Abstract
The triple negative breast cancer (TNBC) accounts for 15% to 20% of the total number of breast cancer diagnosed. A number of clinical studies have shown that TNBC has a high risk of early recurrence and distant metastasis, and a low rate of disease free survival and total survival. The premise of TNBC deterioration was abnormal proliferation and migration of tumor cells, and this study firstly showed that GATS gene could promote proliferation of MDA-MB-231 breast cancer cells. Through lentiviral expression system, the GATS gene was konckdown by shGATS lentivirus infection in the MDA-MB-231 cells, and the result indicated it could remarkably decrease the ability of cell proliferation and migration. Real-time PCR, western blot and immunofluorescence experiments showed the expressions of protein LC3, and p-Akt in shGATS cell group were lower than the shCtrl group. Therefore, we suggest the GATS could promote the MDA-MB-231 cell proliferation, migration and clonogenicity through cell autophagy by the PI3K/Akt pathway, which paved the way for further study the function of GATS in TNBC, and GATS may potentially be a target for gene therapy against triple negative breast cancer.
Introduction
Breast cancer with high heterogeneity is one of the most common malignant tumors in women, which is the main cause of cancer death in women. According to statistics, the annual incidence of breast cancer is about 167.1 million population, and the death rate is about 52.2 million population [1]. In addition, the age of breast cancer is becoming younger, which has been attracted the worldwide attention [2]. Breast cancer could be divided into four different subtypes according to the expression levels of human epidermal growth factor receptor (HER2), estrogen receptor (ER) and progestin receptor (PR), such as Luminal A, Luminal B, HER2
Autophagy has become a research hotspot in the regulation of malignant phenotype of tumor cells [8]. The study shows that autophagy can promote the expression of tumor proliferation related genes and lead to the high invasion and metastasis of tumor [9]. Autophagy is considered to impede the occurrence of tumor during the early stage of tumor development, but autophagy after radiotherapy and chemotherapy can remove and utilize the harmful substances inside and outside the cell in time to promote the deterioration and metastasis of the tumor. This may be the main cause of the refractory treatment of cancer patients in clinical cancer [10], so several scholars have suggested that inhibiting autophagy can improve the chemosensitivity of breast cancer cells [11]. For the subtypes of breast cancer Luminal A, Luminal B and HER2
The gene encoding human GATS protein is a single copy gene located on the 22 arm of chromosome 7 (7q22.1), consisting of 9 exons, containing 163 amino acids. The GenBank accession number of GATS is NM_178831, and its database analysis showed that the molecular weight of GATS protein was 17.8 kDa and the isoelectric point was 8.78. Transcriptional sequence and proteomics studies show that the GATS gene is expressed in all tissues of the human body, especially in the brain, testis, ovary and endometrium [14], but until now there is no specific report about the function of GATS gene. Therefore, in this study we firstly found the downexpression of GATS could decrease the cell autophagy in MDA-MB-231 cells by the the PI3K/Akt signal transduction pathway. And the molecular experiments showed that GATS could promote the proliferation and metastasis of MDA-MB-231 cells, this may provide an effective method for developing targeted drug to treat TNBC.
Materials and methods
Cell lines and cell culture
Human breast cancer cell lines MDA-MB-231, MCF-7, T47D, 293T were maintained in our laboratory. Cell lines were cultured in RPMI 1640 (Gibco, USA) supplemented with 10% fetal calf serum (Gibco), and 1% penicillin and streptomycin (Invitrogen, USA) at 37
qRT-PCR analysis of mRNA expression
The RT-PCR primer sequences used for GATS were as follows: F: 5’-TATTGGCATG ACCAAGATCG-3’; R: 5’-TTGAGCACCAGGATGAAGTCT-3’, and the relative amount of each mRNA was normalized to GAPDH, which was amplified by the primers: F: 5’-TGACTTCAACAGCGACACCCA-3’; R: 5’-CACCC TGTTGCTGT AGCCAAA-3’. Total RNA was isolated with TRIzol reagent (Invitrogen) according to the manufacturer’s protocol, then the cDNA was synthesized by the RevertAid First-Strand cDNA Synthesis Kit (Fermentas, USA). The expression levels of each analyte compared with untreated controls was assessed using the 2
ShRNA design of GATS and its lentivirus construction
The shRNA interference sequence of GATS was designed according to the selected target sequence as 5’-CACCAGAGGATTACACTAT-3’, and the restriction sites Age I and EcoR I were added at both ends to complete the construcion of the lentivirus vector GV115-GATS-shRNA (Genechem Co., Ltd., Shanghai, China). The recombinant plasmid was tranfected into the prepared Escherichia coli receptive cell, and the positive clone was selected by the PCR method. Then the extracted plasmid was sent to sequencing to verify there was no mutation in the shRNA sequence.
The logarithmic growth phase 293T cells were inoculated in 10 cm cell culture dishes, and adjusted the cell density to about 70%–80%. Then the vectors 20
Cell proliferation assay
The Cell Counting Kit 8 (CCK-8) was conducted as follows: briefly, 1,000 cells of each group were plated per well in 96-well microplates in 150
For the proliferation assay of cells with green fluorescence were calculated by the Celigo machine (Nexcelom Bioscience LLC., MA, USA). By adjusting the input parameters of analysis settings, the data and the cell proliferation curve were plotted for 5 days.
Clonogenicity assay
The cancer cells were transfected with GATS- shRNA or control RNA for 12 h, and then transferred to six-well plates and seeded at a density of 1.0
Scratch-healing assay
MDA-MB-231 cells were seeded into six-well plates at a density of 0.8
Cell cycle detection by FCM (flow cytometry)
The cells (1
Immunofluorescence
The cells were seeded in a 6-well plates after digestion, then fixed in 4% paraformaldehyde, permeabilized, blocked with 3% BSA, and incubated with LC3 antibody (Santa Cruz, CA, USA) for 1 h at 37
High expression of GATS in triple negative breast cancer cells MDA-MB-231. A, RT-PCR analysis of GATS expression in breast cancer cell lines MDA-MB-231, MCF-7 and T47D; B, the protein expression levels of GATS in breast cancer cell lines MDA-MB-231, MCF-7 and T47D. Data are shown as means 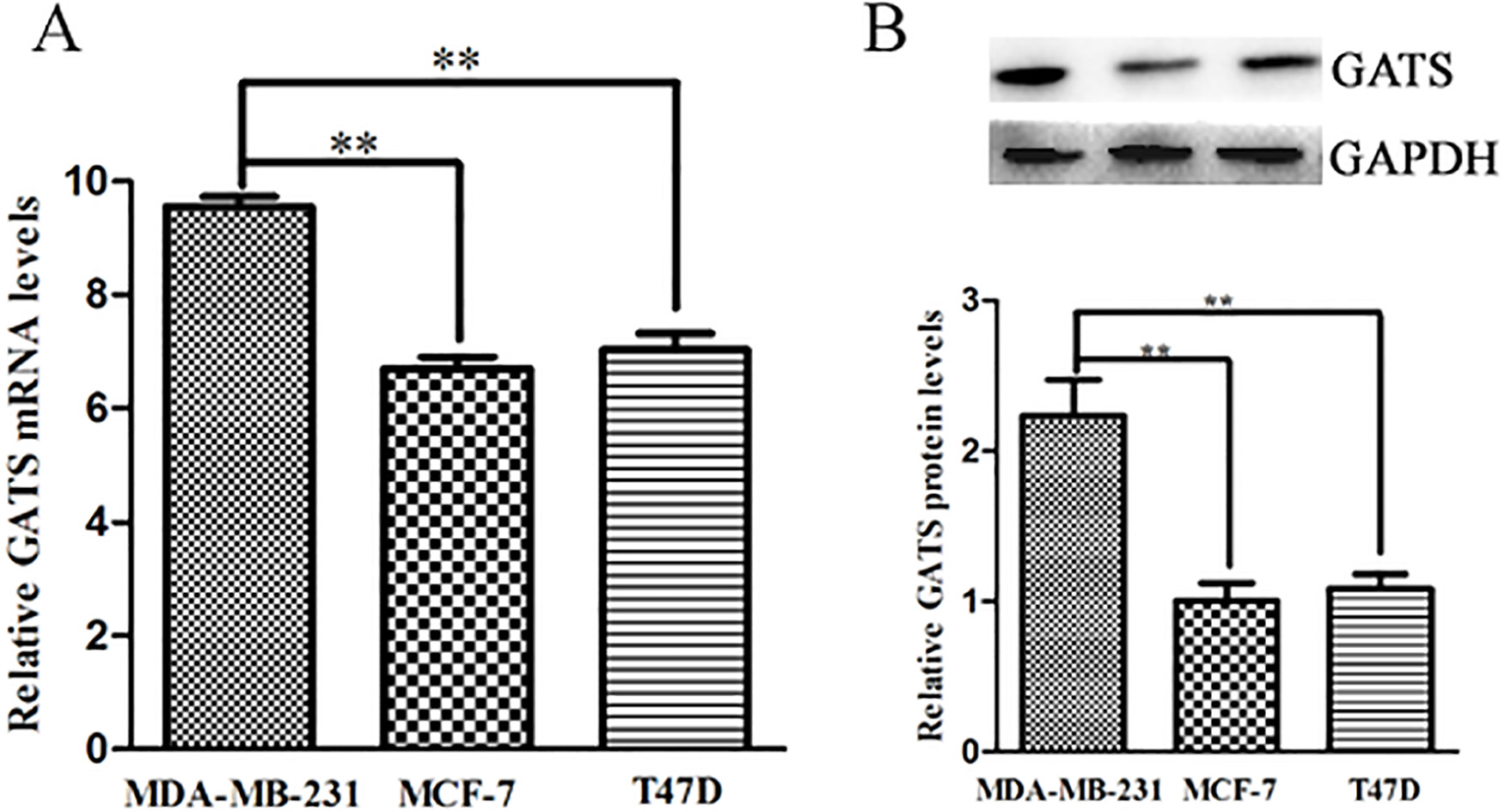
Downexpression of GATS inhibited colony formation of MDA-MB-231 cells. A, the number of colonies in shGATS group were much less than those in the control group, and figures of soft agar cloned experimental plate were obtained by digital camera, 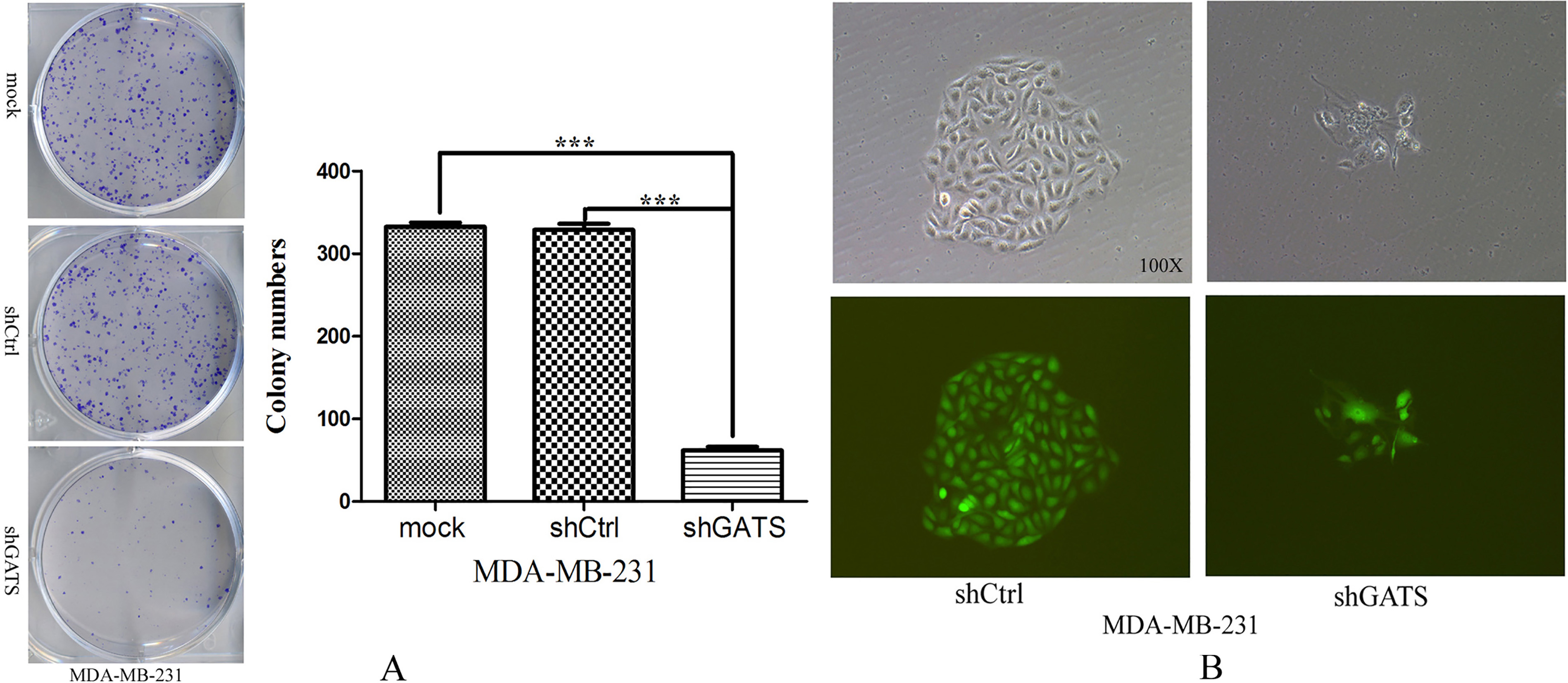
Downexpression of GATS inhibited proliferation and migration of MDA-MB-231 cells. A, proliferation curve of MDA-MB-231 cells in shCtrl and shGATS group under the fluorescence field; B, wound closure assay revealed a clear inhibiting effect on cell migration by observation at 24 h in shGATS group (magnification, 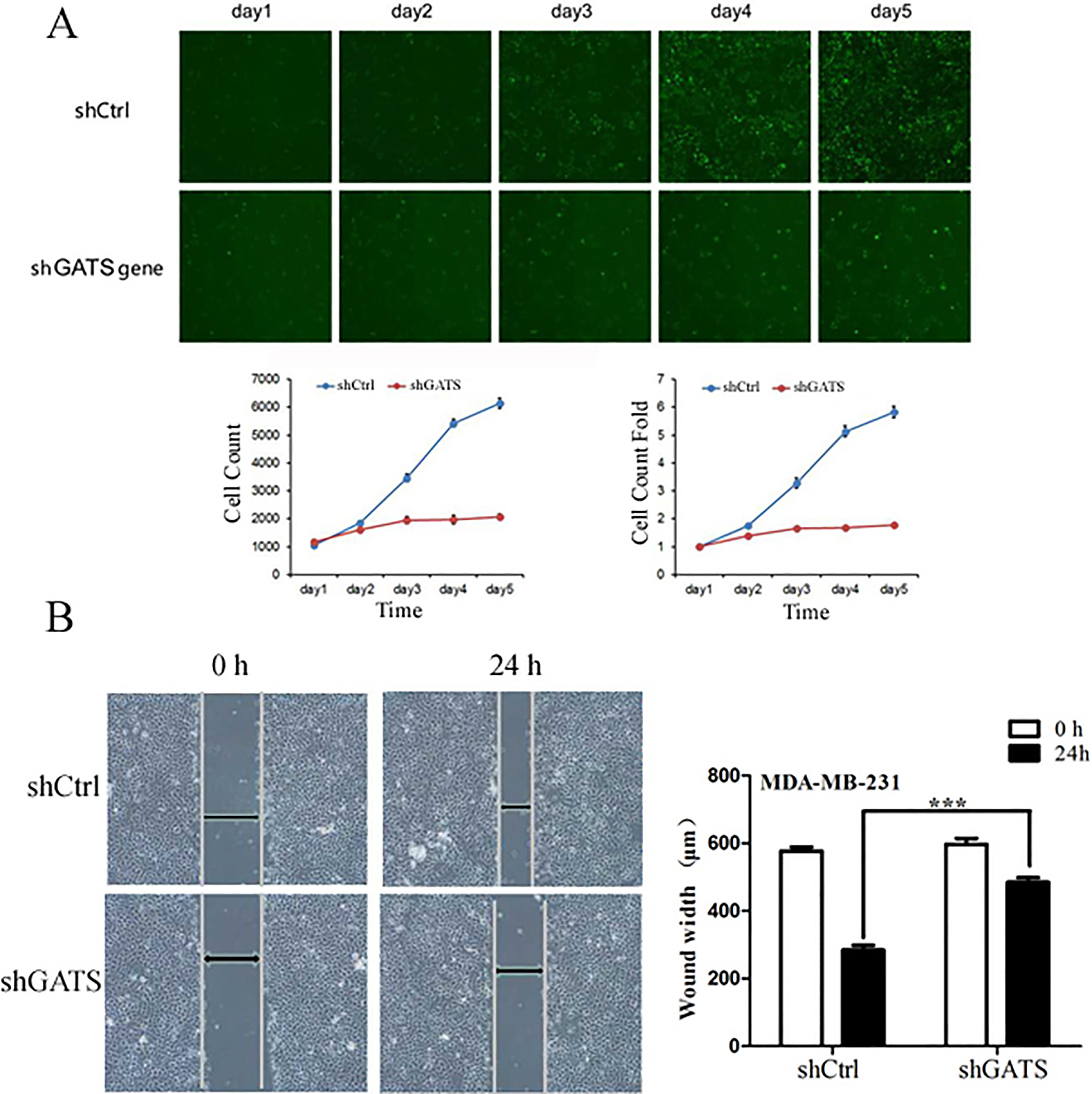
Effect of GATS downexpression on the MDA-MB-231 cell cycle. shGATS infection MDA-MB-231 cells showed an increased percentage of G0/G1 phase cells and a decreased percentage of S phase cells. 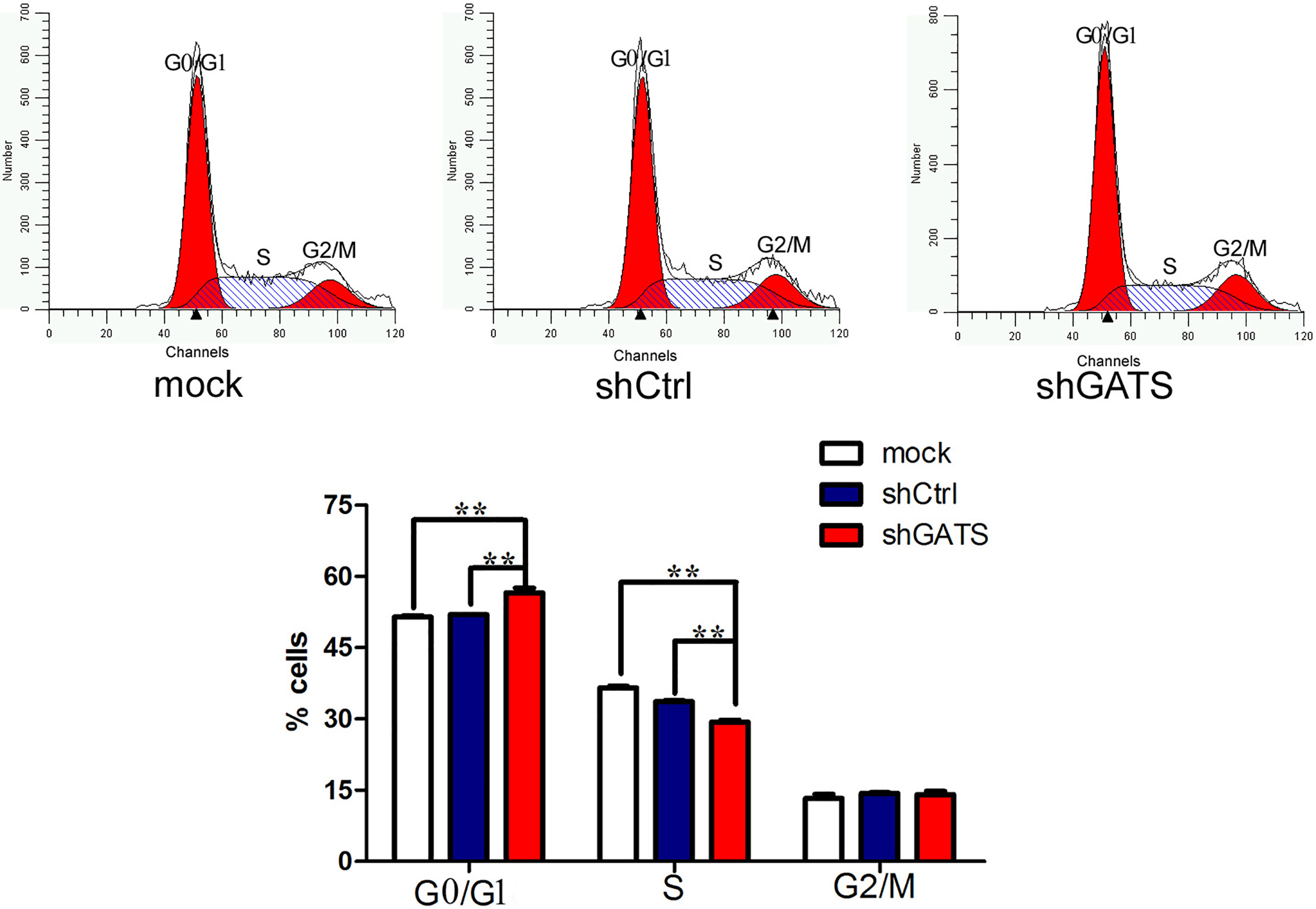
Downexpression of GATS inhibited the level of autophagy in MDA-MB-231 cells after 6 h starvation. A, the immunofluorescence images of MDA-MB-231 cells. The puncta were visualized by fluorescence microscope imaging of cells immunostained for nucleus and LC3 using DAPI (blue) and Alexa Fluor 488 secondary antibody conjugates (green), respectively. Arrow indicates the LC3 puncta, and the histogram indicated the relative average fluorescence density of each group cells, 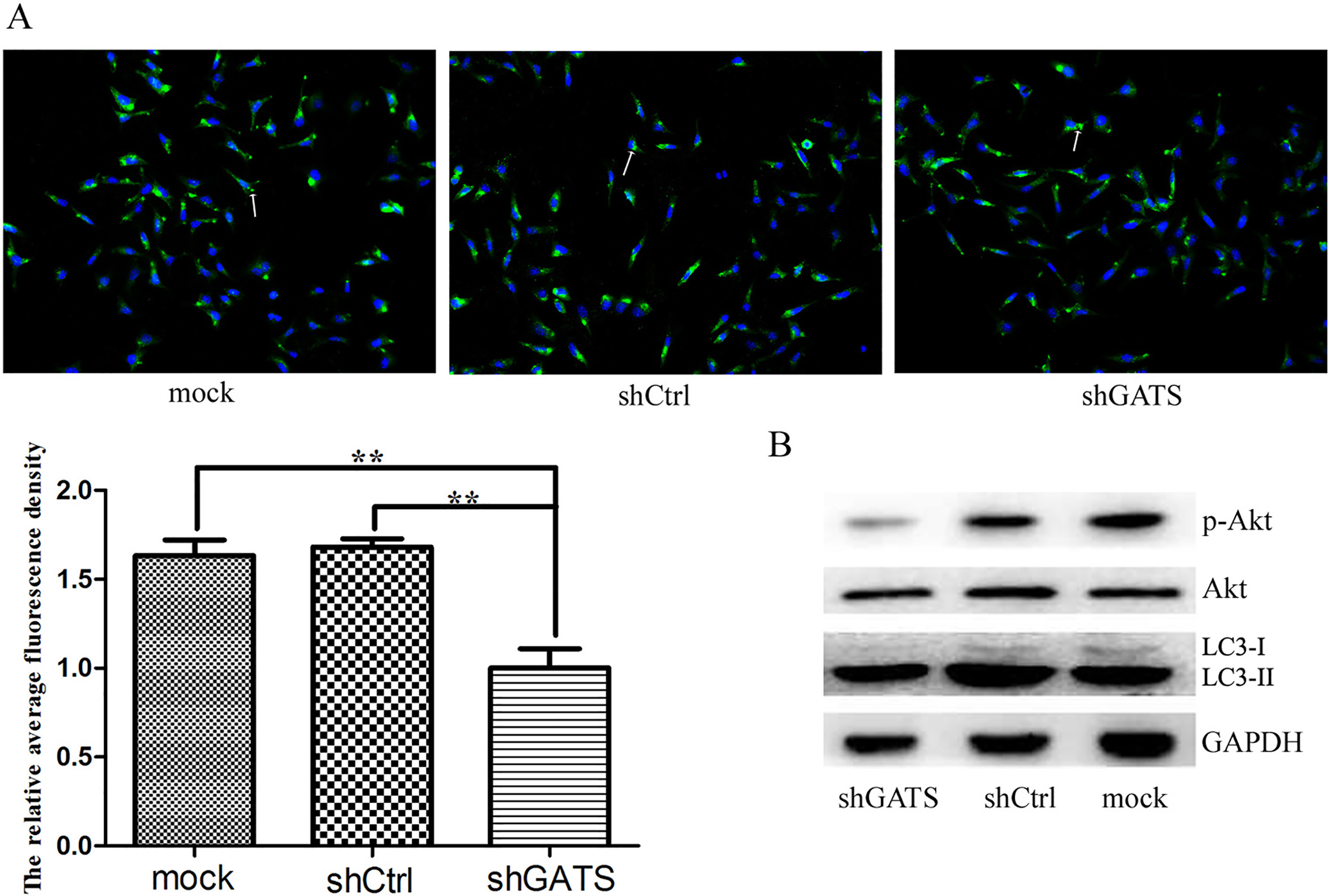
Total cellular extracts were prepared by homogenization of 3
Statistical analysis
Data was shown as mean
Results
GATS was highly expressed in the triple negative breast cancer cells MDA-MB-231
To investigate the expression level of GATS in TNBC cells, the real-time PCR of GATS was conducted in MDA-MB-231 cells. Compared with the other cell lines, the GATS mRNA level was obviously upregulated (Fig. 1A). The protein analysis by western blotting revealed that GATS protein was in accordance with the RT-PCR result, it was upregulated about 2-fold compared with the other control cells (Fig. 1B). Therefore, GATS was confirmed that it was highly expressed in the triple negative breast cells MDA-MB-231.
Downregulation of GATS inhibits colony formation of MDA-MB-231 cells
For analyzing the reason that GATS gene was highly expressed in TNBC cells, 5
Downregulation of GATS inhibits cell proliferation and migration of MDA-MB-231
For further study the functon of GATS gene, the lentivirus 5
Next, the cell migration ability was analyzed using a wound closure experiment in MDA-MB-231 cells following shGATS lentivirus infection. The wound-closing procedure was serially observed for 72 h following the introduction of the wound on the plate. As shown in Fig. 3B, the speed of wound-closing was slower in shGATS-infected cells, compared with that in the control groups (uninfected and mock-infected GATS cells). This indicated that the downregulation of GATS expression inhibited cell migration in vitro.
Downregulation of GATS induces cell cycle arrest in MDA-MB-231 cells
The cell cycle fraction was investigated using flow cytometry based on shGATS infection for 96 h for TNBC cell lines. In the MDA-MB-231 cells, shGATS infection caused a significant increase in the G0/G1 fraction (56.23 vs. 51.98 and 51.95%; the mock and control groups, respectively) with concurrent decline in S (29.14 vs. 36.02 and 33.80%) and G2/M fractions (14.62 vs. 12.00 and 14.26%; the mock and control groups, respectively), compared with the controls (Fig. 4). Overall, these results indicated that the downregulation of the GATS gene by shGATS lentivirus induces G0/G1 phase arrest.
GATS regulates cell autophagy in MDA-MB-231 cells by the PI3K/Akt signaling pathway
To verify the relationship between GATS expression with cell autophagy, immunofluorescence expriment was conducted to detect the level of autophagy after the MDA-MB-231 cells infected with shGATS lentivirus. As shown in Fig. 5A, the level of autophagy in shGATS group was obviously decreased compared with the NC group after starvation for 6 hours, suggesting GATS could regulate cell autophagy in TNBC cells.
Next, to explore the cell autophagy mechanism of breast cancer cell regulated by GATS, the Akt signal pathway was confirmed to participate in autophage by western blotting experiment (Fig. 5B), and this indicated that GATS regulate cell autophagy through the Akt signal pathway.
Discussion
The triple negative breast cancer accounts for about 20% of the breast cancer, and the treatment of non-targeted drugs is easy to appear the malignant transformation of the tumor [17]. Therefore, it is necessary to screen the effective target for the deterioration and development of breast cancer. The premise of breast cancer deterioration is abnormal proliferation and migration of tumor cells [18]. In this study, GATS gene was firstly proved that it could promote proliferation, clonogenicity and migration of triple negative breast cancer cells MDA-MB-231. The structure and functional domain of bioinformatics showed that the 86-152 amino acid region of GATS was the aspartate kinase-chorismate mutase-tyrA (ACT) domain, which played an important role in the process of metabolic control, solute transport and signal transduction. At present, the studies about proteins containing the ACT domain were concentrated in the secondary metabolites of bacteria and plants, and many research results suggested that the encoded protein containing the ACT domain could promote the survival of the cells in the adversity environment [19, 20], which was similar to the malignant transformation of tumor cells under the action of chemotherapeutic drugs. The protein interaction prediction analysis showed that the interaction protein of GATS was nephronophthisis 3 (NPHP3). It was found that NPHP3 was closely related to the development of prostate cancer and the poor prognosis in the whole genome association of the tumor [21]. All these results indicated that GATS played an important role in the transformation of human tumor, and we firstly showed the GATS gene could promote the cell proliferation and migration.
The role of autophagy in regulating malignant transformation and chemoresistance is a hot topic in recent years. A large number of studies have shown that autophagy is considered to impede the occurrence of tumor during the early stage of tumor development, but autophagy could increase cancer cell proliferation and migration in time to promote the deterioration and metastasis of tumor after radiotherapy and chemotherapy, which may be the main failure reason for the treatment of cancer patients [22, 23]. Inhibition of autophagy is proposed to enhance chemosensitivity of breast cancer cells [24]. The study showed that the combination of rapamycin and resveratrol could effectively block autophagy and induce apoptosis in ER
The study about the transformation of the TNBC malignant phenotype on the signal pathway is mainly focused on the PI3K/Akt signaling pathway, and the treatment of TNBC is also reported in many research practices, proving that the PI3K/Akt signaling pathway is closely related to the malignant proliferation of TNBC [27, 28]. Studies have found that hyaluronic acid, PI3K and epidermal growth factor receptor family can activate PI3K downstream molecules and mediate tumor cell proliferation [29]. And it is found that dietary inorganic sulfur can inhibit the expression of ErbB2 and ErbB3 through the Akt pathway, thereby reducing the proliferation of triple negative breast cancer cells [30]. The results are in agreement with the experimental results of GATS gene knockout expression, indirectly indicating that GATS molecules may induce the expression of ErbB2 regulated by PI3K/Akt pathway, thus stimulating the malignant proliferation of TNBC.
In conclusion, the GATS gene was proved highly expressed in the triple negative breast cancer cells. Using the lentivirus knockdown system, the effects of downregulating the GATS gene were explored in multiple biological features, including cell proliferation, migration, and the cell cycle. Further study showed the GATS gene regulated the cell proliferation by the cell autophagy through PI3K/Akt signal pathway. These findings provide evidence that GATS may potentially be a target for gene therapy against triple negative breast cancer. The downregulation of GATS may be a therapeutic strategy for the suppression of breast cancer progression, which provide an effective therapeutic method that could significantly improve the treatment of triple negative breast cancer.
Footnotes
Acknowledgments
This study was supported by the Provincial Natural Science Research Project of Anhui Colleges (KJ2017 ZD36). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
