HGSOC. Older age was significantly associated with high DDR2 expression. DDR2 expression was associated with FIGO stage, CA125 level, post-surgery residual disease, and response to platinum-based chemotherapy. Advanced stage, post-surgery residual disease, and high expression of DDR2 were independently associated with worse PFS. Only post-surgery residual disease remained an independent prognostic factor of worse OS after adjustment for other variables. MiR-182 expression levels were significantly lower in patients harboring tumors with higher expression of DDR2.
In a recently published cohort of 103 ovarian cancer patients of mixed histological types, Fan et al. 8 concluded that DDR2 expression was an independent prognostic marker of OS. In their study, tumors with moderate or strong immunostaining (scores 4–12) were labeled as high DDR2 expression; using that threshold, roughly 60% (63) had high DDR2 expression. One of the major limitations for the clinical use of DDR2 is the lack of standardization regarding staining and image analysis. Authors disagree with regard of cutoff points for DDR2 positivity, and many studies failed to determine the DDR2 positivity thresholds using receiver operating characteristic (ROC) curve analysis or any other technical procedure.6,8,15
In our study, miR-182 levels were significantly lower in tumors with high DDR2 expression. MiR-182 levels were not independently associated with PFS, but tumors with lower miR-182 and higher DDR2 expression levels had a worse PFS. MiR-182 and miR-96 belong to the miR-183 family. 26 Some authors reported miR-182 overexpression in aggressive subtypes of ovarian cancer, but those studies were based on in vitro experiments or orthotopic xenografts.18,27 Yang et al., 9 examining 459 cases of HGSOC in The Cancer Genome Atlas (TCGA) data, identified miR-182 along with other eight miRNAs as part of a regulatory network for the mesenchymal subtype. The mesenchymal subtype was determined through miRNA expression analysis as a predictor of poor patient OS. MiR-182 expression was associated with increased survival in TCGA cohort. Indirectly, these results are in perfect alignment with ours, since we detected that a reduced miR-182 expression was associated with higher DDR2 expression, which was per se a marker of worse PFS.
In our cohort of women with HGSOC, as expected, FIGO stage and post-surgery residual disease were important independent prognostic factors. Although we did not assess gene expression of DDR2 with in vitro and in vivo tests, we depicted a significant number of consecutive HGSOC women with available FFPE sample and annotated clinical data. Our data show that further mechanistic studies are needed to establish the association of these molecules in HGSOC progression and response to platinum-based chemotherapy. To the best of our knowledge, this is the largest cohort study of Brazilian women with HGSOC, in which all cases were reviewed by an expert pathologist, DDR2 expression and miR-182 expression levels were evaluated, and platinum resistance was assessed. In essence, DDR2 expression was independently associated with worse PFS but not OS. Lower miR-182 expression levels were associated with high DDR2 expression.
Footnotes
Acknowledgements
Susana Ramalho, Rodrigo de Andrade Natal, Amanda Canato Ferracini, Marina Pavanello, and Sophie Derchain provided and analyzed clinical data and wrote the paper. Luis Otávio Sarian, Luis Felipe Sallum, and Cássio Cardoso Filho wrote and corrected the manuscript. Liliana AL De Angelo Andrade performed the immunohistochemical evaluation. Susana Ramalho, Rodrigo de Andrade Natal, and Amanda Canato Ferracini performed the qRT-PCR analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by two grants from the National Council for Scientific and Technological Development (CNPq) number 306583/2014-3 and the São Paulo Research Foundation (FAPESP) number 2016/07822-4.
