Abstract
BACKGROUNDS:
The purpose of this study was to investigate clinical role and functional effects of miR-431 expression in papillary thyroid carcinoma (PTC).
METHODS:
Expression of miR-431 in PTC patient tissue samples and plasma samples was examined by using qRT-PCR methods. Cell migration and invasion capacity were evaluated using transwell assays. Western blot analysis was performed to detect protein expression after miR-431 overexpression in PTC cells.
RESULTS:
We demonstrated that miR-431 expression was lower in PTC tissues and plasma samples compared to their corresponding controls. MiR-431 expression was particularly lower in PTC patients with lymph node (LN) metastasis. In vitro, miR-431 overexpression significantly inhibited cell migration, invasion and EMT process by upregulating E-cadherin and downregulating Vimentin expression. Additionally, wedemonstrated that miR-431 overexpression suppressed Hedgehog (Hh) signaling pathway by downregulating Gli1 expression.
CONCLUSION:
Our results indicated that miR-431 could serve as a predictor for PTC patients with positive lymph node metastasis and a potential target of PTC treatment.
Introduction
Papillary thyroid carcinoma (PTC) is the common type of thyroid malignant tumor [1]. Treatments after surgical resection combined with radioiodine and levothyroxine treatment present good survival rate for patients [2]. However, due to tumor relapse and distant metastasis, more than 10% of PTC patients show bad response to standard treatments [1, 3]. Thus, to investigate the molecular mechanisms underlying PTC progression is urgently needed.
Recently, microRNAs, a class of endogenous, small (19–25 nucleotides) RNA, could regulate gene expression by targeting the 3’-untranslated regions (3’-UTRs) of their target mRNAs and lead to mRNAs degradation or translational suppression [4, 5]. In previous findings, miR-431 is involved in tumor development and progression. Downregulation of microRNA-431 by human interferon-
In the study, we demonstrated that miR-431 expression was lower in PTC tissue and plasma samples compared to their corresponding controls. MiR-431 overexpression inhibited cell migration, invasion and EMT process. Additionally, we demonstrated that miR-431 overexpression suppressed hedgehog (Hh) signaling pathway by downregulating Gli1 expression. Therefore, these results implied that miR-431 could serve as a predictor for PTC patients with lymph node metastasis and a potential target of PTC treatment.
Materials and methods
Patients and tissue samples
Sixty-six PTC patient tissue samples and adjacent normal tissue samples were obtained from PTC patients who underwent resection at the First Affiliated Hospital of Zhengzhou University from January 2013 to March 2015. In additional, the above 66 PTC patient plasma samples (before surgery) and 40 patients with nodular goiter (NG) were also obtained at the First Affiliated Hospital of Zhengzhou University from January 2013 to March 2015. No patient received radiation therapy or chemotherapy before surgery. The study was approved by Ethics Committee of First Affiliated Hospital of Zhengzhou University. Written informed consents were obtained from all patients.
Cell line culture
Two human PTC cell lines including PTC-1 and BCPAP were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). PTC-1 and BCPAP cells were maintained at 37
RNA extraction and quantitative RT-PCR (qRT-PCR)
Total RNA was isolated from tissues or plasma samples and cells tissues using Trizol (Invitrogen Technologies, Carlsbad, CA, USA) according to the manufacturer’s instructions. RNA was reversed transcribed to cDNA by Prime Script™ RT reagent kit (Takara, Dalian, Liaoning, China). The SYBR Premix Ex Taq
Cell transfection
Mir-431 mimic, miR-431 inhibitor and miR-negative control are purchased from Ribobio (Guangzhou, Guangdong, China). The vectors were transfected into PTC-1 and BCPAP cells using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions.
Cell migration and invasion assays
Cell migration or invasion assay was performed by using 8
MiR-431 expression is significantly downregulated in PTC tissue and plasma samples. (A) The expression of miR-431 was detected using qRT-PCR analysis in 66 pairs of PTC patient tissue samples and adjacent normal tissue samples. (B) The expression of miR-431 was detected using qRT-PCR analysis in 66 cases of PTC patient tissue samples and 40 cases of patients with nodular goiter (NG). (C)–(D) MiR-431 expression in both tissues and plasma samples were lower in patients with lymph node metastasis than that in patients without lymph node metastasis. *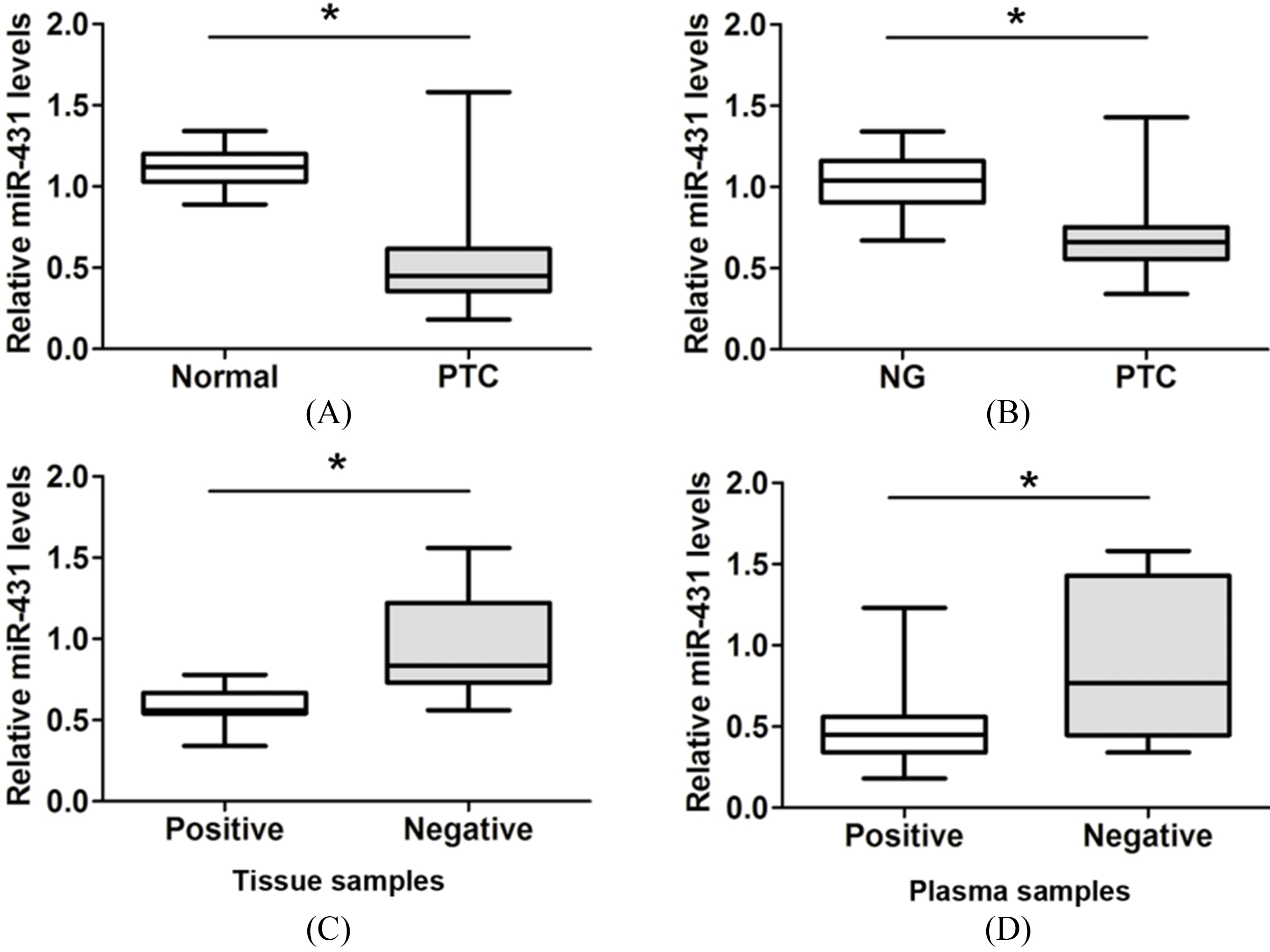
Protein was extracted with lysis buffer (0.5 M Tris-HCl at pH 6.8, 2 mM EDTA, 10% glycerol, 2% SDS, and 5%
Statistical analyses
All data are shown as mean
MiR-431 inhibits cell migration and invasion in PTC-1 cells. (A)–(B) Cell migration and invasion assays were shown after PTC-1 cells were transfected with miR-431 mimic, miR-431 inhibitor or miR-NC. (C)–(D) Cell migration and invasion number was calculated after PTC-1 cells were transfected with miR-431 mimic, miR-431 inhibitor or miR-NC. *
MiR-431 inhibits cell migration and invasion in BCPAP cells. (A)–(B) Cell migration and invasion assays were shown after BCPAP cells were transfected with miR-431 mimic, miR-431 inhibitor or miR-NC. (C)–(D) Cell migration and invasion number was calculated after BCPAP cells were transfected with miR-431 mimic, miR-431 inhibitor or miR-NC. *
MiR-431 inhibits cell EMT process and Hedgehog signaling by affecting Gli1 expression in PTC. (A) The protein expression of Gli1, E-cadherin or Vimentin was detected after PTC-1 cells were transfected with miR-431 mimic or miR-NC. (B) The protein expression of Gli1, E-cadherin or Vimentin was detected after BCPAP cells were transfected with miR-431 mimic or miR-NC. *
MiR-431 expression is significantly downregulated in PTC tissue and plasma samples
To determine the clinical role of miR-431 expression in PTC, we examined the relative expression of miR-431 in 66 pairs of PTC patient tissues samples and adjacent normal tissue samples from PTC patients who underwent resection. As shown in Fig. 1A, our results demonstrated that miR-431 expression was obviously lower in PTC tissue samples compared to adjacent normal tissue samples (
MiR-431 inhibits cell migration and invasion of PTC
To further explore whether miR-431 expression associated with cell migration and invasion, the gain and lose function assays were performed by transfecting with miR-431 mimic or miR-431 inhibitor in PTC-1 and BCPAP cells. Cell migration and invasion ability tested by transwell assay showed that overexpression of miR-431 dramatically inhibited cell migration and invasion ability compared to the control groups in PTC-1 cells, but reduce miR-431 expression led to a significant enhance of cell migration and invasion capacities (Fig. 2A and D). Otherwise, cell migration or invasion ability was inhibited in BCPAP cells after overexpression of miR-431 compared to the control group, but reduce miR-431 expression led to a significant enhanced migration or invasion ability (Fig. 3A and D). These results indicated that miR-431 inhibited cell migration and invasion of PTC.
MiR-431 inhibits cell EMT process and Hedgehog signaling by affecting Gli1 expression in PTC
EMT plays a pivotal role in normal physiological development and enables the cancer cells to gain invasive properties and activation of Hedgehog signaling were reported to promote tumor EMT process [8, 9]. Herein, western blot analysis showed that overexpression of miR-431 dramatically upregulated E-cadherin expression, but downregulated the vimentin expression, compared to the control group in PTC-1 and BCPAP cells (Fig. 4A and B). Moreover, miR-431 overexpression also significantly downregulated Gli1 protein expression in PTC-1 and BCPAP cells compared to control groups (Fig. 4A and B), which indicated the miR-431 overexpression suppressed the Hedgehog signaling in PTC cells. Thus, these results indicated that miR-431 overexpression could inhibit cell EMT process and Hedgehog signaling by affecting Gli1 expression in PTC cells.
Discussion
MicroRNAs regulate gene-expression in biological and pathophysiological processes of tumors including PTC and serve as important biomarkers for prognosis. MicroRNA-451 is identified as a prognostic marker for diagnosis and lymph node metastasis of papillary thyroid carcinoma [10]. MiR-199a-3p downregulation in thyroid tissues is associated with invasion and metastasis of papillary thyroid carcinoma [11]. MiR-577 inhibits papillary thyroid carcinoma cell proliferation, migration and invasion by targeting SphK2 [12]. Our results demonstrated that miR-431 expression was lower in thyroid carcinoma tissues compared to adjacent normal tissues. Furthermore, miR-431 expression was significantly downregulated in plasma samples of PTC patients compared to patients with nodular goiter (NG). Moreover, miR-431 expression was particularly lower in PTC patients with lymph node (LN) metastasis compared with no lymph node (LN) metastasis. Thus, these results indicated that reduced miR-431 expression functioned as a predictor for patients with lymph node (LN) metastasis, which provided an important clinical value.
EMT is characterized as a switch from an epithelial phenotype of polarized cells to a mesenchymal phenotype of cells that lack polarity, which is crucial for tumor invasion [13]. Our results indicated that overexpressed miR-431 inhibited cell EMT process by upregulating epithelial maker E-cadherin expression, but downregulating mesenchymal maker Vimentin expression in PTC cells. In previous study, microRNA-431 was found to inhibit migration and invasion of hepatocellular carcinoma cells by targeting the ZEB1-mediated epithelial-mensenchymal transition [7]. Herein, we verified that miR-431 could inhibit cell invasion of PTC by suppressing the EMT process.
The hedgehog (Hh) signaling pathway is a crucial mediator of tumor cell growth, invasion and metastasis [14]. Inhibition of Hh pathway could reverse the EMT process. CCL2/CCR2 axis induces hepatocellular carcinoma invasion and epithelial-mesenchymal transition in vitro through activation of the Hedgehog pathway [15]. SHH gene silencing suppresses epithelial-mesenchymal transition, proliferation, invasion, and migration of cervical cancer cells by repressing the hedgehog signaling pathway [16]. Galectin-1 induces invasion and the epithelial-mesenchymal transition in human gastric cancer cells via non-canonical activation of the hedgehog signaling pathway [8]. In the study, we found that increased miR-431 inhibited Hedgehog signaling by affecting Gli1 expression PTC, which indicated that miR-431 play an important role in regulating Hedgehog signaling. Of course, the underlying molecular mechanism between miR-431 and Hedgehog signaling need to be investigated in the further.
In conclusion, our results indicated that miR-431 expression was significantly downregulated in tissues and plasma samples of PTC patients. Lower miR-431 expression acts as predictor of patients with lymph node (LN) metastasis. In vitro, we demonstrated that miR-431 inhibited cell invasion and EMT process. In addition, we found that the hedgehog (Hh) signaling pathway was inhibited by miR-431 overexpression. Thus, these results indicated that miR-431 functions as a target of PTC treatment.
Footnotes
Acknowledgments
The study was supported by foundation of Henan Educational Committee (18A320080).
Conflict of interest
The authors declare that they have no competing interests.
