Abstract
The treatment of multiple myeloma has evolved markedly in the last decade, but mortality remains high, emphasizing the need for more effective therapies. Daratumumab, a fully human monoclonal antibody targeting CD38, has shown clinical efficacy in relapsed/refractory multiple myeloma both as monotherapy and in combination with other drugs, including novel agents. More recently, promising results have been reported in patients with untreated newly diagnosed multiple myeloma (NDMM). Clinical trials thus far have shown enhanced efficacy and tolerability of several daratumumab-based combinations in both transplant ineligible and eligible patients, without compromising transplant ability. However, benefit in high-risk subpopulations is still unclear. A subcutaneous formulation of daratumumab has been introduced to decrease the risk of infusion reactions, with preliminary results showing non-inferior efficacy. The antimyeloma activity of daratumumab is achieved through multiple mechanisms including direct, Fc-dependent, and immunomodulatory mechanisms. Enhanced efficacy of daratumumab in combination with immunomodulatory drugs and proteasome inhibitors is supported by preclinical data showing synergism. This review will focus on the role of daratumumab in untreated NDMM patients, highlighting the results of major clinical trials, and listing ongoing trials that are evaluating various daratumumab-based combinations in this setting.
Introduction
Multiple myeloma (MM) is a neoplastic clonal plasma cell disorder characterized by secretion of a monoclonal protein in the blood or urine and organ dysfunction manifesting as anemia, hypercalcemia, renal failure, and lytic bone lesions. 1 MM is more commonly diagnosed in men and older patients between the age of 65 and 74 years, and accounts for 1.8% of all new cancer cases in the US. 2 Treatment strategies in MM have evolved markedly in the past decade with the introduction of novel treatment agents.3–5 This has expanded the treatment options for both newly diagnosed multiple myeloma (NDMM) and relapsed or refractory (R/R) disease. These include immunomodulatory drugs (IMiD) like thalidomide, lenalidomide, and pomalidomide; and proteasome inhibitors (PI) like bortezomib, carfilzomib, and ixazomib. The heterogeneity of drug classes and nonoverlapping mechanisms of action has allowed their use in combination to achieve greater responses.3–5 In the absence of direct comparisons among all available combinations, the choice of initial treatment is currently guided by physician and patient preference, taking into account disease stage, transplant eligibility, risk stratification based on cytogenetic abnormalities, comorbidities, frailty, and functional status. 6 Despite advances in treatment, MM mortality remains high, with 5-year overall survival estimated at 52.2%. 2 This highlights the need for more effective therapeutic options. The CD38 targeted monoclonal antibody (mAb) daratumumab has shown clinical efficacy in R/R MM both as monotherapy, and in combination with other drugs, including novel agents, leading to its approval for use in this setting. More recently, it has been explored in untreated NDMM patients, with promising results in both transplant eligible and ineligible patients. The role of daratumumab in untreated NDMM patients will be the primary focus of this review.
Mechanisms of daratumumab activity
Daratumumab is an IgG1κ fully human mAb that targets CD38, a type II transmembrane glycoprotein composed of extracellular, transmembrane, and intracellular domains.
7
CD38 is widely expressed on nonhematopoietic and hematopoietic tissues, including plasma cells (PCs), natural killer (NK) cells, lymphoid cells, and myeloid cells. Its high expression in MM has made it an attractive target for treatment.
8
CD38 serves as a receptor for CD31. Upon ligand binding, CD38 interacts with other cell surface receptors, resulting in activation of intracellular signaling pathways, leading to cellular responses ranging from proliferation to apoptosis.7,9 In addition, CD38 is considered an ectoenzyme using NAD+ and NADP+ as its main substrates.7,9 The antimyeloma activity of daratumumab is exerted through several mechanisms: direct, Fc-dependent, and immunomodulatory mechanisms.
9
Direct effects are mediated by inhibition of intracellular signal transduction, and by inhibition of CD38 enzymatic activity, which leads to decreased levels of immunosuppressive adenosine. Fc-dependent mechanisms include complement-dependent cytotoxicity (CDC),
8
antibody-dependent cellular cytotoxicity (ADCC),
8
antibody-dependent cellular phagocytosis (ADCP),
10
and induction of apoptosis through cross-linking
The rationale for daratumumab-based combinations
The benefit of adding daratumumab to other treatment regimens, particularly IMiD- and PI- based combinations, is supported by preclinical data. Daratumumab has exhibited synergistic effects in enhancing MM cell lysis
Role of daratumumab in R/R MM
Since initial use in 2015,
19
there has been clear evidence for daratumumab efficacy in pretreated MM patients both as monotherapy and in combination with other agents. The GEN501 and SIRIUS trials provided the earliest reports on efficacy and safety of daratumumab as monotherapy in heavily pretreated R/R MM patients. A combined analysis showed durable responses after 36.6 months of median follow up, with an overall response rate (ORR) of 30.4%, median overall survival (OS) of 20.5 months (CI 16.6–28.1), and 3-year OS rate of 36.5% (28.4–44.6).19–21 Results from these studies led to the approval of daratumumab monotherapy in patients with at least three lines of prior therapy including an IMiD and PI, or double refractory to an IMiD and PI. Evidence for clinical efficacy in combination with bortezomib and lenalidomide in R/R patients was provided by the CASTOR and POLLUX trials, respectively.22,23 In the CASTOR trial, the combination of daratumumab with bortezomib and dexamethasone (DVd) was superior to bortezomib and dexamethasone (Vd) alone, achieving significantly longer progression-free survival (PFS) (median PFS: 16.7
Role of daratumumab in untreated MM
The efficacy of daratumumab observed in R/R MM, particularly among patients with fewer lines of prior therapy, 24 has been replicated in untreated MM, expanding its labeled use to include transplant-ineligible NDMM. 32 This was based on results from the ALCYONE and MAIA trials, showing improved responses when daratumumab is combined with Rd or melphalan-bortezomib-prednisone (MVP) respectively.33,34 In addition, encouraging results are being reported in other combinations with standard-of-care regimens, and deeper responses are being achieved even in transplant-eligible patients, without compromising the ability to proceed to transplant. We discuss below the most important clinical trials evaluating daratumumab combinations in NDMM.
Daratumumab in transplant-ineligible NDMM
The dosing and safety of daratumumab, in combination with standard regimens in NDMM patients, were established in a phase Ib trial where four different daratumumab-based combinations were used, including melphalan, bortezomib, thalidomide, and pomalidomide.
35
Subsequently, the phase III ALCYONE trial evaluated the efficacy of daratumumab in combination with bortezomib-melphalan-dexamethasone (D-VMP), compared with bortezomib-melphalan-dexamethasone alone (VMP), among 706 transplant ineligible NDMM patients. After a median follow up of 16.5 months, the D-VMP group had significantly improved PFS compared with the VMP group with a median PFS NR
Given the lack of direct head-to-head comparisons, there is little evidence to guide treatment choice between the various standard regimens at this time. A recently published metanalysis by Cao and colleagues compared the efficacy of currently used treatment regimens in this group of patients using Rd as reference. In terms of PFS, three combinations showed superiority to Rd: DRd (HR: 0.57, 0.43–0.73), D-VMP (HR, 0.59, 0.36–0.91), and VRd (HR, 0.72, 0.56–0.90). VRd showed superior OS, but the analysis excluded daratumumab-based regimens given immaturity of OS data. 39 The efficacy of daratumumab is also being evaluated in other PI-based doublet regimens, specifically in frail patients with NDMM. The phase II HOVON 143 study is evaluating the efficacy of daratumumab in combination with ixazomib and low-dose dexamethasone in this subset of patients. In its first planned safety analysis, the combination was tolerated with manageable side effects. A preliminary ORR of 70% has been reported, including 20% VGPR. Updated efficacy results are awaited. 40 Ongoing trials of daratumumab-based combinations in transplant ineligible NDMM patients are listed in Table 1. 41
Trials evaluating daratumumab combinations in transplant ineligible patients with NDMM.

Bort, bortezomib; Car, carfilzomib; CR, complete response; D-KRd, daratumumab, carfilzomib, lenalidomide, dexamethasone; D-VMP, daratumumab, bortezomib, melphalan, prednisone; D-VRd, daratumumab, bortezomib, lenalidomide, dexamethasone; Dara, daratumumab; Dex, dexamethasone; DLT, dose-limiting toxicity; DRd, daratumumab, lenalidomide, dexamethasone; DVd, daratumumab, bortezomib, dexamethasone; IV, intravenous; Ixa, ixazomib; KRd, carfilzomib, lenalidomide, dexamethasone; Len, lenalidomide; Mel; melphalan; MRD, minimal residual disease; NCT, National Clinical Trial; NDMM, newly diagnosed multiple myeloma; ORR, overall response rate; PFS, progression-free survival; PO, by mouth; Pred, prednisone; QD, daily; QOD, every other day; Qw, every week; Rd, lenalidomide; dexamethasone; SC, subcutaneous; SCT, stem cell transplant; dexamethasone; VGPR, very good partial response; VMP, bortezomib, melphalan, prednisone; VRd, bortezomib, lenalidomide.
Daratumumab in transplant-eligible NDMM
Favorable results, including deeper responses, observed in transplant ineligible patients have stimulated efforts to evaluate daratumumab in transplant eligible patients with NDMM. In the phase III CASSIOPEIA trial,
42
daratumumab was combined with bortezomib, thalidomide, and dexamethasone (DVTd) during induction and consolidation, showing increased rates of sCR at 100 days post-transplant; the sCR rate was 29% in the DVTd group compared with 20% in the bortezomib, thalidomide, and dexamethasone (VTd) group [odds ratio (OR): 1.60, 1.21–2.12
Trials evaluating daratumumab combinations in transplant eligible patients with NDMM.

Bort, bortezomib; Car, carfilzomib; CR, complete response; CTD-Dara, cyclophosphamide, thalidomide, dexamethasone, daratumumab; Cyclo, cyclophosphamide; CYBORD, cyclophosphamide, bortezomib and dexamethasone; D-KRd, daratumumab, carfilzomib, lenalidomide, dexamethasone; D-RVd, daratumumab, lenalidomide, dexamethasone, bortezomib; D-VCd, daratumumab, bortezomib, cyclophosphamide, dexamethasone; D-VRd, daratumumab, bortezomib, lenalidomide, dexamethasone; D-VTd, daratumumab, bortezomib, thalidomide, dexamethasone; Dara, daratumumab; Dex, dexamethasone; IV, intravenous; Ixa, ixazomib; Kd, carfilzomib, dexamethasone; KTd, carfilzomib, thalidomide, dexamethasone; Len, lenalidomide; MRD, minimal residual disease; MTD, maximum tolerated dose; NCT, National Clinical Trial; NDMM, newly diagnosed multiple myeloma; PACE, cisplatin, doxorubicin, cyclophosphamide, etoposide; PFS, progression-free survival; PO, by mouth; PR, partial response; QD, daily; Qw, every week; Rd, lenalidomide, dexamethasone; RVd, lenalidomide, dexamethasone, bortezomib; SC, subcutaneous; sCR, stringent complete response; SCT, stem cell transplant; Thal, thalidomide; VGPR, very good partial response; VRd, bortezomib, lenalidomide, dexamethasone; VTd, bortezomib, thalidomide, dexamethasone.
Daratumumab in NDMM irrespective of transplant eligibility
In addition to the aforementioned studies, other trials are investigating daratumumab combinations among NDMM patients, irrespective of transplant eligibility. A phase II clinical trial at Mayo Clinic was designed to evaluate the efficacy of the daratumumab in combination with ixazomib, lenalidomide and dexamethasone (D-IRd) as an induction regimen in this group of patients with a median age of 62 years (41–81). Early reports have shown encouraging results. Among 38 patients, 90% achieved ⩾PR after only two cycles, and all patients (32 patients) who completed four cycles achieved ⩾PR, including VGPR in 50%. Stem cell yield was not adversely influenced by treatment, and there were no reports of treatment discontinuation related to adverse events. Updated analysis including results on MRD is awaited. 47 The phase II Lyra trial studied the efficacy of daratumumab in combination with CYBORD in a heterogeneous group of MM patients including transplant eligible and ineligible NDMM patients and patients who relapsed after one line of therapy. Among patients with newly diagnosed MM (87 patients), 44% achieved ⩾VGPR after four induction cycles, including 5% with CR, with an ORR of 79%. Responses further improved by the end of induction with longer treatment; at a median of six induction cycles, ORR, ⩾VGPR rate and CR rate increased to 81%, 56%, and 9% respectively. At 12 months, PFS was 87% and OS 99%. Adverse events, including infusion reactions, were consistent with previous studies. 48 Ongoing trials of daratumumab-based combinations in NDMM patients irrespective of transplant eligibility are listed in Table 3. 41
Trials evaluating daratumumab combinations in NDMM irrespective of transplant eligibility.
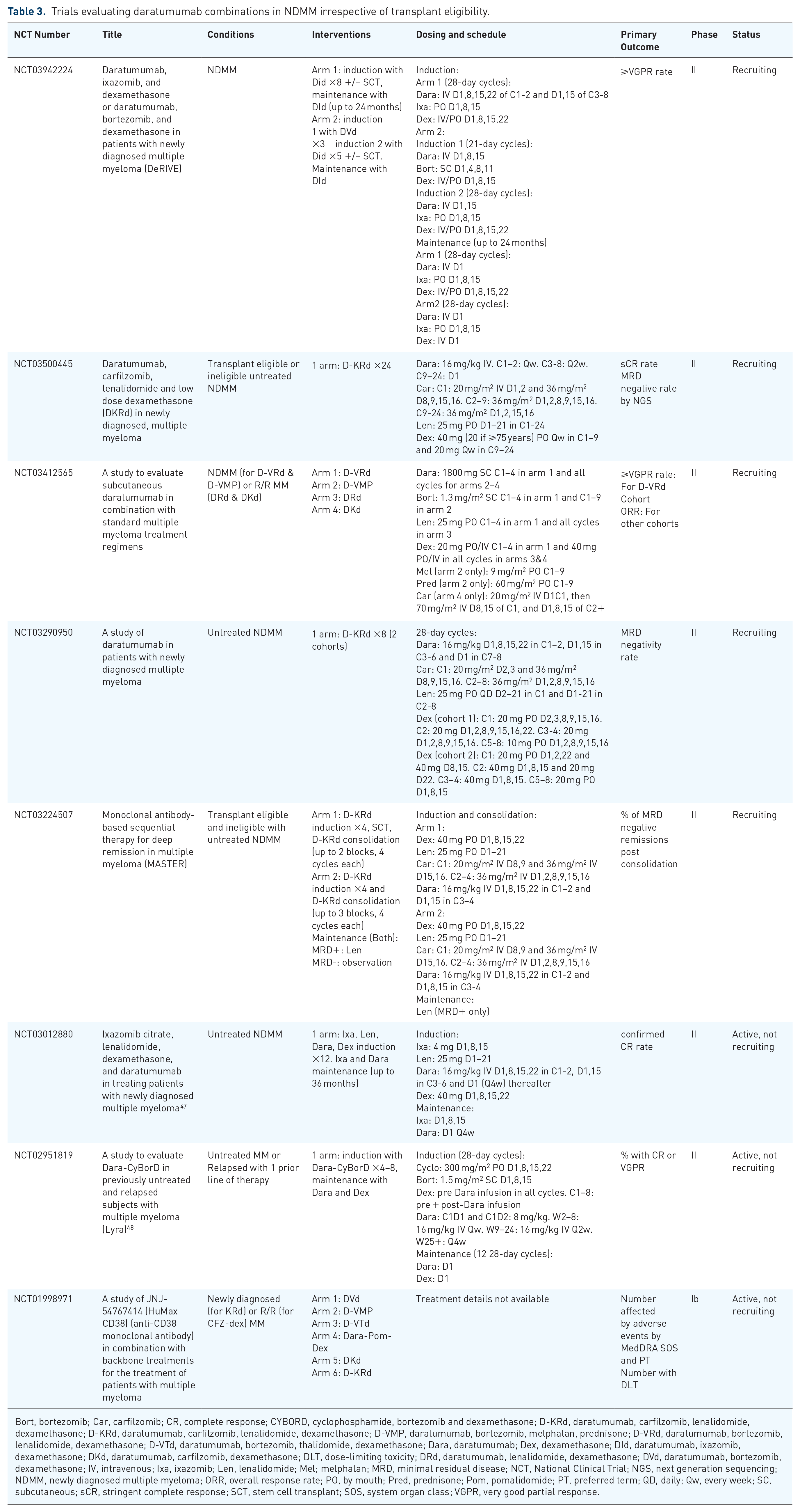
Bort, bortezomib; Car, carfilzomib; CR, complete response; CYBORD, cyclophosphamide, bortezomib and dexamethasone; D-KRd, daratumumab, carfilzomib, lenalidomide, dexamethasone; D-KRd, daratumumab, carfilzomib, lenalidomide, dexamethasone; D-VMP, daratumumab, bortezomib, melphalan, prednisone; D-VRd, daratumumab, bortezomib, lenalidomide, dexamethasone; D-VTd, daratumumab, bortezomib, thalidomide, dexamethasone; Dara, daratumumab; Dex, dexamethasone; DId, daratumumab, ixazomib, dexamethasone; DKd, daratumumab, carfilzomib, dexamethasone; DLT, dose-limiting toxicity; DRd, daratumumab, lenalidomide, dexamethasone; DVd, daratumumab, bortezomib, dexamethasone; IV, intravenous; Ixa, ixazomib; Len, lenalidomide; Mel; melphalan; MRD, minimal residual disease; NCT, National Clinical Trial; NGS, next generation sequencing; NDMM, newly diagnosed multiple myeloma; ORR, overall response rate; PO, by mouth; Pred, prednisone; Pom, pomalidomide; PT, preferred term; QD, daily; Qw, every week; SC, subcutaneous; sCR, stringent complete response; SCT, stem cell transplant; SOS, system organ class; VGPR, very good partial response.
Daratumumab dosing and formulations
The currently recommended dosing of intravenous (IV) daratumumab is 16 mg/kg administered on a weekly basis for 8 weeks, then biweekly for 16 weeks, and every 4 weeks thereafter, a schedule established based on efficacy and safety data from clinical trials.
49
The pharmacokinetic properties of daratumumab were found to be unchanged when used in combination with other regimens, compared with monotherapy and regardless of the backbone regimen and of baseline patient or disease characteristics.
50
Infusion reactions related to the IV formulation of daratumumab are very commonly encountered, especially with the first dose. They have been reported at a rate between 28% and 54% in major clinical trials, with the majority being grade 1–2.20,23,29,33,34,42,48 Thus, a subcutaneous (SC) formulation of daratumumab with recombinant human hyaluronidase enzyme (DARA + rHuPH20) has been developed in attempts to mitigate this reaction. Indeed, significantly decreased rates of infusion reactions have been reported with this formulation at a daratumumab dose of 1800 mg in a phase Ib safety study.
51
Subsequently, the phase III COLUMBIA trial compared the DARA + rHuPH20 SC formulation (1800 mg) with the IV formulation (16 mg/kg) among 522 MM patients with at least 3 prior lines of therapy. The SC formulation was non-inferior to the IV formulation in terms of efficacy (ORR 41%
Conclusion
Despite significant advances in MM treatment options, mortality remains high and more efficacious regimens are needed. Daratumumab has shown encouraging results both as monotherapy and in combination with other regimens in both R/R MM and untreated disease. Clinical trials so far have shown enhanced efficacy and tolerability of several daratumumab-based combinations in NDMM for both transplant ineligible and eligible patients, without hindering transplant. Although benefit in high-risk subpopulations is still unclear, data may be limited by the smaller number of participants in these subgroups, and thus more evidence is needed. Efforts are underway to explore other combinations with daratumumab, improve drug formulation, gain understanding of response predictors and mechanisms of resistance, 53 and identify patient subgroups more likely to respond to these treatments.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and publication of this article.
Conflict of interest statement
S.K.K. served as a consultant for Celgene, Millennium Pharmaceuticals, Onyx Pharmaceuticals, Janssen, and Bristol-Myers Squibb and received research funding from Celgene, Millennium Pharmaceuticals, Novartis, Onyx Pharmaceuticals, AbbVie, Janssen, and Bristol-Myers Squibb. N.A. has no conflicts of interests to disclose.
