Abstract
This article has been retracted, and the online PDF replaced with this retraction notice.
Introduction
Endometrial cancer (EC) is one of the common malignant tumors of female reproductive system. At present, there is no satisfactory treatment method for advanced or recurrent patients [1]. PI3K/Akt/mTOR signaling pathway involves in various biological processes, including cell proliferation, apoptosis, migration, invasion, and drug resistance, which is closely related to tumor occurrence, progression, metastasis, and treatment [2, 3]. Drugs targeting PI3K/Akt/mTOR signaling pathway also received more and more attention [2, 4, 5]. Mammals rapamycin target of rapamycin (mTOR), as a downstream molecule of PI3K/Akt pathway, belongs to the serine/threonine protein kinase family. It can promote eIF4E activating target genes through phosphorylating 4EBP1 to remove the combining inhibition between 4EBP1 and eIF4E, thus to play a critical role in regulating cell proliferation, apoptosis, autophagy, and chemotherapy resistance [6]. The enhancement of mTOR expression and activity is correlated with the multiple cancer cell occurrence, progression, and prognosis, such as lung cancer [7], breast cancer [2], prostate cancer [8], and bladder cancer [4]. Several studies demonstrated mTOR overexpression and PI3K/Akt/mTOR signaling pathway enhancement in EC tissue, suggesting that PI3K/ Akt/mTOR pathway abnormal activation is associated with EC [1, 9, 10].
MicroRNA (miRNA) is a kind of small non-coding RNA in eukaryotes at about 22
Materials and methods
Main reagents and materials
EC cell line HEC-1A was purchased from Cellcook (Guangzhou, China). Human normal endometrial epithelial cell (HEEC) was obtained from PriCells (Wuhan, China). DMEM and RPMI 1640 medium were got from Hyclone (USA). Fetal bovine serum was bought from Gemini Bio-Products (USA). Penicillin-streptomycin, L-Glutamine, and sodium pyruvate were provided by CellGro (USA). TriPure RNA Isolation Reagent was purchased from Roche (USA). Lipofectamine RNAiMAX was obtained from Thermo Fisher Scientific. QuantiTect SYBR Green RT-PCR Kit was got from Qiagen (Germany). MiR-101 mimics, miR-NC, and PCR primers were provided by GE Dharmacon. siRNA sequences were synthetized by Genechem (Shanghai, China). Mouse anti human p-mTOR and mTOR monoclonal antibodies were got from CST (USA). Rabbit anti human p-4EBP1 antibody was bought from Abcam (USA). Mouse anti human
Clinical information
A total of 37 cases of EC patients received surgical treatment in Jinan maternity and child care hospital between Nov 2015 and Aug 2016 were enrolled. The median age was 57.2
The study protocol was approved by the Research Ethics Committee of Jinan maternity and child care hospital, and all patients gave their informed consent before study commencement.
Immunofluorescence
The tissue section was washed by PBS and blocked by serum. Next, it was incubated in mouse anti human mTOR monoclonal antibody (1:300) at 4
Cell culture
HEC-1A cells were maintained in DMEM containing 10% FBS, 1% penicillin-streptomycin, and 2 mM L-Glutamine. HEEC cells were cultured in RPMI 1640 medium supplemented with 10% FBS, 1% penicillin-streptomycin, and 1% sodium pyruvate.
Dual-luciferase activity detection
The PCR product containing the full length fragment of mTOR 3’-UTR was cloned to psi-CHECK2 plasmid and named as mTOR-UTR-wt. Another luciferase reporter vector containing the mutant of mTOR 3’-UTR was constructed and named as mTOR-UTR-mut. Lipofectamine RNAiMAX was used to co-trans- fect 1
HEC-1A cell transfection
HEC-1A cells were divided into five groups, including miR-NC, miR-101 mimic, si-NC, si-mTOR, and miR-101
qRT-PCR
Total RNA was extracted using the TriPure RNA Isolation Reagent kit. QuantiTect SYBR Green RT-PCR Kit was applied for qRT-PCR detection. The qRT-PCR reaction system contained 10.0
Western blot
The cells were lysed by SDS containing 1 mM PMSF and transferred to the 1.5 ml EP tube. After centrifuged at 10000 g and 4
Flow cytometry
The cells were collected by 0.25% enzyme without EDTA and transferred to 15 ml centrifuge tube. Next, the cells were added to 300
Cell invasion assay
Boyden Chamber Assay was used to test cell invasion. A total of 1
Colony formation assay
The cells were seeded in the 10 cm dish at 100/dish. After 2–3 weeks, the cells were fixed by 4% parafor- maldehyde and stained by Giemsa. The colony formation rate
Statistical analysis
All data analysis was performed on SPSS 18.0 software. All population has a normal distribution. The measurement data were depicted as mean
MiR-101 declined, while mTOR upregulated in EC tissue and cell line. (A) qRT-PCR detection of gene expression; (B) Immunofluorescence detection of mTOR protein expression; (C) The binding site between miR-101 and the 3’-UTR of mTOR mRNA; (D) Dual luciferase reporter gene assay.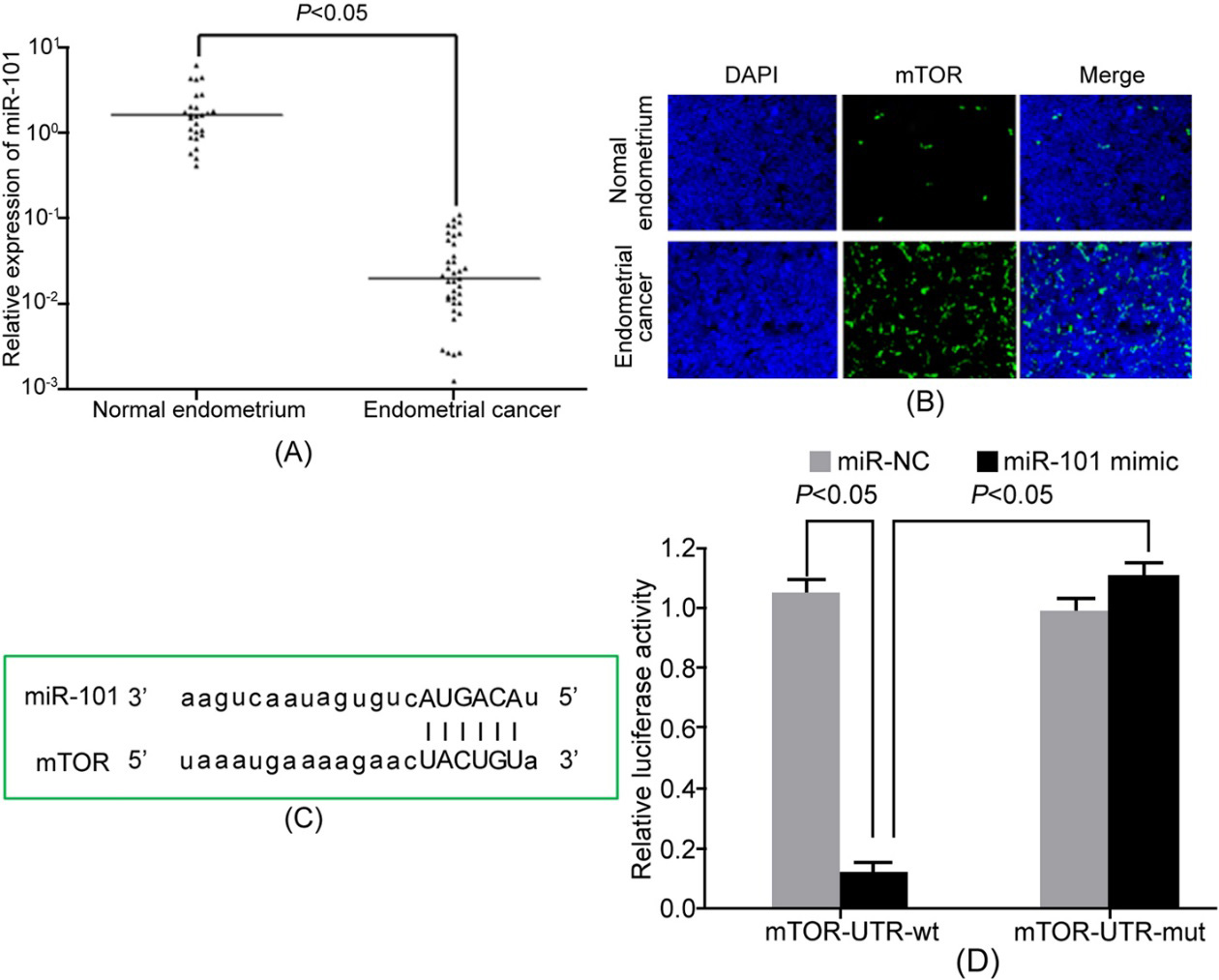
MiR-101 downregulation was related to the enhancement of mTOR and 4EBP1 phosphorylation in HEC-1A cells. (A) qRT-PCR detection of gene expression; (B) Western Blot detection of protein expression; (C) qRT-PCR detection of gene expression; (D) Western Blot detection of protein expression.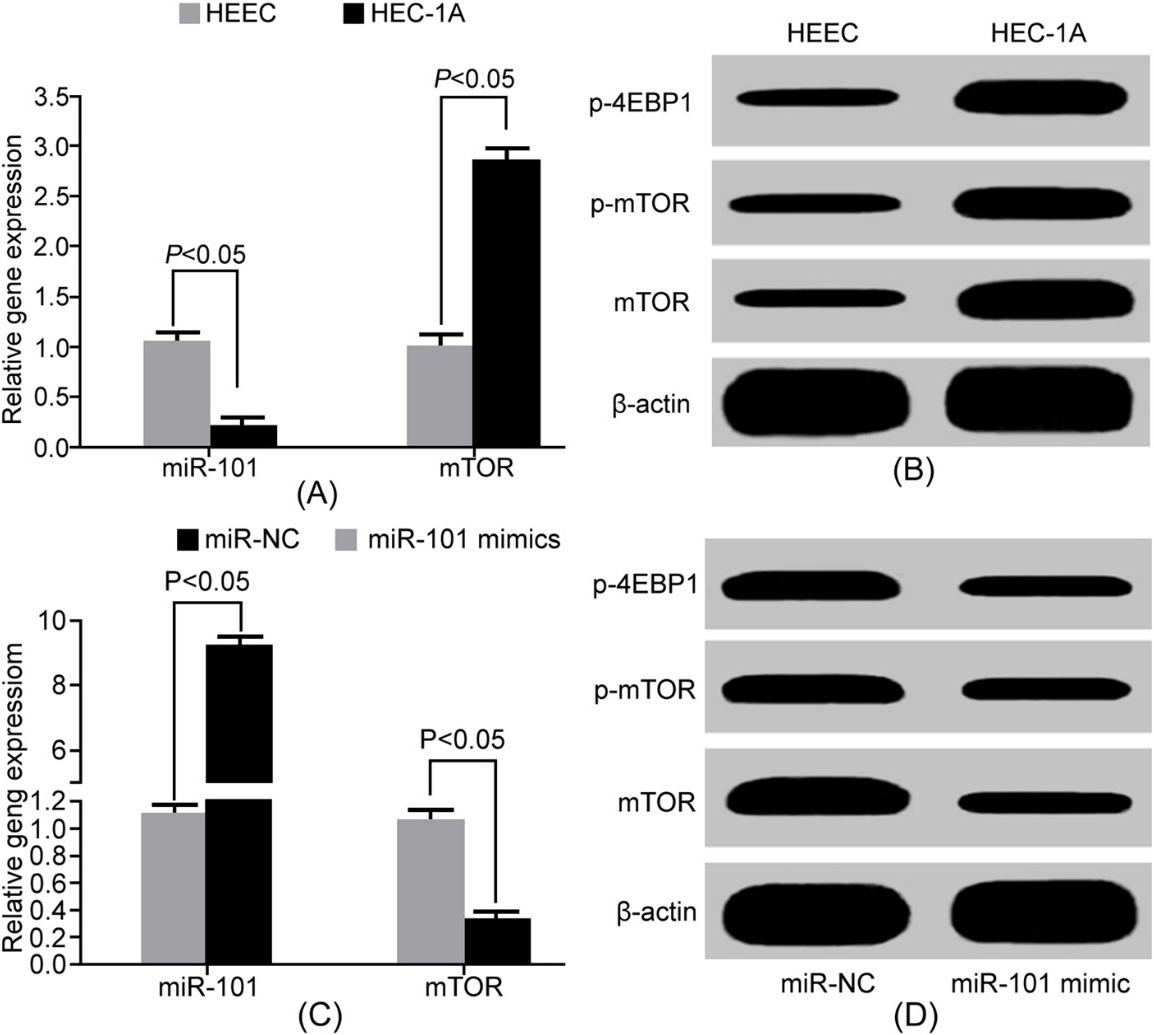
MiR-101 declined, while mTOR upregulated in EC tissue and cell line
qRT-PCR detection showed that miR-101 expression significantly declined in EC tissue compared with normal endometrial tissue (Fig. 1A). Immunofluorescence detection revealed that mTOR expression was obviously higher in EC tissue than the normal control (Fig. 1B). It suggested that miR-101 may target mTOR. MiR-101 downregulation may play a role in elevating mTOR expression and promoting EC pathogenesis. microRNA.org online prediction demonstrated the complimentary binding site between miR-101 and the 3’-UTR of mTOR mRNA (Fig. 1C). Dual luciferase reporter gene assay exhibited that miR-101 overexpression markedly reduced the relative luciferase activity in HEK293T cells (Fig. 1D), confirming the targeted relationship between miR-101 and mTOR.
MiR-101 upregulation attenuated mTOR activity, cell invasion, and proliferation, and facilitated cell apoptosis. (A) qRT-PCR detection of gene expression; (B) Western Blot detection of protein expression; (C) Colony formation assay; (D) Transwell assay detection of cell invasion; (E) Flow cytometry detection of cell apoptosis. a. 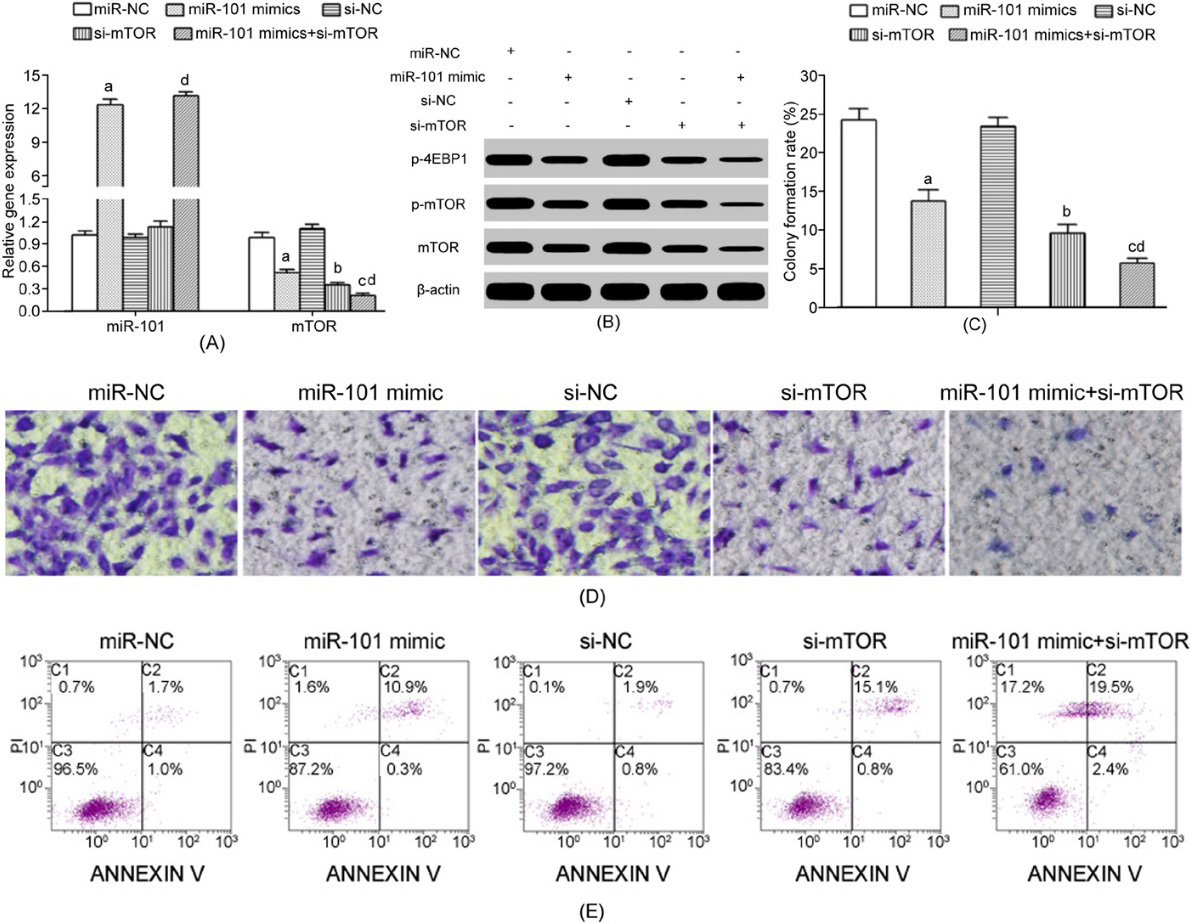
qRT-PCR results showed that miR-101 reduced, while mTOR mRNA upregulated in HEC-1A cells compared with HEEC cells (Fig. 2A). Western blot exhibited that mTOR, p-mTOR, and p-4EBP1 levels markedly elevated in HEC-1 cells compared with HEEC cells (Fig. 2B). MiR-101 mimics significantly decreased mTOR mRNA expression in HEC-1A cells (Fig. 2C), leading to mTOR, p-mTOR, and p-4EBP1 protein levels reduction (Fig. 2D). It indicated that mTOR activity enhancement in EC cells was based on mTOR upregulation.
MiR-101 upregulation attenuated mTOR activity, cell invasion, and proliferation, and facilitated cell apoptosis
MiR-101 mimic and/or si-mTOR transfection obviously reduced mTOR (Fig. 3A), p-mTOR, and p-4EBP1 (Fig. 3B) expressions, decreased colony formation (Fig. 3C), decreased invasion (Fig. 3D), and enhanced apoptosis (Fig. 3E) in HEC-1A cells.
Discussion
The pathogenesis of EC is complicated and has not been fully elucidated. Therefore, in-depth investigation of the EC pathogenesis is of great significance to help diagnose, enhance the curative effect, and improve prognosis. Signaling pathway composed of extracellular ligands, cell receptors, and a series of downstream molecular cascade can regulate a variety of biological processes. The role of abnormal signal transduction in tumor receives more and more attention. PI3K/Akt is the most important protein located at the upstream of mTOR. PI3K/Akt/mTOR signaling pathway is the main dependent pathway to help mTOR plays a regulatory role. PI3K/Akt/mTOR signaling pathway has been confirmed to play an important role in tumor pathogenesis as it participates in the regulation of multiple biological processes, such as cell proliferation, cycle, apoptosis, and invasion [2, 3]. Phosphatidylinositol-3 kinase (PI3K) auto-conformation change was activated under the stimulation of growth factor, mitogen, and other signaling molecules. Activated PI3K can promote phosphatidylinositol(4,5)-bisphosphate (PIP2)phosphorylation to phosphatidylinositol(3,4,5)-trisph- osphate (PIP3) through its p110 catalytic subunit. PIP3 can act on the PH domain of Akt/PKB to change conformation. Moreover, it can phosphorylate Akt at Ser473 and Thr308 under the effect of PDK1 and PDK2. Activated Akt kinase further phosphorylated mTOR (mTORC1 and mTORC2), leading to it binding with the eIF-4E binding protein1 (4EBP1) on eukaryotic initiation factor 4E (eIF-4E). The 4EBP1 was phosphorylated and reduced affinity with eIF-4E, thus blocking the suppression of 4EBP1 on eIF-4E binding and translation. Dissociative eIF-4E band with eIF-4G/eIF-4A/eIF-4B to form eIF-4F complex and further coupled to the cap structure of mRNA 5’-terminal. It promoted mRNA translation and protein synthesis, thus accelerating cell cycle and mitosis proliferation [18]. In addition to cell proliferation and cycle, PI3K/Akt/mTOR also plays an inhibitory role on cell apoptosis through acting on Bax/Bcl-2 [19]. Moreover, mTORC2 participates in Akt protein activation via Ser473 phosphorylation, thus forming the positive feedback of PI3K/Akt/mTOR pathway [20]. Several studies showed that mTOR enhancement exhibited tumorpromoting role in EC occurrence [21, 22]. The inhibitors targeting mTOR demonstrated the antitumor effect on EC [23]. the expression and function of mTOR increase in the incidence of endometrial carcinoma showed the role that promote cancer, and with the targets of mTOR inhibitors are showed on the anti-tumor effect of endometrial carcinoma [23]. It was revealed that miR-101 markedly decreased in EC tissue [17], suggesting that miR-101 also played a tumor suppressor role in EC. Bioinformatics analysis showed the binding site between miR-101 and the 3’-UTR of mTOR mRNA. This study investigated the role of miR-101 in affecting EC cell line HEC-1A proliferation, apoptosis, and invasion, and mediating mTOR expression.
Our results demonstrated that miR-101 level obviously reduced, while mTOR significantly upregulated in EC tissue compared with normal endometrium, suggesting their potential targeted regulatory relationship. Dual luciferase report gene assay confirmed the targeted regulatory relationship between miR-101 and mTOR. Hiroki et al. [24] reported that miR-101 level significantly declined in EC tissue compared with normal endometrium. Patients with lower expression level showed shorter progression free survival. Thus, miR-101 could be treated as the independent predictor of survival and prognosis. Konno et al. [25] exhibited that miR-101 expression markedly downregulated in EC tissue compared with para-carcinoma tissue. Hiroki et al. [26] also found miR-101 downregulation in EC tissue through microarray screening. Our study demonstrated miR-101 was significantly decreased in EC tissue compared with control, which was in accordance with Hiroki [24] and Konno [25]. In addition, miR-101 abnormal reduction was also found in EC cell line HEC-1A. Konno et al. [25] discovered that miR-101 declined in EC cell line SPAC-1-L, HEC-50, and HOUA-I compared with normal endometrial epithelial cells, which was similar to our results. Elevated miR-101 markedly reduced mTOR, p-mTOR, and p-4EBP1 expressions in HEC-1A cells, indicating that mTOR phosphorylation enhancement was the reason of mTOR upregulation in EC cells. Further investigation found that miR-101 mimic and/or si-mTOR transfection obviously reduced mTOR, p-mTOR, and p-4EBP1 expressions, decreased colony formation, decreased invasion, and enhanced apoptosis in HEC-1A cells. Hiroki et al. [24] showed that miR-101 upregulation apparently inhibited EC cell line SPAC-1-L proliferation. Konno et al. [25] reported that miR-101 overexpression obviously restrained EC cell proliferation and induced apoptosis and senescence. Konno et al. [25] also demonstrated that miR-101 can suppress the epithelial-mesenchymal transition (EMT) and attenuated tumor cell migration and invasion. Bae-Jump et al. revealed that mTOR siRNA significantly enhanced Ishikawa and ECC-1 cell apoptosis induced by cisplatin. All of these researches reported that miR-101 upregulation or mTOR reduction may attenuate the malignant biological characteristic of EC cells. This study demonstrated that miR-101 downregulation promoted EC pathogenesis by enhancing PI3K/Akt/mTOR signaling pathway.
Conclusion
MiR-101 downregulated and mTOR elevated in EC tissue. MiR-101 decreased HEC-1A cell proliferation and invasion, and upregulated apoptosis through targeting mTOR to attenuate PI3K/Akt/mTOR signaling pathway activity.
