Abstract
MiRNAs regulated most genes expression, which were proved important in various tumors. In this study, we want to investigate miR-101 effect and molecular mechanism on pancreatic cancer (PC), the research about this was blank now. RT-PCR analysis showed that miR-101 expression was declined in PC. MTT assay found that miR-101 mimic suppressed cell viability, while suppressing miR-101 facilitated cell proliferation. Transwell assay showed that miR-101 mimic inhibited cell invasion, but promoted cell invasion by miR-101 inhibitor. With TargetScanHuman’s help, we verified STMN1 as a specific target of miR-101 and luciferase reporter assay was carried out to further confirm this discovery. STMN1 expression was reduced by miR-101 mimic and increased by miR-101 inhibitor. We next found that STMN1 was elevated in PC and its expression was negatively correlated with miR-101 expression. Furthermore, STMN1 siRNA curbed cell proliferation and invasion, which was opposite to miR-101 inhibitor effect on PC progression and STMN1 siRNA attenuated miR-101 inhibitor effect on cell proliferation and invasion. In conclusion, miR-101 inhibited PC cell proliferation and invasion via regulating STMN1, which provided a potential therapeutic for PC patients.
Introduction
Pancreatic cancer (PC) is a malignant tumor of digestive tract, which is difficult to diagnose and treat, and about 90% of the patients are ductal adenocarcinomas originating from the epithelium of the gland. The morbidity and mortality of PC have risen significantly in recent years and 5-year survival rate is lower than 5% [1, 2] and it is one of the worst prognosis of malignant tumors [3]. Although there is a great improvement in the new methods of treatment PC, the cure rate is low. Therefore, searching for the specific biomarkers for treating PC has become much more urgent [4].
MiRNAs were reported quite important in most malignancy studies due to its regulatory effect on tumors progression as oncogenes and tumor suppressors [5, 6]. Recently, many studies have elucidated that miRNAs and PC tumorigenesis has a close connection [7, 8]. For instance, miR-27a facilitated PC cell proliferation and suppressed apoptosis as an oncogene [9], while miR-126 and miR-34a showed antitumor efficacy in PC [10]. In this study, our research was focused on miR-101. Because miR-101 was reported to have close connection with many different cancers, such as cervical cancer [11], colorectal cancer [12], bladder cancer [13] and breast cancer [14]. However, the role of miR-101 and its underlying mechanism in PC remains uncertain as far as we known, which need us to further explore it.
MiR-101’s mechanism in regulating malignant tumor was most through targeting its mRNA. Deng et al. showed that miR-101/VEGF-C signaling involved in the inhibitory effect on intrahepatic cholangiocarcinoma progression [15]. Furthermore, it had a report stated that miR-101 modulated bladder cancer development via COX-2 [16]. Nevertheless, the mechanism of miR-101 in modulating PC proliferation and invasion has not reported until now.
STMN1 is a kind of microtubule depolymerization phosphorin which has been widely studied in recent years. It affects the balance of microtubule power system by self regulation of phosphorylation level, and it is involved in the regulation of many signaling pathways. STMN1 is up-regulated in many tumor tissues and is associated with the malignancy, invasion and migration of the tumors, including breast cancer [17], gallbladder carcinoma [18], acute myeloid leukemia [19], lung cancer [20] and prostate cancer [21]. However, STMN1 role in PC proliferation and invasion was not studied now and whether it acted as a target of miR-101 in regulating PC progression was not reported too.
In our research, we started to study miR-101 and STMN1 role in PC and the mechanism of miR-101 in modulating PC cell proliferation and invasion. We confirmed that miR-101 had a close connection with STMN1, and miR-101 played a suppressive effect on PC progression while STMN1 done the opposite effect. We also verified STMN1 as a specific target of miR-101 and STMN1 could attenuate miR-101 inhibitory effect, suggesting that miR-101/STMN1 signaling took part in regulating PC development and might provide a target for treating PC.
Materials and methods
Tissue specimens
Make sure ethics committee of Hospital approved this project and all patients with PC signed the informed consent before tissues acquisition. Forty-two pairs of tissue specimens and normal tissues were collected from PC patients who underwent surgery in Hospital. The specimens were instantly put into liquid nitrogen when the tumor tissues were removed and then stored in
Cell culture
Four PC cell lines (PANC-1, AsPC-1, BxPC-3 and SW1990) and normal cell line (HPNE) were purchased from ATCC (USA). All cells were cultured in DMEM with 10% FBS, penicillin (100 U/ml) and streptomycin (100
Cell transfection
MiR-101 mimic/inhibitor or negative control was provided by GenePharma (Suzhou, China) and they were used to intervene miR-101 expression. STMN1 siRNA was synthesized by Shanghai Genechem Co., LTD and it was used to silence STMN1. We added PANC-1 and AsPC-1 cells in 48-well plates respectively and performed transfection with the help of Lipofectamine 2000 (Invitrogen) and then incubated for 48 h with 5% CO
RNA extraction and quantitative RT-PCR
Total RNA was extracted from cells or tissue specimens using Trizol reagent (Invitrogen). The measurement and calculation of qRT-PCR was performed with the help of SYBR green qPCR assay (Takara, China). U6 was used as a control to normalize the expression of miR-101 and
miR-101’s low expression in PC. (A) miR-101 average expression measured in PC and normal tissues (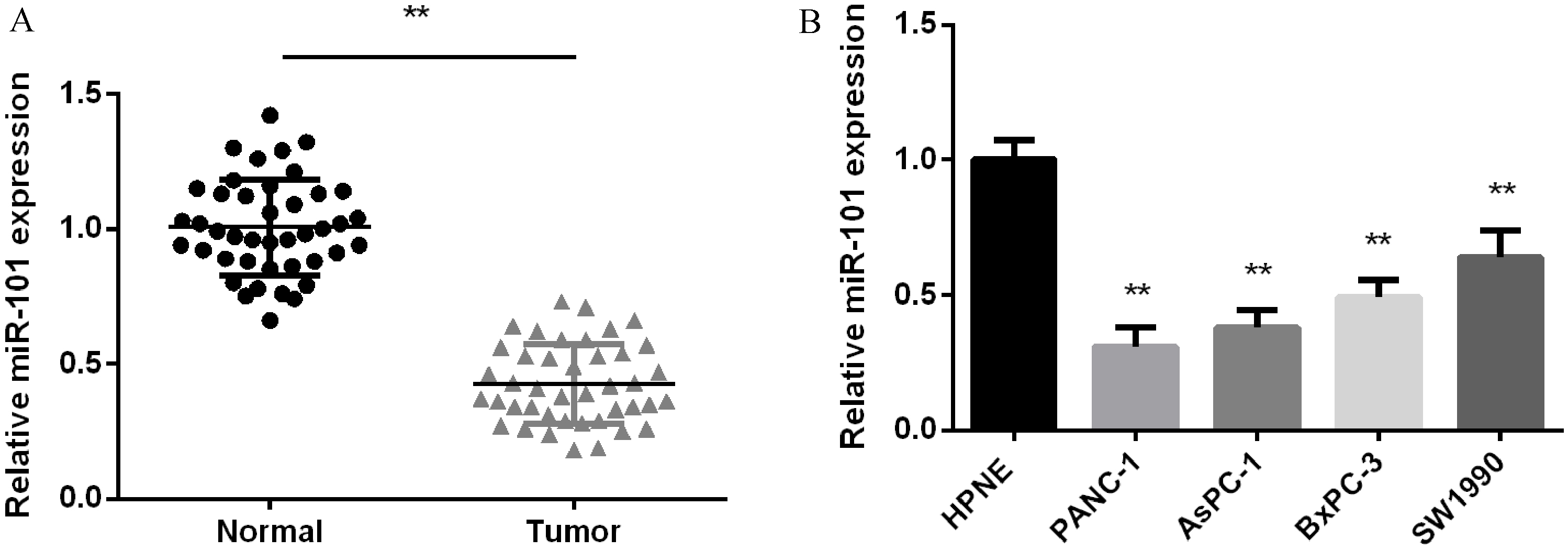
Immunoblotting was carried out to detect STMN1 protein level in PC tissues and cells. Lysis buffer containing 1% PMSF was added into PC to lyse tissues or cells. After centrifugation, the supernatant proteins were adjusted to the same level by BCA kit. 50
MTT assay
Cell viability was evaluated by MTT assay with a slight modification. We seeded PC cells (1
Transwell assay
The invasive ability of PC cells was detected by Transwell assay. The transwell chamber with 8
Luciferase reporter assay
Luciferase reporter assay was applied to measure the STMN1 luciferase ability. PNCA-1 cells were seeded into 24-well plate and cultured overnight, then miR-101 mimic or inhibitor and STMN1 3’ UTR WT or MT luciferase reporter vector were co-transfected into PNCA-1 cells with the help of Lipofectamine 2000 (Thermo, USA). After transfection for 48 h, the Dual Luciferase Reporter System (GeneCopoeia) was used to measure the luciferase activity in different transfection cells.
Statistical analysis
The experiments were conducted three times separately. SPSS 17.0 statistical software (SPSS, USA) was applied to process the data and the data was displayed as mean
miR-101’ suppressive effect on PC cell proliferation and invasion. (A) miR-101 mRNA expression tested in PANC-1 and AsPC-1cells after over-expressing or silencing miR-101. (B) Cell viability detected in PC two cell lines after over-expressing or silencing miR-101. (C) Relative invaded cells detected in PC two cell lines after over-expressing or silencing miR-101. 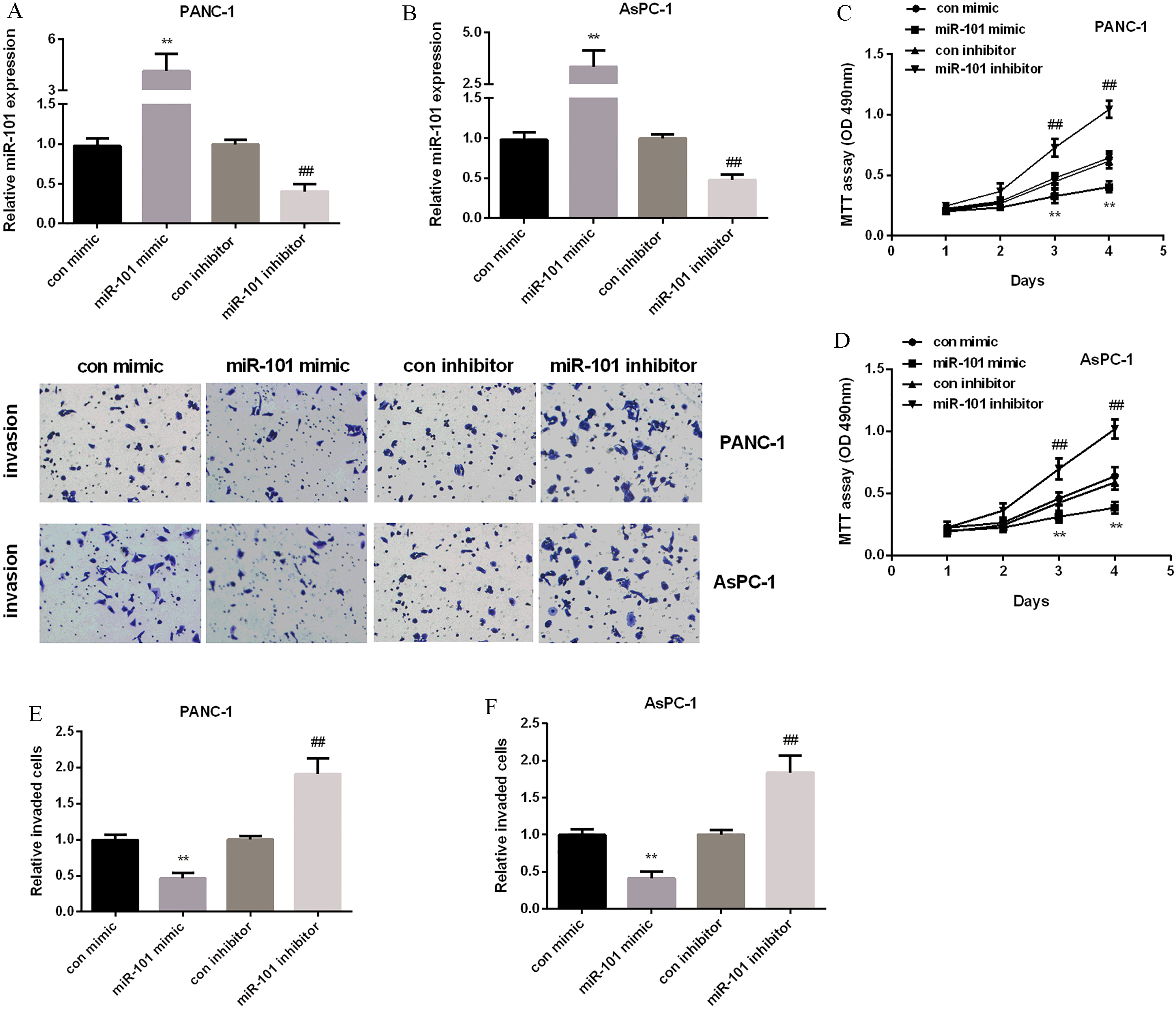
miR-101’s target, STMN1 in PC. (A) The binding sites of miR-101 and STMN1. (B) STMN1’s luciferase activity detected in PANC-1 cells after treated with miR-101 mimic or inhibitor. (C) STMN1 protein expressional level measured in PC two cell lines after treated with miR-101 mimic or inhibitor. (D) STMN1 mRNA expressional level measured in PC two cell lines after treated with miR-101 mimic or inhibitor. 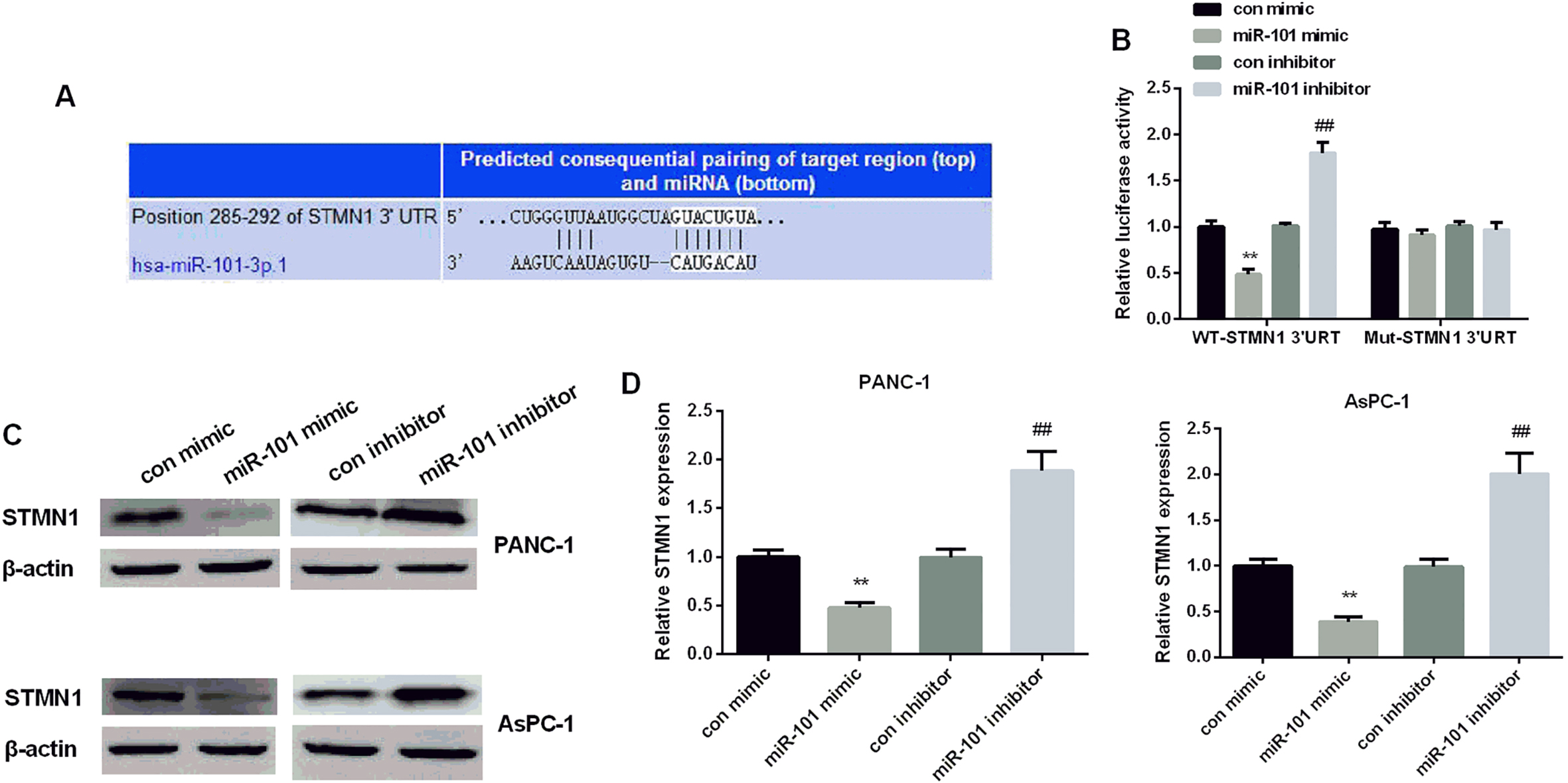
(A) STMN1 mRNA expressional level detected in PC and normal tissues (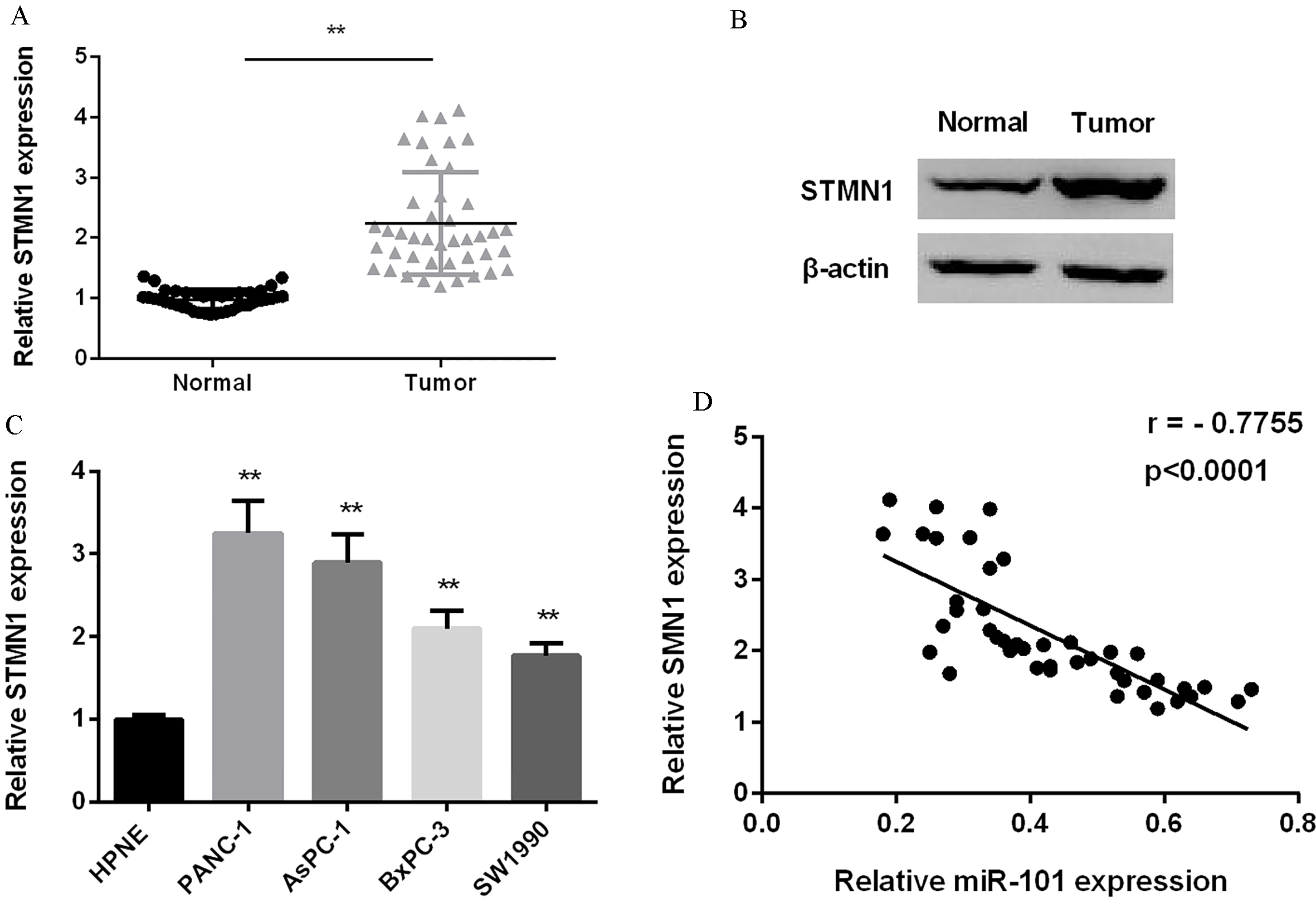
STMN1’s promoting effect and reversion the miR-101 suppressive effect on PC progression. (A) STMN1 expression tested in PC two cell lines after silencing STMN1. (B) Cell viability detected in PC two cell lines after inhibiting miR-101, silencing STMN1 and inhibiting both miR-101 and STMN1. (C) Relative invaded cells detected in PC two cell lines after inhibiting miR-101, silencing STMN1 and inhibiting both miR-101 and STMN1. 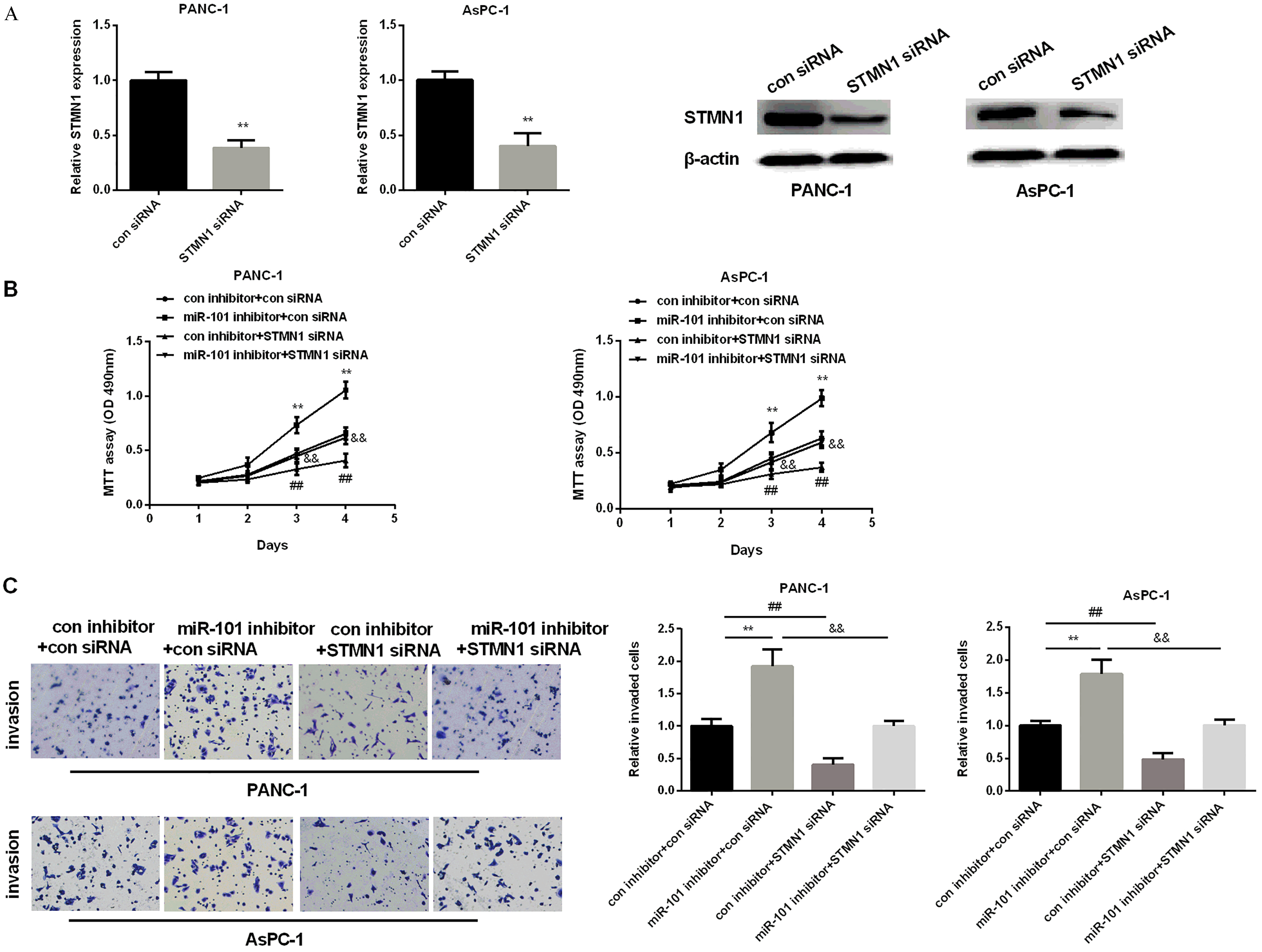
MiR-101 expression was declined significantly in PC
QRT-PCR was applied to quantify miR-101 expression in PC tissues and cells. As we saw in Fig. 1A, miR-101 average expression in 42 cases of PC tissues was obvious lower than normal tissues. Besides, miR-101 expression in all PC cell lines was declined significantly in comparison with normal cell line HPNE (Fig. 1B).
MiR-101 hindered PC cell proliferation and invasion
MiR-101 expression was down-regulated in both PANC-1 and AsPC-1 cells and had a different expression, so we chose the two cell lines for further investigation. Then, the effect of miR-101 on PC cell progression was then detected. We transfected miR-101 mimic or inhibitor into the two cell lines to achieve miR-101 re-expression or inhibition, separately. As we expected, the transfection efficiency was very successful, miR-101 expression was raised after treated with miR-101 mimic, but decreased after treated with miR-101 inhibitor in both two cell lines (Fig. 2A and B). MTT assay was applied to monitor PC cell proliferation. MTT results showed that miR-101 mimic inhibited cell viability while miR-101 inhibitor promoted that in two cell lines (Fig. 2C and D). In addition, Transwell assay was carried out to check cell invasion. Results showed that the invaded cells in miR-101 mimic group were reduced while they were increased in miR-101 inhibitor group (Fig. 2E and F). The results above indicated that miR-101 was tumor-suppressive in regulating PC proliferation and invasion.
MiR-101 suppressed STMN1 expression by targeting its 3’UTR
TargetScanHuman 7.1 was applied to preliminarily predict the targets of miR-101 to clarify miR-101’s mechanism in inhibiting PC cell proliferation and invasion. As Fig. 3A showed, the predicted consequential pairing of target region and miR-101 was at position 285-292 of STMN1 and STMN1 was predicted as a possible target of miR-101. The luciferase reporter assay was applied to further confirm the accuracy of prediction that STMN1 was a target gene of miR- 101 in PC. Co-transfected with miR-101 mimic and STMN1 3’-UTR WT showed a declined expression of luciferase activity, whereas con-transfected with miR-101 inhibitor and STMN1 3’-UTR WT showed an increased expression of luciferase activity in PANC-1 cells compared with control. However, there were no significant differences in MUT neither when the cells transfected with miR-101 mimic nor inhibitor (Fig. 3B). Subsequently, we used western blot and RT-PCR to study whether STMN1 expression was affected by intervention miR-101 expression in two cell lines. As the results exhibited, elevated miR-101 expression reduced STMN1 protein and mRNA level significantly in both two cell lines, while silencing miR-101 increased STMN1 level compared with control (Fig. 3C and D), suggesting that miR-101 repressed STMN1 expression in PC cells.
MiR-101 and STMN1 expression was negatively correlated in PC
QRT-PCR and western blot were applied to quantify STMN1 expression in PC tissues and cells. As shown in Fig. 4A and B, STMN1 mRNA and protein average expression in 42 cases of PC tissues were increased remarkably. And, its expression in all PC cell lines was elevated significantly in comparison with normal cell line HPNE (Fig. 4C). As Fig. 4D showed, the correlation between miR-101 and STMN1 expression was inverse.
STMN1 involved in the progression of PC regulated by miR-101
We first silenced STMN1 using STMN1 siRNA to study STMN1 role in PC. As we expected, STMN1 expression was reduced significantly in two cell lines after treated with STMN1 siRNA (Fig. 5A). Transwell and MTT assays were applied to test cell invasive ability and cell viability. As results shown in Fig. 5B, STMN1 siRNA reduced invaded cells of two PC cell lines, whereas miR-101 inhibitor increased the invaded cells. Moreover, STMN1 siRNA could reverse miR-101 inhibitor promoting effect on PC cell proliferation (Fig. 5B). Besides, cell invasion results showed that the invaded cells of two PC cell lines were reduced by STMN1 siRNA but raised by miR-101 inhibitor. Also, STMN1 siRNA partially attenuated the promoting effect of miR-101 inhibitor on PC cell invasion (Fig. 5C). All results indicated that miR-101 could curb PC cells proliferation and invasion by targeting STMN1.
Discussion
In most fields of malignant biology, miRNAs’ expression was observed almost abnormally. They involved in cell proliferation, invasion, metastasis and apoptosis as oncogenes or tumor suppressors [22]. MiR-101 was reported to participate in the progression of multiple tumors, but not reported in PC. So, we selected miR-101 as the aim of our research. Yang et al. stated that hepatocellular carcinoma progression was regulated by miR-101 [23]. Also, Imamura T and his colleagues suggested that cell migration and invasion was suppressed by increased miR-101 expression in gastric cancer [24]. However, miR-101’s role and its detailed molecular mechanism in PC cell proliferation and invasion remain unclear. In our study, we found that miR-101 expression was obviously reduced in PC, which in line with Jiang W’s study that miR-101 functioned as a tumor suppressor in PC cells [25]. We also showed that PC cell proliferation and invasion was inhibited by manual enhancing miR-101 expression. On the contrary, inhibiting miR-101 accelerated cell viability and invasive ability. All results above demonstrated that miR-101 negatively regulated PC progression as a tumor suppressor.
Previous studies had been showed that miRNAs effect on tumors depended on their downstream targets [26, 27]. TargetScanHuman was carried out to predict the candidate target of miR-101 and STMN1 was verified as a specific target of miR-101. Furthermore, STMN1 expression was inhibited when miR-101 expression increased but promoted by miR-101 inhibitor. We further confirmed that STMN1 was a directly target of miR-101 by luciferase reporter assay in PCNA-1 cells. Nevertheless, STMN1 expressional level in PC specimens was significantly raised and it was negatively correlated with miR-101 expression.
STMN1’s effect on malignancy was tumor- promoting according to previous studies. As a results showed, deprivation of STMN1 reduced cell viability and invasion in oesophageal squamous cell carcinoma while increased STMN1 expression facilitated cell progression [28]. STMN1 was also proved to play stimulative effect on prostate cancer development [21, 29]. Furthermore, Lu et al. showed that STMN1 was related to gallbladder cancer development [30]. To further explore whether STMN1 was involved in PC cell proliferation and invasion regulated by miR-101, we first detected STMN1 role in PC and found that STMN1 was increased and silencing STMN1 repressed cell proliferation and invasion, which consistent with the result of Li that STMN1 expression was elevated in PC patients and it was correlated with poor prognosis [31]. STMN1 siRNA attenuated miR-101 inhibitor promoting effect on PC cell proliferation and invasion.
In conclusion, we identified that via targeting STMN1, PC cell proliferation and invasion were suppressed by miR-101, suggesting that miR-101/STMN1 signaling involved in modulating PC cell viability and invasive ability was very essential and might provide a new idea for treating PC treatment in the future.
Footnotes
Acknowledgments
This work was supported by the National Nature Science Foundation of China (Grant No. 81671679), Shanghai Municipal Science and Technology Development Fund (Grant No. 15411952000) and the Shanghai Shenkang Hospital Development Center Foundation (Grant No. SHDC12014227).
