Abstract
PIWIL2 is a human Argonaute protein, which is guided by small RNAs to its targets, plays a role in germ cell maintenance and has been proposed to be expressed in precancerous stem cells and tumor stem cells. However, the significance of PIWIL2 expression in oral cancer and precancerous lesions has not been investigated. In this study, we analyzed the expression of the stem cell protein PIWIL2 in oral squamous-cell carcinoma (OSCC) and in premalignant oral leukoplakia (OL) with predominant expression in malignant and premalignant tissues. In the evaluated patients, we found that PIWIL2 was associated significantly with OSCC prognosis and OL. Furthermore, PIWIL2 was found to be expressed in tumor epithelial cells and macrophages in the tumor microenvironment, which are not derived from enlarged lymph nodes. Cytological experiments confirmed that the human squamous cell carcinoma cell line SCC-25, can promote the PIWIL2 and Nanog level in THP-1 cells, which are extensively used to study the modulation of monocytes and macrophages. Our findings showed that PIWIL2 can predict effectively OSCC prognosis and OL with a high risk of OSCC development and substantiate the deduction that cancer stem(-like) cells in oral cancer have the ability to reconstitute the heterogeneity of the bulk tumor and contribute to poor outcome and immunosuppression.
Introduction
Worldwide, 405,000 new cases of oral cancer are diagnosed each year, 90% of which are squamous cell carcinomas and causes significant morbidity and mortality [1]. Oral leukoplakia (OL) is the best-known potentially malignant disorder of oral squamous-cell carcinoma (OSCC), and generally is thought with a frequency of malignant transformation between 11.6% and 35% [2, 3]. Pathological assessment of epithelial dysplasia is currently an effective mean for identifying the risk of OL transformation; however, this histologic classification is insufficient and subjective [4]. Furthermore, an appropriate treatment for oral leukoplakia is more required, which will enable successful differentiation between surgical and observation cases.
Multiple observations support that the initiation, progression, recurrence and metastasis of OSCC are related to the behavior of a small subpopulation of cancer stem cells (CSCs) [5, 6, 7]. Shrivastava et al. found that the CSC markers CD133, NANOG, and NOTCH1, which were identified in head and neck squamous cell carcinoma cells, have been shown to increase invasion, angiogenesis, and proliferation and to accelerate disease progression in head and neck squamous cell carcinoma [8]. Remarkably, Gao et al. have experimentally identified a new type of cancer cell from murine lymphoma [9], representing an early stage of CSC development, but similar to pre-cancer in clinical origin, having the potentials of both benign and malignant differentiation; these cells were named ‘pCSCs’ [10]. It has been suggested that PIWIL2 might be a molecular marker of precancerous stem cells and may play an important role in the regulation of tumorigenesis, such as from OL to OSCC [10, 11, 12].
P-element induced wimpy testis like 2 (PIWIL2), also known as cancer/testis antigen 80 (CT80), is a small RNA-binding protein that plays a key role in germ cell maintenance in the testis and is widely expressed in colon, breast, prostate, gastrointestinal, ovarian, soft tissue, and endometrial cancers, but not in normal somatic cells and stem cells [13, 14, 15, 16].
The underlying mechanism of PIWIL2 in tumorigenesis may differ among various cancer cells. However, little attention has focused on CSCs markers expressed on the cells comprising tumor microenvironment. The purpose of this paper is to study PIWIL2 expression in 137 OL and 144 OSCC samples from patients who received long-term follow-up, and determined their prognostic values for OL patients for subsequent development of OSCC for OL patients. Moreover, we also preliminarily analyze the potential reason and implication of PIWIL2 expression in tumor stroma. CD133, Nanog, and Notch1 were used as reference markers.
Material and methods
Patients and tissue specimens
A cohort of 208 patients with the clinical and pathologic diagnosis of OL between 1998 and 2016 was reviewed at the Department of stomatology, First and the Second Affiliated Hospitals of Harbin Medical University in our previous study. Meanwhile, We enrolled 198 patients who underwent surgical resection at the Department of Oral and Maxillofacial Surgery at the First Affiliated Hospitals of Harbin Medical University between 2005 and 2016. All the OL patients underwent biopsy or surgery, and samples were obtained from formalin-fixed paraffin-embedded tissues. Because of lack of adequate tissues or follow-up data, the 101 patients with OL were reviewed in this study. Also, for the foregoing reasons or death caused by perioperative complications, 137 patients OSCC patients were left for inclusion in this study. None had distant metastases or underwent neoadjuvant therapies, such as radiotherapy or chemotherapy, before surgery. Clinicopathological characteristics are summarized in supplementary Table 1. Periodic follow-up examinations at intervals of every 6 months were recommended for patients with OL [17]. Latency of the progressor was defined as the interval from first biopsy of OL to subsequent OSCC development. Clinicopathological characteristics are summarized in supplementary Table 4. The sites of all lesions were classified as tongue and nontongue. In this retrospective follow-up study, malignant transformation versus non-transformation was considered as the surrogate for clinical outcome of patients with OL. Of the 101 patients, 27 (26.7%) developed OSCC that was confirmed by histopathology at the same sites. All patients provided written informed consent. Procedures were approved by the First Affiliated Hospitals of Harbin Medical University Institutional Research Board and were consistent with the Declaration of Helsinki.
Tissue processing and immunohistochemistry
Four-micrometer sections of formalin-fixed, paraffin-embedded tumor tissue were consecutively cut onto silanized glass slides, and were dewaxed and rehydrated using standard protocols. Antigens were retrieved by incubation in a EDTA buffer (pH 9.0) for 15 min in a pressure cooker (106 KPa) according to manufactures’ instructions. The slides were rinsed with phosphate-buffered saline (PBS), after which they were immersed in 3% hydrogen peroxide solution for 10 min to block endogenous peroxidases. Non-specific antibody binding was blocked by incubation with 5% normal goat or rabbit serum for 20 min in a humidified chamber. The sections were then incubated with a polyclonal anti-CD133 antibody (1:200; catalog no. Bs-4770R), polyclonal anti-Nanog mAb (diluted 1:200; catalog no. Bs-10408R), polyclonal anti-Notch1 mAb (diluted 1:200; catalog no. Bs-1335R), monoclonal anti-CD68 antibody (1:200; catalog no. Bs-33056R) (above all from Biosynthesis Biotechnology, Beijing, China) and polyclonal anti-PIWIL2 mAb (diluted 1:200; catalog no. Ab85084 Abcam) overnight at 4
Evaluation of immunohistochemical variables
All samples were reviewed by two independent investigators who were blinded to the clinical outcomes. Evaluation of immunohistochemical variables were conducted as described previously [18, 19].
Cell line culture
THP-1 cells are extensively used to study the modulation of monocytes and macrophages in which the monocyte state can be differentiated into a macrophage-like phenotype stimulated by phorbol-12-myristate-13-acetate (PMA) [20, 21]. THP-1 cells were cultured in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% FBS, 100 units/ml, and 100 ng/ml streptomycin. The human squamous cell carcinoma cell line SCC-25 was cultured in a medium containing DMEM:Ham’s F12, and 20% fetal calf serum.
Co-culture assay
2
Western blot analysis
THP-1 cells in control and co-culture were washed with ice-cold PBS and lysed by RIPA buffer containing PMSF (Beyotime Biotechnology, Shanghai, China) and phosphatase inhibitors (Beyotime Biotechnology). Whole cell lysates were collected after centrifugation at 14000 rpm for 15 min at 4
Expression of CD133, Nanog, Notch1 and PIWIL2 and cancer-free survival of PIWIL2 in oral squamous cell carcinoma (OSCC). Positive cells with brownish granules are shown in the nuclei or the cytoplasm. CD133, Nanog and PIWIL2 expressed in the tumor epithelial and stroma, but Notch1 almost not in the stroma. Representative 10 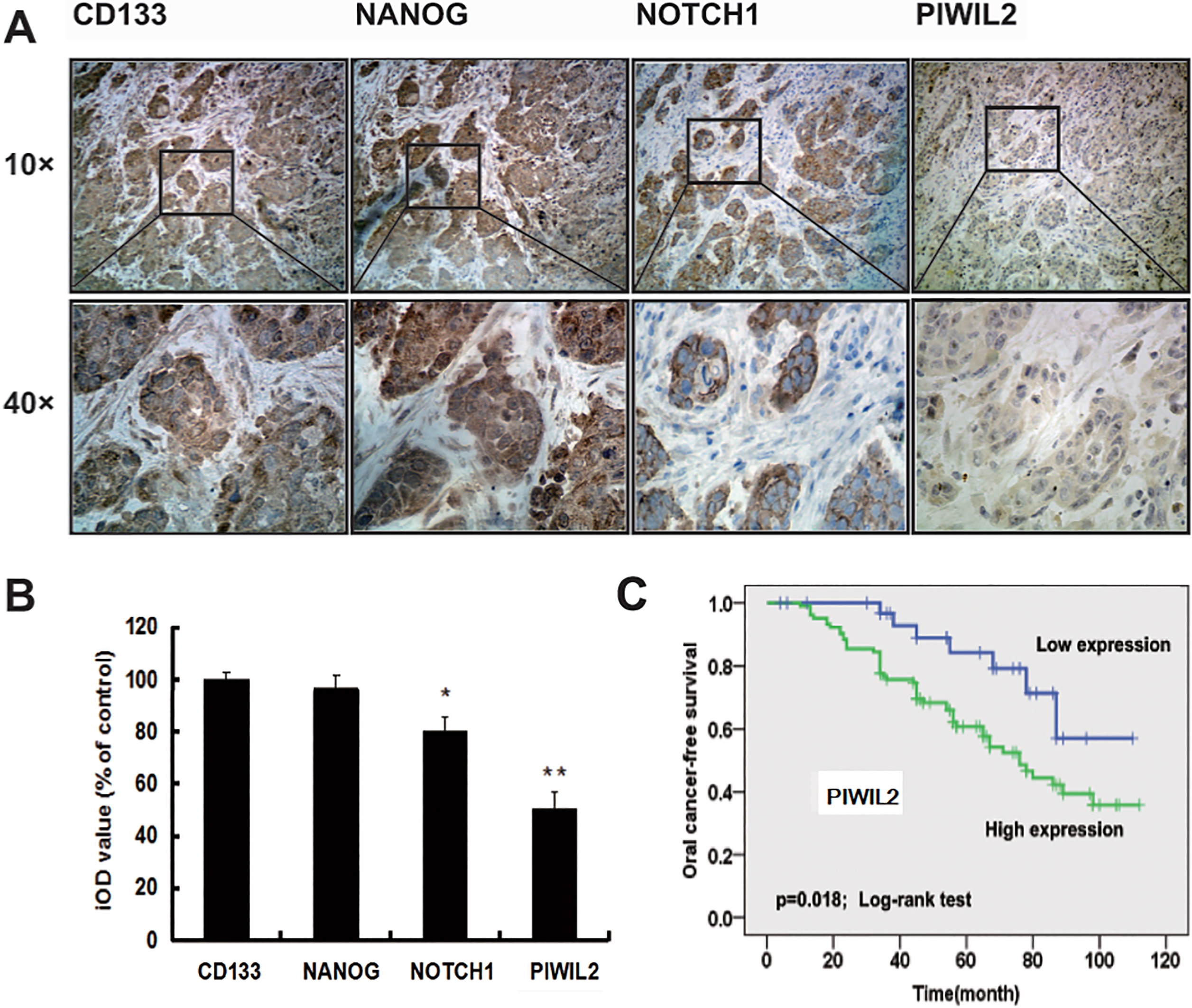
Statistical analysis was carried out with the chi-square test among qualitative variables. Student’s
Results
Expression of PIWIL2 analysis predicts OSCC survival
Previously, it has been shown that human PIWIL2 is expressed specifically in testis and various types of solid tumors, such as breast, liver, uterine cervix cancer [15, 23, 24]. However, there is little information on PIWIL2 in OSCC and prognosis. To confirm this, we analyzed its expression through immunohistostaining on human 137 OSCC sections, and positive expression of PIWIL2 was 75.2%. Specific expression of PIWIL2 in OSCC could be found in island shape cancer tissues and cytoplasm and nuclei had been colored with brown (Fig. 1A). Expression of CD133 and Nanog was similar to that of PIWIL2 (Fig. 1A), however, Notch1 was detected only in cytoplasm (Fig. 1A). Staining intensity of PIWIL2 was lower than those of other three markers (Fig. 1B). PIWIL2 may be identified as independent prognostic factors for oral cancer-free survival (Fig. 1C). Patients data and univariate and multivariate Cox hazard regression analysis in these cases were presented in Tables S1–S3.
Expression and of CD133, Nanog, Notch1 and PIWIL2 and cancer-free survival of PIWIL2 in oral leukoplakia patients some of whom eventually converted into OSCC. Positive cells with brownish granules are shown in the nuclei or the cytoplasm. Representative 10 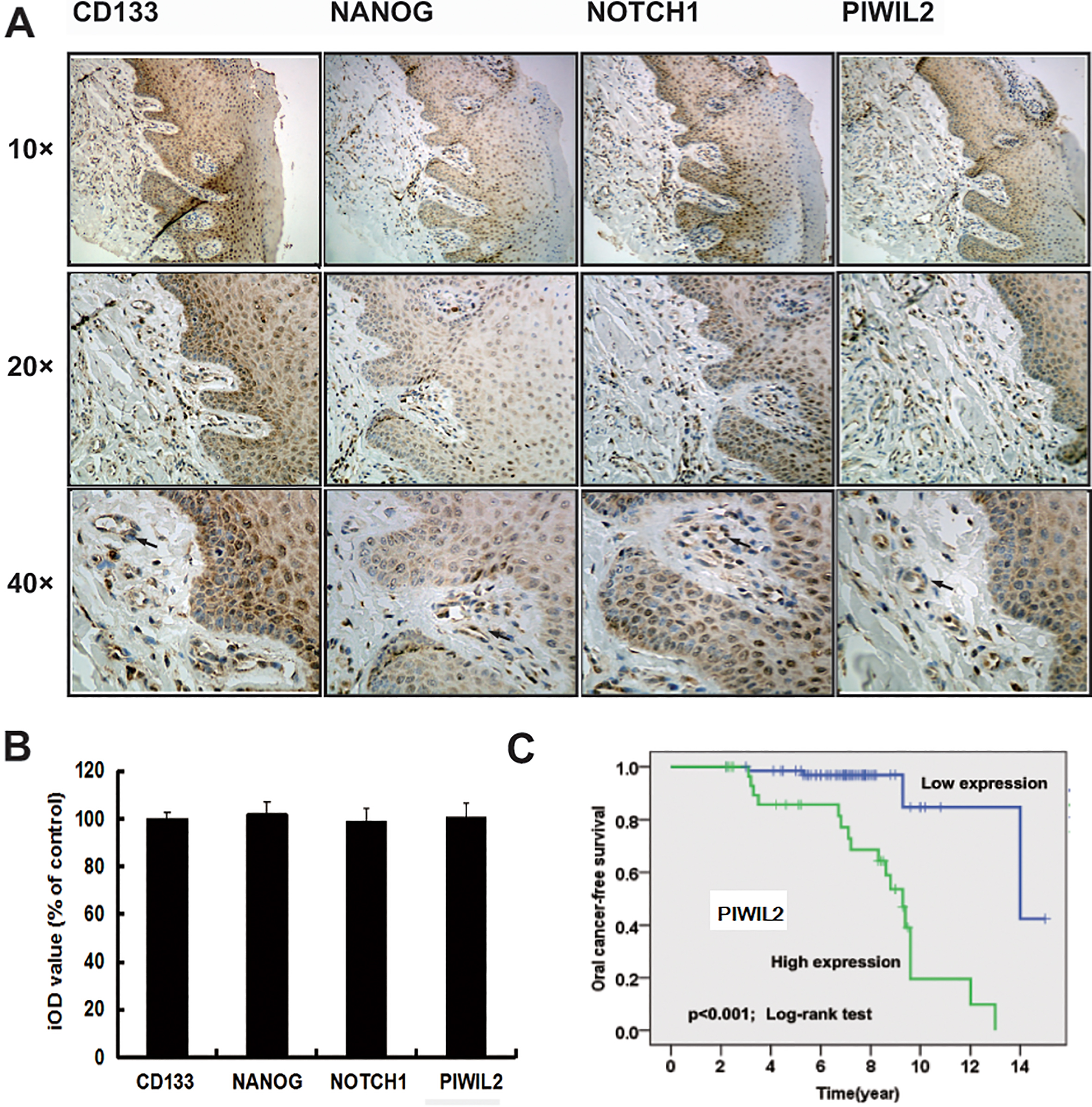
We next analyzed whether PIWIL2 can predict OSCC risk in OL patients. One hundred and one patients with OL were included in this retrospective follow-up study; twenty-seven cases were ultimately, with malignant-transformed tumors after several year latency. The characteristics and PIWIL2 expression patterns of these patients with OL is summarized in Table S4. Oral epithelial dysplasia and expression of PIWIL2 were associated significantly with OSCC risk (Table S5). Positive cells of CD133, Nanog, Notch1, and PIWIL2 were apparent in epithelial tissues and lamina propria (Fig. 2A). Staining intensity of the four markers was also similar (Fig. 2B). Expression of PIWIL2 in patients with OL was found to be strongly prognostic of malignant transformation by Kaplan-Meier method (Fig. 2C) Univariate and multivariate analysis revealed that grade of dysplasia and PIWIL2 were significant (Table S6). These data suggest that PIWIL2 might play a role in the early steps of oral cancer tumorigenesis.
Expression of PIWIL2 in tumor epithelial and stroma. PIWIL2 positive cells appeared in tumor epithelial and stroma. Figure 3 (top and left) showed perineural infiltration by tumor cells (arrow). Figure 3 (top and right) showed tumor cell with pyramid or long shuttle or irregular in shape. PIWIL2+cell with light brown cytoplasm also shaped in vascular endothelial-like cell and fibroblast-like structure in Fig. 3 bottom. Certain cells with brown, similar to certain monocytes or granulocyte, are lobate nuclei widely existed in stroma (Fig. 3 bottom arrow).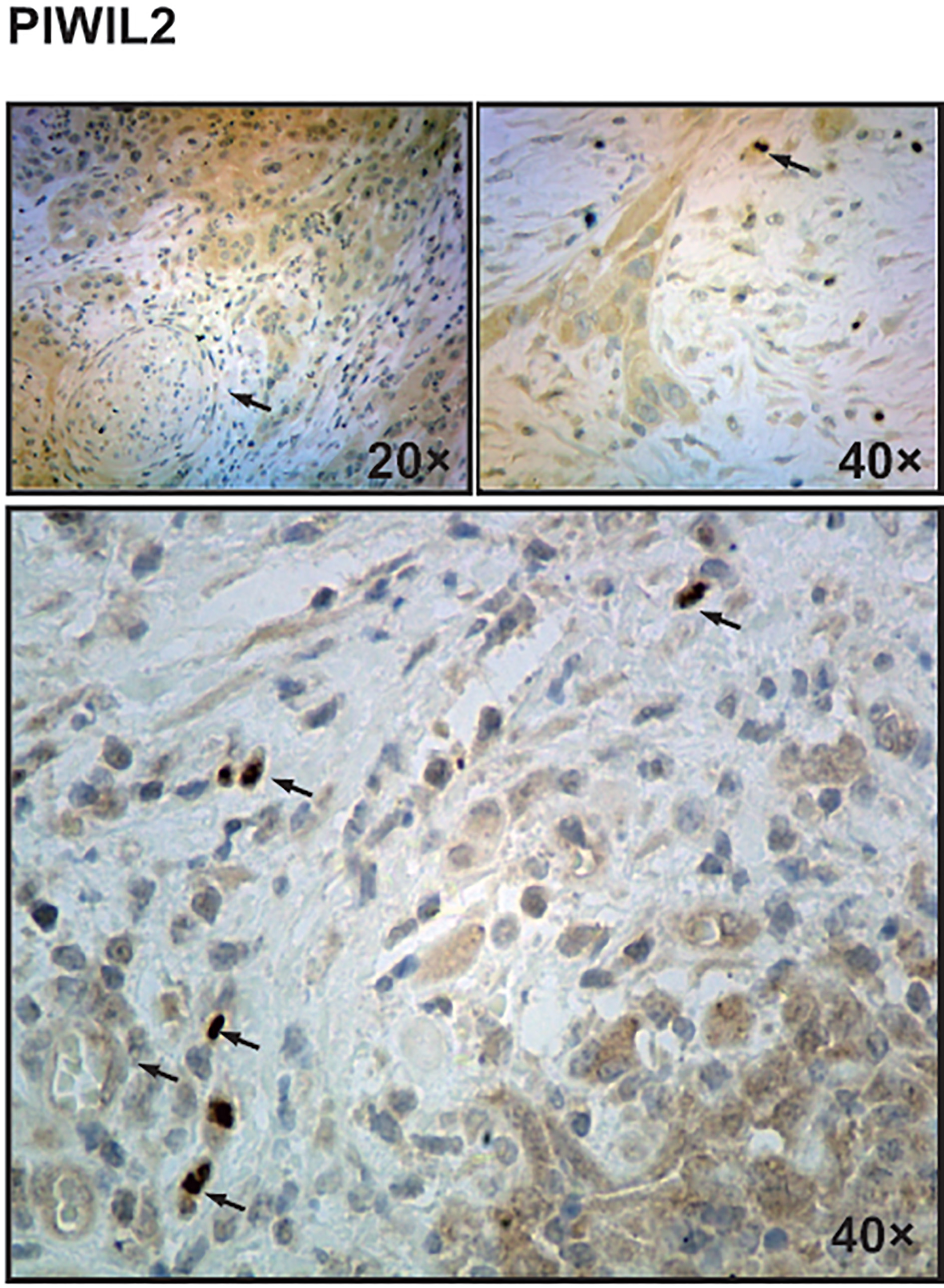
PIWIL2 positive cells appeared in tumor epithelium and stroma. Cells were pyramidal, long shuttle or irregular in shape in invasive OSCC (Fig. 3). It was difficult to distinguish cancer cells and stroma cell morphologically. Light brown PIWIL2 also appeared in vascular endothelial-like cells and fibroblast-like structures (Fig. 3 bottom). Wu et al. found that PIWIL2 is expressed in mesenchymal stem cells [25]. Possibly, PIWIL2 is expressed in vascular endothelial cells or fibroblasts, as mesenchymal stem cells can transform into both of these cell types; Of interest, some immune cells in stimulitumor microenvironment were also stained by PIWIL2 (Fig. 3 bottom). Certainly, CD133 and Nanog were also similar to PIWIL2 (Fig. 1A), however, no Notch1 expression was apparent in stroma tissue (Fig. 1A).
Enlarged lymph nodes are not the source of PIWIL2 positive immune cells
To confirm the source of positive PIWIL2 immune cell, we examined the expression of PIWIL2 in enlarged lymph nodes of OSCC including with lymph nodes metastasis (65 cases) and without lymph nodes metastasis (10 cases) using immunostaining analysis that represented one branch of immune cells. We found that CD133+cells and Nanog+ cells mainly appeared in the lymph sinus and rarely in lymphoid nodules (Fig. 4). Notch1 widely and mildly located in lymph nodule, but less so in lymph sinus. Interestingly, PIWIL2 did not appear in the lymph node. The foregoing results appeared in 100% of cases (75 cases). According to the experimental results 3 and above, it was assumed that PIWIL2 positive immune cells expression may be associated with tumor microenvironment. To confirm this deduction, First, we examined the expression of PIWIL2 in human hypersinia in OSCC tissue and OL using immunostaining analysis that represented benign, in situ, and invasive tumor microenvironment. We found PIWIL2 expression located in hypersinia epithelial and stroma in OSCC, but less so in simple hyperplasia in OL (Fig. 5). This indicated tumor microenviornment might possess the capacity to shape and maintain the CSCs traits, compared with the generally inflammatory state. Second, we would find out whether there are PIWIL2 positive immune cells in tumor microenvironment.
Enlarged lymph nodes is not the source of PIWIL2 positive immune cells. CD133+cells and Nanog+ cells mainly appeared in the lymph sinus and rarely in lymphoid nodules. Notch1 widely and mildly located in lymph nodule, but less so in lymph sinus. PIWIL2 did not appear in the lymph node. Representative 10 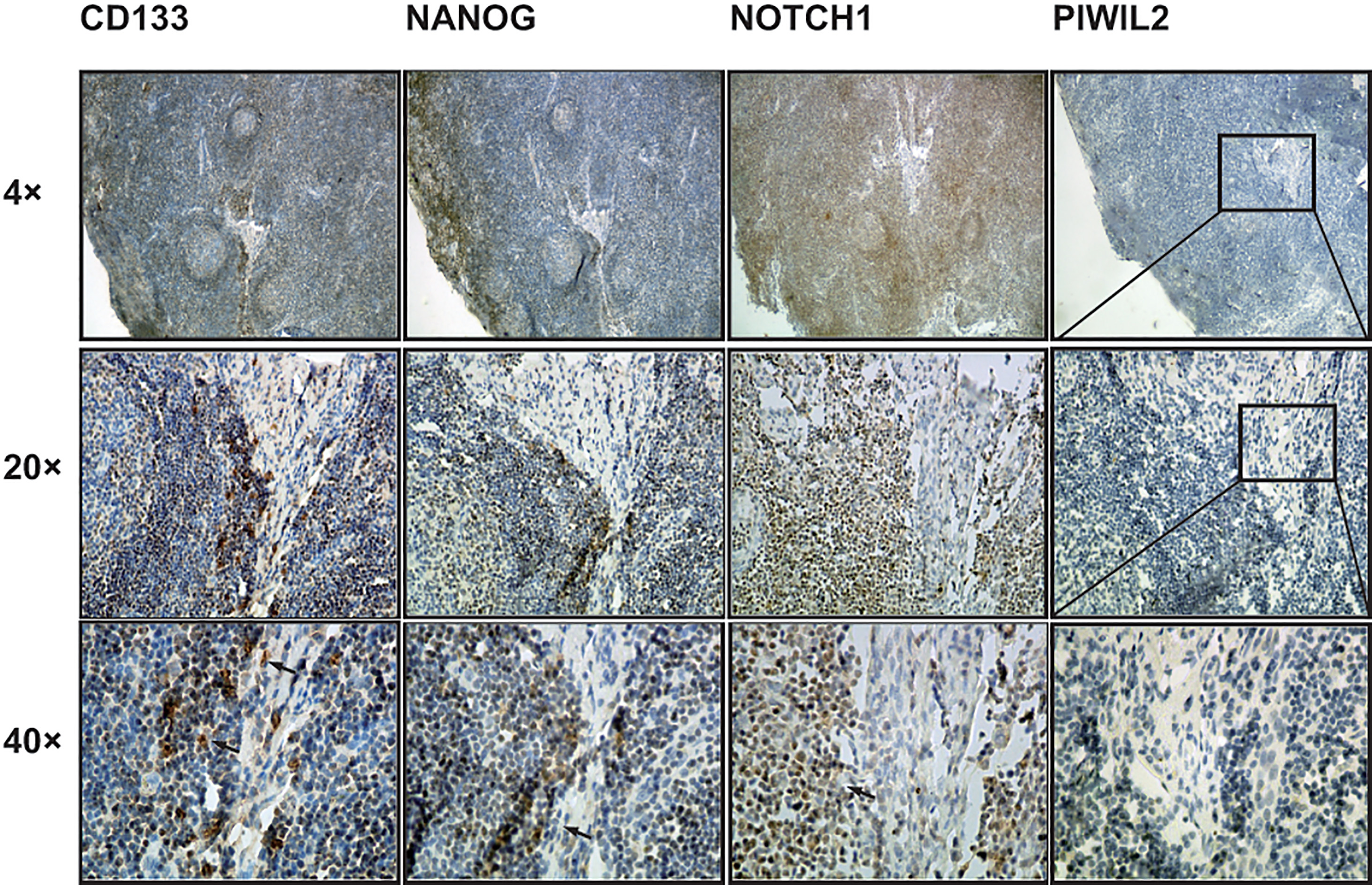
Expression of PIWIL2 in oral hyperphlasia of OSCC and OL. Hyperplasia is a common preneoplastic response to stimulus. Microscopically, cells resemble normal cells but are increased in numbers. PIWIL2 positive expression appeared in hyperphlasia of oral cancer but did not in hyperphlasia of OL. Representative 4 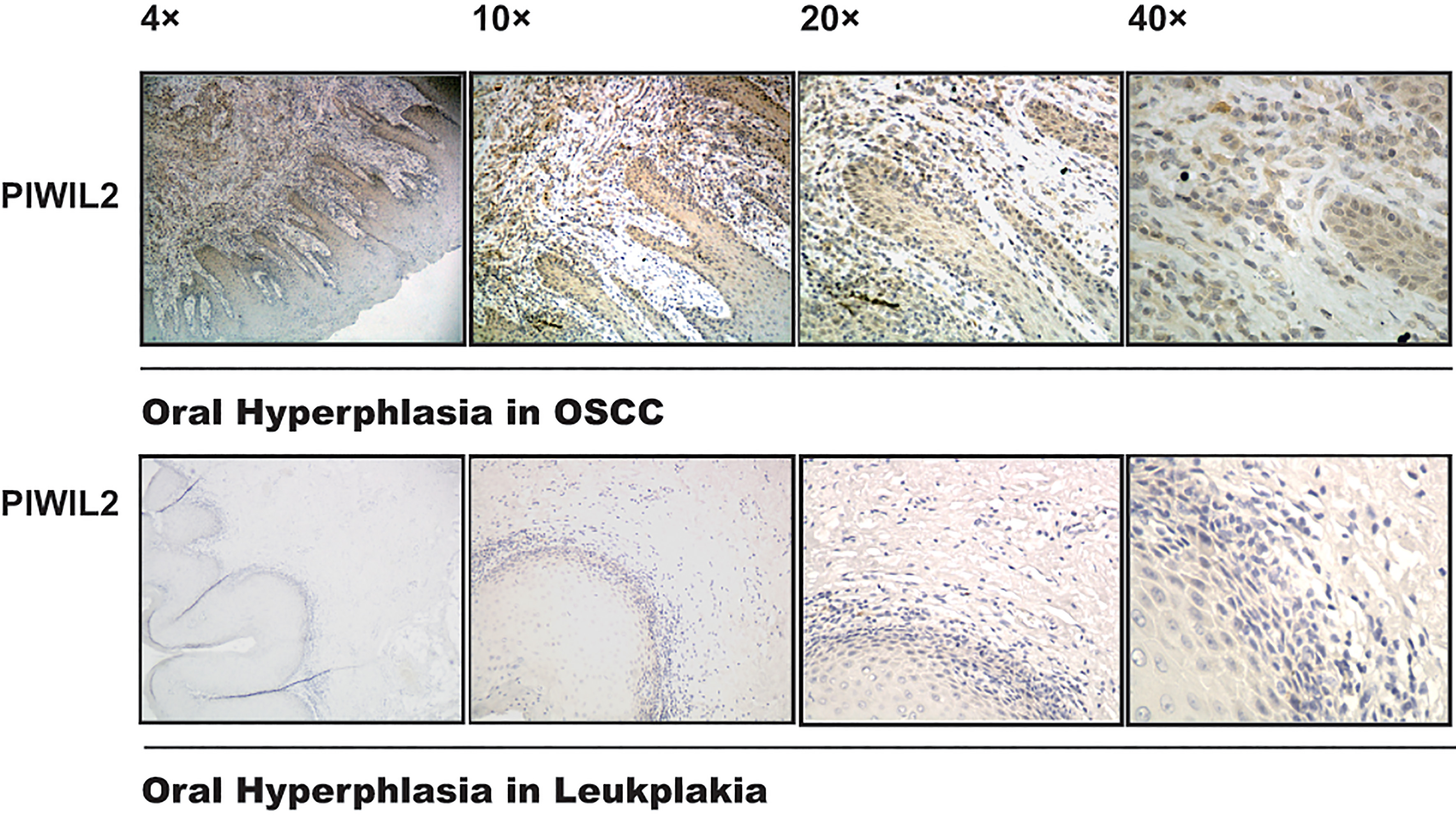
Macrophages are a major component of the leukocyte infiltrate of tumors [26, 27]. Tumor-associated macrophages (TAMs) have complex dual functions in their interaction with neoplastic cells, and evidence suggests that they are part of inflammatory circuits that promote tumor progression [26, 27]. CD68 has been used widely as a means of identifying cells of monocyte/macrophage lineage in normal and pathologic conditions and as a TAMs marker [28]. To confirm that the PIWIL2 positive cells in stroma is a source of TAMs, we examined the expression of PIWIL2 and CD68 in advanced OSCC tissues of clinical stage III
Expression of PIWIL2 and CD68 in tumor epithelial and stroma. PIWIL2 (brown) and CD68 (red) double positive cells appeared in tumor epithelial and stroma. Figure 6 (left) showed double positive macrophages in tumor stroma (arrow). Figure 6 (right) showed double positive macrophages in tumor block mass (arrow). Representative 10 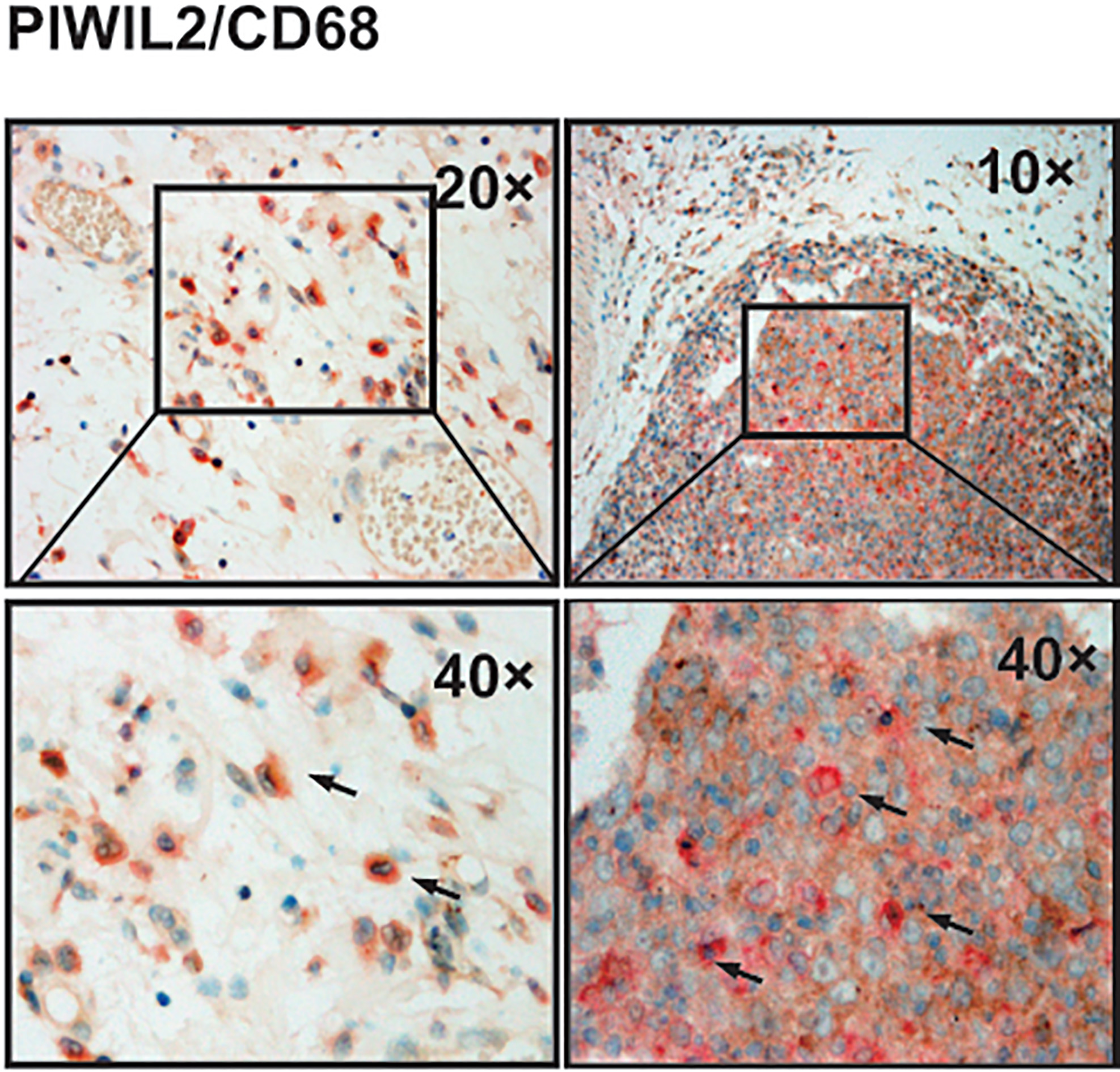
To confirm that tumor microenvironment can induce PIWIL2 expression in macrophages, we set up a co-culture between human squamous cell carcinoma cell line SCC-25 and THP-1 cells, which are extensively used to study the modulation of monocytes and macrophages [29], and examined the expression of PIWIL2 in THP-1 cells using by western blotting. We found that After 6 d of SCC-25 co-culture, THP-1 cells were subjected to expressed PIWIL2 and Nanog CSCs markers. As shown in Fig. 7A, however, CD133 and Notch1 had no significant changes (Fig. 7B).
SCC-25 cells influence CSCs markers on THP-1 cells. A showed higher PIWIL2 and Nanog protein level in THP-1 cells in co-cultured with SCC-25. B showed no change of CD133 and Notch1 protein level in THP-1 cells between control and co-cultured with SCC-25.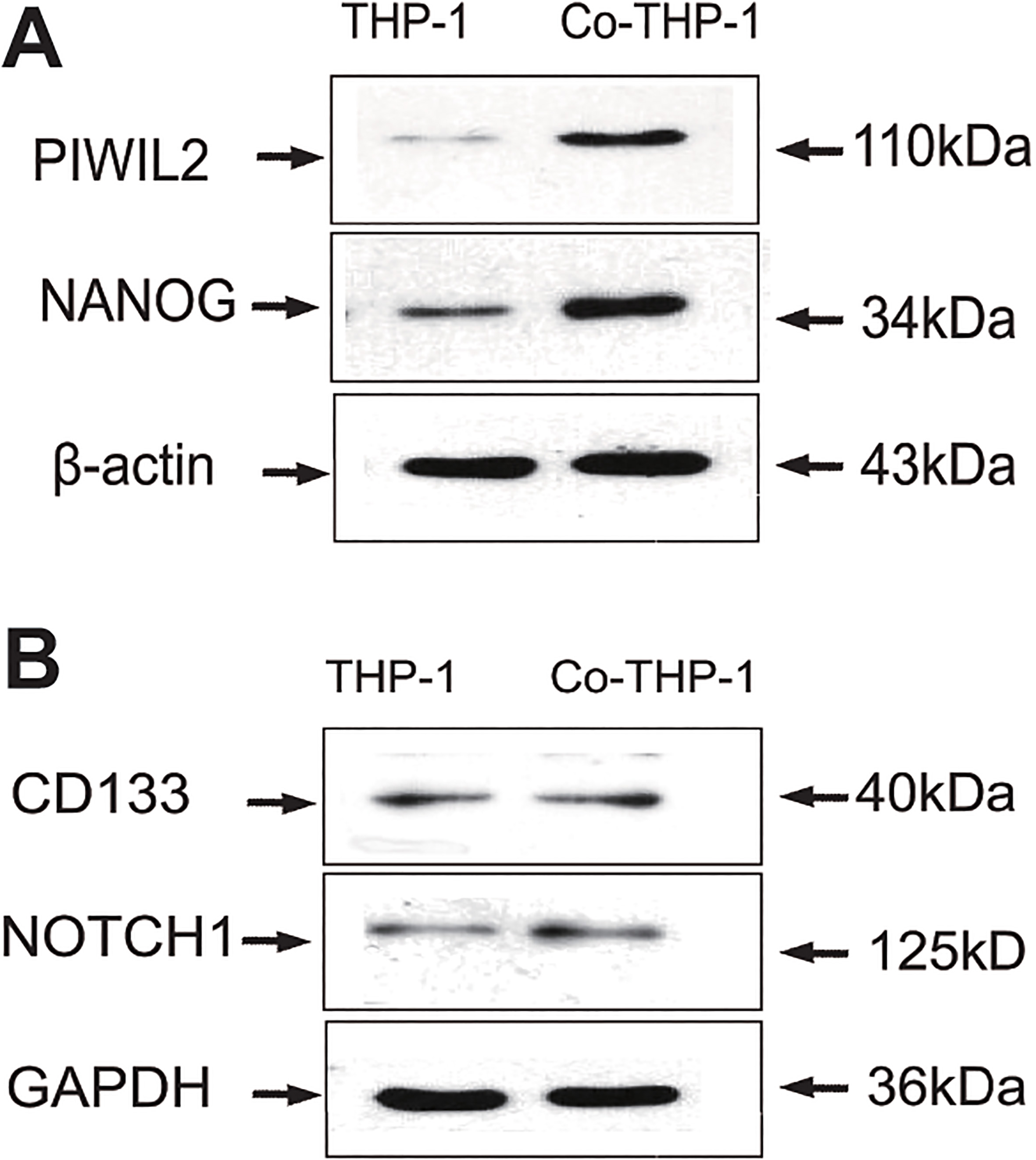
In a previous study, we reported that CD133, Nanog, and Notch1, as CSCs markers, can predict the prognosis for high-risk patients with occult metastases from early stage OSCC [30]. We now extend these findings and demonstrate that PIWIL2, a novel PCSCs and CSCs marker is a predictive biomarker for oral cancer free survival in OSCC and risk of malignant transformation of OL (Fig. 2). Retrospective clinical studies have previously revealed that the PIWIL2 expression correlates with increased lymph node metastasis, histological type, and clinical stage in various solid malignant tumors [15, 22, 23, 24]. In this article, we revealed that not only does increased PIWIL2+cells density correlate with poor outcome and risk of malignant transformation of OL (Figs 1 and 2), but also, PIWIL2+cells only occur in cancer and precancer-associated stroma (Fig. 3), which is an important finding when considering that PIWIL2+ cells did not appear in hyperphlasia of OL without risk of malignant transformation (Fig. 5). Moreover, we also found that it was difficult to distinguish cancer cells and stromal cells morphologically due to the similar shape in invasive OSCC. These findings indicate that PIWIL2 may be a useful predictor of recurrence and poor outcome for OSCC, and, may serve as predictor to identify OL with a high risk of oral cancer development.
One hypothesis for the expression of PIWIL2+ cells in invasive OSCC stroma. PIWIL2+ cells in stroma might be considered tumor associated fibroblast, immune cells, such as macrophages, immature dendritic cells, or tumor neovascularity. Supportive of this hypothesis was light brown PIWIL2 appeared in vascular endothelial-like cell and fibroblast-like in stroma (Fig. 3 bottom). Furthermore, to confirm that some immune cells in stroma were also stained by PIWIL2 (Fig. 3 bottom), we examined the expression of PIWIL2 and CD68 in advanced OSCC using double immunohistochemical staining. Co-culture results also showed that cancer cells can activate PIWIL2 and other CSCs marker, such as Nanog, in THP1 cells. These results showed that PIWIL2+ macrophages might appear in tumor epithelial and stroma. Of interest, these PIWIL2+ macrophages were not from regional lymph nodes. This suggests that the tumor microenvironment may be involved in stroma cells differentiation. This hypothesis could be supported to some level by the following two conclusions. Chen and colleagues revealed that PIWIL2 regulates precancerous stem cells based on environmental factors [10], and Shen et al. found that precancerous stem cells could serve as tumor vasculogenic progenitors [31].
Most (67/70 cases), but not all, invasive carcinoma samples express PIWIL2/CD68, and this may represent a specific mechanism of disease progression in a discrete cohort. Here, we want to usher in a conception to explain above results. CSCs, a heterogeneous population, can be distinguished in predominantly non-cycling CSCs and cycling CSCs [32, 33, 34]. Cycling subpopulation accelerates tumor growth, whereas the non-cycling promotes invasion promotes migration and metastasis [34, 35, 36]. Cycling sub-population has a predominantly epithelial phenotype in contrast to the predominantly mesenchymal phenotype of the non-cycling subpopulation [34, 37]. Marcucci et al. proposed that CSCs develop into either phenotype depending on stimuli from the tumor microenvironment [38], and CSCs with predominantly epithelial phenotype rely mainly on oxygen metabolism for energy production, while several others show that CSCs rely on glycolysis for energy production [39]. Therefore, the presence of heterogeneous traits [38] suggests that PIWIL2 expression in invasive samples has different outcomes from PIWIL2 expression in early stage samples. PIWIL2+/CD68+ cells mainly appeared in invasive samples, suggesting that hypoxia, or low oxygen tension, may be a major regulator of PIWIL2 expressed in macrophages.
Considerable interest in the cancer field is focused on the specific characteristics of the tumor microenvironment and how these phenomena depend on intercellular communication of malignant and nonmalignant cells of the host. It is a worthy question to explore why TAMs can express PIWIL2. After all, past research suggested that PIWIL2 is not expressed in normal tissues, including peripheral blood, and lymphocytes [15]. Possibly, this is due to various signal transducion pathways converging at the level of transcriptional activation to produce specific patterns of gene expression in response to environmental stimuli. Alternatively, exosomes, which can carry PIWIL2 biological information consisting of mRNAs, miRNAs, as well as soluble and transmembrane proteins between cancer cells and components in stroma might be responsible for this phenomenon. The next step of our research will focus on the function of PIWIL2 in macrophages.
High sensitivity and high specificity are essential criteria for a bio-marker to be used for detection of cancer. Although some non-invasive methods, such as saliva testing, brush biopsy and DNA diploidy analysis, have appeared. scalpel biopsy is currently essential method in identification of early cancers. Scalpel biopsy depends heavily on the experiences of pathologists in the differential diagnosis of dysplasia and early oral cancer. PIWIL2, as a novel CSCs marker, is expressed predominantly in OSCC and OL of risk malignant transformation influencing their outcome, suggesting that PIWIL2 as a biomarker is more sensitive and specific than other known OSCC bio-markers. It would improve the predictive power of the assays if PIWIL2 was used to detect the tissue of complicated lesions by scalpel biopsy.
In conclusion, our results show that PIWIL2, as a novel CSCs marker, is expressed predominantly in OSCC and OL with risk of malignant transformation influencing their outcome. PIWIL2 expression in stroma, especially on macrophages, has potential research value and would be useful for application in diagnostic and therapeutic purposes.
Footnotes
Acknowledgments
This work was supported by grants from Foundation of First Affiliated Hospital of Harbin Medical University (No: 2019M20), Health Commission of Heilongjiang Province (No: 2018035), Natural Science Foundation of Heilongjiana Province of China (No. YQ2019H020) and Chinese Postdoctoral Science Foundation (No: 2018M641872).
Conflict of interest
The authors declare that they have no competing interests.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-182009.
