Abstract
Carfilzomib is the second proteasome inhibitor approved for relapsed multiple myeloma. Since its approval in 2012, carfilzomib has been an active and versatile drug, based on its efficacy as a single agent; superiority as a doublet with dexamethasone compared with bortezomib and dexamethasone; and as a partner in diverse three drug combinations such as with lenalidomide or daratumumab. While it has an established place in relapsed disease, clinicians should be aware of its cardiovascular and renal adverse event profile, which is manageable, in order to optimize outcomes. This review will provide a perspective on the current and future role of carfilzomib in relapsed/refractory multiple myeloma.
Keywords
Introduction
The treatment of multiple myeloma has improved significantly in the past two decades with the introduction and use of several core drug classes: proteasome inhibitors (PI; e.g., bortezomib, carfilzomib, and ixazomib); immunomodulatory (IMiD) drugs (e.g., lenalidomide and pomalidomide); and, more recently, anti-CD38 monoclonal antibodies (e.g., daratumumab and isatuximab). These new classes have led to gains in quality of life and overall survival. While significant advances have been made with treatment, disease relapse continues to be a central issue in myeloma. A challenge in sequencing myeloma treatment is the diminishing effectiveness of each successive line of treatment, with a shorter period of disease response with each regimen as well as patient attrition.1,2 Carfilzomib received accelerated approval by the United States (US) Food and Drug Administration (FDA) in 2012 and has established a core role for treating relapsed/refractory multiple myeloma. This review will discuss the clinical development of carfilzomib in multiple myeloma and its use in the current treatment landscape.
Background
A principal strategy in multiple myeloma is inhibiting the ubiquitin proteasome pathway with PIs like bortezomib, carfilzomib, and ixazomib. Multiple myeloma cells are particularly sensitive to proteasome inhibition due to their production of large amounts of monoclonal immunoglobulin. Inhibition of the proteasome leads to accumulation of unfolded proteins and endoplasmic reticulum stress, followed by activation of the unfolded protein response and arrest of the cell cycle. 3 Bortezomib is a peptide boronic acid that potently but reversibly binds to the chymotrypsin-like, β5 subunit of the 20S subunit, and it was the first PI to be approved in multiple myeloma in 2003. 4 However, a key limitation of bortezomib is peripheral neuropathy. In the initial studies where bortezomib was given twice-weekly intravenously, peripheral neuropathy was common, occurring in 35% of patients, including 13% where it was grade ⩾3. 5 When given subcutaneously, the rate of grade ⩾3 peripheral neuropathy decreases to 6%. 6 The rate of neuropathy decreases further when administered weekly. 7
Carfilzomib (previously known as PR-171) is the second PI approved for multiple myeloma. The origins of carfilzomib begin with epoxomicin – an epoxyketone and natural compound isolated from an unidentified strain of actinomycete that was found to have activity in the B16 mouse melanoma cell line. 8 It was later determined that epoxomicin is a highly specific inhibitor of the proteasome, 9 and motivated by the emerging benefit seen with bortezomib, further chemistry work led to carfilzomib. 10 Carfilzomib, like bortezomib, binds to the chymotrypsin-like subunit of the proteasome, but, unlike bortezomib, carfilzomib binds irreversibly to the site thereby allowing for more sustained inhibition. 11 Moreover, carfilzomib is more potent than bortezomib, with activity in bortezomib-resistant cell lines. 12 Additional work into understanding the mechanism for peripheral neuropathy with bortezomib showed that bortezomib also inhibits the serine protease HtrA2/Omi, which is involved in neuronal survival. In contrast, carfilzomib does not inhibit this serine protease, potentially explaining the relative lack of peripheral neuropathy observed with carfilzomib. 13
Initial studies
The dosing and schedule of carfilzomib have evolved since the initial trials of carfilzomib conducted over 10 years ago. 14 Based on animal model data supporting better proteasome inhibition with longer drug exposure, 11 carfilzomib development began with a twice-week schedule on days 1, 2, 8, 9, 15, 16 of a 28-day cycle. (A dosing schedule for five consecutive days of a 14-day cycle was also evaluated initially. 15 ) In addition to the schedule, additional details that are relevant in interpreting subsequent studies include the duration of the infusion, if dexamethasone was given, as well as hydration. Carfilzomib was initially given as an infusion over 2–10 min, as a single agent, without corticosteroid. In the phase I studies, adverse events (AEs) analogous to tumor lysis syndrome with worsening renal function were occasionally observed. 16 This was managed with hydration with 250–500 ml prior to each dose and as needed after each dose. Hydration could be continued beyond cycle 2. “First dose” reactions with pyrexia, chills could be mitigated with a very low dose of dexamethasone, 4 mg, prior to each dose of carfilzomib in cycle 1.
The FDA accelerated approval of carfilzomib in July 2012 was based on the findings of the PX-171-003-A1 trial. 17 This phase II study evaluated carfilzomib as a single agent in relapsed disease in 266 patients with a median of five prior lines of treatment, with nearly all patients having had prior treatment with bortezomib (99.6%) and lenalidomide (94%). 18 Carfilzomib was given as an infusion over 2–10 min at 20 mg/m2 on days 1, 2, 8, 9, 15, 16 of a 28 day cycle; with cycle 2, the dose increased to 27 mg/m2. Dexamethasone 4 mg was given as premedication in cycle 1 and as needed thereafter, and patients received hydration.
The overall response rate (ORR) was 23.7% (see Table 1). Unlike bortezomib, peripheral neuropathy was less common and less severe, with grade ⩾3 neuropathy occurring in 1.1% of patients. However, toxicities that occurred more frequently in carfilzomib included cardiac failure, any grade, in 3.8% of patients. Dyspnea was reported in 34% of patients.
Selected trials with carfilzomib (95% of patients in control arm received C). PFS is in months. 95% of patients in control arm received cyclophosphamide).
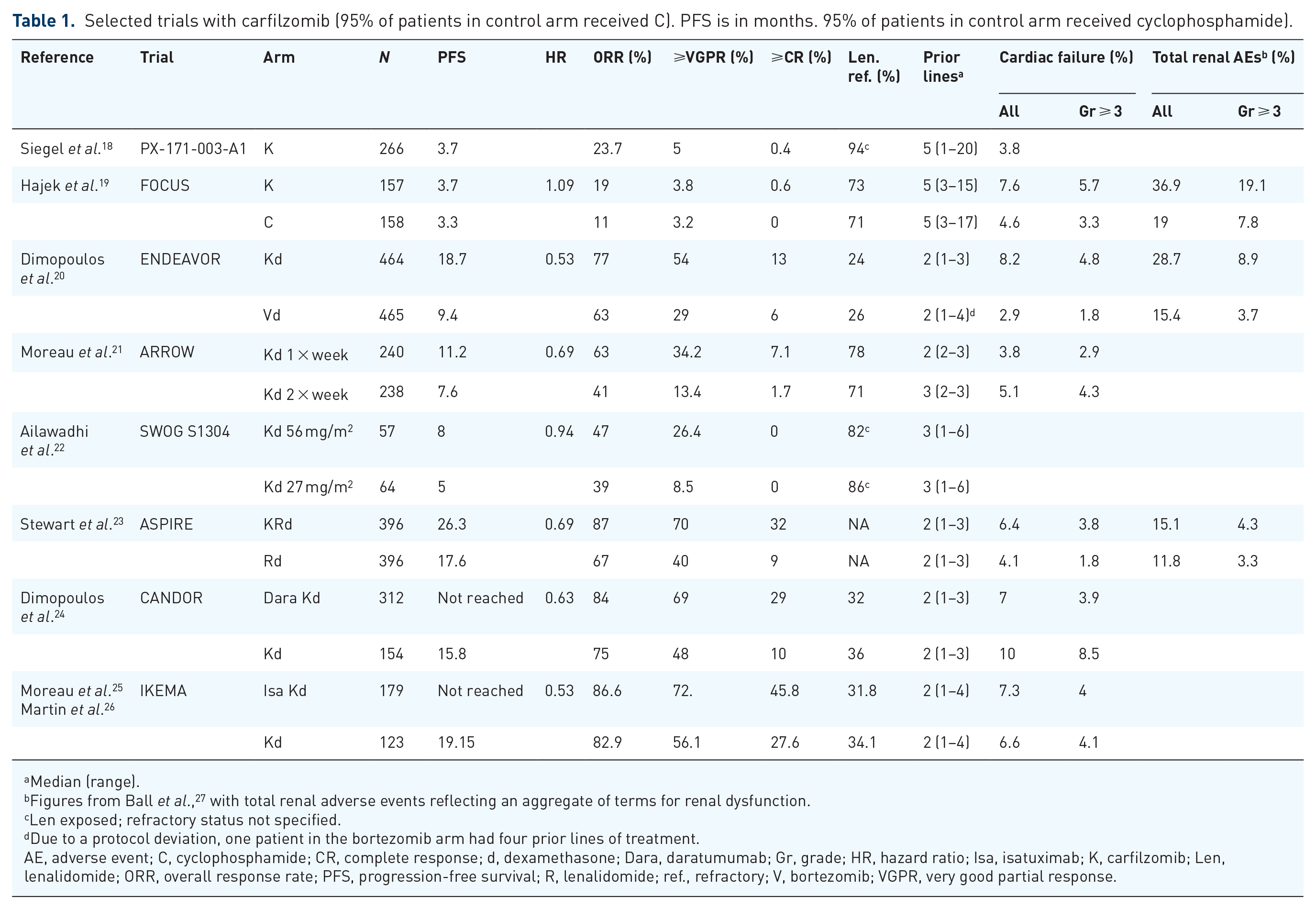
Median (range).
Figures from Ball et al., 27 with total renal adverse events reflecting an aggregate of terms for renal dysfunction.
Len exposed; refractory status not specified.
Due to a protocol deviation, one patient in the bortezomib arm had four prior lines of treatment.
AE, adverse event; C, cyclophosphamide; CR, complete response; d, dexamethasone; Dara, daratumumab; Gr, grade; HR, hazard ratio; Isa, isatuximab; K, carfilzomib; Len, lenalidomide; ORR, overall response rate; PFS, progression-free survival; R, lenalidomide; ref., refractory; V, bortezomib; VGPR, very good partial response.
FOCUS trial
Motivated by the findings of PX-171-003-A1, the FOCUS trial compared carfilzomib as a single agent with a best supportive care regimen of corticosteroids and cyclophosphamide in a randomized study of relapsed patients.19,28 This trial randomized 315 patients with a median of five prior lines of treatment, all having received prior bortezomib and 82% had prior lenalidomide. The schedule and dose of the carfilzomib arm was similar to the phase II trial that led to its approval, though the increase in carfilzomib from 20 to 27 mg/m2 occurred earlier on cycle 1 day 8 instead of at cycle 2. Dexamethasone 4 mg was given before each dose. In the control arm, patients received prednisone 30 mg every other day or dexamethasone 6 mg every other day. Cyclophosphamide 50 mg daily was also permitted in the control arm, which occurred in 95% of the control arm. While the ORR was higher in the carfilzomib arm versus control arm (19.1% versus 11.4%, p = 0.0305), unexpectedly, there was no significant difference in progression-free survival (PFS) (3.7 versus 3.3 months) or overall survival (OS), which was the primary endpoint (10.2 versus 10 months). The AE profile was similar in both arms, though there was more hypertension (any grade, 15% versus 6%) and grade ⩾3 renal AEs (24% versus 9%) in the carfilzomib arm.
There are several possible explanations for the lack of difference in PFS and OS in the FOCUS study. More patients in the control arm were censored for progression, due to starting a new treatment (20.3% versus 7.6%). The carfilzomib arm performed similarly to prior trials, and it is possible that cyclophosphamide and corticosteroid may be more active than expected in this patient population. Finally, carfilzomib was given as a single agent. The dose of dexamethasone used in the carfilzomib arm was minimal, 4 mg, to prevent reactions. Use of a higher dose of dexamethasone in combination with carfilzomib could have led to deeper responses and better outcomes, as is commonly seen when dexamethasone is added to various myeloma therapies.29–32
Carfilzomib with dexamethasone
ENDEAVOR trial
The ENDEAVOR trial is a pivotal, randomized, phase III trial that compared the doublet of carfilzomib and dexamethasone with bortezomib and dexamethasone in patients with 1–3 prior lines of treatment. 20 This trial evaluated higher doses of carfilzomib, where it was stepped up from 20 mg/m2 to 56 mg/m2 on cycle 1 day 8 and given over 30 min. Dexamethasone 20 mg was given on the days of carfilzomib infusion and also on days 22 and 23. Hydration (250–500 ml before and after each dose) was given during cycle 1.
The change in dosing was based on studies in rats that showed that efficacy was driven by total dose rather than Cmax, and that treatment by a longer infusion rather than bolus was better tolerated as well, attributed to lower Cmax. 33 This raised the possibility of being able to give a higher dose of carfilzomib over a longer infusion than the initially described 2–10 min infusion. These observations were confirmed in a phase I trial of carfilzomib, where it was given as a 30 min infusion and which determined the maximum tolerated dose (MTD) as 56 mg/m2. 34
Bortezomib was given according to the traditional twice-week schedule of 1.3 mg/m2 on days 1, 4, 8, and 11 on a 21-day cycle with dexamethasone 20 mg on the day of, and day after, treatment. Bortezomib was given either intravenously (IV) or subcutaneously (SC) by investigator choice; most patients (79%) received SC bortezomib throughout the study. The ORR was significantly higher in the carfilzomib arm, 77% versus 63% in the bortezomib arm (p < 0.0001), and median PFS was also higher, 18.7 versus 9.4 months (p < 0.0001). Of note, while 54% of patients had prior bortezomib, in a subgroup analysis, patients who were bortezomib-naïve also showed significantly improved PFS in the carfilzomib arm. Furthermore, treatment with carfilzomib improved OS, with a median of 47.6 compared with 40 months in the bortezomib arm [hazard ratio (HR) 0.791, one-sided p = 0.01]. 35
The incidence of grade ⩾2 peripheral neuropathy was significantly higher in the bortezomib group compared with the carfilzomib group, 32% versus 6% respectively; grade ⩾3 peripheral neuropathy was 8% versus 2%. Dose reductions due to AEs were more common in the bortezomib group (48%) than in the carfilzomib group (23%), which may have compromised the true efficacy of bortezomib. Even though the majority of the patients in the bortezomib arm received it SC, peripheral neuropathy continued to be an ongoing finding, and the majority of the dose reductions in the bortezomib group (62%) were due to peripheral neuropathy.
Additional notable differences in toxicity between the two groups included renal dysfunction. Acute renal failure, all grades, was higher in the carfilzomib arm than the bortezomib arm, 8% versus 5%; grade ⩾3, 4% versus 3%. Hypertension, grade ⩾3, was seen more frequently with carfilzomib compared with bortezomib, 9% versus 3%. Cardiac failure (which also included decreased ejection fraction and pulmonary edema), all grades, was more frequent in the carfilzomib group compared with the bortezomib group (8.2% versus 2.9%); grade ⩾3 (4.8% versus 1.8%). Quality of life was also assessed. 36 While there were differences in patterns of AEs, there were statistically significant improvements in quality of life in the carfilzomib arm across various domains, including globally, fatigue, pain, and neuropathy, though the mean differences did not meet threshold for clinical significance. Overall, the ENDEAVOR study demonstrated the benefit of carfilzomib over bortezomib in terms of responses and survival, though with important differences in the AE profile.
SWOG S1304 trial
The SWOG S1304 trial compared the dosing of carfilzomib in the ENDEAVOR study, 56 mg/m2, versus the initially approved 27 mg/m2 dosing, in a randomized phase II study. 22 This study enrolled patients with between one and six prior lines of therapy; the median was three prior lines and 24% had four to six prior lines of treatment. Notably the dosing of carfilzomib 20 mg/m2 was for the entire first cycle, and the treatment was fixed at 12 cycles (similar to the earlier studies with carfilzomib rather than to progression in ENDEAVOR). The median PFS was 8 versus 5 months for high versus low dose carfilzomib, but the difference was not statistically significant, p = 0.3842. Importantly, the increased dose of carfilzomib did not lead to increased rates of cardiopulmonary AEs overall, though grade ⩾3 hypertension was higher in the higher carfilzomib dose cohort, 7% versus 3%. The lack of apparent improvement with the higher dose of carfilzomib could be attributed to the step up in dosing of carfilzomib occurring later at cycle 2 (rather than at cycle 1 day 8) and the fixed duration of treatment at 12 cycles rather than treatment until progression.
Carfilzomib combinations with IMiD drugs
ASPIRE trial
Therapy in multiple myeloma has evolved from doublet to triplet regimens as the components of these regimens have become better tolerated and more effective. The ASPIRE trial was one of the trials that ushered in triplet therapy for relapsed disease. ASPIRE was a phase III trial that compared the combination of carfilzomib with lenalidomide and dexamethasone to lenalidomide and dexamethasone. 23 Patients were eligible to participate if they received between one and three prior lines of therapy. Prior lenalidomide and bortezomib treatment were permitted if there was no disease progression with these drugs; the majority of patients (80.2%) had not received prior lenalidomide therapy. Carfilzomib was given as a 10 min infusion at 20 mg/m2 with ramp up to 27 mg/m2, following the schedule of its initial approval, though the increase to 27 mg/m2 occurred at cycle 1 day 8. From cycles 13–18, the second week of carfilzomib was omitted, and, after cycle 18, carfilzomib was discontinued. Lenalidomide was given 25 mg on days 1–21 with dexamethasone 40 mg weekly; the same conventional schedule of lenalidomide and dexamethasone was given in the control group. In the carfilzomib arm, hydration (250–500 ml) before and after treatment was also required during cycle 1.
The ORR was significantly higher in the carfilzomib arm compared with the control arm, 87.1% versus 66.7% (p < 0.001), and, similarly, the carfilzomib arm had a higher complete response rate, 31.8% versus 9.3%. The median PFS was higher, 26.3 versus 17.6 months (HR 0.69, p = 0.0001). An updated analysis showed improvement in OS with carfilzomib, 48.3 versus 40.4 months (HR 0.791, one-sided p = 0.0045). 37 The depth and duration of response in the treatment arm were unprecedented at the time, and serious AEs were uncommon. However, grade 3–4 dyspnea (2.8%), hypertension (4.3%), and cardiac failure (3.8%) were higher in the carfilzomib group, showing a similar toxicity profile seen in other studies, such as the ENDEAVOR trial. In July 2015, the FDA approved carfilzomib in combination with lenalidomide and dexamethasone in relapsed multiple myeloma.
Carfilzomib, pomalidomide, and dexamethasone
In addition to lenalidomide, carfilzomib has also been combined with pomalidomide and dexamethasone. A phase I study evaluated this regimen in patients with disease refractory to prior lenalidomide treatment. 38 A total of 32 patients were enrolled; they had received a median of six prior lines of treatment (range 2–12); 100% were refractory to lenalidomide and 97% to bortezomib. Dosing of carfilzomib was similar to the ASPIRE study, 20 mg/m2 with ramp up to 27 mg/m2 beginning on cycle 1 day 8; pomalidomide was 4 mg on days 1–21; and dexamethasone was given 40 mg weekly. The MTD was dose level 1, likely reflecting the extensive treatment history of this patient population. The ORR was 50% with a median PFS of 7.2 months, which is notable given that all patients were refractory to lenalidomide and nearly all were refractory to bortezomib. The regimen thus showed significant activity in a heavily pretreated, double refractory cohort, with a side effect profile typical for an IMiD and PI combination.
The same regimen was evaluated in a less heavily treated population by the European Myeloma Network in the EMN011/HOVON 114 trial. 39 This study evaluated carfilzomib, pomalidomide, and dexamethasone in patients with relapsed disease after participating in EMN02/HO95. As background, EMN02/HO95 is a trial in newly diagnosed disease where all patients received induction with bortezomib, cyclophosphamide, dexamethasone; followed by randomization to bortezomib, melphalan, prednisone versus high dose melphalan and autologous second transplant; followed by a second randomization to consolidation with bortezomib, lenalidomide, and dexamethasone versus no consolidation. Following induction, all patients received lenalidomide maintenance. In EMN011/HOVON 114, nearly all patients (95%) entered the trial with lenalidomide-refractory disease. This trial evaluated a higher dose of carfilzomib than in the prior phase I study of the combination: carfilzomib was given at 36 mg/m2 twice weekly. The results of 60 patients were presented, and this regimen showed an ORR of 87% and median PFS of 18 months. Notably, the study was able to show that a higher dose of carfilzomib was able to be given with pomalidomide and tolerated well.
Weekly carfilzomib
A practical consideration for carfilzomib is the twice-weekly schedule, especially since patients may be on therapy for a prolonged duration. The CHAMPION-1 trial evaluated a weekly schedule of carfilzomib in a phase I–II trial. 40 Carfilzomib was given as a 30 min infusion on days 1, 8, and 15 with dexamethasone 40 mg weekly. The 70 mg/m2 dose level was determined to be the MTD.
The ARROW study compared weekly carfilzomib with the conventional schedule of twice-weekly carfilzomib at 20 and 27 mg/m2 (10 min infusion; step up at cycle 1 day 8) in patients with relapsed disease and two to three prior lines of treatment. 21 The dosing in ARROW followed the dosing regimen determined by CHAMPION-1, with the first dose at 20 mg/m2 and subsequent weekly doses at 70 mg/m2. Hydration 250–500 ml was given before each dose in both arms for cycle 1. The once-weekly schedule showed higher responses (ORR 62.9% versus 40.8%, p < 0.0001) and PFS (HR 0.693, 11.2 versus 7.6 months, p = 0.0029). AEs in both arms were comparable. For example, for grade ⩾3 cardiac failure, the rates were similar in the once-weekly and twice-weekly arms (2.9% versus 4.3%). These findings thus serve as the basis for weekly combinations with carfilzomib, which significantly improves the convenience of a carfilzomib-based regimen.
Weekly carfilzomib has been evaluated with lenalidomide and pomalidomide. Carfilzomib was evaluated at 56 mg/m2 and 70 mg/m2 weekly with lenalidomide and dexamethasone in relapsed and refractory multiple myeloma in patients with a median of one prior line of treatment (range 1–4). 41 The efficacy of this weekly combination was similar to ASPIRE. However, there were two cardiac-related deaths at the 70 mg/m2 dosing level out of 46 patients. The 56 mg/m2 weekly dosing level is undergoing further evaluation in the ARROW2 study, which is an ongoing study comparing once-weekly versus twice-weekly carfilzomib in combination with lenalidomide and dexamethasone described in ASPIRE [ClinicalTrials.gov identifier: NCT03859427].
A phase I/II study based in Italy evaluated weekly carfilzomib with pomalidomide and dexamethasone in relapsed/refractory disease with one to three prior lines of treatment. 42 However, the MTD at this dosing level was only 27 mg/m2. There were five dose limiting toxicity events at the 36 and 45 mg/m2 dose levels: two of these events included sudden death and heart failure, and four of these events were related to hypertension. Using a lower dose of carfilzomib at 27 mg/m2 and increased attention to blood pressure led to significant improvement in tolerability, with a reduction of grade ⩾3 cardiovascular AEs from 56% to 6%. To better evaluate the dosing of carfilzomib in combination with pomalidomide, the SELECT study is evaluating weekly carfilzomib at 56 mg/m2 with pomalidomide and dexamethasone in patients with prior lenalidomide and daratumumab treatment [ClinicalTrials.gov identifier: NCT04191616].
Anti-CD38 monoclonal antibody and additional combinations with carfilzomib and dexamethasone
CANDOR trial
Carfilzomib has been studied in combination with the anti-CD38 monoclonal antibodies daratumumab and isatuximab. The CANDOR study was a randomized phase III study that compared the combination of daratumumab with carfilzomib and dexamethasone versus carfilzomib and dexamethasone in patients with relapsed disease and 1–3 prior lines of treatment. 24 Carfilzomib was given on the twice-weekly schedule at 56 mg/m2 along with weekly dexamethasone as described in the ENDEAVOR study. In the carfilzomib and dexamethasone arm, patients received pre-hydration of 250 ml during cycle 1 but no post-hydration (unlike ENDEAVOR). The trial accrued patients with a median of two prior lines of treatment. The addition of daratumumab improved ORR, 84% versus 75%, and median PFS, 28.6 versus 15.2 months [HR 0.59, 95% confidence interval (CI) 0.45–0.78].24,43 This regimen with carfilzomib was approved by the FDA in August 2020.
Daratumumab has also been evaluated with weekly carfilzomib and dexamethasone, with similar findings. 44 In this study, carfilzomib was given at a dose of 70 mg/m2 weekly on days 1, 8, and 15, as described in the ARROW study.
IKEMA trial
The IKEMA study evaluated the recently approved anti-CD38 monoclonal antibody isatuximab in a similar patient population and trial design as CANDOR, with one to three prior lines of treatment and where carfilzomib was given according to the ENDEAVOR schedule. 25 Patients had a median of two prior lines of treatment. Results were comparable to CANDOR, where the addition of isatuximab improved ORR, 86.6% versus 82.9% and median PFS, not reached versus 19.15 months (HR 0.531, 95% CI 0.318–0.889). The FDA approved this combination with isatuximab in March 2021.
Carfilzomib, cyclophosphamide, dexamethasone
The Spanish Myeloma Group evaluated weekly carfilzomib and dexamethasone with cyclophosphamide. 45 This was a phase II randomized study that compared carfilzomib, cyclophosphamide, and dexamethasone with carfilzomib and dexamethasone in relapsed disease with one to three prior lines of treatment. Carfilzomib was given weekly at 70 mg/m2 on days 1, 8, and 15; cyclophosphamide was 300 mg/m2 IV on days 1, 8, 15; and dexamethasone was given 40 mg weekly, split over 2 days. The median PFS was higher in the three drug arm, 20.7 versus 15.2 months, but the difference was not statistically significant (HR 1.2, p = 0.24). However, when evaluated in patients with lenalidomide-refractory disease (36% of the overall study), there was a significant difference in outcome, with median PFS 26.2 versus 9.3 months (HR 0.4, p = 0.02).
Additional combinations with carfilzomib
Carfilzomib is a versatile drug that has been partnered with several other agents as well. These include bendamustine, 46 panobinostat, 47 and venetoclax. 48 More recently, weekly carfilzomib and dexamethasone has been evaluated in combination with the oral XPO1 inhibitor selinexor as one of the arms of the STOMP trial. 49 The ORR was 75% in a patient population with a significant exposure history: pomalidomide (67%) and daratumumab (63%). The recommended dose going forward is carfilzomib 56 mg/m2 weekly with selinexor 80 mg weekly and dexamethasone 40 mg weekly. Carfilzomib is also undergoing evaluation with the next generation of cereblon targeting drugs, the cereblon E3 ligase modulators (CELMoD) iberdomide [ClinicalTrials.gov identifier: NCT02773030] and CC-92480 [ClinicalTrials.gov identifier: NCT03989414].
Adverse events
Cardiovascular
The principal limitation with carfilzomib is cardiovascular AEs, which is a distinguishing characteristic among the agents used in myeloma (Table 1). In the initial studies of carfilzomib as a single agent, aggregated cardiac failure events (including congestive heart failure, pulmonary edema, and decreased ejection fraction) were reported in 7.2% (any grade) and 5.7% (grade ⩾3) of patients. 50 However, rates of cardiovascular AEs during the initial experience with carfilzomib may have been higher. For example, in a series of 130 patients mostly treated with carfilzomib on a compassionate basis, 20% were hospitalized for a cardiac-related complication during the first two cycles of treatment. 51 Of note, nearly all of these patients had prior treatment with doxorubicin, which may have led to increased susceptibility to cardiac dysfunction, and 77% received carfilzomib as a 2–10 min infusion versus 30 min. Interpretation of above AEs is limited given the single arm nature of these studies.
In an analysis of three randomized trials in relapsed myeloma (ASPIRE, ENDEAVOR, FOCUS), the rates of these AEs – cardiac failure, dyspnea, and hypertension – were higher in the carfilzomib arm compared with the control arm, though the frequency of discontinuation or death to these cardiac events was low and comparable between these arms. 52 Pooling data across carfilzomib trials, grade ⩾3 cardiovascular AEs included hypertension (5.9%), dyspnea (4.5%), and cardiac failure (4.4%). A meta-analysis by a different group across a wide range of carfilzomib trials found all grade cardiovascular events at 18.1% and grade ⩾3 cardiovascular events at 8.2%. 53 Moreover, the relative risk for all-grade and grade ⩾3 cardiovascular AEs in three randomized trials was 1.8 and 2.2, respectively.
Furthermore, a substudy of the ENDEAVOR study looked more closely at cardiac function, and echocardiograms were performed at baseline, every 12 weeks, and end of study; 17% of participants in the ENDEAVOR trial participated in the cardiac substudy. 52 There was no reduction in ejection fraction and no treatment-related effect on ejection fraction. The PROTECT study prospectively performed comprehensive cardiac assessment in patients undergoing treatment with carfilzomib or bortezomib. 54 Similar to the ENDEAVOR substudy, there were no significant findings with echocardiography. This suggests that the heart failure related to carfilzomib is not from direct cardiotoxicity, as seen with trastuzumab or anthracyclines. Moreover, routine echocardiography did not play a significant role in predicting heart failure from carfilzomib. In the PROTECT study, there were 20% grade 3–4 heart failure events with carfilzomib, compared with 13% in bortezomib. Notably, patients receiving carfilzomib-based therapy with a baseline elevated brain natriuretic peptide (BNP) level >100 pg/ml or N-terminal proBNP > 125 pg/ml had increased risk for cardiovascular adverse events (odds ratio, 10.8; p < 0.001). Elevated natriuretic peptides occurring mid–first cycle of treatment with carfilzomib were associated with a substantially higher risk of cardiovascular adverse events (odds ratio, 36.0; p < 0.001). Even with these cardiovascular adverse events, the majority of patients (89%) in the study (treated with either carfilzomib or bortezomib) were able to resume treatment.
The mechanism of these cardiovascular side effects continues to not be well understood. Some of these events may be a class effect of proteasome inhibition.55,56 In a rat myocyte model, exposure to either bortezomib or carfilzomib resulted in myocyte damage and apoptosis, which was potentiated by doxorubicin. 57 However, interestingly, bortezomib was found to induce more myocyte damage than carfilzomib, 58 which is in contrast to the decreased frequency of cardiovascular events seen with bortezomib clinically. 59 Endothelial toxicity has also been proposed as a possible mediator, and the irreversible proteasome inhibition and higher potency of carfilzomib compared with bortezomib may explain why these types of events have been observed more with carfilzomib.60,61
Renal
In the initial experience with carfilzomib, grade ⩾3 renal impairment occurred in 7.2% of patients, though half of all renal AEs were associated with disease progression. 50 Some of the renal impairment was due to tumor lysis-like syndrome, which motivated hydration in the early stages of the clinical development of carfilzomib. In a meta-analysis of four randomized trials with carfilzomib (ASPIRE, ENDEAVOR, FOCUS, and CLARION), the cumulative rate of kidney toxicity was 21.3% for all grades and 8.3% for grade ⩾3 events. 27 (CLARION is a randomized trial that compared carfilzomib, melphalan, prednisone with bortezomib, melphalan, and prednisone in newly diagnosed, transplant-ineligible patients. 62 ) The pooled relative risk for kidney toxicity all grades was 1.79 (95% CI, 1.43–2.23) in the carfilzomib arm compared with the control group and 2.29 (95% CI, 1.59–3.30) for grade ⩾3 events.
Some of the toxicities that have emerged with further use of carfilzomib include thrombotic microangiopathy. Thrombotic microangiopathy has also been described with bortezomib, though less commonly. 63 In the ENDEAVOR study, thrombotic microangiopathy occurred in 0.4% of patients and none in the control arm, 20 and, in an analysis of 114 consecutive patients treated with carfilzomib-based regimens, thrombotic microangiopathy occurred in 5%. 64 In a series of three patients with thrombotic microangiopathy attributed to carfilzomib, two patients were heterozygous for CFHR3–CFHR1 deletion, suggesting a predisposition for developing atypical hemolytic syndrome for patients with prolonged carfilzomib exposure. 65 The mechanisms for thrombotic microangiopathy may overlap with the mechanisms for some of the cardiac events noted above.
Even though renal impairment was more common in carfilzomib-treated patients than in controls, patients with underlying renal dysfunction who received carfilzomib in the ENDEAVOR trial had better outcomes. 66 The ENDEAVOR trial enrolled patients with GFR as low as 15 ml/min, and it is the largest trial to include patients with this degree of renal impairment. For patients with impaired renal function, carfilzomib improved PFS and OS regardless of baseline renal function. Rates of complete renal response in this trial (i.e., creatinine clearance ⩾60 ml/min in any two consecutive visits if baseline creatinine clearance <50 ml/min) were similar between the carfilzomib and bortezomib arms (15.3% versus 14.1%). On the other hand, in an analysis of real world data, after baseline covariate adjustment, patients receiving carfilzomib and dexamethasone compared with bortezomib and dexamethasone were more likely to have a complete renal response (incidence rate ratio 1.68, 95% CI, 1.24–2.28). 67
Current practice with carfilzomib
The treatment landscape for relapsed multiple myeloma has become increasingly more complex, given the increasing number of treatment options and combinations. Whereas doublets have been historically used in relapsed disease over 10 years ago, beginning with bortezomib and dexamethasone and lenalidomide and dexamethasone, the practice has moved to three drug combinations, as exemplified by carfilzomib, lenalidomide, and dexamethasone in the ASPIRE trial. Furthermore, lenalidomide and bortezomib are increasingly used as first line treatment as RVd followed by lenalidomide maintenance. With this treatment history, carfilzomib is more of a consideration for relapsed disease. For second line treatment, in patients relapsing on lenalidomide treatment, carfilzomib-based options include: daratumumab, carfilzomib, dexamethasone; isatuximab, carfilzomib, dexamethasone; carfilzomib, pomalidomide, dexamethasone; and carfilzomib, cyclophosphamide, dexamethasone. Similarly, as daratumumab is increasingly used as first-line treatment and earlier in lines of treatment, carfilzomib also plays a role for treating patients with this type of relapse. In a retrospective analysis of patients with disease refractory to CD38 monoclonal antibody therapy like daratumumab, the best outcomes were observed with carfilzomib-based therapy, such as a combination of carfilzomib and alkylators like cyclophosphamide. 68
Moreover, use of carfilzomib has progressively moved to weekly dosing, which maintains efficacy and improves on convenience. With weekly dosing, it is important to emphasize that with IMiD drug combinations like pomalidomide, the recommended weekly dose of carfilzomib is lower at 56 mg/m2 instead of 70 mg/m2. Similar to the evolution from two to three drug combinations, four drug combinations with carfilzomib are under evaluation. This includes for example daratumumab with carfilzomib, pomalidomide, and dexamethasone. 69 In this trial, carfilzomib is given twice-weekly at a dose of 20 and 27 mg/m2. In patients with a median of one prior line of treatment, the ORR was 86% with a 24-month PFS of 76%. A similar trial of dara-KPd is ongoing with a weekly schedule of carfilzomib [ClinicalTrials.gov identifier: NCT04176718].
Among agents currently used in myeloma, the cardiovascular and renal side effects of carfilzomib are notable. With attention to hypertension and volume status, these cardiovascular adverse events can be reduced. The prescribing information for carfilzomib has evolved since its initial approval in 2012 to reflect these considerations. The original version advised hydrating patients prior to and after each dose of carfilzomib in cycle 1 with 250–500 ml of normal saline. 70 In July 2015, the hydration requirement was changed to “as needed” and also recommended adjusting total fluid intake to take into account risk of cardiac failure. More recently in August 2020, the language under administration precautions was further modified from recommending hydration to “consider hydration.”
Overall, it is important to note that, even with these cardiovascular and renal considerations, carfilzomib improves overall survival in relapsed disease, based on the ASPIRE and ENDEAVOR trials. Finally, this benefit extends to frailer patients, based on an analysis of a frail subset of patients (30%) participating in ASPIRE, ENDEAVOR, and ARROW. 71
Conclusions
Carfilzomib is a core drug in multiple myeloma therapy. Its use has evolved from single agent to doublet regimens with dexamethasone to now routine three drug combinations with IMiD drugs, anti-CD38 monoclonal antibodies, or cyclophosphamide. Along the way have been changes in dosing and schedule to weekly administration to improve efficacy and convenience along with gains in how to better manage the cardiovascular AEs. These developments have led to significant improvements in outcomes for patients, with more gains expected in the future with newer strategies that incorporate carfilzomib.
Footnotes
Conflict of interest statement
Consulting for Adaptive Biotechnologies, Amgen, BMS, Celgene, GSK, Janssen, Karyopharm, Oncopeptides, Sanofi, Takeda. Research funding from Amgen, BMS, Celgene, Janssen.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
