Abstract
Multiple myeloma (MM) is a common hematological malignancy that is often associated with osteolytic lesions, anemia and renal impairment. Deregulation of miRNA has been implicated in the pathogenesis of MM. It was found in our study that miR-19b and miR-20a as members of crucial oncogene miR-17-92 cluster were differentially expressed between patients with MM and normal controls by genechip microarray, and this result was further confirmed in sera of patients with MM by qRT-PCR. The functional effect of miR-19b/20a was analyzed and results showed that miR-19b/20a promoted cell proliferation and migration, inhibited cell apoptosis and altered cell cycle in MM cells. PTEN protein expression was reduced after transfection of miR-19b/20a, suggesting that PTEN was a direct target of miR-19b/20a. In addition, over-expression of miR-19b/20a reversed the anti-proliferation and pro-apoptosis effect of PTEN in MM cells. Finally, our in vivo experiment demonstrated that lentivirus-mediated delivery of miR-20a promoted tumor growth in murine xenograft model of MM, which provide evidence that miR-20a inhibitor exerts therapeutic activity in preclinical models and supports a framework for the development of miR-19b/20a-based treatment strategies for MM patients.
Introduction
Multiple myeloma (MM) is a plasma cell malignancy characterized by aberrant expansion of plasma cells within the bone marrow, and is the second most common hematological malignancy after non-Hodgkin’s lymphoma [14, 20]. The pathogenesis of MM is complex and remains elusive despite novel insights into the pathobiology of the disease and the availability of new research platforms and therapeutics. Therefore, innovative treatment strategies are urgently needed [18, 25, 30]. Recent studies [3, 11] have demonstrated abnormal expression of microRNAs (miRNAs or miRs) in MM. MiRNAs are known to be a group of small, non-coding RNAs with 18–25 nucleotides, which bind to the 3’-untranslated region (3’-UTR) of their target mRNAs to regulate gene expression, causing target degradation or inhibiting translation [9, 21, 28]. MiRNAs have been demonstrated to be essential regulators of cellular functions, including proliferation, differentiation and apoptosis, in almost all tumor types [2].
Phosphatase and tensin homolog deleted on chromosome 10 (PTEN) is a key tumor-suppressor gene that is disrupted or suppressed in various cancer types [1, 26, 27], including MM [6, 31]. Previous results [17] demonstrated that PTEN could inhibit cell proliferation and migration, and promote cell apoptosis in MM. However, PTEN may be regulated by several miRNAs in MM, resulting in a reduction in its expression and loss of its original function. For example, miR-21, miR-221 and miR-222 downregulated PTEN and reactivated important signaling pathways to enhance oncogenicity in MM [10, 24].
The miR-17-92 gene is expressed as a polycistronic primary transcript containing six tandem stem-loop hairpin structures that ultimately yield six mature miRNAs (miR-17, miR-18, miR-19a, miR-19b, miR-20a and miR-92a). Recent studies [7, 13, 23] have indicated that primary members of the miR-17-92 cluster are dysregulated in MM and associated with disease development and progression, but the mechanisms of the six miRNAs require further investigation. In study by our group, abnormally expressed miRNAs in MM were identified by microarray. The results indicated that the expression of miRNAs containing miR-138, miR-1260b, miR-148a, miR-339, miR-138, miR-29b, miR-19b, miR-4449, miR-20a, let-7i and miR-152 was markedly increased in the patient group compared with that in the control group. MiR-19b and miR-20a are members of the miR-17-92 cluster, which has been identified as an oncogene [16]. We confirmed the increased expression of miR-19b and miR-20a in the sera of patients and further studied the regulation in MM in vivo and vitro. Comprehensive using three miRNA predicted software-Targetscan, miRanda and Pic Tar, we found that PTEN was predicted as a target of miR-19b and miR-20a. The regulation between PTEN and miR-19b/20a was further identified in MM.
Materials and methods
Cell lines and antibodies
MM cell lines U266, 8226, H929 and MM1S (Chinese Academy of Sciences, Shanghai, China) were cultured in complete DMEM (Corning, USA) supplemented with 10% fetal bovine serum (FBS) (Gibco, USA), 100 U/mL penicillin, 100 mg/mL streptomycin, and 2 mmol/L L-glutamine (Beyotine, China). Cell cultures were kept in cell incubators with a humidifed atmosphere and 5% CO
Patients and serum samples
Serum samples were obtained from patients with newly diagnosed multiple myeloma (NDMM) (
Quantitative real-time reverse transcription-PCR (qRT-PCR)
Total RNA was extracted from 400
Cell transfection
U266 cells were seeded in six-well plates at a density of 5
WST cell proliferation assay
U266 cells were transfected with miR-19b/20a mimics, miR-19b/20a inhibitor and negative control for 48 h. 100
Transwell cell migration assay
U266 cells were transfected with miR-19b/20a mimics, miR-19b/20a inhibitor and negative control for 48 h. 100
Cell apoptosis assay
U266 cells were transfected with miR-19b/20a mimics, miR-19b/20a inhibitor and negative control for 48 h, and 100
Cell cycle analysis
U266 cells were transfected with miR-19b/20a mimics, miR-19b/20a inhibitor and negative control for 48 h, and cells were harvested by centrifugation, washed with PBS, and fixed with cold 70% ethanol overnight. The fixed cells were then stained with PI solution and 0.1% Triton X-100. After 1 h incubation in dark, fluorescence-activated cells were sorted by flow cytometry.
Luciferase reporter assay
For luciferase reporter experiments, 3’-UTR segment containing the miR-19b and miR-20a binding sites of PTEN was amplified by PCR from human genomic DNA and inserted into the psiCHECK-2 vector using the XhoI and NotI site immediately downstream from the stop codon of luciferase. DNA segments with scrambled target sites (PTEN-MUT) designed to interfere with seed sequence recognition were also cloned to serve as control for specificity. 293 T cells were transfected in 48-well plates using Lipofectamine 2000 according to the manufacturer’s instructions. For each well, 100 nM has-miR-19b, has-miR-20a or negative control was used. Firefly and renilla luciferses activities were measured using a dual-luciferase reporter assay 48 h after transfection on a GloMax 96 Microplate Luminometer.
Protein extraction and Western blotting analysis
U266 cells were transfected with miR-19b/20a mimics, miR-19b/20a inhibitor and negative control for 72 h. Then, cells were collected, washed with PBS and lysed in RIPA buffer (20 mM Tris, 150 mM NaCl, 1 mM EDTA, 1 mM EGTA, 1% Triton X-100, 2.5 mM sodium pyrophosphate, 1 mM-glycerolphosphate), plus 0.5 mM Phenylmethanesulfonyl fluoride (PMSF) on ice for 20 min. The resulting lysate was clarified by centrifugation at 12000 r/min for 20 min at 4
Animals and in vivo models of multiple myeloma
Nude mice (6–8 weeks old, 18–20 g weight) were housed and monitored in Experimental Animal Center of Nantong University (Nantong, China). To establish subcutaneous human multiple myeloma xenograft models, 5
Bioinformatic analysis
The option of Targetscan (
Statistical analysis
Statistical analyses were performed using Graphpad Prism 7.0 software. Each experiment was performed at least three times and all values are reported as mean
Expression of miR-19b/20a in samples from patients with MM. (A) Genechip microarray was analyzed in order to search potential tumor suppressor or tumor promoter miRNA genes. Sample MM-1, MM-2, MM-3 and MM-4 obtained from NDMM patients. Sample NC-1, NC-2 and NC-3 obtained from healthy subjects. (B) Expression levels of miR-19b/20a in 50 NDMM patients, 22 RRMM patients, 10 patients at CR and 30 healthy subjects were assayed by qRT-PCR. RNAs were isolated from serum samples. (C) Expression of miR-19b and miR-20a in bone marrow CD138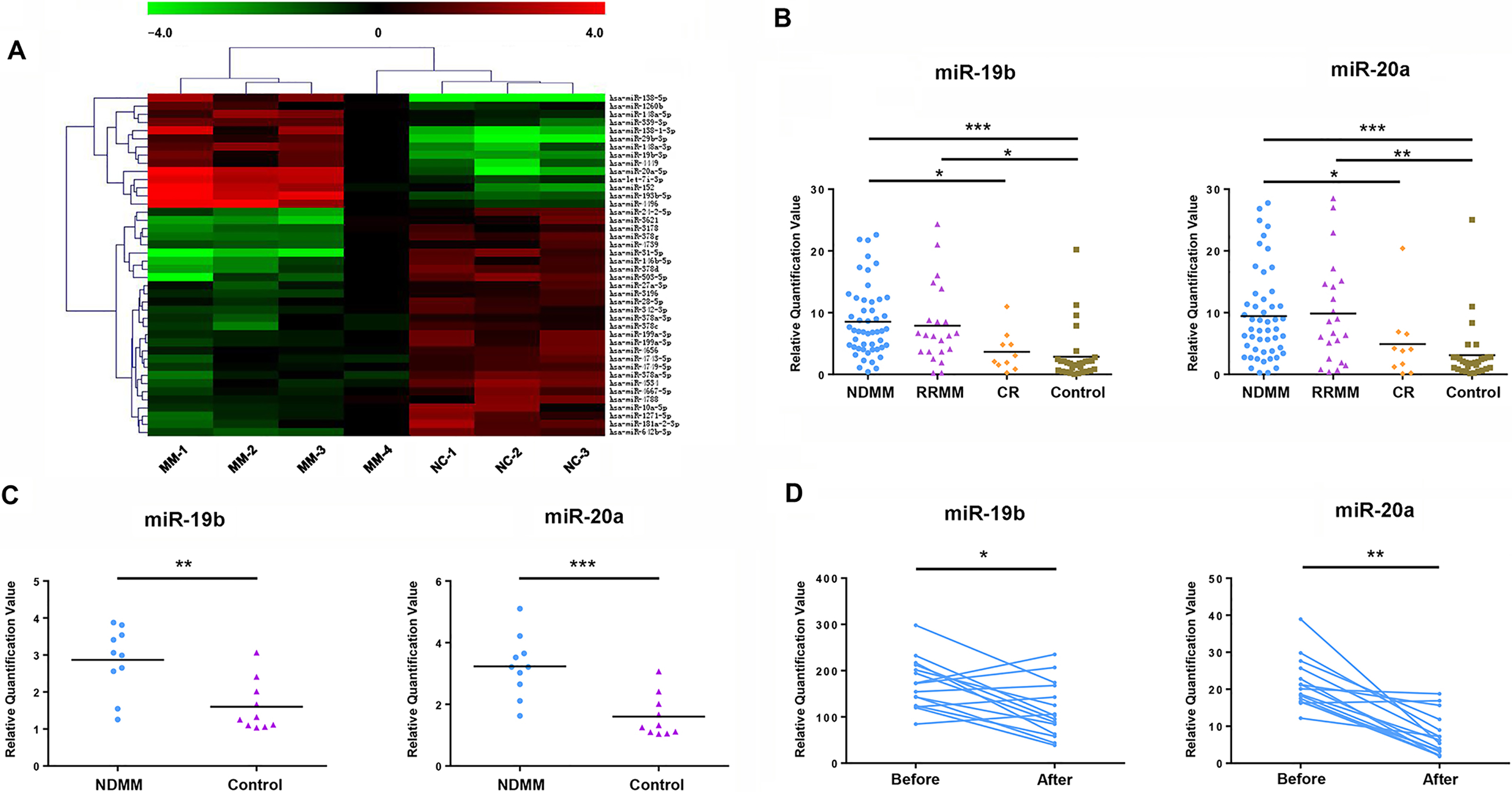
Clinical characteristics of patients with NDMM
Ig, immunoglobulin; ISS, international staging system.
Expression of miR-19b/20a in samples from patients with MM
To identify potential tumor suppressor or tumor promoter miRNA genes, differential expression of miRNAs between patients with MM and normal controls was analyzed by microarray (Fig. 1A). The results demonstrated that the expression of miR-19b/20a in patients with MM was significantly higher compared with normal controls. To further confirm the tumorigenic effect of miR-19b/20a in MM, we analysed the expression levels of miR-19b/20a in serum samples from 50 newly diagnosed MM (NDMM) patients (Table 1), 22 relapsed refractory MM (RRMM) patients, 10 patients at complete remission (CR) and 30 healthy subjects. QRT-PCR analysis indicated that the expression of miR-19b/20a in patients with NDMM and RRMM was significantly higher compared with normal controls (Fig. 1B). Moreover, compared with NDMM patients, the expression of miR-19b/20a was significantly lower in patients at CR. We further studied the expression levels of miR-19b/20a in primary CD138
Correlation between clinical parameters and miR-19b/20a
Correlation between clinical parameters and miR-19b/20a
LDH, lactate dehydrogenase;
MiR-19b/20a regulates proliferation, migration and apoptosis of MM cells. (A) Expressions of miR-19b and miR-20a in 4 MM cell lines (H929, U266, RPMI8226 and MM1S) were assayed by qRT-PCR. (B) U266 cells were transiently transfected with synthetic miR-19b/20a mimics, 19b/20a inhibitors and scrambled miRNA (negative control, NC). (C–E) After 48-h transfection treatment, transfected cells were examined for cell proliferation using WST assay and migration using transwell assay, counted migration cell numbers under a microscope. (F) After 48-h transfection, U266 cells were examined for apoptosis using Annexin/propidium iodide double staining by flow cytometry. (G) Apoptosis-related protein Bcl-2, Bax and Bad were examined for confirming the apoptosis result by Western blot after 72-h transfection of U266 cells. (H) Cell cycle distribution was detected by flow cytometry using propidium iodide staining after 48-h transfection of U266 cells. 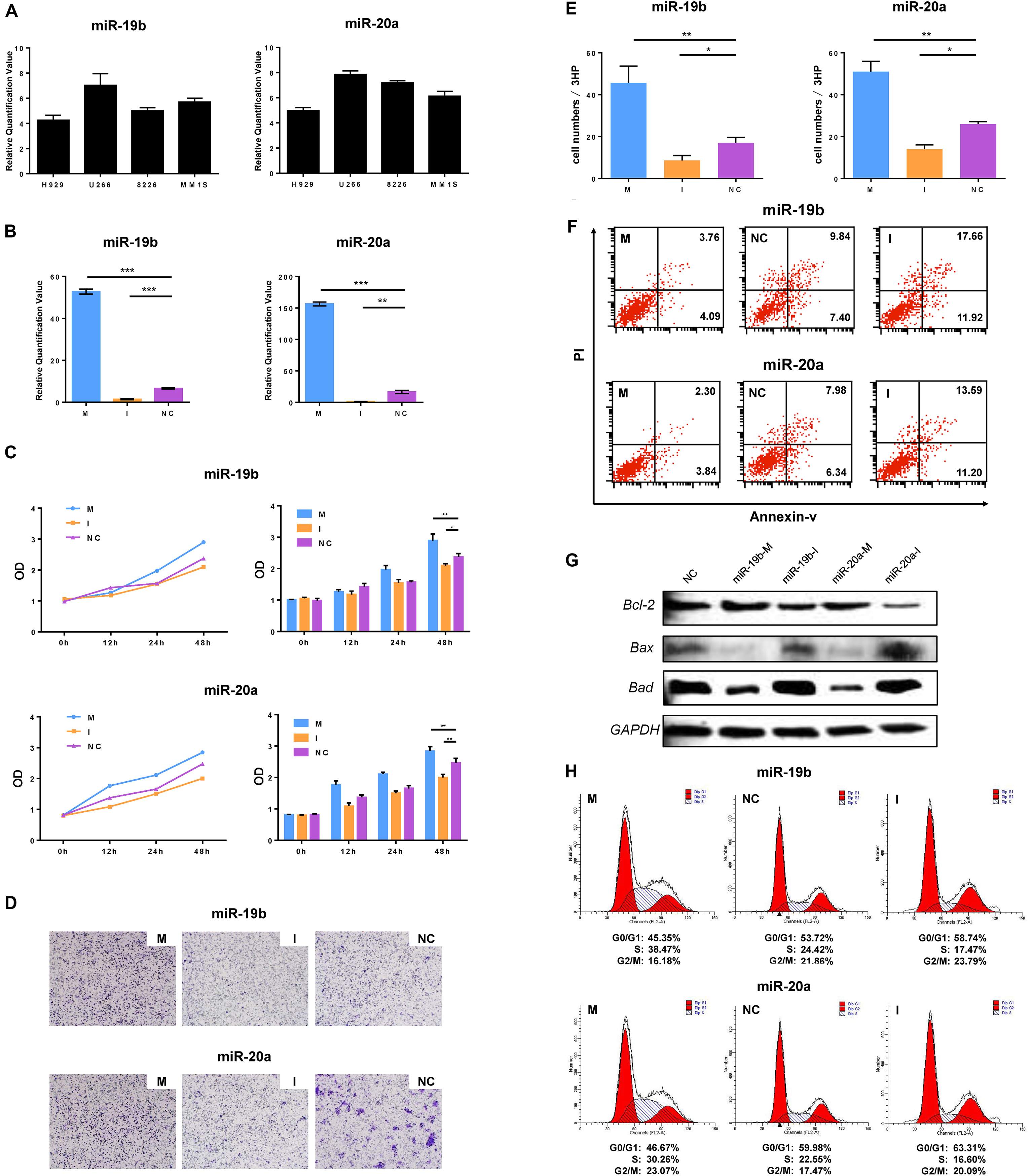
MiR-19b/20a expression was first evaluated in four MM cell lines (H929, U266, RPMI8226 and MM1S). It was identified that the expression of miR-19b and miR-20a in U266 was significantly higher compared with the other three MM cell lines (Fig. 2A). To determine the role of miR-19b/20a in MM cells, synthetic miR-19b mimics, miR-19b inhibitors, miR-20a mimics, miR-20a inhibitors and scrambled miRNA (negative control, NC) were transfected into U266 cell lines. Given the high transfection efficiency, related cellular function experiments were performed subsequently (Fig. 2B). The WST cell proliferation assay indicated that the growth of miR-19b mimic-transfected U266 cells was significantly higher compared with NC-transfected cells in a
MiR-19b and miR-20a directly target PTEN. (A) The possible binding region between miR-19b/20a and PTEN 3’-UTR was predicted using bioinformatics software. Matched nucleic acid base pairs were linked as “ 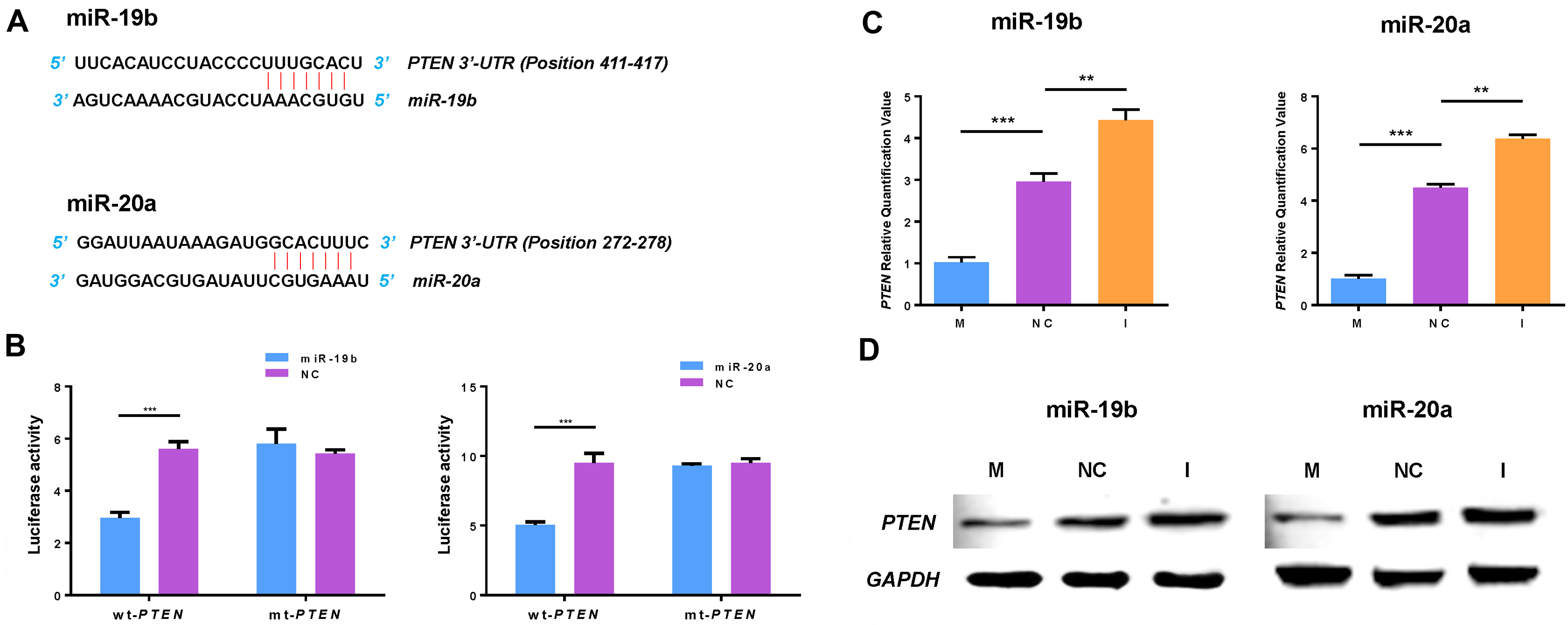
time-dependent manner. The same result was obtained following transfection of miR-20a mimics into the cells (Fig. 2C). The Transwell migration assay demonstrated that cell migration ability was increased following transfection of miR-19b mimics and miR-20a mimics into U266 cell lines compared with the NC group (Fig. 2D–E). In addition, overexpression of miR-19b/20a in U266 cells decreased apoptosis of U266 cells as indicated by flow cytometry. By contrast, transfection of miR-19b or miR-20a inhibitor promoted apoptosis of U266 cells (Fig. 2F). Subsequently, these results were confirmed by western blot analysis. Three apoptosis-associated proteins were detected. The results demonstrated that the expression of Bcl-2 protein was increased following transfection of miR-19b mimics and miR-20a mimics into U266 cell lines, but the expression of Bax and Bad proteins was decreased (Fig. 2G). A cell cycle assay indicated that G0/G1 phase cells were decreased and S phase cells were increased following transfection of miR-19b mimics and miR-20a mimics into U266 cells as compared with the NC group (Fig. 2H). These data further confirm the role of miR-19b/20a as positive regulators of cell growth in MM cells.
MiRNA prediction software programs TargetScan, miRanda and Pic Tar predicted PTEN as a target of miR-19b and miR-20a, and identified a possible binding region of miR-19b and miR-20a in the 3’-UTR of PTEN (Fig. 3A). Subsequently, the 3’-UTR sequence of PTEN was cloned into the luciferase-expressing vector psiCHECK-2, downstream of the luciferase stop codon. Pre-miR-19b, pre-miR-20a and scrambled oligonucleotide were transfected into U266 cells. The results indicated that miR-19b and miR-20a significantly reduced luciferase activity compared with the scrambled control miRNA in wild type (Fig. 3B). There was no significant difference between the two groups in the mutant type. Synthetic miR-19b/20a mimics or inhibitors were transfected into U266 cells. QRT-PCR and western blotting analyses confirmed a regulatory interaction between miR-19b/20a and PTEN (Fig. 3C and D), suggesting that miR-19b and miR-20a bound to the 3’-UTR of PTEN and impaired its mRNA translation.
Overexpression of miR-19b/20a down-regulates PTEN and reverts cell proliferation and apoptosis in MM cells. (A) Relative expression levels of PTEN in 28 newly diagnosed MM patients and 10 healthy subjects were assayed by qRT-PCR. RNAs were isolated from serum samples. (B) The expression of PTEN in 4 MM cell lines (H929, U266, RPMI8226 and MM1S) was assayed by qRT-PCR. (C) U266 cells were transiently cotransfected with expression plasmid of PTEN 3’-UTR and miRNAs (miR-19b mimics, miR-20a mimics, or scrambled miRNA). After 48-h transfection treatment, cell proliferation was detected by WST assay. (D) Cell apoptosis was detected using Annexin/propidium iodide double staining by flow cytometry after 48-h transient co-transfection. 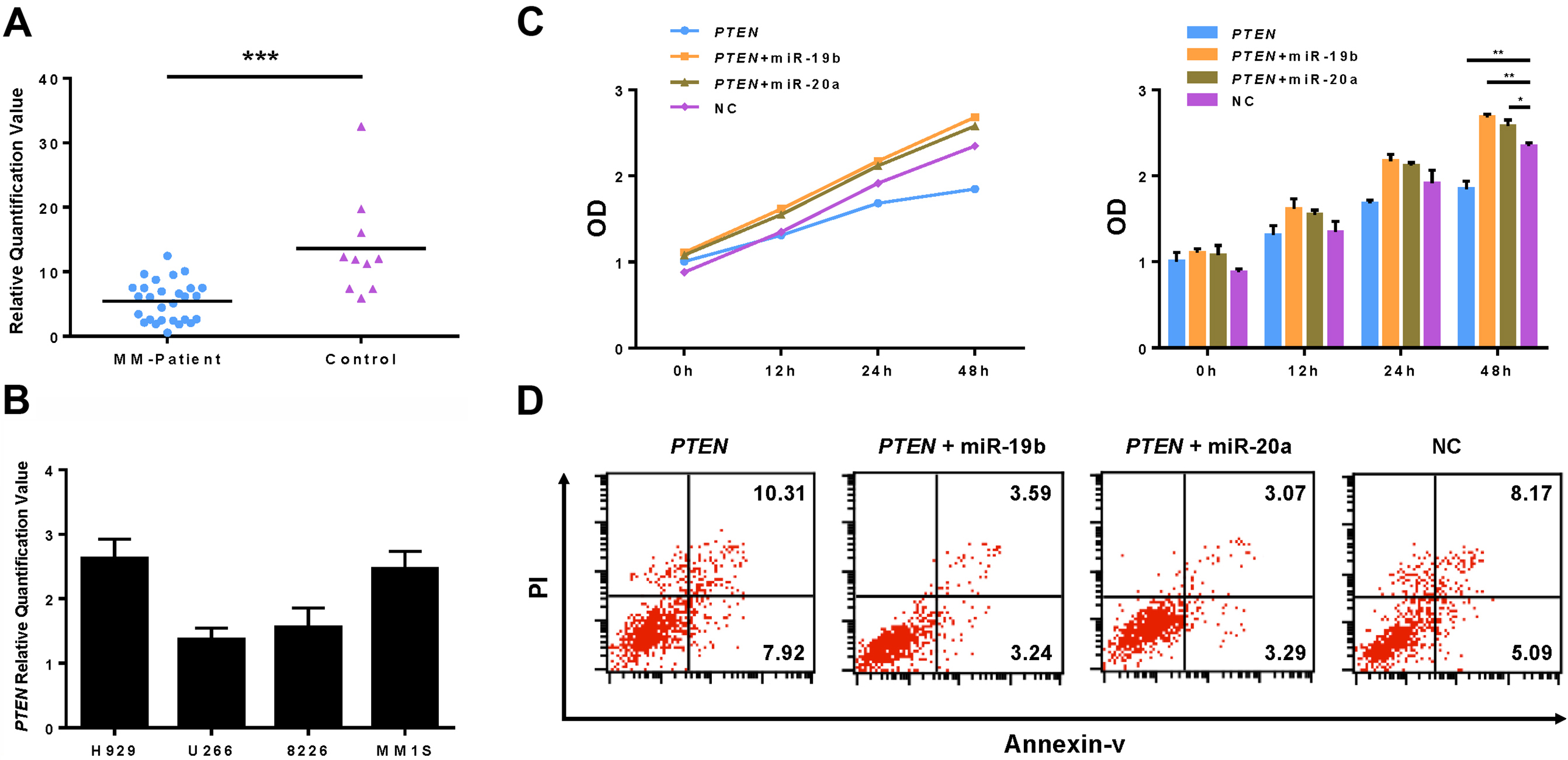
To investigate the effect of tumor suppressor gene PTEN in MM, serum samples from 28 patients with MM and 10 normal controls were assayed via qRT-PCR. The results indicated that the expression of PTEN was significantly higher in normal controls, compared with in patients with MM (Fig. 4A). In addition, the expression of PTEN was higher in the MM1S cell line compared with in the U266 cell line (Fig. 4B). The expression level of PTEN was inversely correlated with that of miR-19b/20a in the four cell lines tested. To establish a more definitive functional association between PTEN and miR-19b/20a, U266 cells were transiently co-transfected with expression plasmids of PTEN 3’-UTR and miRNAs (miR-19b mimics, miR-20a mimics or scrambled miRNA) for proliferation and apoptosis analysis. The WST cell proliferation assay demonstrated that overexpression of PTEN inhibited cell proliferation in U266 cells (Fig. 4C). However, proliferation in these cells was significantly enhanced when miR-19b or miR-20a was overexpressed. In addition, the increase in cell apoptosis induced by overexpression of PTEN was suppressed by overexpression of miR-19b or miR-20a in U266 cells (Fig. 4D). These results indicated that overexpression of miR-19b/20a reverted the effect of PTEN in MM cells, and PTEN-mediated cell death was attenuated by miR-19b/20a.
Inhibition of miR-20a exerts an anti-MM effect in vivo
For the in vivo study, the effect of transduced lentiviral-miR-20a on the tumorigenic potential of MM cells engrafted in nude mice was examined. It was observed that either overexpression of miR-20a alone or co-transfection with expression plasmid of PTEN promoted tumor growth in mice injected with U266 cells, while lentivirus-transfected miR-20a inhibitor suppressed tumor formation (Fig. 5A and B). The weight of the mice increased gradually due to tumor growth (Fig. 5C). Notably, the life span of mice injected with lentiviral-miR-20a inhibitor-transfected U266 cells was significantly longer compared with mice receiving empty vector U266 cells (Fig. 5D). Immunohistochemistry analysis of the excised tumors indicated lower expression of Ki67 and higher expression of Bcl-2 in miR-20a inhibitor-transduced mice compared with the empty vector-transduced mice (Fig. 5E). In summary, these results indicate that inhibition of miR-20a suppressed the proliferation and stimulated the apoptotic cascade of U266 xenografts.
Inhibition of miR-20a exerts an anti–MM activity in vivo. Mice were divided into four groups (lentiviral-miR-20a inhibitor group, lentiviral-miR-20a overexpression group, lentiviral-miR-20a and expression plasmid of PTEN cotransfected group and negative control) and injected with U266 cells with or without lentiviral-miR-20a, lentiviral-miR-20a inhibitor or expression plasmid of PTEN, and then tumor growth, body weight and survival were observed. (A–B) Mice were dead 30 days later, and there was a significant difference of tumor volume in lentiviral-miR-20a inhibitor group, lentiviral-miR-20a overexpression group, and lentiviral-miR-20a and expression plasmid of PTEN cotransfected group compared with negative control. (C) Body weight of mice was measured every 2 days until death. (D) Pairwise comparison of animal survival was carried out between lentiviral-miR-20a inhibitor group and negative control (both 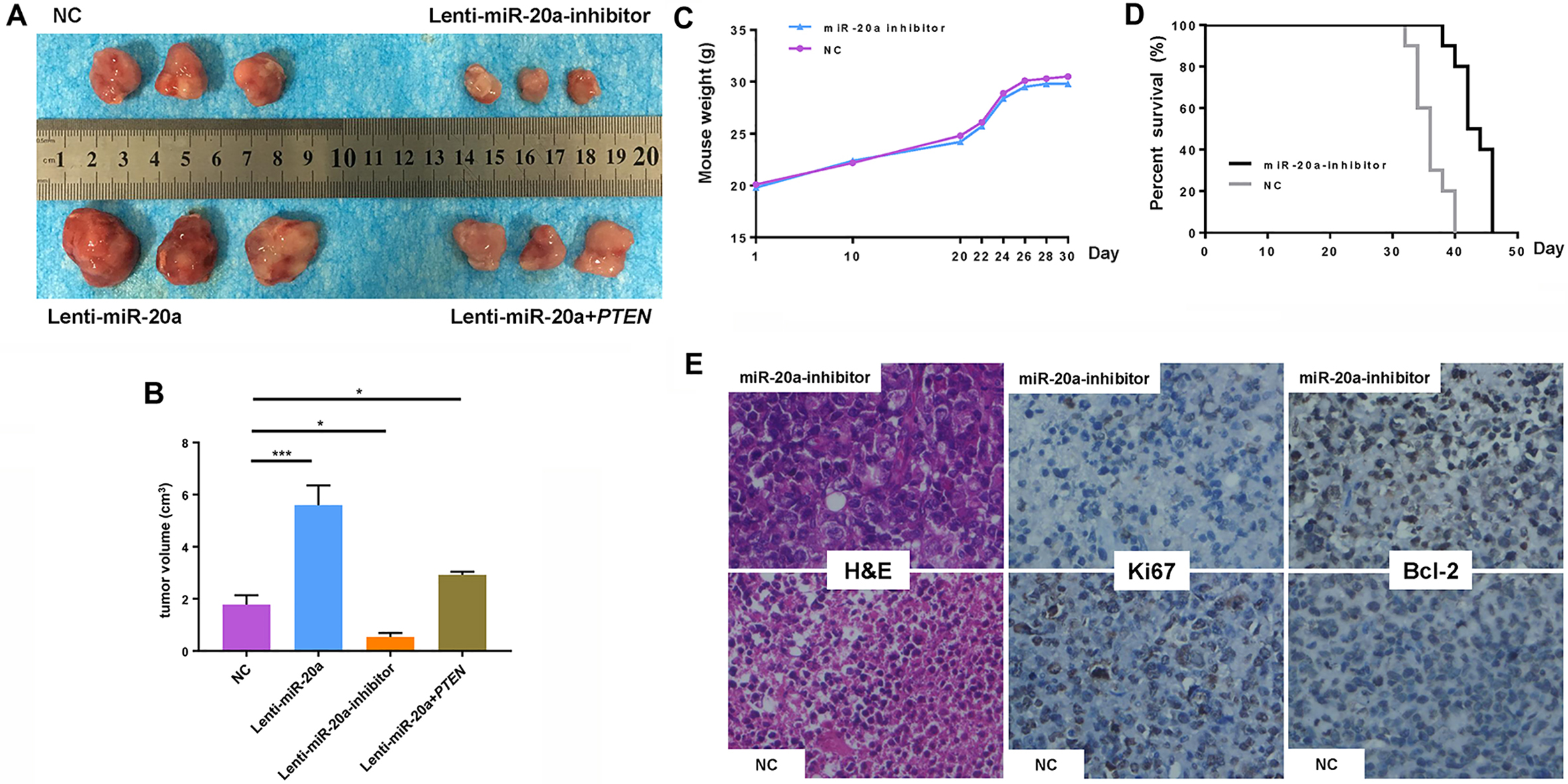
MM is one of the most common hematological malignant malignancies, and is often complicated by osteolytic lesions, anemia and renal impairment [29]. Abnormal expression of miRNAs has been demonstrated in MM and speculated to be associated with the pathogenesis and progression of the disease. The effects of miRNAs are characterized by binding to the 3’-UTR of target mRNAs to regulate gene expression, causing mRNA degradation or translation inhibition. In addition, large numbers of studies have demonstrated that the pathogenesis of MM is closely associated with the activation of multiple signaling pathways with regulation of miRNAs. For example, adhesion of bone marrow stromal cells promotes the secretion of IL-6 to activate JAK and STAT3. Activation of STAT3 signaling pathway promotes cell proliferation and migration, while it is inhibited by suppressor of cytokine signaling 1 (SOCS1) and PTEN [5, 12, 19]. Overexpression of miR-19a and miR-19b suppresses the expression of SOCS1 protein and reactivates the STAT3 signaling pathway [23].
The miR-17-92 cluster, which has been identified as an oncogenic miRNA cluster implicated in several cancer types [4, 8, 15], including MM, contains six mature miRNAs (miR-17, miR-18, miR-19a, miR-19b, miR-20a and miR-92a). Previous studies [7, 13, 23] have indicated that primary members of the miR-17-92 cluster are dysregulated in MM and associated with disease development and progression, but the mechanisms of the six miRNAs require further investigation. In addition, one study reported conflicting results [22]. A microarray in our previous study indicated that there was a significant difference in the expression of miR-19b and miR-20a between patients with MM and normal controls. It was identified that the expression of miR-19b/20a in patients with NDMM was significantly higher compared with normal controls whether in serums or in primary CD138
To date, miR-19b/20a expression has only been evaluated in human serum and bone marrow biopsy samples. To investigate the potential role of miR-19b/20a as an oncogene and to study its mechanism, the expression of miR-19b/20a was assayed in MM cells in the current study. It was identified the expression of miR-19b/20a in the U266 cell line was significantly higher compare to the other myeloma cell lines. Furthermore, enforced expression of miR-19b/20a promoted the proliferation, migration and anti-apoptosis capacities of U266 cells. In addition, changes in the levels of apoptosis-associated proteins confirmed the anti-apoptotic effect of miR-19b/20a. Furthermore, miR-19b/20a altered the cell cycle by shortening G0/G1 phase and prolonging S phase of U266 cells, whereas suppression of miR-19b/20a using synthetic miRNA inhibitors in U266 cells led to the opposite result.
Since miR-19b/20a was identified to regulate myel- oma cells, particularly the U266 cell line, a search for targets of miR-19b and miR-20a in MM was conducted. A possible binding region of miR-19b and miR-20a in the 3’-UTR of PTEN was predicted by bioinformatics software. It was observed that enforced expression of miR-19b/20a resulted in significant downregulation of PTEN at the mRNA and protein level. Subsequently, a luciferase reporter assay confirmed that PTEN was a direct target of miR-19b and miR-20a. PTEN is an important tumor suppressor gene that plays anti-proliferative and pro-apoptotic roles in MM, according to numerous studies. The anti-cancer effect of PTEN could be inactivated by certain regulator genes, such as miRNAs, therefore the regulatory relationship between miR-19b/20a and PTEN was investigated. Following co-transfection with expression plasmid of PTEN and miR-19b/20a mimics, overexpression of miR-19b/20a downregulated PTEN and inhibited the anti-proliferative and pro-apoptotic effects of PTEN. This suggested that the anti-MM effect of PTEN was regulated by miR-19b/20a. Therefore, inhibition of miR-19b/20a could enhance the anti-neoplastic effect of PTEN.
Finally, the potential therapeutic significance of miR-20a inhibitor was studied in a mouse subcutaneous xenograft model. It was demonstrated that stable transfection with the lentivirus vector of miR-20a inhibitor into U266 cells and subcutaneous injection into the mice resulted in miR-20a silencing, thus inhibiting tumor growth and prolonging survival of the mice. This demonstrated an oncogenic role of miR-20a and suggested that miR-20a could be a therapeutic target of MM in vivo.
In summary, the current study demonstrated that miR-19b/20a were oncogenes that may serve critical functions in the pathogenesis of MM by inducing cell proliferation and migration, inhibiting cell apoptosis and altering cell cycle. These findings support the development of miR-19b/20a inhibitors as a potential pharmacological intervention strategy in MM. Effective inhibition of miR-19b/20a to promote expression of PTEN protein may have potential as a targeted and effective therapeutic approach for the control of MM cell growth and survival.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China [81301498, 81271920] and Nantong Science and Technology Program [MS22015055].
