Abstract
Long noncoding RNAs (LncRNAs) are involved in the occurrence and progression of human tumors including ovarian cancer (OC). Long noncoding RNA HOTTIP has been found to be involved in several human tumors development. However, the role of HOTTIP in OC remains large unknown. In the present study, our results observed that lncRNA HOTTIP expression levels were notably higher in ovarian cancer tissue samples compared to adjacent normal tissue samples. Increased lncRNA HOTTIP expression levels were significantly associated with advanced FIGO stage and lymph node metastasis of ovarian cancer patients. Survival plots analysis results showed high lncRNA HOTTIP expression levels in ovarian cancer patients showed a poor prognosis compared to patients with low lncRNA HOTTIP expression levels. Function assays showed that lncRNA HOTTIP knockdown in ovarian cancer cells decreased cell proliferation and cell invasion capacities. Furthermore, we demonstrated that inhibition of lncRNA HOTTIP suppressed Wnt/
Introduction
Ovarian cancer is listed as the one of the most severe tumor types affecting the female reproductive tract [1]. Therapeutic methods including surgery and chemotherapy have improved the 5-year survival rate of this disease [2]. However, due to a lack of reliable diagnostic biomarkers, tumor recurrence and tumor metastasis at late stage, the diagnosis and therapy of ovarian cancer remain large challenge [3, 4]. Thus, to explore novel diagnostic or prognostic biomarkers and therapeutic targets is needed.
Long noncoding RNA (lncRNAs) have been found to be involved in a series of biological function including cell proliferation, migration, invasion and tumor metastasis in ovarian cancer [5]. Microarray profile of lncRNAs identifies many lncRNAs as novel biomarkers in ovarian cancer [6]. For instance, lncSOX4 serves an oncogenic role in the tumorigenesis of epithelial ovarian cancer by promoting cell proliferation and inhibiting apoptosis [7]. Long non-coding RNA LSINCT5 promotes ovarian cancer cell proliferation, migration and invasion by disrupting the CXCL12/CXCR4 signaling axis [8]. Upregulation of the long non-coding RNA SPRY4-IT1 indicates a poor prognosis and promotes tumorigenesis in ovarian cancer [9]. Yan et al. showed that long noncoding RNA NBAT-1 suppresses tumorigenesis and predicts favorable prognosis in ovarian cancer [10]. However, the role of lncRNA HOTTIP involved in ovarian cancer developments remains unknown.
In the study, we found that lncRNA HOTTIP expression levels are higher in ovarian cancer tissues. Higher lncRNA HOTTIP expression levels were significantly associated with advanced FIGO stage, lymph node metastasis and poor prognosis for ovarian cancer patients. Moreover, reduced lncRNA HOTTIP expression level inhibited cell proliferation, cell invasion and Wnt/
Methods
Human tissue samples
Sixty-nine paired ovarian cancer tissues and adjacent non-cancerous tissues were obtained from ovarian cancer patients who underwent surgical resection between 2010 and 2012 at Department of Gynaecology, Guizhou Provincial People’s Hospital. Patients did not receive preoperative chemotherapy, radiotherapy, or other treatments. All tissues were immediately frozen in liquid nitrogen and stored at
RNA isolation and quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA was extracted from tissues and cells by using the trizol reagent (Takara Biotechnology Co., Ltd., Dalian, China) according to the manufacturer’s instructions. A total of 1
Cell lines culture
The human ovarian cancer cell lines including OVCAR3, A2780 and SKOV3 cells and human ovarian surface epithelial cells (HOSEpiC) were all purchased from the American Type Culture Collection (ATCC; Manassas, VA, USA). All of cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM) and supplemented with 10% fetal bovine serum (FBS; Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA). The cells were cultured at 37
Cell tranfection
Cells were seeded in 6-well plates and then incubated for 24 h. A 200 ng/ml specific siRNAs against for HOTTIP or negative control (NC) were transfected into the cells using Lipofectamine
MTT assay
The cell proliferation capacity was assessed using MTT assay. Transfected cells (2000 cells/well) were seeded in 96-well plates. After cell transfection at 24, 48, 72, or 96 h, a 20
The HOTTIP expression levels were higher in OC tissues and cells. (A) The expression levels of HOTTIP in ovarian cancer tissues was increased compared with adjacent normal ovarian tissues.(B) QRT-PCR analysis of HOTTIP expression levels in human ovarian cancer cell lines including SKOV3, A2780 and OVCAR and a human ovarian surface epithelial cell line, HOSEpiC. Data represent the mean 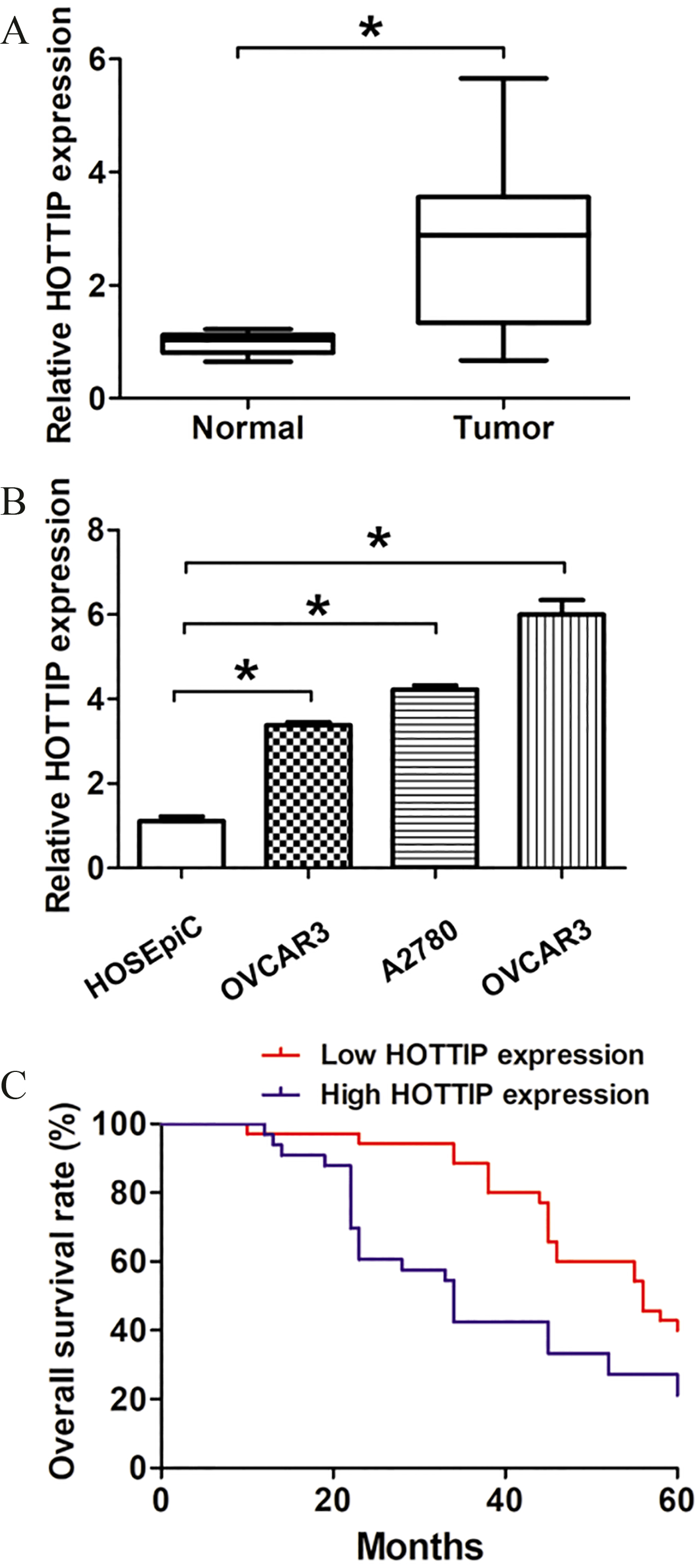
Protein samples were lysed from transfected cells by using a radioimmunoprecipitation assay (RIPA) lysis buffer (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany). Protein samples were quantified using the Pierce BCA Protein assay kit (Pierce; Thermo Fisher Scientific, Inc.). Equal protein samples (40
Cell cycle analysis
Briefly, 48 h post transfection, cells were harvested in a 6-well plate at 1
Transwell assay
Transwell invasion assay was performed using 24-well plates (8.0-
Statistical analysis
All results were shown as mean
The association between HOTTIP expression and clinicopathological parameters
The association between HOTTIP expression and clinicopathological parameters
*
The reduced HOTTIP expression levels suppressed OC cell proliferation. (A)–(B) QRT-PCR analysis of HOTTIP expression levels of si-NC, si-HOTTIP-1 or si-HOTTIP-2 transfected OVCAR and A2780 cells. (C)–(D) MTT assay showed cell proliferation rates of si-NC, si-HOTTIP-1 or si-HOTTIP-2 transfected OVCAR and A2780 cells. (E)–(F) Cell cycle analysis showed cell proliferation rates of si-NC or si-HOTTIP transfected OVCAR and A2780 cells. Data represent the mean 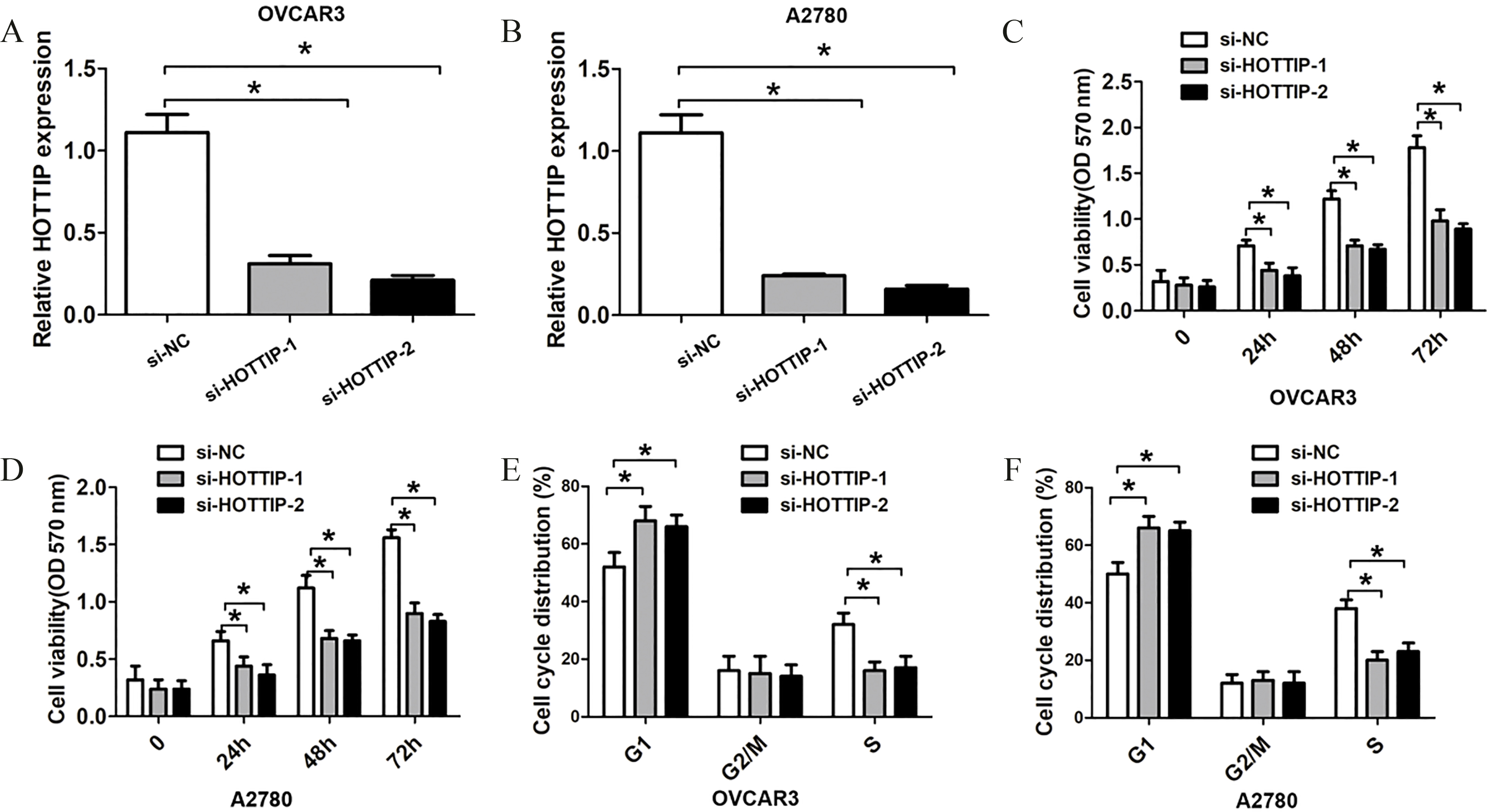
Expression of lncRNA HOTTIP is higher in tumor tissue samples in ovarian cancer patients
To confirm the clinical role of lncRNA HOTTIP expression in ovarian cancer, we detected the relative expression of lncRNA HOTTIP in OC tissue samples and adjacent normal tissue samples by qRT-PCR analysis. The results indicated that the expression of lncRNA HOTTIP was significantly higher in tumor samples than that in adjacent normal tissue samples (Fig. 1A,
The reduced HOTTIP expression levels suppressed OC cell invasion. (A)–(B) Transwell cell invasion showed cell invasive number of si-NC, si-HOTTIP-1 or si-HOTTIP-2 transfected OVCAR cells. (C)–(D) Transwell cell invasion showed cell invasive number of si-NC, si-HOTTIP-1 or si-HOTTIP-2 transfected A2780 cells. Data represent the mean 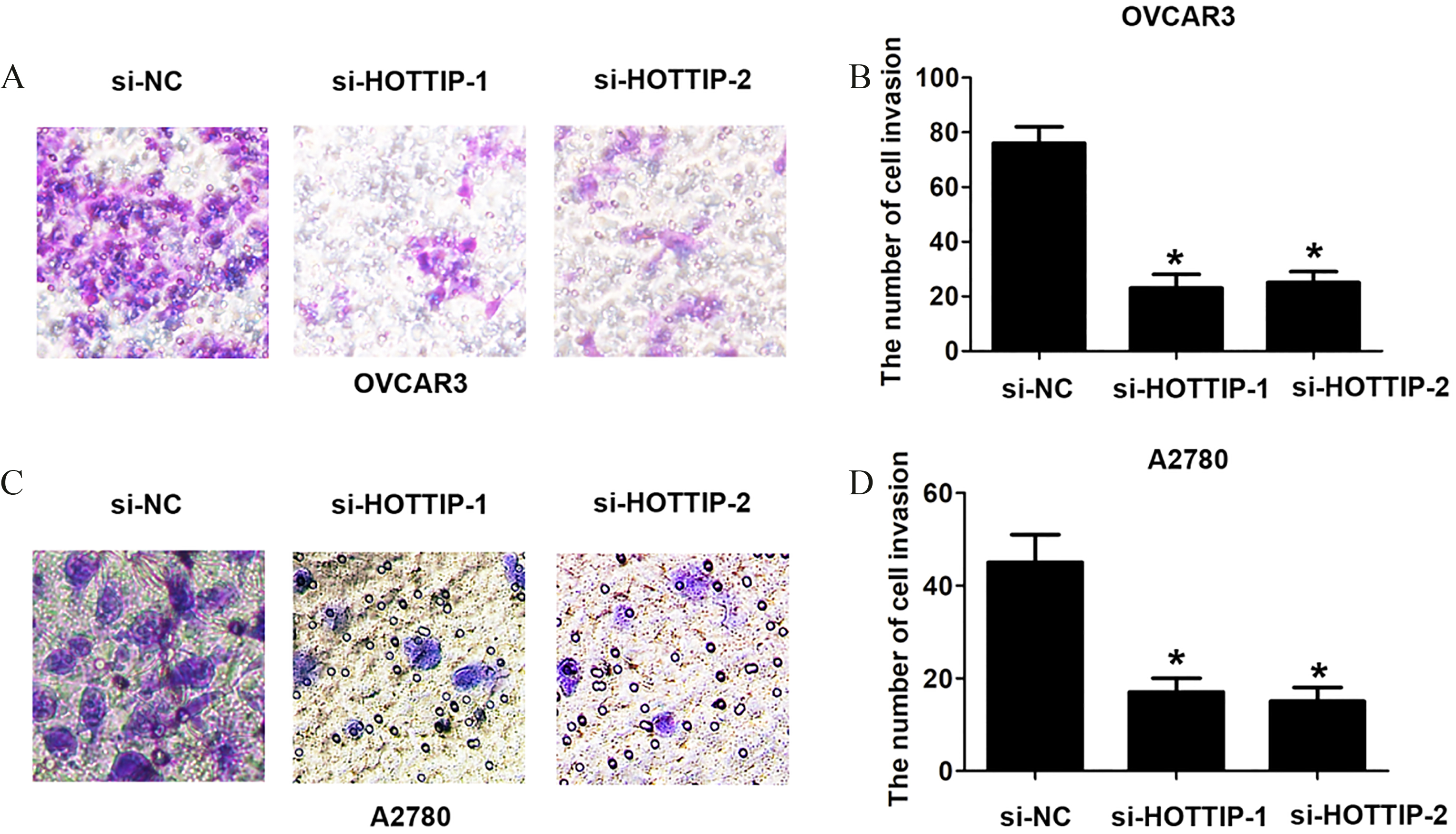
The reduced HOTTIP expression levels suppressed Wnt/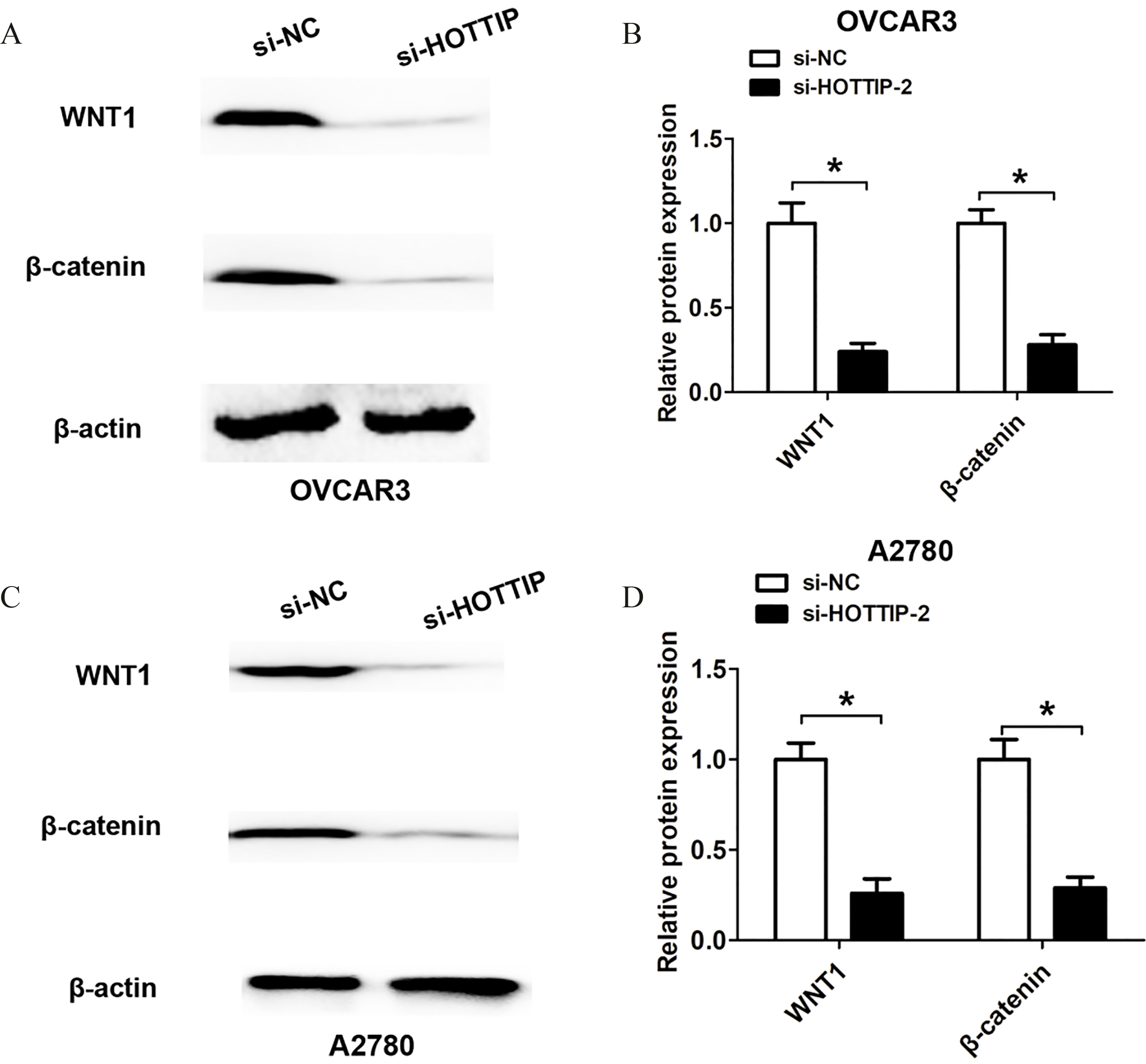
Next, we analyzed the association between lncRNA HOTTIP expression and clinical factors in OC patients. We divided patients into lncRNA HOTTIP low and lncRNA HOTTIP high groups using the median value of 3.22 as a cut off value. As shown in Table 1, the results indicated that higher lncRNA HOTTIP expression significantly associated with advanced FIGO stage (
Knockdown of lncRNA HOTTIP suppresses OC cell proliferation and invasion capacity
Furthermore, we transfected with two si-HOTTIP oligos or si-NC into A2780 or OVCAR3 cells, and the transfection efficiency was detected using qRT-PCR analysis. We found two si-HOTTIP oligos had higher efficiency of knockdown for lncRNA HOTTIP and were used for the following experiments (Fig. 2A and B). To investigate the association between lncRNA HOTTIP expression and cell growth and invasion of OC, The MTT assays and transwell assay were performed. The results indicated that knockdown of lncRNA HOTTIP significantly decreased cell proliferation compared to the control groups in OVCAR3 and A2780 cells (Fig. 2C and D). Furthermore, we observed that knockdown of lncRNA HOTTIP significantly decreased S phase cell number compared to the control groups in OVCAR3 and A2780 cells (Fig. 2E and F). Besides, the knockdown of lncRNA HOTTIP significantly reduced the cell invasion ability compared to control groups in OVCAR3 and A2780 cells (Fig. 3A and D). Thus, these results indicated that inhibition of lncRNA HOTTIP significantly reduced cell proliferation and invasion in OC.
Reduced lncRNA HOTTIP inhibits WNT signaling pathway in OC cells
Given the finding that lncRNA HOTTIP could affect OC cell proliferation and invasion, we attempted to explore the underlying molecular mechanisms. As we known, WNT signaling pathway plays crucial pathways in regulating tumor growth and metastasis including ovarian cancer. Thus, we detected the state of the WNT pathway after lncRNA HOTTIP silencing using si-HOTTIP-2 due to its higher knockdown efficiency. After lncRNA HOTTIP knockdown, we found that the protein levels of WNT1 and
Discussion
Recent studies have reported that lncRNA HOTTIP could play crucial roles in tumor development of many human malignant cancers. For example, Chen et al. showed that upregulation of long non-coding RNA HOTTIP promotes metastasis of esophageal squamous cell carcinoma via induction of EMT [11]. Sang et al. found that up-regulation of long non-coding HOTTIP functions as an oncogene by regulating HOXA13 in non-small cell lung cancer [12]. Ye and his team demonstrated that overexpression of long non-coding RNA HOTTIP promotes tumor invasion and predicts poor prognosis in gastric cancer [13]. Long non-coding RNA HOTTIP promotes BCL-2 expression and induces chemoresistance in small cell lung cancer by sponging miR-216a [14]. Knockdown of the long non-coding RNA HOTTIP inhibits colorectal cancer cell proliferation and migration and induces apoptosis by targeting SGK1 [15]. However, the clinical significance as well as underlying mechanism of lncRNA HOTTIP in OC is still unclear.
In this study, we found that lncRNA HOTTIP was higher in ovarian cancer tissues samples. Higher lncRNA HOTTIP expression associated with advanced FIGO stage and lymph node metastasis of ovarian cancer patients. Survival plots analysis results showed high lncRNA HOTTIP expression levels in ovarian cancer patients showed a poor prognosis compared to patients with low lncRNA HOTTIP expression levels. Thus, these results indicated that lncRNA HOTTIP could serve as a promising biomarker for monitoring ovarian cancer.
Moreover, by performing function assays, we showed that lncRNA HOTTIP silencing suppressed cell proliferation, cell cycle progression and cell invasion ability in OC. These results indicated that lncRNA HOTTIP expression was involved in OC progression. After lncRNA HOTTIP knockdown, we found that the protein levels of Wnt1 and
In conclusion, we found that lncRNA HOTTIP is higher in ovarian cancer and associated with poor prognosis. Reducing lncRNA HOTTIP expression suppresses cell proliferation, invasion and WNT signaling. Thus, these results indicated that lncRNA HOTTIP could serve as a promising biomarker for monitoring ovarian cancer and target of OC treatment.
