Abstract
Photodynamic therapy (PDT), defined as “the light-induced inactivation of cells, microorganisms, or molecules,” combines the use of a photosensitive agent or photosensitizer (PS) activated by irradiation with a light source to produce reactive oxygen species and highly reactive singlet oxygen. PDT has been used for the treatment of different oral diseases with promising results. This review describes the basic principles of PDT, including the nature of PS and the light sources, and focuses on evaluating the efficacy of this method, according to the existing data, for the treatment of different oral diseases. PubMed was used as the source of the relevant literature, about the clinical application of PDT in actinic cheilitis (AC), leukoplakia, oral lichen planus, oral lichenoid lesions, and oral cancer. Complete response to PDT as high as 100% has been reported not only in cases of AC but also in dysplasias/carcinomas in situ, T1N0, and T2N0. According to the available data, PDT appears to be a safe, well-tolerated therapy with limited adverse effects and excellent cosmetic outcome. Although PDT is a very promising therapy, further research is needed in order to investigate the cases that fail to respond, to examine the long-term prognosis for cases of reported complete response and thus avoid the future recurrence of diseases.
Introduction
Photodynamic therapy (PDT) is a type of treatment defined as “the light induced inactivation of cells, microorganisms, or molecules,” which is characterized by rapid development in the past decades. 1 The term photodynamic was coined by Jodlbaner and Von Tappeiner in 1904 for the description of oxygen-dependent chemical reactions as a result from photosensitization that could cause bacterial deactivation. 2 In 1999, US Food and Drug Administration approved for the first time the PDT for the treatment of precancerous skin lesions on the face or the scalp. 2 PDT includes the use of a photosensitive agent or photosensitizer (PS) and a specific wavelength light source. 3 The PSs that are used in PDT are chemical compounds which after absorption of light are capable of being promoted to an excited state. 3 The PS is either applied topically or through intravenous injection, which depends on its type, and concentrates mainly in abnormal or neoplastic cells. 4
The activation of PS from its initial state to a highly energized triplet one occurs after it has been irradiated with a specific wavelength of light. 5 This highly energized triplet state has a longer lifetime, and as a result, the excited PS can interact with the surrounding molecules, and it is believed that in this state, species that are toxic for the cells are being produced. 5 After a photon of light is absorbed, a molecule of the photosensitive agent is excited to singlet state (S*) from its former state (S) through absorption of the photon energy. 6 This molecule can either return to its initial state through emission of a photon as light energy (fluorescence) or turn into heat, or can be promoted to the triplet excited state, by the determinant factors such as the molecular structure of the PS and the environment. 6,7 Molecules in the triplet excited state are capable of emitting light (phosphorescence) by returning to their initial state or by further reaction through one or both of two pathways (known as type I and II process) where oxygen is necessary. 6 The PS in this state reacts with the biomolecules with two different pathways (type I and II). 5 In the type I reaction, there is production of ions through direct transfer of electron/hydrogen from the photosensitive agent, or removal of electron/hydrogen from a substance molecules for the production of free radicals. There is a rapid reaction between free radicals and oxygen thus generating highly reactive oxygen species (superoxide, hydroxyl radicals, and hydrogen peroxide). 2 These species can harm decisive cell components: for example, structural proteins, enzymes, DNA, or phospholipids. 8
PDT causes tumor destruction through three main mechanisms. The first involves direct destruction of tumor cells, the second involves damaging of the tumor vasculature, causing formation of thrombus and subsequent tumor infarction, while the third mechanism is characterized by the activation of an immune response against the cancer cells. 9 In the type II reaction, the PS, in its triplet state, reacts with oxygen to create singlet oxygen (highly reactive oxygen state) that is capable of interaction with various biological molecules due to its high chemical reactivity. 5 Cytotoxic effects are being produced on subcellular organelles and molecules (including mitochondria, lysosomes, cell membranes, and tumor cells’ nuclei), and the PS promotes not only apoptosis in mitochondria but also necrosis in lysosomes and cell membranes. 2
Porphyrins are a group of natural macrocyclic compounds which are ubiquitous in nature and are decisive for the metabolism of living organisms as they participate in various procedures such as oxygen transport, electron transfer, as well as oxidation catalysts and photosynthesis. 10,11 Porphyrins are pigments with crucial roles in nature, known as “the pigments of life.” 12 Heme is an iron-containing porphyrin, which is a cofactor in various cellular processes. 13
Porphyrins, chlorins, bacteriochlorins, and phthalocyanines are widely used as PS. They consist of one tetrapyrrole ring structure (porphin) and its derivatives (porphyrins). 14 Porphyrins such as chlorin e6 and hematoporphyrin derivatives concentrated mainly along the plasma membrane and the intracellular sites especially the nucleus are far more sensitive to photodynamic damage. 15
Types of PS
Various types of PS are (1) dyes, (2) chlorines, (3) porphyrines, (4) xanthenes, and (5) monoterpene. 2 Commonly used PS are as follows.
5-Aminolevulinic acid
5-Aminolevulinic acid (ALA) is a metabolite of the heme biosynthesis pathway, which is associated with active absorption and concentration mainly in tumor cells. 16 This substance is the biological precursor of the PS, which is protoporphyrin IX (PpIX; activation possible by red, green, and blue light), 17,18 in the heme biosynthesis pathway. 9 Within 48 h, this substance is removed both from the tissue and the body with the skin photosensitivity lasting for less than 1 day. 19 PpIX absorbs light at a wavelength of 400–700 nm (visible range) and has 10-nm-wide absorption peaks (at a wavelength of 410, 500, 532, 585, and 635 nm), while by high-energy external light illumination, at a wavelength of the above absorption peaks, PpIX can produce singlet oxygen and other free radicals. 16 ALA (Levulan®, DUSA Pharmaceuticals, Inc., Wilmington, Massachusetts, USA) is used in cases of actinic keratosis (AK) of the face or the scalp and is approved in the United States at a wavelength of 632 nm for topical use. 6
Methyl 5-aminolevulinate
Methyl 5-aminolevulinate (MAL) is an esterified derivative of ALA. 3 This PS is characterized by lipophilicity and is more selective than ALA for specific cells, resulting in higher phototoxicity. 3 MAL is an effective PS, which demonstrates deep penetration into the lesion due to its increased lipophilicity. 20 MAL (Metvix, Galderma, Switzerland) is approved for use in cases of AK, basal cell carcinoma (for topical use in the European Union, Australia, New Zealand, Brazil), and AK (for topical use in the United States) at a wavelength of 632 nm. 6
Methylene blue
Methylene blue (MB) is a phenothiazine dye, whose administration in high doses orally or through intravenous injection in humans is not associated with any side effects. 21 This PS, a redox indicator, demonstrates a blue color in an oxidizing environment, while it turns colorless upon reduction. 5 MB has the advantage that can be administered either topically or orally, and it could be used for superficial lesions in skin and oral cavity. 22 MB demonstrates natural antifungal and antibacterial properties, which can be increased through light activation. 3 It shows better absorption at wavelengths longer than 620 nm. 23
Photolon
Photolon is a new chlorin e6-derived PS, which has been used with success for the treatment of precancerous lesions. 24 Better therapeutic results are obtained by the use of light at longer wavelengths in cases of deeper localized lesions with no increase of phototoxicity. 25 Photolon consists of chlorine e6 (96.5% purity) and polyvinylpyrrolidone at a ratio of 1:1. 25 A study by Ali-Seyed et al. 15 found that photolon preferentially localized in subcellular organelles such as nucleus, mitochondria, lysosomes, and, finally, the Golgi apparatus. The subsequent photodamage of the mitochondria and lysosomes was found to be crucial in mouse colon cells (CT-26) apoptosis, which is mediated by PDT. 15 Photolon® (Rue Belmedpreparaty, Republic of Belarus) is used at a wavelength of 660–670 nm. 26
Toluidine blue
Toluidine blue, a cationic PS, is a dye, which demonstrates maximum absorbance at a wavelength of 630 nm.
3,27
This PS stains granules within mast cells, proteoglycans, and glycosaminoglycans within connective tissues and has been used for the detection of tissues with dysplasia.
5
It is a PS active against cell membrane, which absorbs red laser light and has bactericidal properties against various species including
Types of light source used in topical PDT

Lasers in PDT (modified from Brancaleon and Moseley 30 ). PDT: photodynamic therapy.

Lamps in PDT (Modified from Brancaleon and Moseley 30 ). PDT: photodynamic therapy.
Light protection guidelines
Gradual exposure to light over a period of approximately 2 weeks is necessary to avoid phototoxic side effects after PDT, in which the PS was administered systemically (intravenously). 31 –35
Search strategy
PubMed was used as the source of the corresponding literature by combining the following keywords: PDT, actinic cheilitis (AC), leukoplakia, oral lichen planus (OLP), oral lichenoid lesions, and oral cancer. The search was completed at the beginning of June 2016 and from the initial 514 articles according to the following inclusion criteria, a total of 42 articles were further assessed.
Inclusion criteria
The inclusion criteria include: clinical studies, case reports, original articles, articles in English language or with an English version available, articles with a full text available, and adequate information about the PS and the light source used.
We also included three letters to the editor. 36 –38 Following are the reasons for the inclusion of these articles: first, in all of these case reports, the initial diagnosis as well as the complete response was evaluated not only clinically but also histologically; second, adequate information about the PS and the light source used was reported; and third, the last case report 38 is the first case report in which daylight-activated PDT (DA-PDT) was applied successfully in AC.
An exception to the previous criteria is also the review of Biel 4 as it provides information for the photodynamic treatment outcome in 161 cases of oral cancer treated by the author. Finally, we further excluded duplicate studies.
Clinical application of PDT in oral potentially malignant disorders and oral cancer
PDT consists of two stages. The first stage includes the delivery and the concentration of the photosensitizing agent in the cells, which require treatment (cells that divide rapidly), and the second stage includes the exposure of these cells to a specific wavelength light source which complies with the absorption peak of the PS. 3
Actinic cheilitis
AC is a chronic premalignant condition caused by the protracted sun exposure, which affects especially the vermilion border of the lower lip as it is more exposed to the sunlight compared to the upper lip. 38,39 The fair-skinned (especially Caucasian) individuals are mainly affected and also the outdoor workers. 39 The clinical features of AC lesions include dryness, scaliness, and color variation on the lips. 39 Other characteristics of these lesions include erythema and edema, progressing with atrophy, hyperkeratosis, erosions, or ulcerations as well as infiltration and blurring. 40,41 AC is associated also with commonly asymptomatic white, red, or white with red areas localized or diffused lesions, from which the whitish ones are mainly hyperkeratotic lesions and the red erosive or atrophic lesions imply cheilitis. 42 The main histological criteria include atypia loss of the keratinocytes’ polarity of the lower portion of the epithelium, solar elastosis, and inflammatory infiltrate. 40 In a study by Markopoulos et al. 42 on 65 patients with AC, hyperkeratosis was found in 13.8% of the patients, mild epithelial dysplasia in 27.7%, and moderate dysplasia to invasive squamous cell carcinoma (SCC) in 72.3%. 42 Without treatment, a malignant transformation of the AC lesions into SCC is possible. 38 The malignant transformation rate is reported to be 16.9%, while the estimated time needed for lip cancer development is 20–30 years or less. It is known that SCC of the lower lip resulting from malignant transformation of AC has a metastatic potential four times higher than that of SCC in general, and so it is crucial that AC lesions are being identified early and treated properly. 38,43,44
There are several available treatments for AC; however, each one of them has different advantages and disadvantages. 39 In vermilionectomy, local or general anesthesia is necessary and an unsightly scar is possible. The treatment with 5-fluorouracil or imiquimod requires several weeks and a very intense inflammatory reaction is not uncommon. 45 Cryotherapy and electrocoagulation are proper for localized AC lesions; however, their use in more extensive lesions could cause hypopigmented scars and longer recovery times. 45 In cases of AK according to the international PDT guidelines, PDT is the first-line therapy although it is not a common choice in AC treatment. 20,43 AC as a pathological condition is comparable to AK; however, the use of PDT is not associated with the same satisfactory clinical and histological results as in cases of AK. 43 The PDT outcomes in AC lesions could be enhanced through a previous skin treatment using fractional laser resurfacing due to the increased bioavailability of MAL in the skin and the better response to PDT. 20 PDT in which ALA is used as a PS in combination with a pulsed dye laser has shown good results in patients with resistance to conventional treatment AC lesions as well as PDT with a fractional thulium laser as light source, which provides less scarring and better cosmetic outcome in comparison to conventional treatments. 46,47 During PDT, no local intralesional anesthesia is needed, while a stinging sensation, which is associated with this therapy, is usually restricted to the area which is being treated. 48
MAL-PDT was reported to be more efficient with a lower recurrence rate in comparison to ALA-PDT in AK; however, more studies are necessary. 20 The most important side effects of PDT in AC include mild or moderate erythema, edema, blistering, hemorrhagic crusting, localized pain, and burning sensation during the light exposure. On the other hand, PDT was also well tolerated by the patients in combination with an excellent cosmetic outcome. 47,48 PDT is an ablative procedure, and so, no tissue specimen can be obtained; as a result, no histopathological examination of the lesion is possible. 1,46
DA-PDT is a promising special type of PDT for AK treatment and potentially for AC, in which daylight is used as the light source that activates continuously the PS. 38 The daylight consists of direct and diffuse sunlight outdoors. 49 DA-PDT has been reported to be equally effective and less painful compared to conventional PDT, maybe because of the constant generation and activation of small portions of the PS PpIX. 38,50 The DA-PDT is characterized as a simpler treatment as there is no occlusion and the light source is the daylight in combination with a reduction of the time that the patients have to spend in the clinic. 49 The geographical location (latitude), weather conditions, and time of year are determinant factors for the PpIX light dose which combined with the outdoor temperature suggest the best place and time for a DA-PDT. 50 Although treatment of AK lesions by DA-PDT requires only one treatment session, AC lesions may require repeated sessions as shown in a study by Levi et al., 38 in which two cases of AC required repeated sessions of DA-PDT in order to achieve a complete response. 38 The adverse effects included mild erythema, edema, and crusting, 38,51 while they were reported to be independent not only from the severity of the AC but also from the period during the year in which the PDT took place. 51
Table 1 includes studies for PDT in AC, while Table 2 includes studies in which patients with AC were treated with DA-PDT.
Methodology and results of studies for PDT in AC

PDT: photodynamic therapy; MAL: methyl 5-aminolevulinate; LED: laser-emitted diodes; ALA: 5-aminolevulinic acid; Er: YAG AFL: erbium-doped yttrium aluminum garnet ablative fractional laser; AC: actinic cheilitis.
Methodology and results of studies for DA-PDT in actinic cheilitis.

DA-PDT: daylight-activated photodynamic therapy; MAL: methyl 5-aminolevulinate.
Leukoplakia
Oral leukoplakia (OL) defined, according to the 2005 World Health Organization workshop, as “white plaques of questionable risk having excluded (other) known diseases or disorders that carry no increased risk for cancer” 53 is the most common oral potentially malignant disorder. 54 OL is reported to occur in 0.2–11.7% or 9.7% of population, approximately three times more often in males than in females aged 40–60 years old compared with the general population. 55 From clinical aspect, there are two types of OL: homogeneous and nonhomogeneous. 19 A type of nonhomogeneous leukoplakia, the oral erythroleukoplakia (OEL), presents a higher malignant transformation rate. 17,18,56 Oral proliferative verrucous leukoplakia (OPVL) is one type of OL with multifocal localization and it demonstrates a very high risk of malignant transformation (approximately 70%). 19 Leukoplakia as a precancerous condition is strongly associated with the oral SCC’s (OSCC) pathogenesis. 8 The malignant transformation rate of homogenous thick leukoplakia is 1–7%, while it is mentioned to be 4–15% for granular or verruciform leukoplakia and 18–47% for erythroleukoplakia. 9
There is a variety of available treatments such as traditional surgery, electrosurgery, cryosurgery, laser surgery, and drug administration. 19 An alternative treatment for the management of OL appears to be PDT with the ALA being one particularly significant PS in this treatment. 19,55 Injection and topical application of ALA in patients with OL treated with PDT showed equivalent results. 16 Achievement of complete response in OEL treated with PDT required less treatment sessions when the lesions had the greatest diameter <1.5 cm or their surface keratin layer was ≤30 μm compared to lesions which had the greatest diameter of ≥1.5 cm or their surface keratin layer was >30 μm. 56 PDT in which 20% or 10% ALA was used as PS compared to cryosurgery in OL treatment was associated with less pain after the treatment. However, more reapplications of the PS for a longer period were reported in the PDT, which showed significantly lower side effects in comparison with cryotherapy. 57 ALA-PDT is reported to be more effective in treatment of oral verrucous hyperplasia (OVH) than OL with the OL treated twice a week showing better response rather than the ones treated only once a week. 9 OEL was found to demonstrate lower complete response rate and higher recurrence rate compared to OVH after ALA-PDT. 56 The advantages of ALA PDT in OL treatment include safety, noninvasion, and less disfiguration compared to other common OL treatments. 16 In OL with dysplasia, more ALA is diffused into the lesions due to the presence of a thinner surface keratin layer and a more permeable epithelium (resulting from wide intercellular spaces) and this epithelium compared to hyperplastic one could retain more PS and the thinner keratin layer may contribute to prevention of the light intensity reduction. 58 Also in dysplastic lesions, the number of epithelial cells in the cell division cycle is higher, which are more sensitive to destruction by singlet oxygen molecules and free radicals that are produced during PDT. 56 However, Chen et al. 9 achieved partial response in four of the five OL lesions with dysplasia (two mild, two moderate dysplasias) treated with ALA-PDT and complete response in one OL with moderate dysplasia. 9 In Table 3, the studies for PDT in leukoplakia are reported.
Methodology and results of studies for PDT in leukoplakia.

OL: oral leukoplakia; OVH: oral verrucous hyperplasia; OEL: oral erythroleukoplakia; OPVL: oral proliferative verrucous leukoplakia; OVL: oral verrucous leukoplakia; PDT: photodynamic therapy; ALA: 5-aminolevulinic acid; mTHPC: meta-tetrahydroxyphenylchlorin.
Oral verrucous hyperplasia
OVH is a common precancerous condition of the oral cavity, which is associated with a rather high malignant transformation potential. 56 PDT was found to be an effective treatment of OVH lesions, with complete response and no recurrences in several studies. 9,56,60 Better results of PDT in OVH lesions were found in lesions with smaller size, pink to red color, with or without minor epithelial dysplasia, and a thinner surface keratin layer. 56
Oral lichen planus
OLP is a rather common, chronic dermatologic disease in which the oral mucosa is often affected. 22 The prevalence of OLP among the population is 0.5–2.2%, mainly affecting females two times more frequently than males between 30 and 60 years old. 22
The OLP pathogenesis is associated with a chronic autoimmune damage to basal keratinocytes, which is mediated by T cells, as a result of the recognition of these cells as antigenically foreign or altered. 23 The OLP types include reticular, erosive, atrophic, plaque-like, papular, and bullous. 23 The malignant transformation rate ranges from 0.4% to >5%, while the reported time needed until malignant transformation varies from 6 months to more than 20 years. 21 OLP occurs more often compared with cutaneous lesions and is reported to be more treatment resistant. 27 In the available treatment, options are included local or systemic corticosteroids or other immunomodulators administration and laser therapy. 23,27 The use of corticosteroids for a long time in combination with the fact that OLP is a chronic disease can lead to several adverse effects such as fungal infections, adrenal insufficiency, gastrointestinal disorders, hypertension, and diabetes. 27 MB-PDT was reported to be effective in the management of signs and symptoms of OLP, with limited side effects presenting no statistically significant association between lesions’ location and treatment response. 21 However, in a study by Sobaniec et al., 25 a 55% decrease in the size of OLP lesions was achieved after PDT which was found to have better results in buccal mucosa’s and lips’ lesions than in tongue’s and gingiva’s lesions. 25 Sadaksharam et al. 23 achieved better results in lesions of shorter duration with no statistically significant difference between males and females or different lesion types. 23 Corticosteroids topical administration compared to PDT was found to be more effective and to lead to a greater improvement of pain by treating erosive-atrophic OLP lesions. 27 However, PDT appears to be a promising therapy in cases of OLP without remarkable side effects. 21,23,61 Studies for PDT in OLP are reported in Table 4.
Methodology and results of studies for PDT in oral lichen planusa.
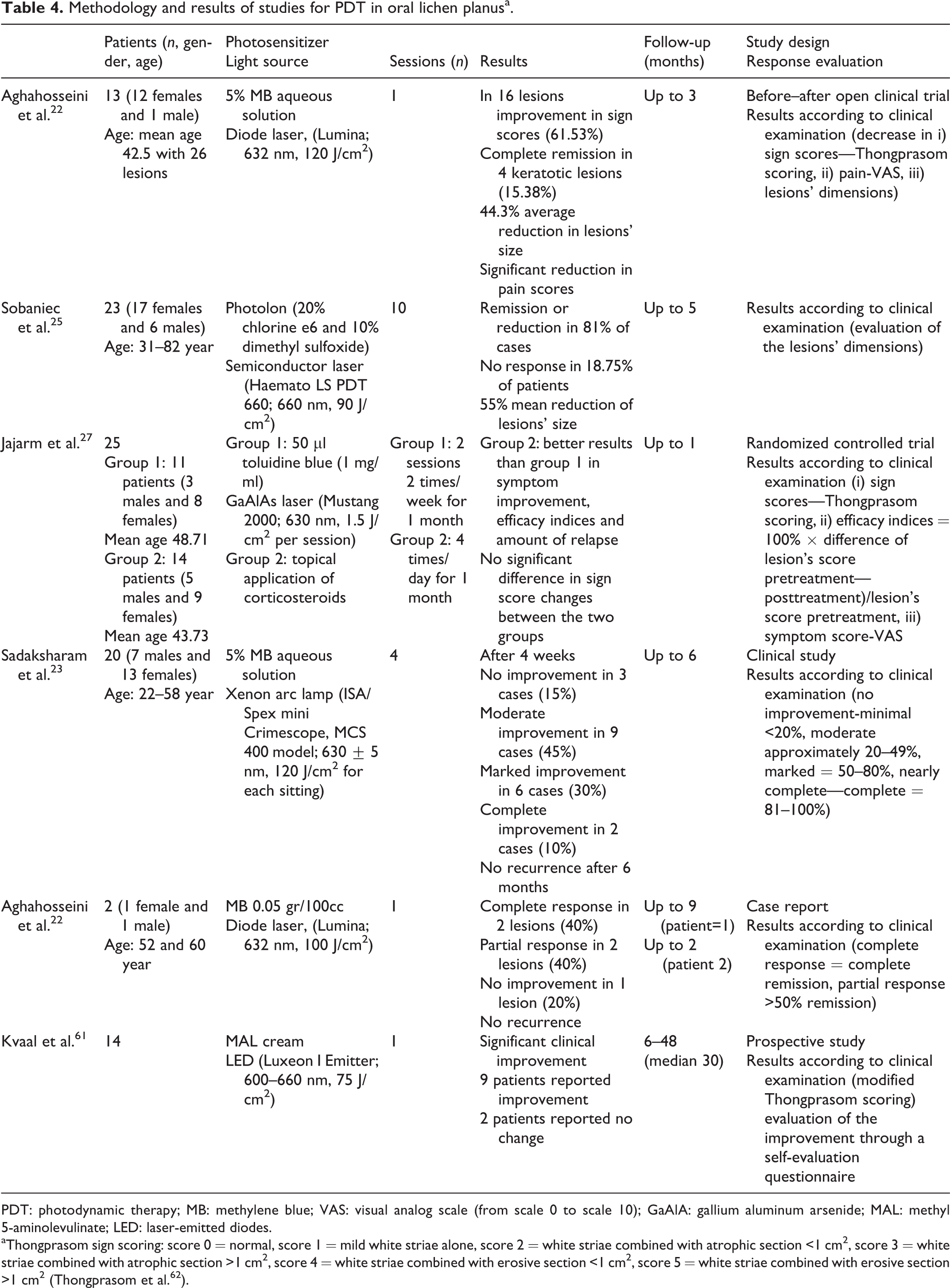
PDT: photodynamic therapy; MB: methylene blue; VAS: visual analog scale (from scale 0 to scale 10); GaAlA: gallium aluminum arsenide; MAL: methyl 5-aminolevulinate; LED: laser-emitted diodes.
aThongprasom sign scoring: score 0 = normal, score 1 = mild white striae alone, score 2 = white striae combined with atrophic section <1 cm2, score 3 = white striae combined with atrophic section >1 cm2, score 4 = white striae combined with erosive section <1 cm2, score 5 = white striae combined with erosive section >1 cm2 (Thongprasom et al. 62 ).
Oral lichenoid lesions
Oral lichenoid lesions are similar to OLP lesions clinically and histologically. 63 They are caused by allergic contact stomatitis due to dental restorative materials, mainly amalgam, or other substances such as cinnamon, various drugs as well as chronic graft-versus-host disease. 64 The treatment includes replacement of the dental restorative materials, modification of the medication, and appropriate management of the chronic graft-versus-host disease. 65 PDT could be of value in cases in which the modification of the medication is not possible as well as in cases persistent to conventional treatments; however, till now, there are no corresponding studies reported.
Oral cancer
The estimated incidence of lip, oral cavity cancer in 2012 was 2.1%, with 1.8% mortality and 2.2% 5-year prevalence (according to Globocan
66
:
Copper et al. 33 achieved complete response in approximately 30% of the patients with multiple primary tumors (T1N0) treated with PDT. 33 Statistically significant difference in local disease-free survival and survival time was found between primary and non-primary neoplasms treated with PDT, with both of these parameters shorter in the case of non-primary neoplasms. 73 However, Ikeda et al. 68 achieved complete response without recurrences during a follow-up period of up to 2 years, in five recurrent lesions (one moderate dysplasia, 2T1N0M0, 2T2N0M0) 68 and Rigual et al. 74 reported equal response rates between primary and recurrent lesions. 74
Significantly better results were found in patients with advanced tumors (advanced incurable SCCs of the head and neck) 10 mm or less in depth and in patients whose lesions were fully illuminated treated with mTHPC-PDT. 31 Ikeda et al. 68 achieved using photofrin-PDT complete response in 7 dysplasias (5 moderate, 2 severe dysplasias) with 1 recurrence (prior to PDT diagnosed as moderate dysplasia) and in 17 SCCs (T1 and T2; 16 well differentiated, 1 moderately differentiated) with 2 recurrences (prior to PDT diagnosed as 1 well differentiated (T1) and 1 moderately differentiated (T1)). 68
Common side effects of PDT involved mild to moderate pain and edema, 31,34,67 while remarkable phototoxicity events were reported only in patients who ignored the light protection guidelines. 33,73 In Table 5, studies for PDT in oral cancer are included.
Methodology and results of studies for PDT in oral cancera.

PDT: photodynamic therapy; mTHPC: meta-tetrahydroxyphenylchlorin; WHO: World Health Organization; OSCC: oral squamous cell carcinoma; HPPH: 3-(1′-hexyloxyethyl) pyropheophorbide α; EGFR: epidermal growth factor receptor; SCC: squamous cell carcinoma; RECIST:
aWHO criteria: complete response = no disease by two measurements ≥4 weeks apart, partial response ≥50% reduction in tumor’s dimensions by two measurements ≥4 weeks apart/no new lesions/no progression of lesions. No change <50% reduction in tumor’s dimensions/<25% tumor’s size increase. Progressive disease ≥25% lesion’s size increase/new lesions (WHO 77 ). RECIST guidelines: complete response = no lesions, partial response ≥30% tumor’s dimensions reduction, progressive disease ≥20% tumor’s dimensions increase, ≥5 mm increase/new lesions, stable disease = neither partial response nor progressive disease (Eisenhauer et al. 78 and Therasse et al. 79 ).
Discussion
In AC, complete response rates after PDT are reported to vary between 29% 20 and 100%, 45,47,52 with a recurrence rate up to 50%, 20 in patients treated with two MAL-PDT sessions, while the same investigators reported only 8% recurrence in patients treated with one MAL-PDT session in which the AC lesions were previously treated with erbium-doped yttrium aluminum garnet ablative fractional laser. 20
In cases of AC, inadequate illumination is possible due to the lower lip anatomy and saliva could reduce the PS absorption leading to poorer results of PDT compared to AK. 37,48
We found only two studies in which patients with AC were treated with DA-PDT. 38,51 In both of them, the PS was 16% MAL cream, while a complete response was reported to be 70% (after two sessions) 51 and 100% (after three to four sessions) 38 with a recurrence rate 28.57% and 0%, respectively. In both of these studies, patients with AC with no response to other treatments (including cryotherapy, vermilionectomy, and imiquimod) were included. 38,51 It is important to mention that Levi et al. 38 achieved complete response (clinically and histologically) without recurrence over a 4-month follow-up period in one patient with AC, who was taught the procedure of the PS application during the first session and he completed the rest of the treatment (the last three sessions) on his own. 38 So it can be concluded that DA-PDT could be in the future a treatment of choice in AC cases, as the first results are very impressive and it is a quite simple procedure which is also well tolerated by the patients.
In leukoplakia, complete response rates vary among 9.09% 59 and 83.33%, 8,57 while recurrence rates are up to 55%. 57 In a case report by Romeo et al., 19 ALA-PDT was found effective in the treatment of one case of OPVL with no recurrence over a 12-month follow-up period. 19 These findings, although limited, are indicative of a possible value of PDT also in challenging cases of OL.
OL and OEL lesions with an increased keratin layer, which could decrease the distribution of the PS or the light into the deeper tissues, are associated with a poorer response to PDT. 9,16,56
In OLP, the mean lesion’s size reduction after treatment with PDT was reported to vary among 44.3% 21 to 55%, 25 while the complete response rate was as high as 10% 23 to 40%. 22 However, we could find only six studies of PDT in OLP, 21 –23,25,27,61 so further research is needed in order to evaluate the utility of PDT in OLP treatment.
The duration of OLP lesions seems to be of importance for the response to PDT as lesions of shorter duration were reported to respond better. 23
In oral cancer, complete response was reported to be as high as 100% in dysplasias/carcinomas in situ, 4,67,68,72 in T1N0, 4,35,69 and in T2N0. 4,68 100% complete response was also reported in T3 tumors 4,33,72 ; however, in these studies, only few patients were included. Only two studies included T4 tumors (seven tumors were treated) 35,72 with only Fan et al. 72 reporting complete response in one of six T4 OSCCs treated with mTHPC-PDT. 72 Biel (2007) 4 achieved complete response in 113 carcinomas in situ/T1N0 and 48 T2N0/T3N0 and in tumors deeper than 3 mm used additionally cylindrical diffusers in order the light to reach also the deeper tissues. 4 Maybe the good results mentioned in this review are strongly associated with this light delivery technique into the deeper tissues which could be of value in treating oral cancer. The overall recurrence rate varied among 3.09% 67 to 50%. 69 However, there is not enough information of which oral cancer lesions were more prone to recur after a complete response to PDT.
The tumor thickness is a crucial parameter for the results of PDT as it has been reported that tumors with a maximum depth of 10 mm demonstrated a better response to PDT compared to tumors with a depth over than 10 mm. 31,69
PDT cannot be used for the treatment of local or distant metastasis. 35
The complicated oral cavity anatomy could lead to a partial illumination of the tumor during PDT especially when it is located in the palate, floor of the mouth, or alveolar process. 68,73
Edema, one of the side effects of PDT, could cause airway blocking in cases of base of the tongue or floor of the mouth (dorsal areas) tumors. 68
Last but not least, in the studies included in Table 5, local or distant metastases after a complete response to PDT were reported in a total of 11 patients during a follow-up period of 3 months to 24 months after the treatment. 4,34,67,68,76 Eight of these died due to metastatic disease. 67,68,76 Consequently, a prolonged follow-up period even in cases with a complete response after PDT is crucial for the optimum survival and quality of life of the patients.
Conclusion
According to the available data, PDT appears to be a very promising therapy for AC, leukoplakia, OLP, and oral cancer. PDT combines safety, limited side effects, and an excellent cosmetic outcome, together with being well tolerated. However, further research is essential in order to establish PDT as the first line therapy of the oral potentially malignant disorders and oral cancer as longer follow-up periods are crucial for evaluating its effectiveness.
Footnotes
Author contribution
Andreadis Dimitrios and Pavlou Achilleia-Maria are equally contributed first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
