Abstract
OBJECTIVE:
To investigate FOXO1 expression in epithelial ovarian cancer (EOC), and to explore its correlation with clinicopathological parameters and prognosis of EOC.
METHODS:
Two hundred and sixteen cases of paraffin-embedded EOC and 41 paratumor tissues from 2009 to 2017 that had been pathologically confirmed at the memorial hospital of Sun Yat-sen University were included in this study, and the expression of FOXO1 was performed by immunohistochemistry using a polyclonal antibody specific for FOXO1.
RESULTS:
FOXO1 protein expression is associated with Recurrence free and overall survival in EOC patients; In addition, FOXO1 expression is associated with age, FIGO stage, intraperitoneal metastasis, intestinal metastasis, vital status, intraperitoneal recurrence and differentiation grade; Moreover, in a multivariate model FOXO1 overexpression was an independent predictor of poor survival in EOC.
CONCLUSION:
FOXO1 may play a candidate oncogenic role in EOC, and FOXO1 is a useful independent prognostic marker in EOC, and it may provide a candidate target therapy in future.
Background
Ovarian cancer is the first leading cause of death from cancers of the female reproductive system [1]. Each year, approximately 238,700 patients are diagnosed with this disease and approximately 151,900 associated mortalities occur globally [2]. And the most type is epithelial. Most of EOC is diagnosed at an advanced stage (the International Federation of Gynecology and Obstetrics (FIGO) III or IV stages) because of the lack of specific symptoms and the absence of effective early diagnostic methods, which leads to a poor prognosis [1, 3]. Recently serum biomarkers such as CA125 and HE4 have been used to monitor the progress and prognosis of ovarian cancer, and also to detect the recurrence of disease after operation or chemotherapy. However, these biomarkers are neither extremely sensitive nor particularly specific for predicting cancer metastasis, recurrence, and prognosis. Therefore, further studies to search new biomarkers and to provide targeted therapy are very important.
The FOXO1 gene located on chromosome 13q4 in human, which is a member of the forkhead box (FOXO) family, and ther are a series of recent studies have demonstrated plays an important role in several intracellular functions, including autophagy [4, 5], cell cycle [6], apoptosis [7], and tumor suppression [8]. Moreover, Many researches have reported that FOXO1 has antineoplastic actions in digestive system cancers [9, 10, 11, 12, 13], prostate cancer [14], breast cancer [15], hematological cancer [16] and others [17, 18]. But in contrary, FOXO1 plays an opposite role in acute myeloid leukemia [19] and renal cell carcinoma [20]. However, there have been no research about the role of FOXO1 in EOC already. Therefore, in this research we aimed to investigate the FOXO1 expression in EOC tissues and paratumor tissues, and to study its clinicopathological and prognostic correlations in EOC.
Material and methods
Tissue specimens
A total of 216 paraffin-embedded ovarian cancer and 41 paratumor tissues from 2009 to 2017 that had been pathologically confirmed at the memorial hospital of Sun Yat-sen University were included in this study. Survival duration was calculated from the operation date until 14 April 2018 when any remaining survivors were included. This trial was obtained approval from the memorial hospital of Sun Yat-sen University Ethics Committee.
Immunohistochemistry (IHC)
The FOXO1 protein expression levels in the human EOC tissues were detected by immunohistochemistry. Briefly, 4
FOXO1 expression in EOC tissues and paratumor tissues (scale bar: 25 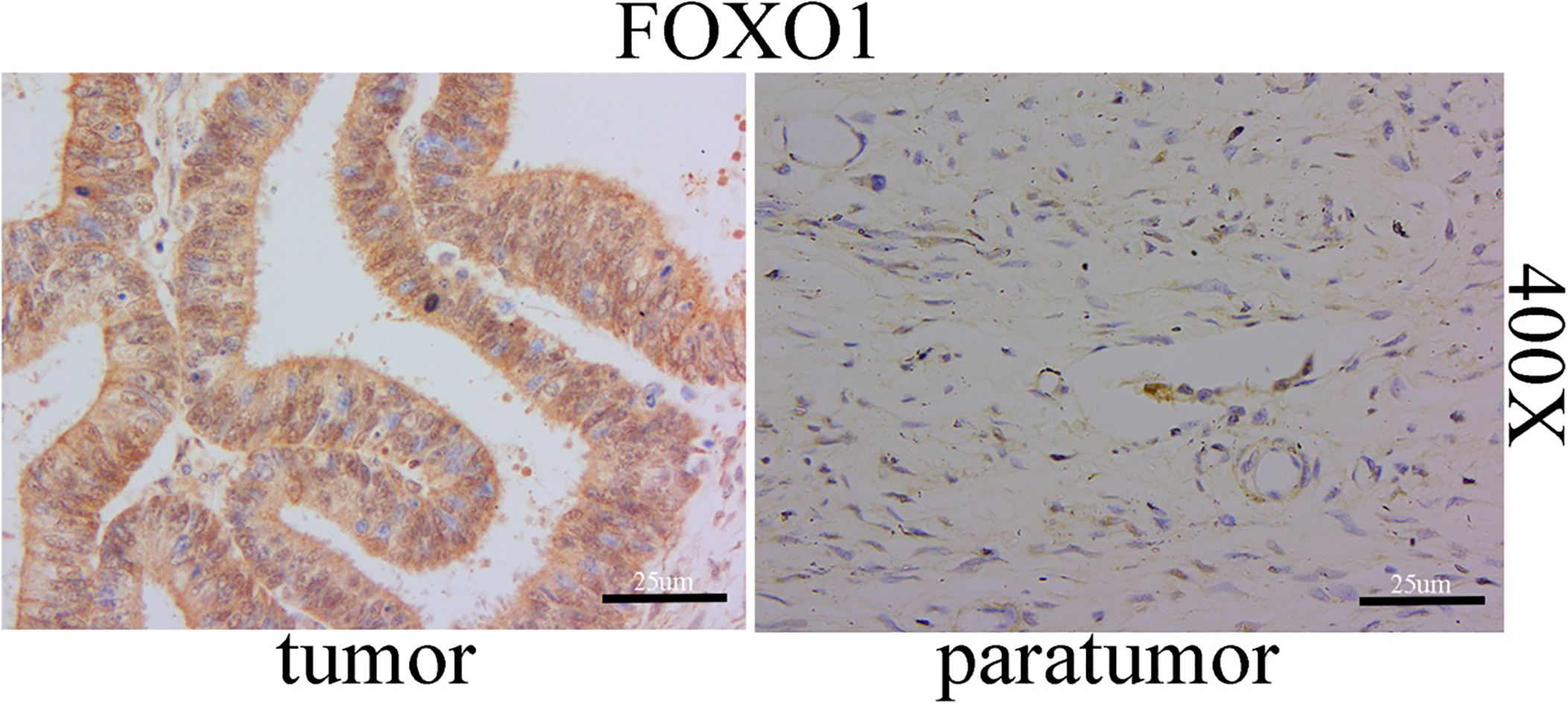
The different straining of FOXO1 expression in EOC (scale bar: 25 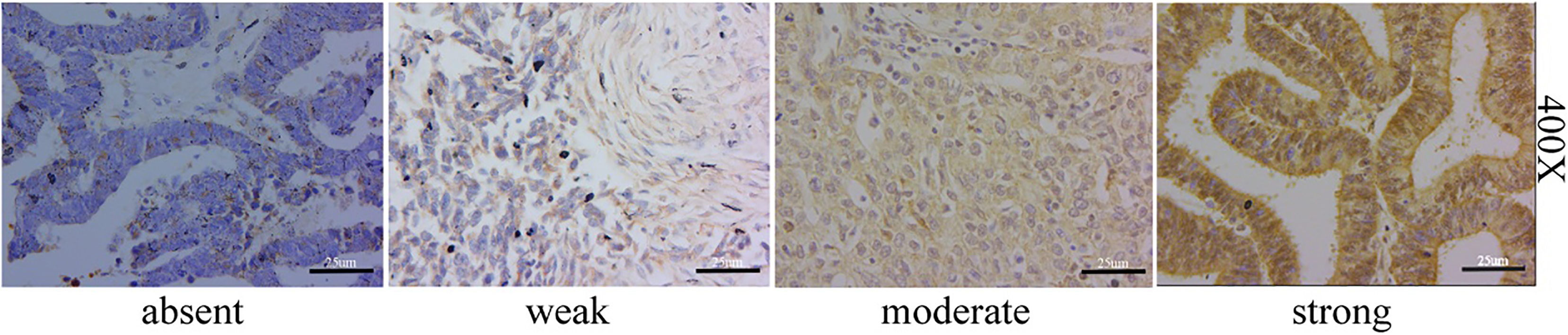
Kaplan-Meier survival of overall survival (OS) and Recurrence free survival (RFS) in dead EOC patients: A. Kaplan-Meier survival of overall survival (OS) in dead ovarian cancer patients; B. Kaplan-Meier survival of Recurrence free survival (RFS) in dead ovarian cancer patients.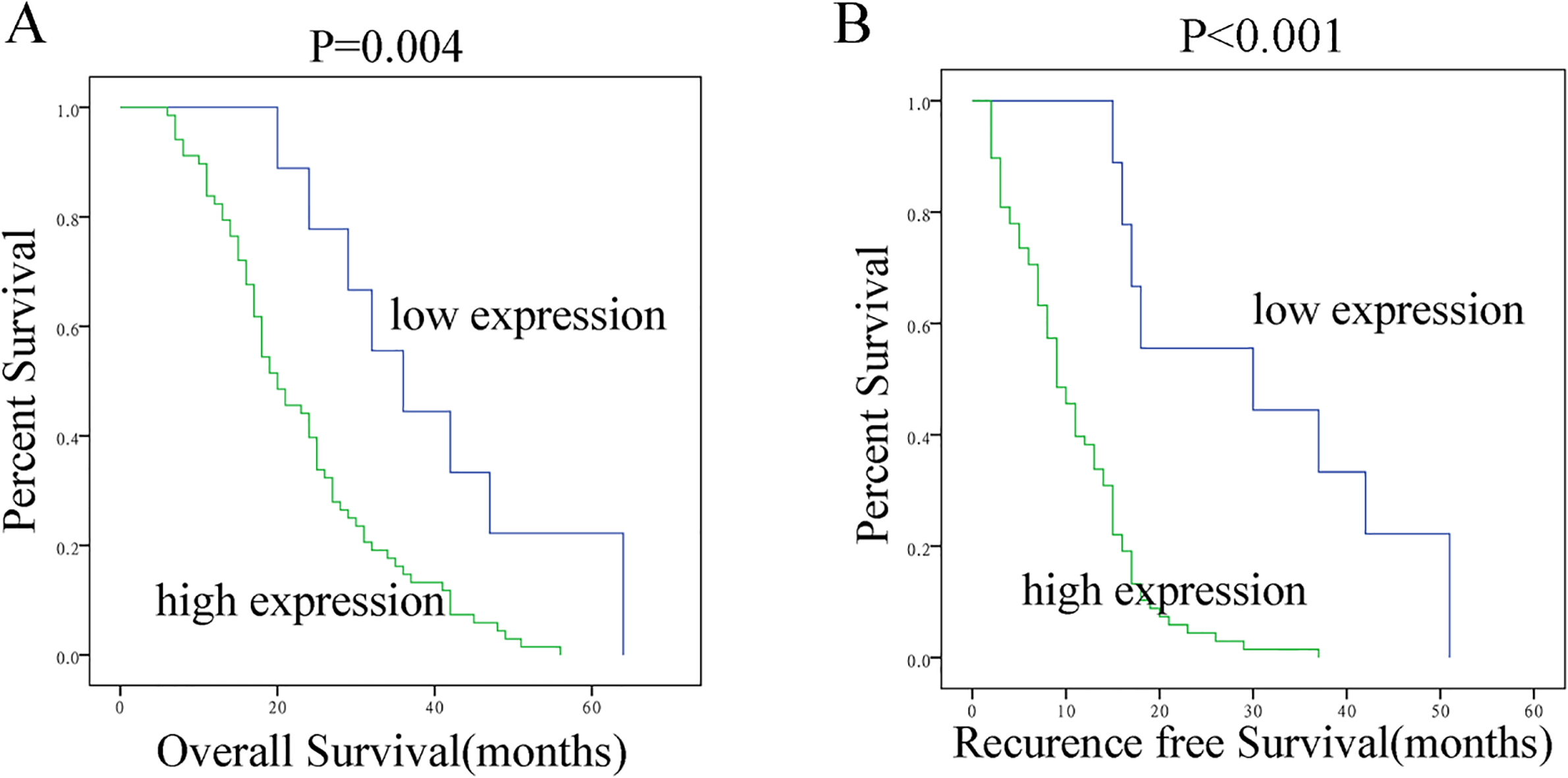
All of the statistical analyses were carried out with the statistical software package SPSS 21.0. The chi-square and Fisher’s exact test were used to analyze the relationship between FOXO1 protein expression levels and clinicopathological characteristics. Additionally, bivariate correlations were computed by Spearman’s rank correlation coefficients. Patient survival was determined by a Kaplan-Meier analysis, and the differences were counted by the log-rank test. Cox’s proportional hazards regression model was applied to the multivariate analysis. A
Results
FOXO1 protein expression is associated with Recurrence free survival and overall survival in EOC patients
To determine whether the FOXO1 protein expression was associated with EOC patients survival, we stained 216 EOC cases and 41 paired paratumor tissues using IHC with a rabbit polyclonal antibody specific for FOXO1. We found that FOXO1 expression staining located at cytoplasmic (Fig. 1).
Moreover, in the 216 evaluable EOC tissues stained, 31/216 (14.4%) had absent/weak staining (also called low expression) and 185/216 (85.6%) had moderate/ strong staining (also called high expression) (Fig. 2).
In addition, Kaplan-Meier survival analysis demonstrated that there was a statistically significance on Recurrence free survival (RFS) between high and low expression of FOXO1 (
Comparison of EOC/paratumor tissues immunohistochemically stained for FOXO1
In 216 EOC tissues stained with the specific FOXO1 antibody, 31/216 (14.4%) had weak/absent staining and 185/216 (85.6%) had strong/moderate staining, while in 41 ovarian paratumor tissues stained with the FOXO1 specific antibody, 35/41 (85.4%) had absent/weak staining and 6/41 (14.6%) had moderate/ strong staining, and there was significantly different between cancer and paratumor groups (
The comparison of FOXO1 expression between tumor and paratumor tissues.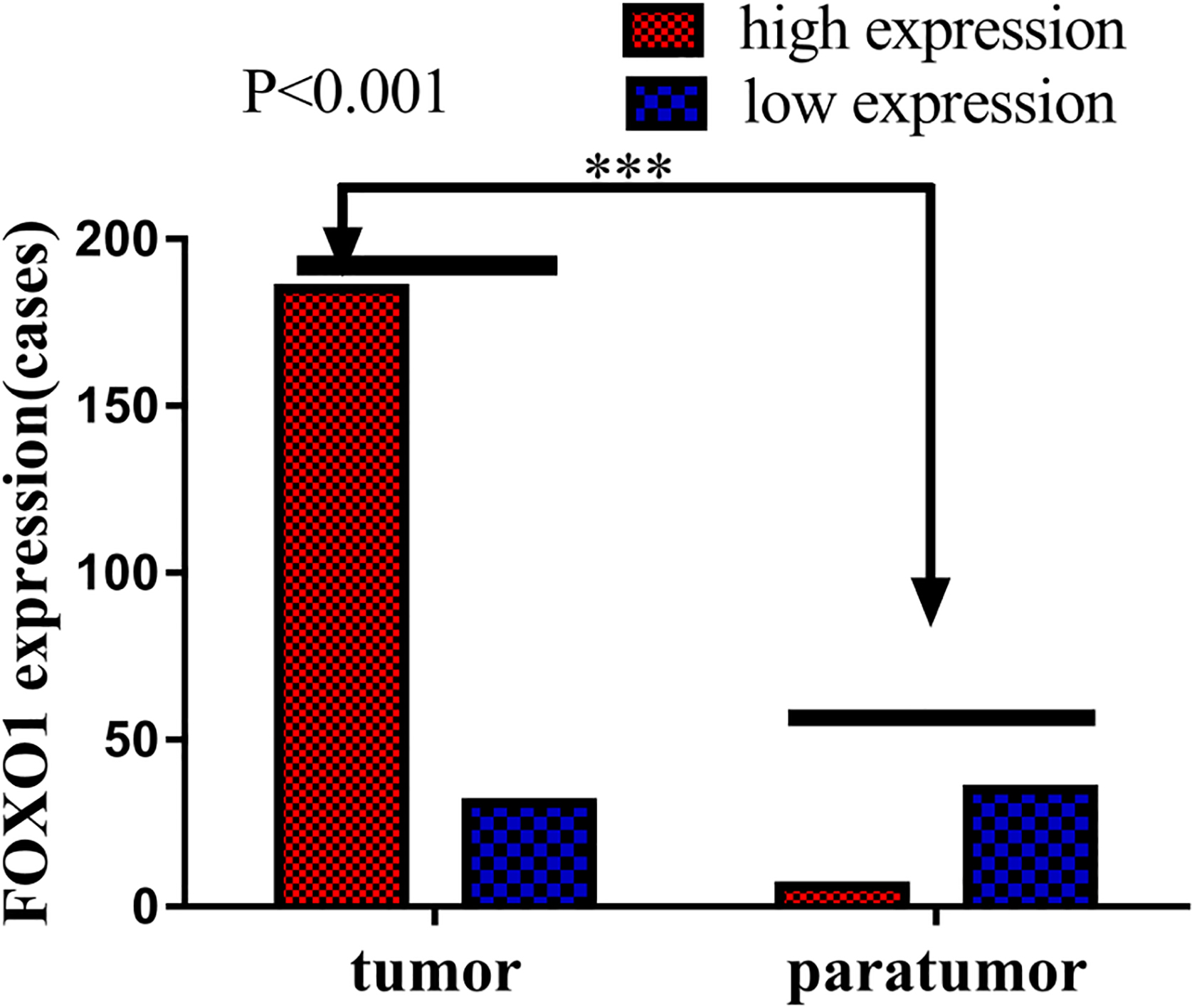
Clinicopathological characteristics and FOXO1 expression in EOC
Subsequently, we studied its correlation with the clinical parameters of EOC. The percentage of patients with stages I–IV tumors were 16.2%, 11.6%, 56.9%, and 15.3%, respectively (Table 1). In this study, patients with high expression of FOXO1 exhibited a median overall survial time of 20 months, while patients with low expression of FOXO1 exhibited a median overall survial time of 36 months. Moreover, patients with high expression of FOXO1 exhibited a median Recurrence free survial time of 9 months, while patients with low expression of FOXO1 exhibited a median Recurrence free survial time of 30 months.
In addition, The results of
FOXO1 expression in association with standard clinicopathological variables using the
or Fisher’s Exact test
FOXO1 expression in association with standard clinicopathological variables using the
Correlation between FOXO1 expression and clinicopathological characteristics of EOC
Cox regression univariate and multivariate analyses of prognostic factors in EOC
Spearman’s correlation analysis revealed that high FOXO1 expression was correlated with the following clinical characteristics: age (
FOXO1 overexpression was significantly associated with a poor prognosis
Furthermore, we also assessed the prognostic value of FOXO1 expression in EOC patient. In a univariate Cox analysis, FOXO1 expression, histological type, intestinal metastasis, hyperthermic intraperitoneal chemotherapy were significant prognostic factors (Table 4). Moreover, in a multivariate Cox regression analysis found that FOXO1 expression, histological type and hyperthermic intraperitoneal chemotherapy were indeed independent prognostic factors of EOC (Table 4), but intestinal metastasis was no longer significant. Taken together, all of these results suggest that FOXO1 expression was an indeed independent prognostic factor, and FOXO1 overexpression may corelate with the prognosis of EOC.
Discussion
Many studies have showed that FOXO1 has antineoplastic actions in a great deal of different cancers [23], such as: digestive system cancers [9, 10, 11, 12, 13], prostate cancer [14], breast cancer [15], hematological cancer [16] and others [17, 18]. But in contrary, FOXO1 plays an opposite role in acute myeloid leukemia [19] and renal cell carcinoma [20]. So there have been some disputes on its negative or positive role in tumors. In this study we for the first time demonstrated that FOXO1 protein expression was elevated in EOC tissues, and in 216 EOC tissues, 185/216 (85.6%) had strong/moderate staining, while only 6/41 (14.6%) had strong/moderate staining in paratumor group, which suggested that FOXO1 may play a candidate oncogenic role in EOC.
Moreover, overexpression of FOXO1 in EOC patients was found to be significantly related with age, FIGO stage, intraperitoneal metastasis, intestinal metastasis, vital status (at last follow-up), intraperitoneal recurrence and differentiation grade. Thus, we concluded that patients with FOXO1 overexpression was more likely to trend for older patients, and this indicated that FOXO1 detection should performed in older people in order to diagnose earlier. Moreover, FOXO1 overexpression was more in stage III/IV patients than that in stage I/II patients, and FOXO1 overexpression was more in grade G3 patients than that in grade G1/G2 patients, and these results showed that FOXO1 overexpression were more likely to exhibit advanced and high grade disease, so we concluded that FOXO1 expression increased with the initiation and progress of ovarian cancer, and it could be recommended as a candidate molecular biomarker.
In addition, FOXO1 overexpression was more likely to exhibit intraperitoneal metastasis, intestinal metastasis and intraperitoneal recurrence, and all of these results showed that FOXO1 was correlated with EOC metastasis and recurrence. To the best of our knowledge, ovarian cancer metastasis and recurrence are the major risk factors of poor survival. Patients with metastasis and recurrence usually have a worse prognosis. Thus, an early diagnosis of intraperitoneal metastasis and recurrence is important for the survival of ovarian cancer patients. However, only a few tumor markers are currently used to predict intraperitoneal metastasis in the clinical work. And according to our results, FOXO1 overexpression was associated with intraperitoneal metastasis and recurrence, and it was meaningful as a biomarker or predictor to diagnose intraperitoneal metastasis and recurrence. Moreover, we concluded that FOXO1 may directly or indirectly involve in the migration and invasion of EOC. And it needs more in vivo and in vitro experiments to identify its role in ovarian cancer.
More importantly, our results showed that FOXO1 overexpression has a poor overall survival (OS) and Recurrence free survival (RFS) of EOC patients. And a survival analysis revealed that the cumulative OS and RFS rates of EOC patients increased with increase in FOXO1 protein expression. In this study, patients with high FOXO1 expression exhibited a median overall survival time of only 20 months and a median RFS time of only 9 months, while these values in patients with low FOXO1 expression were 36 and 30 months, respectively. And in a multivariate Cox regression analysis we found that FOXO1 expression, histological type, and hyperthermic intraperitoneal chemotherapy were indeed independent prognostic factors of ovarian cancer, but intestinal metastasis was no longer significant, which suggest that hyperthermic intraperitoneal chemotherapy should be performed in cytoreductive surgery, because it influences the prognosis of ovarian cancer.
In conclusion, all of these results suggest that FOXO1 overexpression is an independent prognostic factor, and FOXO1 overexpression may corelate with the progression and poor prognosis of EOC. In future, we need more in vivo and in vitro study to demonstrate its role and molecular mechanism in development and progression of EOC.
Conclusions
In short, we demonstrated that FOXO1 overexpression is an independent prognostic factor, and FOXO1 is a useful independent prognostic marker in EOC for the first time. In future, we need more in vivo and in vitro study to demonstrate its role and molecular mechanism in development and progression of EOC.
Footnotes
Acknowledgments
We would like to thank all the participants that contribute to this work. This study was supported by the Guangdong Provincial Medical Research Fund (grant no. A2019096), the President funds of Integrated Hospital of Traditional Chinese Medicine, Southern Medical University (No. 1201902001; No. 1201901002), the High-Level Academic Talent Training Program of Guangzhou Medical University (No. B185004083; No. B195001100), Guangzhou Science and Information Bureau Item (grant no. 201904010013), Natural Science Foundation of Guangdong Province (grant no. 2018A0303130 180) and Hospital achievement transformation and Cultivation Project (grant no. ZH201812).
Conflict of interest
The authors declare no conflict of interest.
