Abstract
BACKGROUND AND AIM:
Relaxin is a short circulating peptide hormone. The aim of this study was to understand the role of relaxin in progression of epithelial ovarian cancer (EOC) and to assess its diagnostic and prognostic significance.
METHODS:
A total of 124 patients with EOC, 46 patients with benign ovarian diseases, and 50 healthy controls were recruited. Serum levels of relaxin were determined by ELISA method. The relationship between serum relaxin level and each of the clinicopathological parameters was analyzed using the
RESULTS:
The results showed that serum relaxin level was significantly higher in patients with EOC than those with benign ovarian diseases and healthy controls (
CONCLUSIONS:
These results indicated that serum relaxin may be a clinically useful indicator for diagnostic and prognostic evaluation in EOC patients.
Introduction
Ovarian cancer (OC) is one of the leading causes of death among all gynecological malignancies. 70% of cases of ovarian cancer are not diagnosed until the cancer has reached an advanced stage, FIGO Stages IIB to IV (spread of tumor within the pelvis or elsewhere in the abdomen).
Primary surgery is the mainstay of treatment for ovarian cancer, followed by adjuvant chemotherapy to destroy any gross or microscopic residual tumour cells. In early-stage ovarian cancer, several retrospective studies have demonstrated that minimally invasive surgery is both feasible and safe compared to laparotomy with seemingly comparable oncologic outcomes [1, 2]. In advanced ovarian cancer, minimally invasive surgery is not currently recommended for primary cytoreduction. However, laparoscopic evaluation may be a useful decision-making tool to identify those patients who are at high risk for suboptimal primary debulking surgery and would thus benefit from neoadjuvant chemotherapy followed by an interval debulking surgery [3].
As to the advanced ovarian cancer, adjuvant chemotherapy is mandatory except for surgery. Although approximately 80% of patients respond to front-line chemotherapy, more than 70% of patients with advanced stage disease recur within 5 years and develop drug resistance [4]. For recurrent disease, the treatment choice is based on the timing and nature of the recurrence and the extent of prior chemotherapy [5]. Platinum-sensitive patients with a treatment-free interval
The bulk of ovarian cancer cells are resistant to chemotherapy prior to the death of the patient. It is not clear if chemoresistance is an acquired property of cells under the selective pressure of chemotherapy or if it is an innate property of a small proportion of cancer cells from the outset [6]. One of the mechanisms is that the cancer cells may have inherent chemoresistance which allows an indefinite expansion, or trransformed ovarian epithelial cells may undergo an immunoediting process or immunoedited ovarian cancer drug-resistant cells escape first-line chemotherapy [7].
Therefore, the early diagnosis and early prediction susceptibility to chemotherapy would provide a better prognosis in ovarian cancer.
As yet there is no approach that justifies a recommendation of general screening for ovarian cancer. The regular transvaginal ultrasound and CA 125 testing, including evaluation that involved complex algorithms have recommended to detect ovarian cancer, but it has not yet been shown whether these screening methods lead to a reduction in mortality [8, 9]. A number of researchers and clinicians have been investigating many new diagnostic markers. However, the number of clinically useful markers is pitifully small [10]. This is partly due to the complex nature of the tumor biomarkers themselves-especially those at the DNA, RNA, and protein levels-and partly due to the lack of acceptable standards for effectively evaluating and incorporating these newer biomarkers in the clinic.
Relaxin (H2 relaxin in humans and relaxin-1 in rats; traditionally referred to as simply “relaxin”) was discovered by Frederick Hisaw in 1926 when he injected serum from pregnant guinea pigs or rabbits into virgin guinea pigs and observed a dramatic relaxation of the pelvic ligament [11]. Relaxin exerts its actions via its leucine-rich-repeat-containing G-protein coupled receptors (GPCR) RXFP1 and RXFP2. As “pregnancy hormone”, relaxin promotes elongation of the interpubic ligament in mammals, inhibits spontaneous contractions of the uterine myometrium in guinea pigs and promotes cervical softening in several species [12]. It is produced in small quantities within the male reproduction tract and has been detected in the human prostate. Further, relaxin is involved in fibrosis, wound healing, cardiac protection and allergic responses [12]. Most recently relaxin has been associated with cancer biology. A number of putative roles, including the modulation of tumor growth, neovascularization, metastasis and oncogenic progression, have been correlated to relaxin overexpression [13, 14, 15, 16].
Elevated relaxin serum concentrations were found in breast cancer patients, especially in patients with metastatic disease [17]. In addition, serum relaxin levels were also significantly higher in patients with osteosarcoma and esophageal squamous cell carcinoma, and higher serum concentrations of relaxin has a poor prognosis for these patients [18, 19]. Kamat et al. has reported that relaxin was overexpressed in EOC, and strong relaxin expression associated significantly with high-grade and depth of myometrial invasion. Notably, strong relaxin expression was associated with a significantly shorter overall survival compared to weak or moderate expression [20].
In this study, we measured the serum levels of human relaxin in EOC patients using an enzyme linked immunosorbent assay (ELISA), and analyzed the association between clinical parameters of EOC and serum relaxin levels.
Materials and methods
Patients
The study complies with the standards of the Declaration of Helsinki and current ethical guidelines and was approved by Ethic Committee of the affiliated hospital of Qingdao University. The Ethic Committee approved that informed consent was not required because data were going to be collected retrospectively and analyzed anonymously.The study included 124 consecutive ovarian cancer patients (age: 60.4
In addition, samples of 46 age-matched patients with benign lesions were analyzed. Of the benign lesions, 18 were classified as mucinous cystadenomas, 6 as benign ovarian teratomas, 8 as ovarian dermoid cysts, 2 as sclerosant tumor, 2 as serous cystadenomas, 3 as fibroma, and 7 as corpus luteum cyst. A total of 50 healthy controls (age: 54.8
Sample collection
Serum samples from all patients were collected presurgically, before initiation of therapy, and stored at
Statistical analysis
The results were expressed as mean
Serum levels of relaxin in EOC patients in relation to clinic-pathological variables of tumor
Serum levels of relaxin in EOC patients in relation to clinic-pathological variables of tumor
Relaxin is elevated in serum of patients with EOC
Mean serum relaxin level was 13.8 pg/mL (range, 0–189.7) in 124 patients with EOC, which was significantly higher compared to patients with 46 cases of benign gynecological conditions [0.63 pg/mL (range, 0–16.7)] and 50 cases of healthy controls [0.56 pg/mL (range, 0–15.4)]. These elevations were found to be statistically significant through the Mann-Whitney test (
Serum relaxin level and clinicopathological features in EOC patients
Mean serum relaxin level was 13.8 pg/mL. We used 13.8 pg/mL as the cutoff value, EOC patients were then divided into group A (
Univariate and multivariate survival analysis in patients with EOC
Univariate and multivariate survival analysis in patients with EOC
We generated ROC curves to assess the potential usefulness of serum relaxin as a noninvasive biomarker for the diagnosis of ovarian cancer. Our ROC analyses revealed that serum relaxin levels were robust in discriminating patients with ovarian cancer from control subjects, with an AUC value of 0.774 (95% CI, 0.645–0.836) (Fig. 1A). Using a cutoff value of 13.8 pg/mL, the sensitivity, specificity, and positive and negative predictive values were 71.8%, 78%, 91.4%, and 70.3%, respectively, to identify a patient with ovarian cancer.
Serum relaxin levels in serum samples. A, Serum relaxin yielded an area under the curve (AUC) value of 0.774 in distinguishing ovarian cancer from healthy control subjects. B, Serum relaxin yielded AUC values of 0.786 in discriminating ovarian from benign gynecological conditions.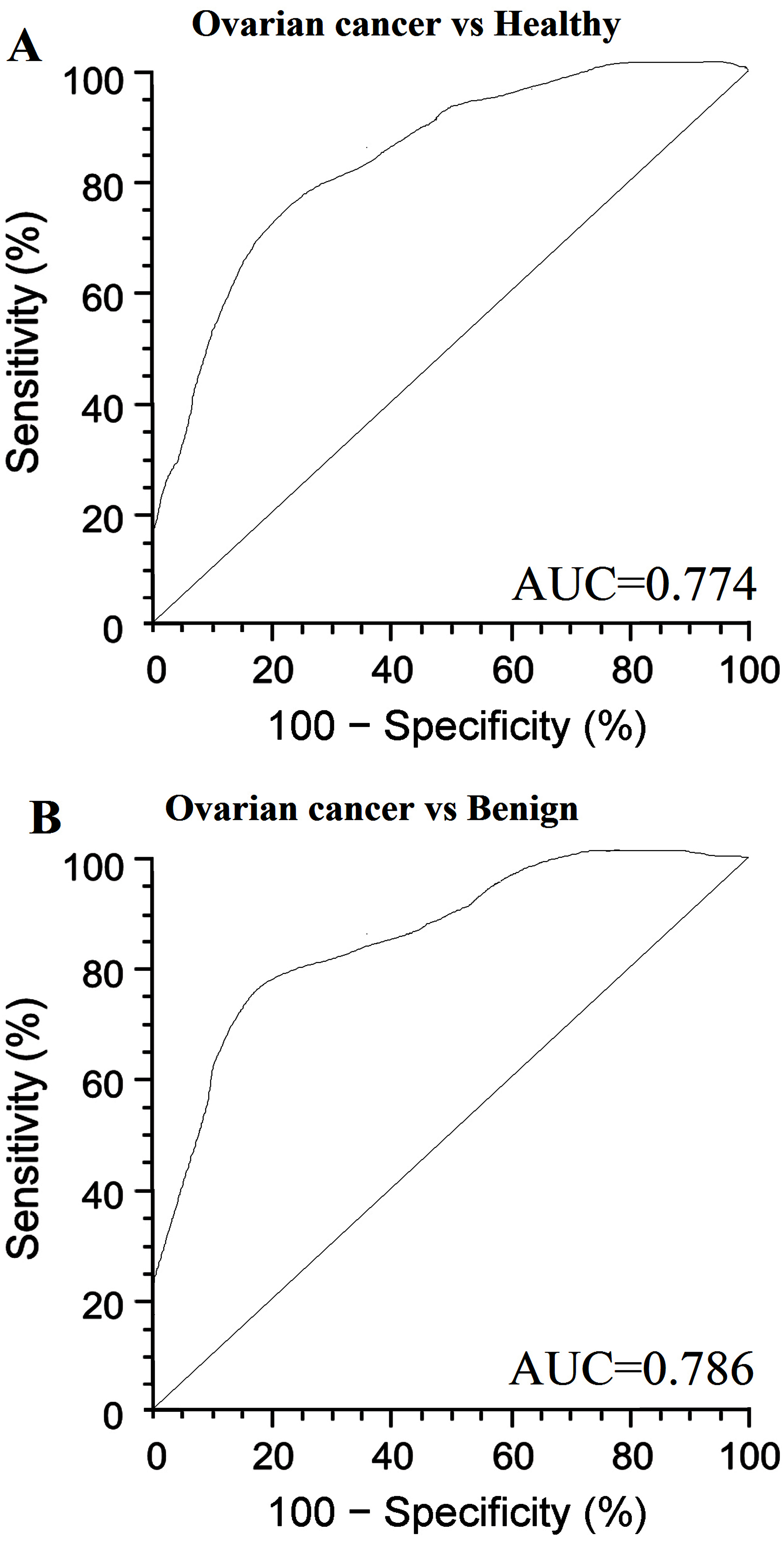
Kaplan-Meier survival curves. Percent survival rate was stratified by relaxin level. A, progression-free survival; B, overall survival of patients.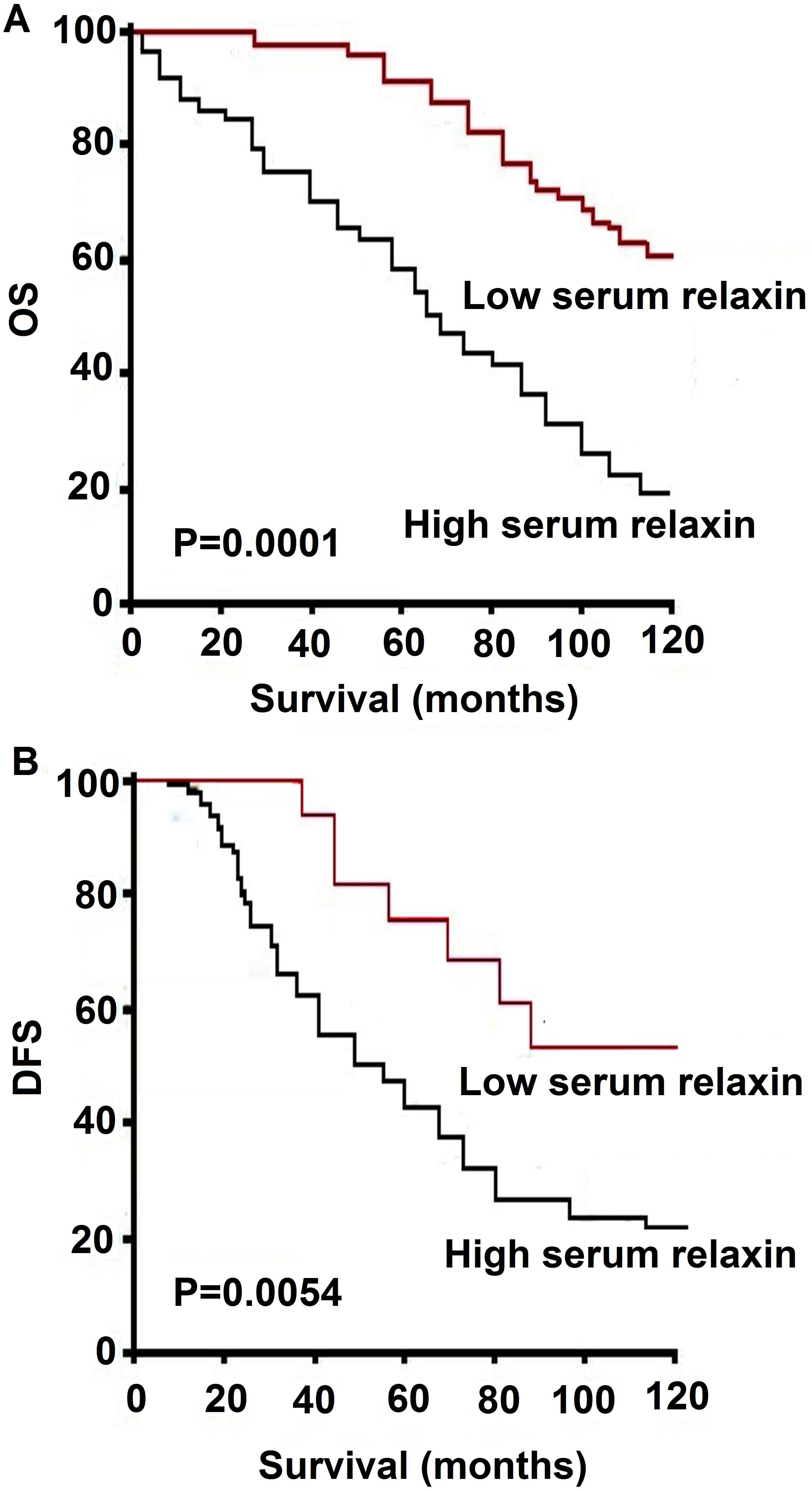
Even more important from a screening perspective, serum relaxin levels could reliably differentiate patients ovarian cancer from benign gynecological conditions, as evidenced by an AUC value of 0.786 (95% CI, 0.663–0.857) (Fig. 1B). The sensitivity, specificity, and positive and negative predictive values were 70.6 % and 82.6%, 88.6%, and 68.2%, respective.
To identify factors with potential prognostic significance in ovarian patients, univariate analysis for each variable was performed in relation to the survival time. In our univariate analysis, stepwise inclusion of variables in the model indicated that significant prognostic factors included serum relaxin level, lymph node metastasis, FIGO stage, recurrence, chemotherapy. In addition, multivariate Cox regression analysis revealed that serum relaxin level, lymph node metastasis, FIGO stage, recurrence, and chemotherapy were indeed independent prognostic markers for ovarian cancer (Table 2).
To further determine the value of serum relaxin in predicting survival of ovarian cancer patients, Kaplan–Meier analysis and the log-rank test were used in this study. The log-rank test showed that the survival time was significantly different between these two groups. As shown in Fig. 2, serum relaxin in ovarian cancer patients was associated with survival time, the patients with low serum relaxin surviving much longer than those with high serum relaxin level. The cumulative OS and disease-free survival (DFS) rates for the patients with high levels of serum relaxin is low than that of the low serum relaxin (
Discussion
Once the diagnosis of gynaecological cancer is established, the patient and his partner will have psychological and physiological adverse consequences. Surgical treatment, chemotherapy and radiotherapy may lead to decreased libido and the decline in the quality of life in both spouses [21]. In addition, gynaecological cancer treatment, including advanced ovarian cancer, can lead to a decline in the quality of life, and depression and anxiety in patients [22]. Therefore, it is necessary to reduce as much as possible the impact of cancer on the sexual and psychological functioning of patients and their partners [23]. The search for an effective screening test for ovarian cancer has been the focus of intensive research efforts.
Blood flows into and out of tumors and circulates tumor-specific protein profiles, making serum or plasma the ideal biological media for finding a screening biomarker [24]. More than a thousand publications have identified potential prognostic markers of epithelial ovarian cancer. However, most of these proposed markers have an uncertain clinical value, their independent prognostic significance is unclear and none are used clinically [10].
The mammalian peptide hormone relaxin is well known for its matrix-modifying capacity. It induces extensive tissue remodeling via upregulation of matrix metalloproteinases (MMP) and angiogenesis [25, 26]. Since this is also a key feature of malignant invasion, they have been implicated in cancer progression. Previous studies showed that serum relaxin concentrations was significantly increased in patients with cancer than the healthy blood donors [18, 19, 20], and higher serum relaxin concentrations has a poor prognosis [18, 19].
In the present study, we measured the serum relaxin levels using an enzyme linked immunosorbent assay (ELISA) to investigate first whether or not relaxin could detect EOC patients and differentiate malignant from benign tumors. The results showed that the levels of relaxin was elevated in ovarian cancer patients compared with benign ovarian diseases and healthy controls. Serum relaxin levels distinguished ovarian cancer patients from normal individuals and/or benign ovarian disease patients very reliably.
The levels of relaxin in the serum of 124 ovarian cancer patients were assessed in relation to clinicopathological features of tumor such as FIGO stage, grading, tumor location, lymph node metastasis, tumor resectability, survival of the patients, recurrence, chemotherapy response and ascites. The results showed that serum relaxin increased significantly with FIOGO stage, lymph node metastasis, survival of patients, tumor resectablility, recurrence and chemotherapy response. Our results are in agreement with the findings of Ren et al. [19], who revealed significant difference of relaxin in lymph node metastasis. As described above, it suggests that the elevated levels of relaxin might reflect the role of this protein as a stimulator of tumor cell metastasis in ovarian cancer progression.
Finally, survival analysis of the ovarian cancer patients was evaluated in this study based on the levels of relaxin. In our study, survival rate of patients with high relaxin level was significantly lower than the patients with low relaxin level. Besides the already established prognostic predictor, such as FIGO stage and lymph node metastasis, serum relaxin was independently associated with prognosis in all patients with ovarian cancer. Thus, serum relaxin can identify EOC patients who are of disease-related death. This could well influence treatment decisions.
In conclusion, serum relaxin may be involved in the progression of ovarian cancer, and that high level of serum relaxin may be related to tumor invasion, recurrence and chemotherapy response in ovarian cancer. As a noninvasive marker, serum relaxin has the potential to clinically expedite the diagnosis of ovarian cancer and to differentiate malignant from benign tumors. Serum relaxin was an independent variable in ovarian cancer. Examining and monitoring serum ovarian cancer levels may be useful in estimating the prognosis of patients with ovarian cancer.
