Abstract
Background:
Macrofocal multiple myeloma (MFMM) is characterized by clonal plasma cells comprising less than 20% of the bone marrow, multiple lytic bone lesions, and the absence of anemia, renal insufficiency, and hypercalcemia. This subtype of multiple myeloma (MM) has a relatively low incidence. Prognostic staging and cytogenetic guidance for MFMM are often insufficient due to the low tumor burden in the bone marrow. Large cohort studies on this subgroup during the era of novel agents are limited.
Objectives:
We aim to describe the clinical characteristics and prognostic markers of MFMM patients undergoing treatment with novel agents.
Methods:
Consecutive cases of MM patients diagnosed at Peking University People’s Hospital and Fu Xing Hospital of Capital Medical University from 2011 to 2023 were screened. A propensity score matching was conducted with a 2:1 ratio, matching classic MM patients to MFMM patients based on clinical variables of age and year of diagnosis.
Results:
We identified 91 cases (4%) of MFMM and 182 matched classic MM among 2291 MM patients. The MFMM cohort had a higher proportion of male patients, those with <90% clonal plasma cells in the bone marrow by multiparameter flow cytometry, and patients with extramedullary disease, along with a lower proportion of patients with high-risk cytogenetics or advanced disease staging. MFMM patients demonstrated better overall responses compared to the control cohort (p = 0.027) in those not receiving upfront autologous stem cell transplantation (ASCT). During a median follow-up of 42.8 months for the entire cohort, the MFMM cohort exhibited significantly superior progression-free survival (PFS) and overall survival (OS) compared to the control cohort. In multivariate analysis of the entire cohort, exposure to immunomodulatory drugs and ASCT consolidation in frontline therapy were independently associated with improved PFS and OS. For the MFMM cohort, a Ki-67 index ⩾20% was associated with inferior PFS, providing valuable prognostic information in a group where staging and cytogenetic guidance are often inadequate.
Conclusion:
We concluded that treatment strategies for MFMM patients should align with those for standard MM, and a Ki-67 index ⩾20% in biopsy samples of plasmacytoma is associated with inferior PFS.
Plain language summary
Macrofocal multiple myeloma (MFMM) is a rare form of multiple myeloma. Our study found that MFMM patients had better outcomes with new treatments compared to typical cases. A high Ki-67 index was linked to worse outcomes, highlighting its importance in prognosis. We also emphasized that treatment strategies for MFMM patients should align with those for standard MM.
Keywords
Introduction
Macrofocal multiple myeloma (MFMM), characterized by multiple lytic lesions and limited percentage of clonal plasma cells in the bone marrow (BM) without other end-organ damage including anemia, renal impairment, and hypercalcemia, has been recognized as a distinct entity with a more favorable prognosis including better responses rate and survivals than classic multiple myeloma (MM).1,2 MFMM accounts only for 3%–5% of all MM with limited cases.1,3,4 Prognostic staging and cytogenetic guidance are often inadequate for this subgroup of patients due to the low tumor burden in the bone marrow. Furthermore, the rapidly evolving treatment paradigm for MM necessitates a reassessment of the characteristics of MFMM within the current treatment modalities, encompassing novel agents, autologous stem cell transplantation (ASCT), and maintenance therapy as the standard approach. Previous large studies on MFMM spanned over 20 years, 1 during which the treatment paradigm underwent significant changes. 5
Given the scarcity of data on this rare form of disease, this study aimed to provide information of the clinical characteristics, genetic abnormalities, treatment responses, and prognosis of patients with MFMM at diagnosis in the era of novel agents.
Materials and methods
Patients
This is a retrospective case–control cohort study based on the clinical database of Peking University Institute of Hematology of Peking University People’s Hospital and Fu Xing Hospital of Capital Medical University. Consecutive cases of MM patients diagnosed from January 2011 to December 2023 were screened. Patients fulfilling the definition of MFMM according to the Greco-Israeli collaborative myeloma working group study, 1 that is, (a) clonal bone marrow plasma cells (BMPCs) <20%; (b) multiple lytic lesions determined by imaging including computed tomography (CT), or 18F fluorodeoxyglucose positron emission tomography (PET/CT), or magnetic resonance imaging (MRI); and (c) absence of anemia, renal insufficiency, and hypercalcemia were included as the case cohort. Propensity score matching (PSM) was performed with a 2:1 ratio, matching classic MM patients (i.e., those meeting SliM CRAB criteria except for multiple lytic bone lesions) to MFMM patients based on clinical variables of age and year of diagnosis.
Diagnosis, treatment, responses, and monitoring protocols
Patients diagnosed with MM meet the International Myeloma Working Group (IMWG) diagnostic criteria 2014. 6 Responses were assessed according to the IMWG response criteria. 7 Extramedullary disease was classified into bone-related masses (EMB) or extraosseous disease (EME). Bone marrow aspiration and biopsy were routinely performed at baseline. For patients with BMPC < 10%, if bone marrow biopsy was missing, at least two bone marrow aspiration at different sites were performed to confirm the BMPC percentage.
Interphase fluorescence in situ hybridization (FISH) of bone marrow were performed at baseline to detect chromosomal abnormalities using CD138-purified plasma cells by magnetic-activated cell sorting, as previously described. 8 All patients were analyzed for 1q21+, del(17p), and IgH rearrangement using gene locus-specific probes (GLP) including GLP 1q21, GLP P53, GLP IgH. If an IgH rearrangement was identified, dual-color and dual-fusion translocation probes such as IgH-FGFR3, IgH-MAF, and IgH-CCND1 were used for the detection of t(4;14)(p16;q32), t(14;16)(q32;q23), and t(11;14)(q13;q32). Three (gain) or ⩾4 (amp) copies of 1q21 were grouped together and indicated as 1q21+. High-risk cytogenetic abnormalities were defined as the presence of del(17p), t(4;14), or t(14;16). 9
All patients received proteasome inhibitors (PIs) during induction therapy, which included doublet regimens of VD (bortezomib-dexamethasone), and triplet regimens including VRD (bortezomib-lenalidomide-dexamethasone), VCD (bortezomib-cyclophosphamide-dexamethasone), VTD (bortezomib-thalidomide-dexamethasone), ITD (ixazomib-thalidomide-dexamethasone), PDD (bortezomib-liposomal doxorubicin-dexamethasone), DVD (daratumumab-bortezomib-dexamethasone). Daratumumab-based quadruplet regimens included DVRD (daratumumab-bortezomib-lenalidomide-dexamethasone), as well as alternating treatment of V-DECP (bortezomib-dexamethasone-etoposide-cyclophosphamide-cisplatin) and Dara-VPD (daratumumab-bortezomib-pomalidomide-dexamethasone) according to an investigator-initiated trial. 10
Patients receiving ASCT was applied after 4–6 cycles of induction therapy. The conditioning regimen for ASCT was with melphalan at a full dose of 200 mg/m2, or a reduced dose of 100–140 mg/m2 in patients with a creatinine clearance of lower than 40 ml/min. For patients receiving tandem ASCT, a second transplant was performed within 6 months of the first transplant. Maintenance strategies were tailored according to patients’ risk stratification, drug availability, and insurance policies. For patients with standard-risk cytogenetics, maintenance therapy consisted of lenalidomide (available in China after 2013) or thalidomide (prior to 2013). In cases where immunomodulatory drugs (IMiDs) were contraindicated, daratumumab was used as an alternative. Patients with high-risk cytogenetics, defined by the presence of del(17p), t(4;14), or t(14;16), 9 received maintenance therapy comprising both a proteasome inhibitor and an IMiD.
Responses were assessed according to the IMWG response criteria. 7 The bone marrow aspiration post-ASCT was routinely evaluated at 100 days (±30 days) after ASCT. Minimal residual disease was assessed by multiparameter flow cytometry (MPFC) panel of CD38/CD138/CD45/CD19/CD56/CD117/cytoplasmic kappa (cκ)/cytoplasmic lambda (cλ). If patients received daratumumab treatment within 3 months prior to FCM test, additional panel of CD38/CD229/CD45/CD19/CD56/CD117/cκ/cλ was tested. The number of cells detected was 1 × 106 at a sensitivity threshold of 10−4.
Statistics
To determine the optimal PSM ratio by clinical variables of age and year of diagnosis, we tested multiple ratios (1:1, 2:1, 3:1, and 4:1) and evaluated balance metrics, including standardized mean differences (SMDs) and variance ratios. The 2:1 ratio provided near-optimal balance (SMD: 0, variance ratio: 1.0) while maintaining a larger matched sample size compared to 1:1. Higher ratios (3:1, 4:1) resulted in worse balance metrics (e.g., SMDs: 0.01 and 0.02, respectively). Visual inspection of histograms and jitter plots further confirmed the suitability of the 2:1 ratio for achieving both balance and statistical power (Supplemental Table S1 and Figure S1). Descriptive statistics were used to summarize co-variates. χ2 or Fisher exact tests were used for categorical co-variates and a nonparametric test for continuous co-variates. Overall response included stringent complete response (sCR), complete response (CR), VGPR (very good partial response), and PR (partial response). Survival functions were estimated using the Kaplan–Meier method and were compared by the log-rank test. Progression-free survival (PFS) and overall survival (OS) were defined as the interval from diagnosis to disease progression or death in the PFS model and death from any cause in the survival model. Patients were censored at last contact. The last follow-up time was June 2024. Cox proportional hazards regression models were used to evaluate factors associated with survival and relapse. For the entire cohort, clinical variables of cohort (MFMM vs control), age, sex, estimated glomerular filtration rate (eGFR), lactate dehydrogenase, presence of immunoparesis, the clonal plasma cell in BM assessed by MPFC, EMD, staging, cytogenetics, induction regimens, and whether the application of ASCT at frontline were included in univariate Cox analysis. For the MFMM cohort, clinical variables included in univariate Cox analysis also included biopsy information such as whether the presence of plasmablastic differentiation of plasma cells, and whether the Ki-67 index above 20% (cutoff determined by median value). Co-variates with p < 0.2 in univariate analyses were included in multivariate analyses and selected using a backward elimination process to fit a Cox regression model. All reported p values are two-sided and considered significant at an overall significance level of 5%. The PSM settings were performed using the R package ‘MatchIt’ (v4.5.5) 11 to perform nearest neighbor matching with a caliper of 0.2. All statistical analyses and graphing were performed with SPSS 26.0 software (SPSS, Chicago, IL, USA) and R version 4.3.1 (R Core Team, Vienna, Austria).
Results
Patient characteristics and treatment
A total of 2291 MM patients were screened, identifying 91 (4.0%) cases of MFMM. The control cohort included 182 classic MM patients, matched to MFMM patients based on age and year of diagnosis. Patients’ characteristics were shown in Table 1. The MFMM cohort had a higher proportion of male patients (p = 0.005), fewer clonal plasma cells in the bone marrow by MPFC (<90% of total plasma cell population, p < 0.001), a higher prevalence of extramedullary disease including EMB and EME (p < 0.001), and more cases of bulky extramedullary disease, that is, longest diameter of EMD ⩾ 5 cm (p = 0.003). Additionally, immunoparesis was less frequent (p < 0.001), and fewer patients harbored high-risk cytogenetics or were classified as high risk based on ISS, RISS, and R2ISS staging criteria (p < 0.001) within the MFMM cohort.
Characteristics of patients by macrofocal and classic MM cohorts.
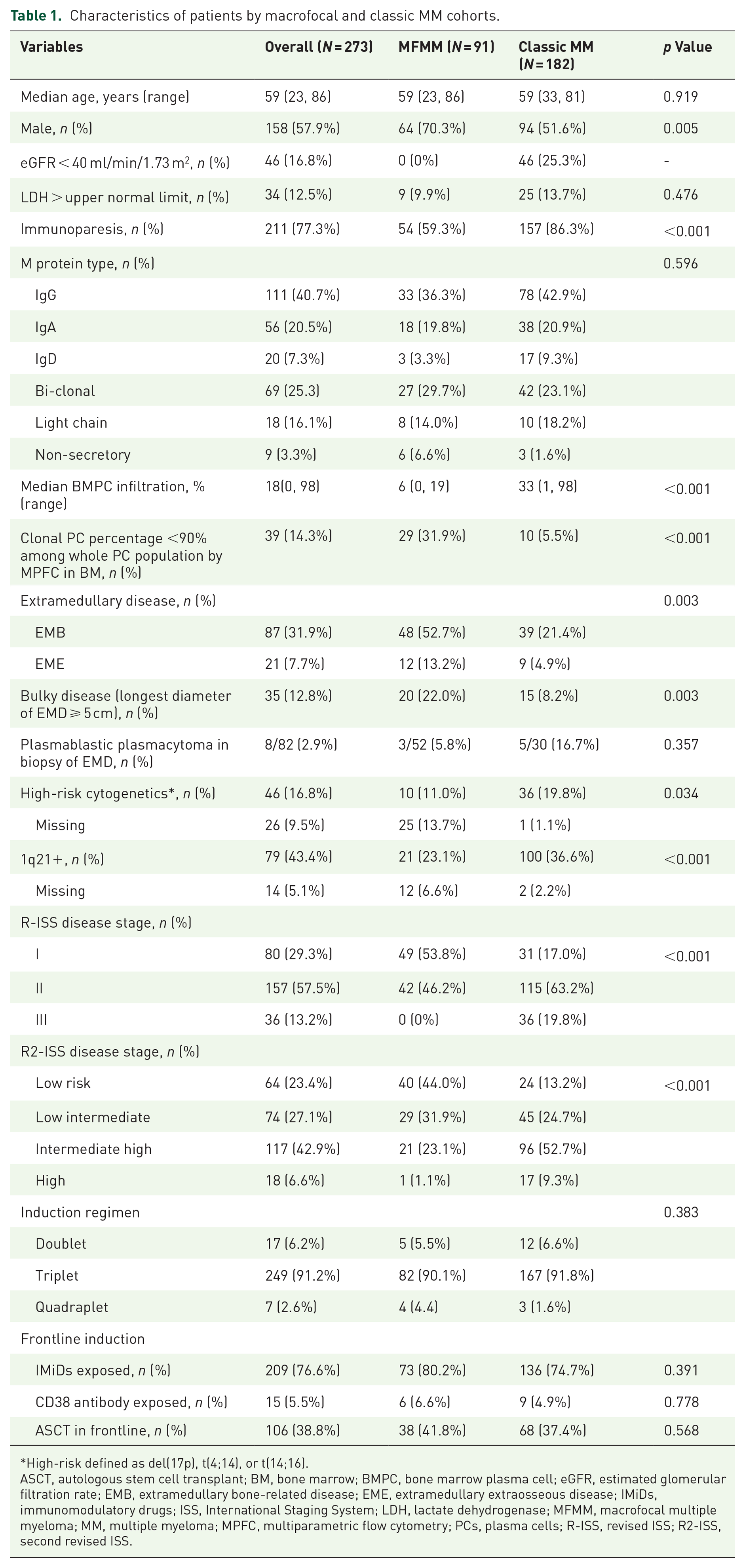
High-risk defined as del(17p), t(4;14), or t(14;16).
ASCT, autologous stem cell transplant; BM, bone marrow; BMPC, bone marrow plasma cell; eGFR, estimated glomerular filtration rate; EMB, extramedullary bone-related disease; EME, extramedullary extraosseous disease; IMiDs, immunomodulatory drugs; ISS, International Staging System; LDH, lactate dehydrogenase; MFMM, macrofocal multiple myeloma; MM, multiple myeloma; MPFC, multiparametric flow cytometry; PCs, plasma cells; R-ISS, revised ISS; R2-ISS, second revised ISS.
Seven (7.7%) patients in the MFMM cohort had a history of solitary plasmacytoma of bone (SBP), compared to none in the control cohort. PET/CT scan was performed in 9 MFMM patients and 16 control MM patients. The median standardized uptake value max of PET/CT was 6.1 (range, 4.2–11.5) in the MFMM cohort, and 4.2 (range, 2.4–9.7) in the control cohort (p = 0.133). In the biopsy specimens of EMD of MFMM patients, the median Ki-67 index was 20% (range, 3%–90%), with three cases (5.8%) of plasmablastic plasmacytoma observed.
In the total cohort, 249 (92.2%) patients received triplet, 17 (6.2%) received doublet, and 7 (2.6%) received quadruplet induction regimens. In frontline treatment, all patients were exposed to PIs. A total of 209 (76.6%) and 15 (5.5%) patients were exposed to IMiDs or CD38 monoclonal antibody-based treatment, respectively. One hundred and six (38.8%) patients underwent upfront ASCT after induction, with 38 (41.8%) in the MFMM cohort and 68 (37.4%) in the control cohort.
Responses
In the total cohort, the overall response rate (ORR) was 86.4% including 78 (28.6%) patients achieving sCR, 46 (16.8%) patients achieving CR, 64 (23.4%) achieving VGPR, and 48 (17.6%) achieving PR. In the MFMM cohort, the ORR was 91.2%, with 31 (34.1%) in sCR, 23 (25.3%) in CR, 15 (16.5%) in VGPR, and 14 (15.4%) in PR. In the control cohort, the ORR was 80.2%, with 18 (10.8%) patients in sCR, 26 (15.6%) in CR, 51 (30.5%) in VGPR, and 39 (23.4%) in PR. Among patients without upfront ASCT, MFMM patients had better responses compared to patients in the control cohort (p = 0.027; Figure 1). Upfront ASCT resulted in significantly better response in both MFMM (p = 0.002) and control (p < 0.001) cohorts.

Rate of responses in MFMM and classic MM patients receiving upfront ASCT or not.
Progression-free survival
The median follow-up for the entire cohort was 42.8 months (95% CI: 34.5–53.7). At the last follow-up, disease progression or death occurred in 38 (41.8%) MFMM patients and 93 (51.1%) in the control cohort. The median PFS for the total cohort was 46.0 months (95% CI: 38.5–53.5), with 73.2 months (95% CI: 35.4–111.0) for MFMM and 44.6 months (95% CI: 36.0–53.2) for the control cohort (p = 0.003; Figure 2). The 2-, 4-, and 6-year PFS probabilities for the total cohort were 72.2% (95% CI: 0.67–0.78), 47.8% (95% CI: 0.41–0.56), and 37.2% (95% CI: 0.30–0.46), respectively. For MFMM, these probabilities were 79.0% (95% CI: 0.71–0.88), 59.5% (95% CI: 0.49–0.73), and 52.8% (95% CI: 0.42–0.67), which were significantly higher than those in the control cohort, which were 68.6% (95% CI: 0.62–0.76), 40.7% (95% CI: 0.32–0.51), and 27.4% (95% CI: 0.19–0.39), respectively. In MFMM cohort, patients received upfront ASCT showed superior PFS than those not (log-rank p < 0.001; Figure 2).
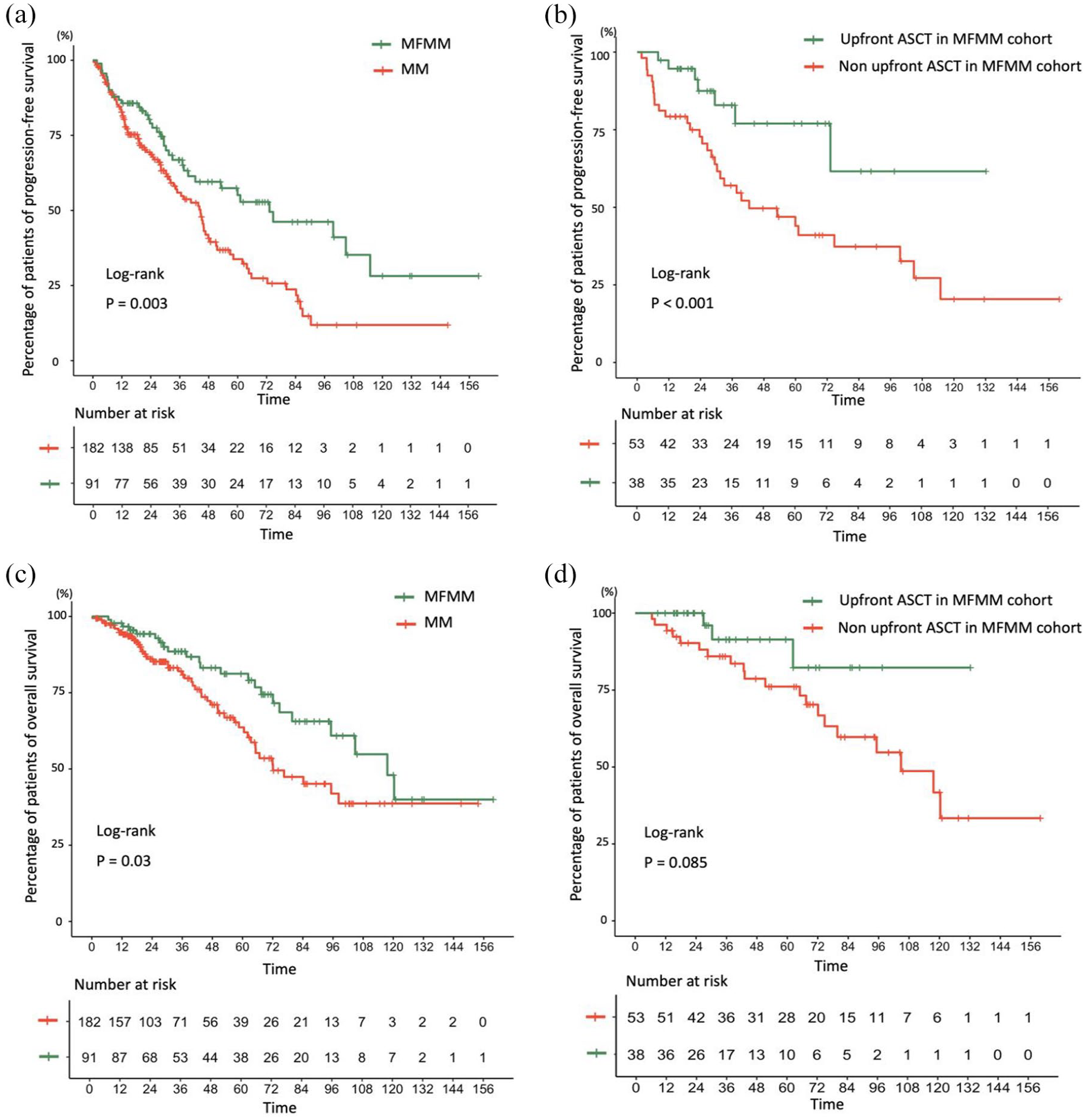
Kaplan–Meier curve of survival. (a) Progression-free survival of MFMM and classic MM patients; (b) progression-free survival of MFMM patients based on upfront ASCT administration; (c) overall survival of MFMM and classic MM patients; (d) overall survival of MFMM patients based on upfront ASCT administration.
For the entire cohort, the detailed results of univariate analysis for PFS were showed in Figure 3. Variables included in the multivariate analysis were MFMM versus control cohort, age, eGFR < 40 ml/min, immunoparesis, clonal BMPC < 90% of the whole plasma cell population by MPFC in BM, 1q21, high-risk cytogenetics, and the use of IMiDs and ASCT in frontline therapy. In multivariate analysis, factors associated with inferior PFS included high-risk cytogenetics, while clonal BMPC < 90% of the whole plasma cell population by MPFC in BM (HR [hazard ratio]: 0.36, 95% CI: 0.19–0.70, p = 0.002), IMiD exposure (HR: 0.57, 95% CI: 0.38–0.85, p = 0.006), and ASCT consolidation (HR: 0.35, 95% CI: 0.22–0.55, p < 0.001) in frontline therapy were associated with superior PFS (Table 2).

Univariate Cox regression analysis for progression-free survival and overall survival for the entire cohort including MFMM and MM patients.
Multivariate Cox analysis of progression-free survival and overall survival for the entire cohort including MFMM and MM patients.

High-risk defined as del(17p), t(4;14), or t(14;16).
ASCT, autologous stem cell transplant; BM, bone marrow; IMiDs, immunomodulatory drugs; MPFC, multiparametric flow cytometry; PCs, plasma cells; PFS, progression-free survival; OS, overall survival; R2-ISS, second revised international staging system.
For the MFMM cohort, additional biopsy sample of plasmacytoma information was incorporated into the univariate analysis, including the presence of plasmablastic differentiation and Ki-67 index values exceeding 20% (Figure 4). Multivariate analysis showed that clonal BMPC < 90% of the whole plasma cell population by MPFC in BM, IMiD exposure, and ASCT consolidation were associated with superior PFS, while the Ki-67 index >20% on biopsy sample of plasmacytoma were associated with inferior PFS (HR: 9.34, 95% CI: 1.63–53.47, p = 0.012; Table 3).

Univariate Cox regression analysis for progression-free survival and overall survival for the MFMM cohort.
Multivariate Cox analysis of progression-free survival and overall survival for the MFMM cohort.

High-risk defined as del(17p), t(4;14), or t(14;16).
ASCT, autologous stem cell transplant; BM, bone marrow; MPFC, multiparametric flow cytometry; IMiDs, immunomodulatory drugs; PCs, plasma cells; PFS, progression-free survival; OS, overall survival; R2-ISS, second revised international staging system.
Overall survival
At the last follow-up, 74 (27.1%) patients in the total cohort had died, including 23 (25.3%) MFMM patients and 51 (28.0%) patients in the control cohort. The median OS for the total cohort was 95.3 months (95% CI: 70.0–120.5), with 117.6 months (95% CI: 93.5–141.8) for MFMM and 72.1 months (95% CI: 45.8–98.4) for the control cohort (p = 0.030). The 2-, 4-, and 6-year OS probabilities for the total cohort were 89.0% (95% CI: 0.85–0.93), 75.7% (95% CI: 0.70–0.82), and 60.9% (95% CI: 0.53–0.70), respectively. For MFMM, these probabilities were 94.3% (95% CI: 0.90–0.99), 83.2% (95% CI: 0.75–0.93), and 74.4% (95% CI: 0.64–0.87), whereas for the control cohort, they were 86.1% (95% CI: 0.81–0.92), 71.0% (95% CI: 0.63–0.80), and 51.5% (95% CI: 0.41–0.64).
For the entire cohort, the detailed results of univariate analysis for OS were shown in Figure 3. In multivariate analysis, factors associated with inferior OS included high-risk cytogenetics and 1q21+. IMiD exposure (HR: 0.49, 95% CI: 0.29–0.82, p = 0.006) and ASCT consolidation (HR: 0.32, 95% CI: 0.15–0.68, p = 0.003) in frontline therapy were associated with superior OS (Table 2).
For the MFMM cohort, additional biopsy sample of plasmacytoma information was incorporated into the univariate analysis (Figure 4). Multivariate analysis showed that IMiD exposure were associated with superior OS, high-risk cytogenetics were associated with inferior OS (Table 3).
Discussion
In this retrospective study, we identified 91 cases (4%) of MFMM among 2291 MM patients diagnosed at Peking University People’s Hospital and Fu Xing Hospital of Capital Medical University over the past 12 years, during which PI-based therapy was widely used in frontline induction treatment. A PSM were performed with a 2:1 ratio of 182 controls to MFMM cases, based on age and year of diagnosis. The MFMM cohort had a higher proportion of male patients, patients with <90% clonal PCs in the BM by MPFC, and patients with extramedullary disease, as well as a lower proportion of patients with high-risk cytogenetics or advanced disease staging. MFMM patients had better overall responses compared to patients in the control cohort (p = 0.027) in patients without upfront ASCT. During the median follow-up of 42.8 months for the entire cohort, the median PFS and OS for MFMM cohort was significantly superior than that in the control cohort. In multivariate analysis in the entire cohort, IMiD exposure and ASCT consolidation in frontline therapy were associated with both superior PFS and OS.
The concept of MFMM was first described by Dimopoulos et al. 2 Initially considered a disease of younger patients, MFMM is now recognized to occur across all age populations, with a incidence of 3%–5%.1,3,4 Due to its low incidence and frequent exclusion from clinical trials, large cohort studies on this subgroup of patients are lacking. The common description and widely accepted opinion regarding MFMM is that patients with this type of disease are generally considered to have a lower risk and better prognosis. Therefore, it remains uncertain whether these patients still require frontline treatment with multiagents combinations or ASCT.
Our findings suggest that MFMM patients, despite having a lower tumor burden and fewer adverse cytogenetic abnormalities, benefit from treatment strategies similar to those used in standard MM, including PIs, IMiDs, and ASCT. This aligns with current clinical guidelines and supports maintaining treatment intensity for MFMM to optimize outcomes. In this study, we reported for the first time that MFMM patients have more bulky extramedullary disease, with the longest diameter ⩾5 cm, and a median Ki-67 index of 20% in EMD biopsies. Importantly, multivariate analysis demonstrated that a Ki-67 index ⩾20% in EMD biopsies is associated with inferior PFS in MFMM patients, highlighting its potential as a prognostic marker. This finding provides valuable prognostic information for MFMM patients, a population where prognostic staging and cytogenetic guidance are often inadequate. Our evidence suggests that the treatment strategies for MFMM cases with elevated Ki-67 levels of EMD biopsies should be tailored to address the higher risk. The prognostic value of these traits warrants further exploration in larger cohorts, as plasmablastic myeloma, known to have a poor prognosis, 12 was not identified as factors associated with poorer PFS in this study, possibly due to the small sample size.
The largest cohort study of MFMM to date was reported by Katodritou et al., 1 with findings that align with some of our observations, including more frequent extramedullary disease, fewer adverse cytogenetics abnormalities, and better treatment responses. However, their study spanned from 2001 to recent years, during which treatment paradigms have significantly evolved, and their multivariate analysis identified only the application of PIs as associated with OS. Modern treatment modality of MM involves novel agents, including PIs, IMiDs, and mAbs, as well as ASCT in eligible patients,5,13 which underscores the need for updated evaluations of prognostic markers within this treatment context. In our cohort, all patients received PIs during induction, 76.6% were exposed to IMiDs, and 38.8% underwent ASCT in frontline settings. Within this framework, our multivariate analysis confirmed the independent prognostic value of IMiDs exposure and ASCT consolidation, supporting treatment strategies consistent with standard MM.
As treatment paradigms evolve with novel therapies, such as CD38 monoclonal antibodies (e.g., daratumumab),14,15 quadruplet regimens,5,13 and advanced immunotherapies including chimeric antigen receptor-T cell therapy and bispecific antibodies, 16 ongoing reassessment of MFMM outcomes is essential to optimize patient care. In our study, limited exposure to these regimens prevented a comprehensive evaluation of their full impact on MFMM outcomes, underscoring the need for continuous monitoring of newer agents and treatment combinations in future research. Furthermore, MM cells promote bone marrow angiogenesis by interacting with endothelial cells, leading to new blood vessel formation that supports tumor growth. Targeting this pathway may inhibit both tumor cell survival and angiogenesis, potentially reducing MM progression. 17 This mechanism is particularly relevant in MFMM, which is often associated with bulky disease in extramedullary lesions, and warrants exploration as a therapeutic target in future studies.18,19
Our study is limited by its retrospective nature, which restricts control over confounding factors. Not all patients underwent bone marrow biopsy, and the FISH test only included specific chromosomal abnormalities. Induction regimens and ASCT consolidation in eligible candidates also varied among patients, which complicates the assessment of their overall impact on MFMM outcomes. For patients with BMPC < 10% lacking bone marrow biopsy, multiple aspiration sites were utilized to improve accuracy in clonal plasma cell measurement, though variability may still remain. The use of a 20% BMPC threshold follows established literature, particularly the findings of Katodritou et al., 1 but remains a topic for further validation against alternative thresholds, such as the 10% criterion described in earlier studies. Notably, all patients in our cohort received PIs, over 75% were exposed to IMiDs, more than 90% received triplet induction, and 40% underwent upfront ASCT consolidation, partially reflecting the standard of care available in some regions in real-world setting.20,21 Other limitations include the use of MPFC with a sensitivity threshold of only 10−4 and the single-country, dual-hospital setting, which may also limit the generalizability of our findings. These factors necessitate careful interpretation of our conclusions. Although larger multicenter, prospective cohort studies could potentially minimize bias and enhance generalizability, the very low incidence of MFMM means gathering a sufficiently large cohort prospectively would require many years, which could reduce the relevance of findings as MM treatment rapidly evolves.
Conclusion
In this study with 91 MFMM and 182 classic MM patients matched by age and year through PSM, the MFMM cohort exhibited more EMD, fewer clonal PCs in the BM, fewer high-risk cytogenetics or advanced disease staging, and better treatment responses. IMiDs exposure and ASCT consolidation were independently associated with superior PFS and OS. This emphasizes that for MFMM, treatment choices and intensity should be consistent with those for standard MM. In the MFMM cohort, the presence of Ki-67 index ⩾20% in EMD biopsies were associated with an inferior PFS.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251314696 – Supplemental material for Macrofocal multiple myeloma in the era of novel agents in China
Supplemental material, sj-docx-1-tah-10.1177_20406207251314696 for Macrofocal multiple myeloma in the era of novel agents in China by Xuelin Dou, Ruixia Liu, Yang Liu, Nan Peng, Lei Wen, Daoxing Deng, Leqing Cao, Qian Li, Liru Wang, Fengrong Wang, Xiaodong Mo and Jin Lu in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
