Abstract
BACKGROUND AND OBJECTIVE:
To investigate the diagnostic potentials of microRNA-451(miR-451) in papillary thyroid carcinoma (PTC) diagnosis and lymph node (LN) metastasis, formalin-fixed, paraffin-embedded (FFPE) tissue specimens corresponding to PTC tumors (
METHODS:
The usefulness of miR-451 expression as a prognostic marker for diagnosis of PTC malignancies was evaluated by Receiver Operating Curve (ROC). We reported that when compared to those in NAT, the levels of miR-451 in FFPE tissues from various stages of PTC patients (
RESULTS:
ROC analyses revealed that AUC was 0.626 for malignancies and was 0.690 for lymph node involvement, respectively, suggesting that miR-451was a modest blood based biomarker for PTC malignancies and lymph node metastasis.
CONCLUSIONS:
We concluded that miR-451 expression is a reliable FFPE tissue biomarker for PTC malignancies and it may have potentials to become a noninvasive, blood-based biomarker for PTC diagnosis and evaluation of LN status.
Introduction
Thyroid carcinoma (TC) is the most common human endocrine malignancies, accounting for 95% of all endocrine tumors. Papillary thyroid carcinoma (PTC) has the most common histotype (approximately 80% of all TCs); its incidence has steadily increased over years [1]. Most PTCs can be effectively treated, with 5 years of survival rate over the 95%. However, a small fraction of patients who failed to response to the treatments could progress to a grave stage with local lymph node metastasis [2]. Despite to recent advances in tumor diagnosis and patient cares; it has not had a major breakthrough with respect to identification of reliable PTC-specific prognostic markers. Genomic analyses of large cohorts of PTC patients have identified quite a few tumor associated molecular markers, including
MicroRNAs (miRNAs) are small non-coding RNAs (19–23 bp in size) well distributed in the whole spectrums of living organisms and are known to participate in the regulation of many physiological and pathological processes [6]. By targeting mostly on 3’ untranslational region (3’UTR) of various Messenger RNAs (mRNAs), miRNA affect gene expression through inhibition of gene translation [7]. Numerous studies have demonstrated that miRNAs play critical roles in oncogenesis of various cancers. Expression of a group of miRNAs, known as oncomiR [8], including but not limited to miR-17-92, miR-21, miR-155 and miR-372, are overexpressed in cancers, while another group of miRNAs (tumor-suppressive miRNAs), including but not limited to Let-7, miR-15a, miR-16-1 are underexpressed in cancers [8, 9]. There are yet another examples such as miRNA-125b, act as an oncomiR in the majority hematologic malignancies and as a tumor suppressor in many solid tumors [10]. These studies indicated a complex network system in which microRNAs play a key role in cell type specific oncogenesis; these findings also implied that comprehensive analyses of microRNA expression could result in the identification of biomarkers useful for diagnosis and prognosis of tumors.
Recent studies have implicated that deregulation of miRNAs was involved in the pathogenesis of thyroid cancers. A comprehensive miRNA profiling indicated that a handful of miRNA were deregulated in tissues of PTC patients when compared to that of normal counterparts [11, 12]. In particular, Lee et al. [13] and Wang et al. [11] have independently demonstrated that higher expression of miR-222 and miR-146b was associated with poor prognosis and lymph node involvement of thyroid cancers. Expression of other species of miRNAs in tissues of PTC, however, has not been well documented.
In the present study, we investigated if miR-451 can be a prognostic marker for malignancies of PTC by retrospective analyses its expression in two most commonly available specimens, formalin-fixed paraffin-embedded tissues (FFPE) and venous serum, from PTC patients prior to their surgeries. The correlation between miR-451 levels and the patients’ clinicopathological features was analyzed.
Materials and methods
Patents and ethics statement
This study was approved by the Ethics Committee of Xinhua Hospital, Shanghai Jiaotong University School of Medicine. Written informed consents were obtained from all participants of this study.
Two groups of patients were recruited for the current study. The first cohort of patients, who participated in the study of miRNA-451 in FFPE preserved tissues, was comprised of 60 patients (13 males and 47 females). These patients were diagnosed with papillary thyroid carcinoma (PTC) and have undergone thyroidectomy at Xinhua Hospital, Shanghai Jiaotong University School of Medicine, between July 2009 and July 2014. The data on patients’ clinical features including gender, age, tumor size, histological types, lymph node status and serum thyroglobulin (GT) levels at diagnosis, were retrospectively collected. Diagnosis of PTC were certified by a group of pathologists in accordance with the Guideline of WHO [14]. The second cohort of patients involved 70 PTC patients (28 males and 42 females) with various stages of PTC. These patients, along with patients determined to have benign nodules (
Clinicopatholgical characteristics of PTC patients in this study
Clinicopatholgical characteristics of PTC patients in this study
For every PTC patient (
For every PTC patient (malignant,
Total RNA extraction
Total RNA was isolated from FFPE tissues using a miRNeasy FFPE Kit (QIAGEN) according the manufactory standard protocol without any modifications. For isolation of miRNAs from patient’s sera, sera stored at 80
Quantitative real-time PCR
For quantitative detection of miRNA, purified serum or tissue total RNA was first used to convert to miRNA gene specific cDNA by reverse transcription reactions using a standard protocol provided by Applied Biosystems, (Grand Island, NY, USA). For PCR amplification, reaction mix was prepared in a 96 well plate; each reaction (5 ml) contained 2.5 ml TaqMan Universal PCR Master Mix II, 0.25 ml miRNA-specific primer/probe mixes, and 2.25 ml cDNA template prepared from above. The PCR reaction was prepared in triplicate. Amplification was performed in an ABI 7900HT thermocycler (Applied Biosystems) using following cycling parameters: 95
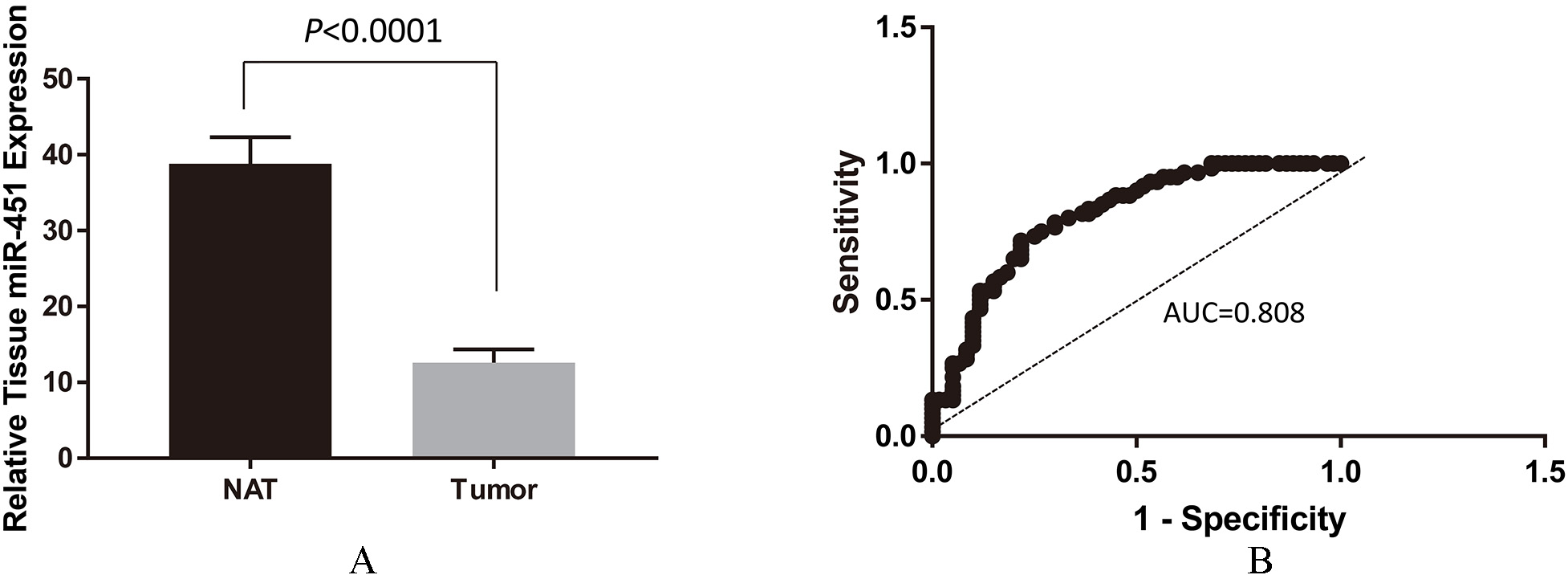
Analysis of miR-451 expression in FFPE tissues from tumors and normal tissues adjacent to tumors (NAT) in 60 PTC patients. (A) Downregulation of miR-451 expression in tumors vs NAT. Total RNA were extracted from paired (tumor and NAT) FFPE samples from patients. Expression of miR-451 was performed by qRT-PCR and normalized by expression of an internal control microRNA RUN-44. The result was shown as Mean
Statistical analyses were performed with the SPSS statistical package, version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows. The images were made with GraphPad Prism version 7.0 (GraphPad Software, San Diego, CA, USA). In the most cases, data were expressed as Whisker Plot to demonstrate the Medium and distribution of values. In some cases, data were also expressed as the Mean
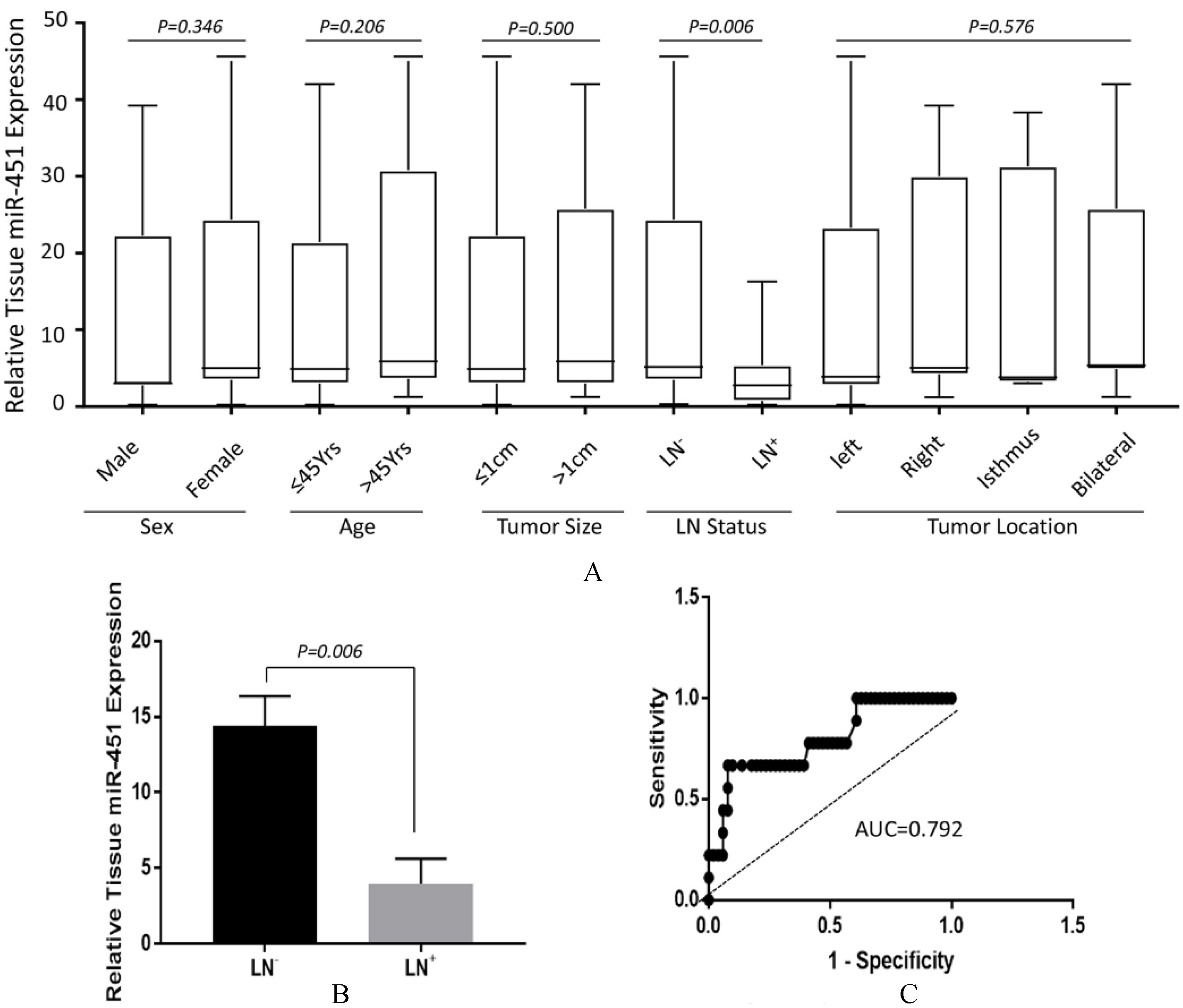
Analysis of correlations between tissue miR-451 expression with clinicopathological features of PTC patients. (A) Expression of miR-451 in PTC tissue with respect to gender, age, tumor size, LN status and tumor location. Data were expressed by Whisker Plot to demonstrate the medium and the distribution of samples. Except for status of lymph node involvement (
Comparison of miR-451 expression between PTC and Normal Adjacent Tissues in FFPE cohort
To explore the expression and significance of miR-451 in oncogenesis of PTC, we analyzed expression of miR-451 in FFPE tissues from patients (
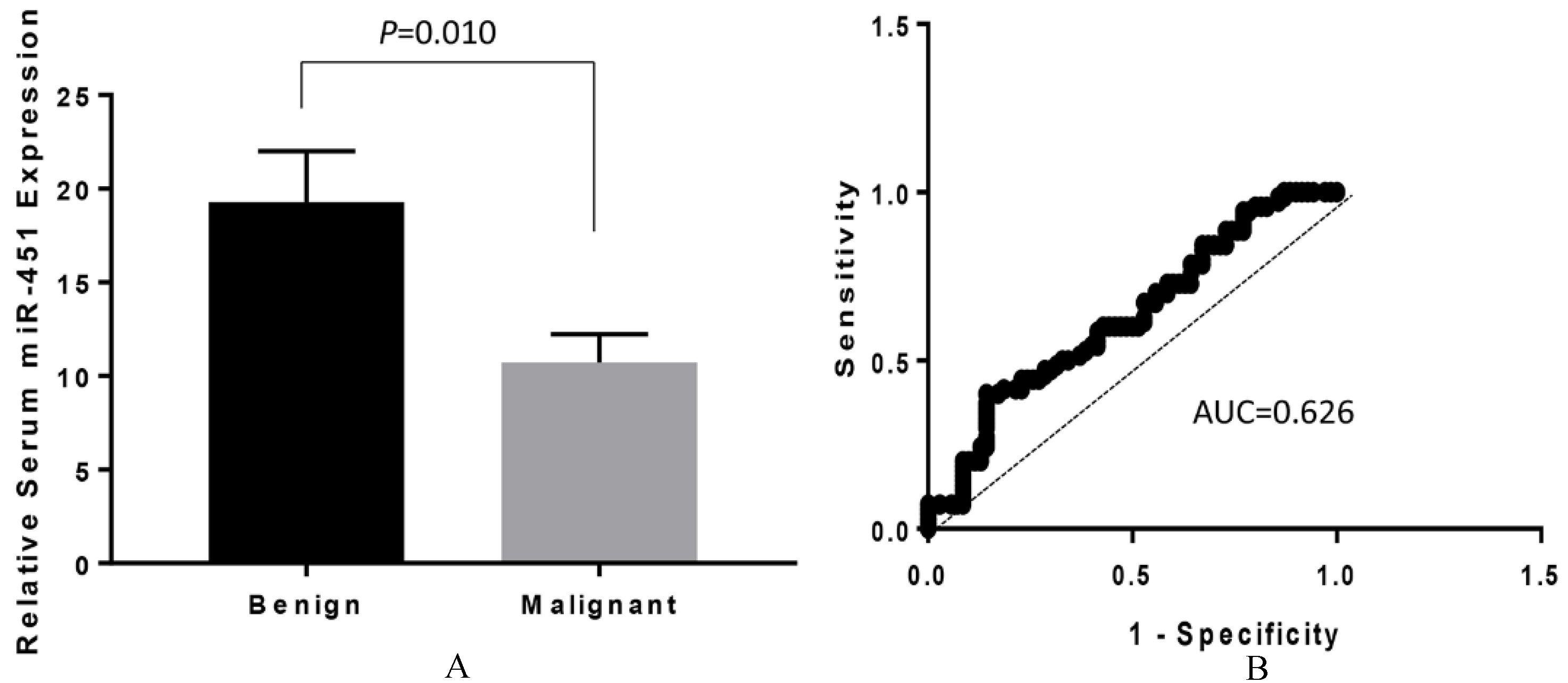
Analysis of miR-451 expression in sera from PTC patients. (A) Downregulation of serum miR-451 in patients with malignant lesions (
Low expression of miR-451 in tumors prompted us to further investigate if its expression pattern is associated with any parameters with respect to clinicopathological features of PTC. We performed miRNA-451 analyses on all participants and retrospectively categorized patients according to different variables, including gender, age, tumor size, lymph node (LN) status and tumor location. Significance of changes of miR-451 level was analyzed with Mann-Whitney U test or one-way ANOVA (tumor location). As shown in Fig. 2A, there were no significant difference in miR-451 levels observed in the sub-group distributed by gender (12.50
Serum miR-451 level was significantly lower in patients with PTC than those with benign nodules
To evaluate the potential of miR-451 as a blood-based biomarker, sera from patients with malignant PTC (
Reduced expression of serum miR-451 correlated with lymph node metastasis
To investigate if serum miR-451 level can be an indicator of aggressiveness of tumor, we performed miR-451 expression in sera from all malignant PTC patients (
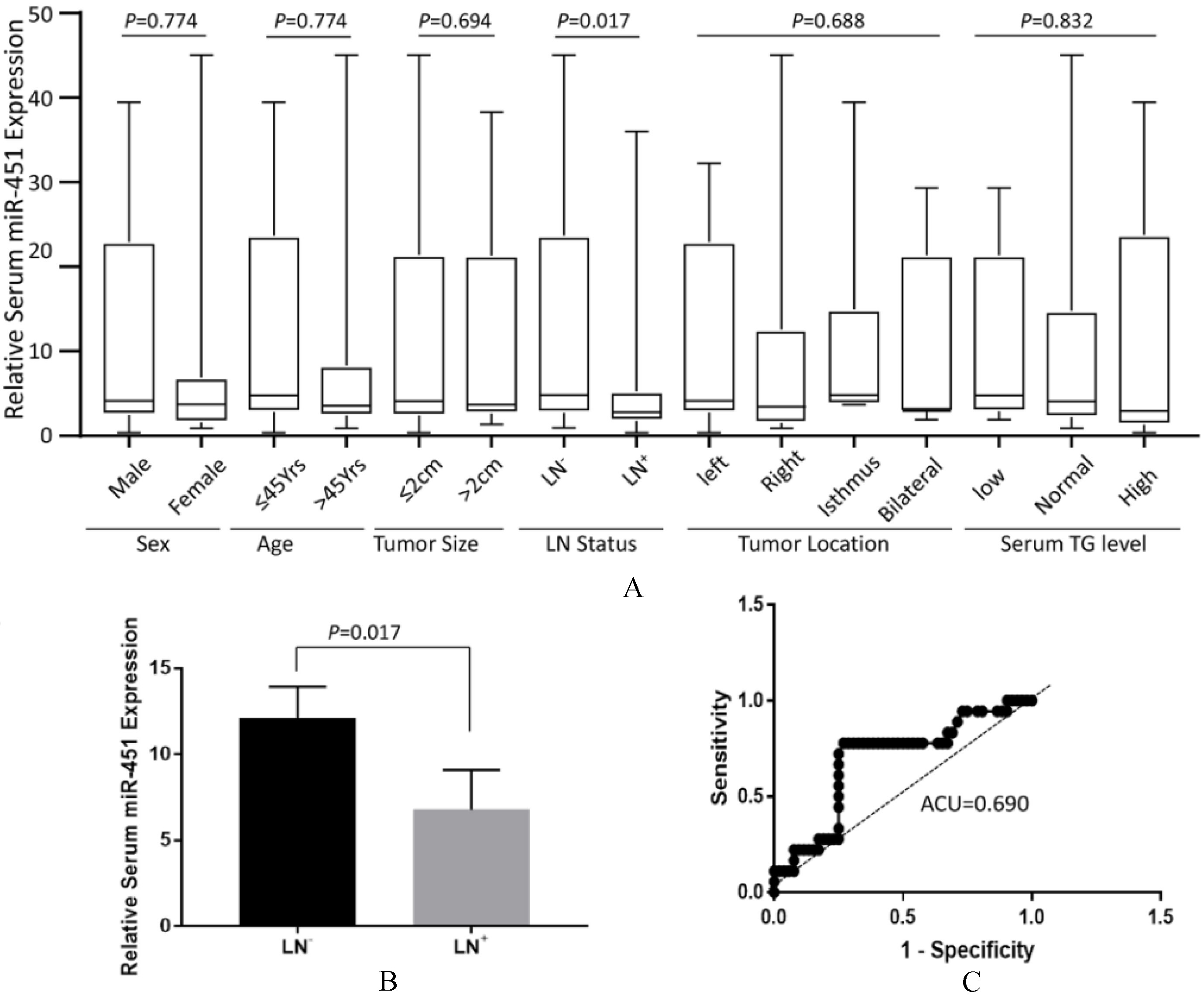
Analysis of correlations between expression of serum miR-451 and clinicopatholgical features of PTC patients. (A) Correlation of miR-451 level in PTC tissue with respect to gender, age, tumor size, LN status, tumor locations and serum TG levels. Data were expressed by Whisker Plot to demonstrate the medium and the distribution of samples. Except for the status of lymph node involvement (
MiRNAs are key regulators of cell proliferation, differentiation, apoptosis and migration. Aberrant expression of miRNAs has been found associated with various types of tumors, including different types of thyroid cancers [15]. Whether these miRNAs can be independent predictors of aggressive clinicopathological features of PTC remains unclear. Searching for additional miRNA biomarkers, which correlate better with the progress of tumors, is urgently needed.
Cellular miR-451 as a signature biomarker for thyroid malignancies
Studies by other groups have shown that expression of miR-451 negatively correlates with aggressiveness of hepatocellular carcinoma [16], Glioblastoma [17] and non-small cell lung cancer [18]In vitro studies showed that overexpression of miR-451 inhibited c-Myc mediated cell proliferation and migration via targeting Akt/mTOR signaling pathway [16, 19]. These studies suggest that miR-451 may function as a tumor suppressor that targets pathways important for oncogenesis. To address a possible role of miR-451 in the oncogenesis of PTC, we performed qRT-PCR analyses on 60 PTC patients FFPE paired samples (tumor and NAT,
We further analyzed the correlations between miR-451 expression and various clinicopathological parameters of our first PTC patient cohort, some of which have been suggested to be prognosticatively relevant [21]. Our results (Figs 2A and B) indicated a strong correlation between lower miR-451 levels in PTC patients with LN metastasis, suggesting that tissue miRNA-451 level is also a “good” biomarker (AUC
Circulating miR-451 as a biomarker for thyroid malignancies
Establishment of a non-invasive and low-cost assay for disease screening and diagnosis is the ultimate goal for the management of cancer patient cares. To investigate if miRNA-451 expression can be developed as a blood based diagnostic/prognostic marker for PTC, we analyzed serum miR-451 level in PTC patients with malignant features and patients with benign features. We found that miR-451 levels in patients with malignant forms was significantly lower than those with benign forms (
In summary, we have clearly demonstrated that miR-451 was down-regulated in both FFPE tissues and sera of PTC patients with malignant features. We have also shown that tissue miR-451 is a good predicting biomarker for malignant PTC. Together with other risk factors, such as overexpression of miR-146b and miR-222 [11, 13] and somatic mutation BRAF
Compliance with ethical standards
Funding
This study was funded by Science and Technology Fund Project of Shanghai Jiaotong University School of Medicine (grant number 13XJ22016).
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the Ethics Committee of Xinhua Hospital, Shanghai Jiaotong University School of Medicine.
Informed consent
Informed consent was obtained from all individual participants included in the study. Written informed consents were obtained from all participants of this study.
Footnotes
Acknowledgments
This work was supported by the Science and Technology Fund Project of Shanghai Jiaotong University School of Medicine, China (Grant No. 13XJ22016).
Conflict of interest
The authors declare no conflict of interests.
