Abstract
Non-small cell lung cancer is the most common malignant tumor in the world. Currently, chemotherapy is still the major method for non-small cell lung cancer treatment, but the problem of cancer drug resistance still exists, so we designed 5 different phosphorothioate oligonucleotides to silence key genes in tumor cell development, which could help avoid inducing cancer cell drug resistance. MicroRNAs have been shown to play a crucial role in the pathogenesis and progression of many malignancies, such as breast, colon, lung, and pancreatic cancer. According to the data from the Gene Expression Omnibus database, miR-21 has been reported to be one of the top 20 differentially expressed microRNAs screened using the Morpheus online tool, and miR-21 has been revealed to regulate a series of biological behaviors in cancer cells, including cell proliferation, migration, invasion, metastasis, and apoptosis. In recent years, gene therapy has emerged as a new therapeutic strategy for cancer treatment. Antisense oligonucleotides have recently been suggested as a novel approach for targeting microRNAs by antisense-based gene silencing. Five phosphorothioate oligonucleotides were designed, synthesized, and screened for anticancer activity. Reverse transcription-polymerase chain reaction was used to detect the relative expression of miR21. Among these 5 sequences, only phosphorothioate oligonucleotide 4 inhibited the proliferation of H1650 cells, and this effect was due to the induction of cancer cell apoptosis by activating the caspase-8 apoptotic pathway. In conclusion, this research confirmed the anticancer activity of phosphorothioate oligonucleotide 4 and revealed the underlying mechanism, which has the potential to be a novel anticancer strategy.
Introduction
Lung cancer has become the most common malignant tumor in the world with high mortality and morbidity, and it is the leading cause of cancer-related death in China. Lung cancer is divided into small-cell lung cancer and non-small cell lung cancer (NSCLC). Non-small cell lung cancer accounts for approximately 85% of all diagnosed lung cancers. 1 Currently, chemotherapy is still the major treatment method for the control of NSCLC, and platinum-based chemotherapy, such as cisplatin, is widely recommended for advanced patients. However, cisplatin treatment can result in cancer cell drug resistance, which limits the clinical application of cisplatin. 2,3 Hence, investigating the molecular mechanisms of NSCLC development and developing a more effective therapy to overcome drug resistance are crucial for clinical NSCLC treatment. In the present study, we aimed to use antisense oligonucleotide drugs to silence the expression of key genes in NSCLC cells without inducing cancer resistance.
MicroRNAs (miRNAs) are a class of endogenous noncoding short-fragment RNA molecules that widely exist in eukaryotes. MicroRNAs can bind to the 3′-untranslated region (3′-UTR) in the messenger RNA (mRNA) of target protein, inhibiting protein translation or mRNA stability, thereby regulating the target gene expression. MicroRNAs are approximately 20 to 22 nucleotides long, and many reports have revealed that miRNAs play important roles in the tumorigenesis and development of certain cancers, such as in tumor invasion, migration, proliferation, metastasis, and cell apoptosis. 4,5 Among them, miR-21 is a molecule of great interest that has been reported to be overexpressed in almost all cancers, such as breast, colon, lung, and gastric cancer, especially in human NSCLC cells, and it may become a new biomarker for tumor diagnosis. 6 -9 Considering the pivotal role of miR-21 in the physiological process of cancer, our research focused on inhibiting the vital regulator miR-21 to inhibit tumor development.
Therapeutic antisense technology has greatly advanced in the last 20 years and has been proven effective in the treatment of certain cancers. Compared with traditional chemotherapy drugs, antisense oligonucleotide drugs have shown higher cancer selectivity and fewer side effects during treatment. Antisense oligonucleotide drugs represent a new strategy for antitumor treatment. 10,11
In the present study, we designed 5 oligonucleotide sequences named phosphorothioate oligonucleotides 1-5 (PS2ODN 1-5) with RNAstructure 4.6 software. All PS2ODNs had a 12-base structure and are modified by sulfur substituents for stability. The relative gene expression levels of miR-21 were detected by real-time quantitative polymerase chain reaction to select the most effective PS2ODN targeting miR-21. The Cell Counting Kit-8 (CCK-8) assay was performed to detect the inhibitory effect of PS2ODNs on H1650 cancer cell proliferation, and the intracellular reactive oxygen species (ROS) accumulation was assessed by an ROS assay kit. In addition, the expression of apoptotic proteins in H1650 cancer cells was detected by western blotting. Generally, in the present study, we aimed to identify a new strategy for human NSCLC treatment by silencing the key regulator miR-21, which would avoid the problem of cancer cell drug resistance.
Materials and Methods
Cell Culture
The human NSCLC cell line H1650 (ATCC, CRL-5883) and normal human normal lung epithelial cells BEAS-2B (ATCC, CRL-9609) were purchased from American Type Culture Collection (ATCC, Rockville, Maryland). The cells were cultured in ATCC-formulated RPMI-1640 medium (Sigma, St. Louis, USA) supplemented with 10% fetal bovine serum (HyClone, New York, USA), 2%
Phosphorothioate Oligonucleotide Target Site Selection and PS2ODN Synthesis
The secondary structure of miR-21 was predicted by RNAstructure 4.6 software. The mRNA-PS2ODN paring parameters were selected according to the data of the binding affinity provided by Oligo Walk 5.0 and the PS2ODNLIGHT program. 12,13 The positions of the PS2ODN target sites are illustrated in Figure 1. The sequences of the PS2ODNs targeting the mRNA of miR-21 were designed with RNAstructure 4.6. Evidence has shown that 12-base antisense PS2ODNs are an effective length, so we designed 5 plausible 12-base length PS2ODNs for antitumor activity screening. All PS2ODNs were synthesized by Shanghai GenePharma (Shanghai, China) with sulfonic modification, and the detailed sequences of the PS2ODNs are listed in Table 1. Purified PS2ODNs were lyophilized and stored at 4°C.

Secondary structure of miR-21 mRNA.
Sequences of Oligonucleotides.

Abbreviation: PS0ODN, phosphorothioate oligonucleotides.
Phosphorothioate Oligonucleotide Transfection
The synthesized PS2ODNs were transfected into H1650 cancer cells as previously reported. 14,15 Briefly, exponentially growing cells were seeded onto 6-well plate and cultured at 37°C in a humidified atmosphere of 95% air and 5% CO2. When the cells reached confluency of 40% to 50%, PS2ODN transfection was performed with Lipofectamine3000 Transfection Reagent (Invitrogen, Carlsbad, California) according to the manufacturer’s protocol. 3 The negative control group was transfected with a control oligonucleotide sequence. The efficiency of gene silencing was detected 48 hours after transfection by using polymerase chain reaction (PCR).
RNA Isolation and Reverse Transcription-Polymerase Chain Reaction Assay
To determine the inhibitory effect of the synthesized PS2ODN sequences on the expression of miR-21, H1650 cancer cells were transfected with a series of concentrations of PS2ODNs with the same volume of diluent used as the negative control. Then, 24 hours after transfection, total RNA was extracted from the cells with TRIzol reagent (Invitrogen) according to the manufacturer’s instructions. 16 The complementary DNA was synthesized using a PrimeScript RT reagent kit (TaKaRa Biotechnology Co., Dalian, China). Reverse transcription-PCR (RT-PCR) was performed using SYBR Premix Ex Taq (TaKaRa Biotechnology Co.). Four nanograms of total RNA were used for each RT-PCR reaction (20 μL). Primers for RT-PCR detection used in this study are listed in Table 2. 17,18 The RT-PCR program was as follows: (95°C, 2 minutes) × 1 cycle and (95°C, 30 seconds; 60°C, 30 seconds; 72°C, 30 seconds) × 40 cycles. Relative expression of miR-21 was calculated by the 2−ΔΔCt method, with normalization to gapdh mRNA levels. The experiment was repeated at least 3 times.
Sequences of Primers.

Cell Counting Kit-8 Assay
After transfection with synthesized PS2ODNs, the proliferation of H1650 cancer cells was determined by a CCK-8 according to the previously provided protocols. 19 In brief, H1650 cells in the logarithmic growth phase were seeded onto 96-well plate at a density of 5 × 103 cells per well. Phosphorothioate oligonucleotid transfection was performed when the cells reached 70% to 80% confluence. After treatment, 10 μL CCK-8 reagent (Sigma) was added into each well for 2 hours of incubation at 37°C in the dark. Then, absorbance at 450 nm was detected for each sample. The cell proliferation rate was calculated according to the OD450 value. All experiments were repeated at least 3 times. The half maximal inhibitory concentration (IC50) values were calculated using SPSS version 22.0.
Cell Apoptosis
To estimate the pathway of H1650 cancer cell death, an Annexin V-FITC/PI apoptosis detection Kit (BioVision, Mountain View, California) was used to detect the percentage of apoptotic H1650 cells. 20 Briefly, H1650 cancer cells were plated onto 6-well plate and transfected with PS2ODNs when the cells reached 70% to 80% confluence. The mismatched sequence and apoptosis inducer kit (Beyotime, Shanghai, China) were used as the negative and positive controls, respectively. After incubation, the cells were harvested with 0.25% wt/vol trypsin for apoptosis detection. Annexin V-FITC and PI were added for a 30-minute incubation at 37°C in the dark to stain the harvested cells, followed by 2 washes with phosphate-buffered saline. The stained cells in each group were detected by flow cytometry (BD, New Jersey, USA) at an excitation wavelength of 488 nm and emission wavelengths of 525 and 625 nm.
Western Blot Analysis
To determine the inhibitory effect of synthesized PS2ODN sequences on the expression of apoptotic proteins, western blotting was performed as previously described. 21 In brief, the H1650 cancer cells were seeded onto 6-well plate to 80% confluence and then transfected with a series of dilutions concentrations of PS2ODNs for 24 hours. After treatment, the cells were harvested and lysed with lysis buffer (1% sodium dodecyl sulfate [SDS], 10 mM Tris-HCl, pH 7.2, 1× protease inhibitor cocktail tablet [Roche, Mannheim, Germany]). Protein concentrations were quantified using the bicinchoninic acid method (Pierce Biotechnology, Inc, Rockford, Illinois). Equal amounts of protein were subsequently separated on SDS-polyacrylamide gels (12%) and transferred to polyvinylidene fluoride (PVDF) membranes (Millipore Corp, Billerica, Massachusetts) for western blotting. Then, the PVDF membranes were blocked with 5% skim milk (in 20 mM Tris-HCl, pH 7.2, 150 mM NaCl, 0.1% Tween 20) overnight at 4°C and incubated with the corresponding primary antibody at 4°C overnight. After that, the membranes were washed with phosphate buffered solution (PBST) buffer 3 times (5 minutes each) at room temperature and then incubated with 1:10 000 diluted anti-mouse immunoglobulin G secondary antibody (Abcam, Cambridge, Massachusetts) for 1 hours at room temperature. After washing with PBST 3 times, the proteins of interest were detected with an enhanced chemiluminescence kit (Amersham, Stockholm, Sweden). ChemiDoc XRS (Bio-Rad, Hercules, California) was used for densitometric analysis of western blots, and the relative band intensities were compared to controls using Quantity One version 4.1.0.
Statistical Analysis
All data in this study are presented as the means ± standard deviations from at least 3 independent experiments and were analyzed with one-way analysis of variance using GraphPad Prism (GraphPad Software, Inc., San Diego, USA). Differences were considered statistically significant at P < .05.
Results and Discussion
Transcript Target Site Selection and PS2ODN Synthesis
RNAstructure software was used to design antisense oligonucleotide drugs targeting miR-21, and the predicted miR-21 secondary structure is shown in Figure1. According to the optimal PS2ODN design characteristics: overall ΔG < −10 kcal/mol, duplex ΔG < −25 kcal/mol, oligo-self ΔG ≥ −1.1 kcal/mol, oligo-oligo ΔG ≥ −8 kcal/mol, and Tm ≥ 50°C, we selected the 5 best antisense PS2ODNs targeting the 3′-UTR of pre-miR-21. The detailed sequences of the antisense PS2ODNs are listed in Table 1.
Inhibitory Effect of Synthesized PS2ODNs on miR-21 Gene Transcripts
After the design and synthesis of PS2ODN 1-5, the anticancer activity of these PS2ODNs was evaluated by measuring their regulatory effect on the expression of key genes in vitro. It has been reported that miR-21 is an important regulator of cancer proliferation and apoptosis by regulating the activation of the PTEN/PI3K/Akt pathway. From Figure 2, we can see that after transfection with PS2ODN 4, the expression of miR-21 was downregulated significantly compared with that in the negative control group (P < .05). Phosphorothioate oligonucleotides 1-3 and PS2ODN 5 hardly affected the miR-21 expression level. Polymerase chain reaction analysis revealed that the relative expression of the PTEN/PI3K/Akt pathway, a downstream target of miR-21, was activated upon treatment with PS2ODN 4. All the results above indicated that the oligonucleotide sequence PS2ODN 4 could target miR-21, inhibit its expression, and inhibit the PTEN/PI3K/Akt pathway.

Phosphorothioate oligonucleotide 4 downregulates miR-21 expression and activates the PTEN/PI3K/Akt pathway. H1650 cancer cells at 40% to 50% confluence were transfected withPS2ODN 1-5. The expression of miR-21 and PTEN/PI3K/Akt messenger RNA was detected by reverse transcription-polymerase chain reaction. Every experiment was performed in triplicate. Differences were considered statistically significant at P < .05. CON represents untransfected H1650 cancer cells; Mis-PS2ODN: H1650 cancer cells transfected with mismatched PS2ODN; PS2ODN 1-5: H1650 cancer cells transfected with the designed PS2ODN 1-5.
Antiproliferation Activity of PS2ODNs
In the previous experiment, we evaluated the inhibitory effect of PS2ODN 1-5 on miR-21 expression and PTEN/PI3K/Akt pathway activation in H1650 cancer cells. Next, we wanted to further explore the inhibitory effect of anti-miR-21 PS2ODN 1-5 on H1650 cancer cell growth, which was examined by assessing cell viability and proliferation. After transfection with PS2ODN 1-5, we observed that PS2ODN 4 inhibited H1650 cancer cell proliferation in culture medium in a time- and concentration-dependent manner. The mismatched sequences, PS2ODN 1-3 and PS2ODN 5, did not show any significant growth inhibitory effects even at higher concentrations (Figure 3). However, unlike its effect on H1650 cancer cells, PS2ODN 1-5 did not affect the growth of normal human cells, indicating the high selectivity of PS2ODN during anticancer treatment. The IC50 values of PS2ODN 1-5 are shown in Table 3. Among the 5 PS2ODNs, PS2ODN 4 had the smallest IC50 (2.13 ± 0.07 µM) compared with the others. These results suggest that anti-miR-21 PS2ODN 4 exerts a concentration-dependent anticancer effect.
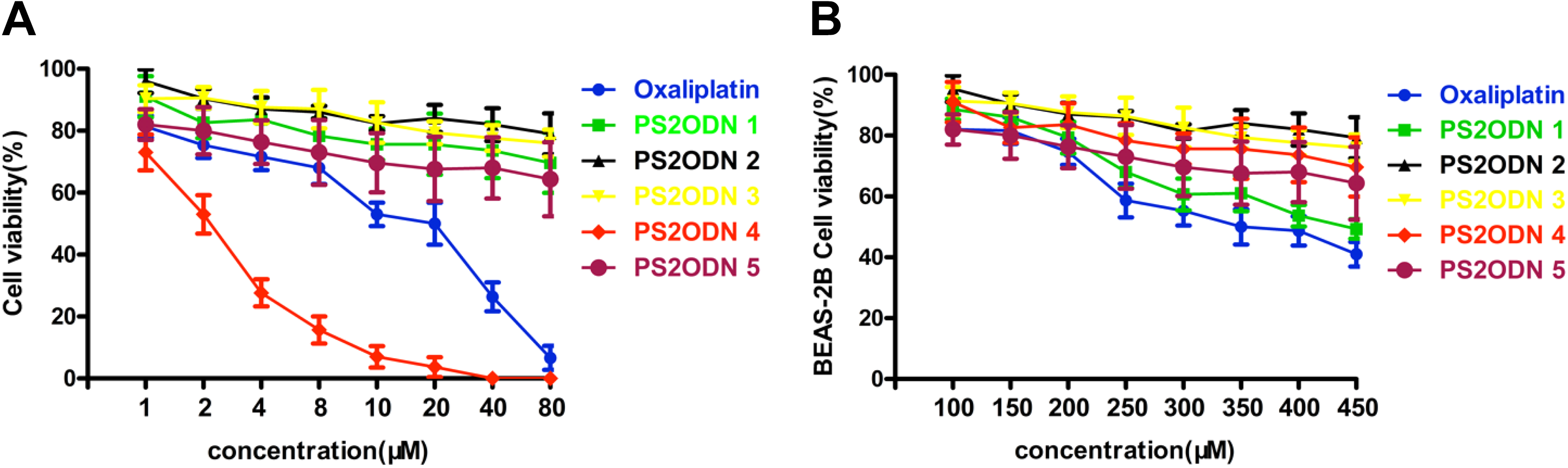
Phosphorothioate oligonucleotide 4 exhibits antiproliferative activity. A, H1650 cancer cells were transfected with PS2ODN 1-5, and cell viability and proliferation were assessed by CCK-8. B, BEAS-2B cells were transfected with PS2ODN 1-5 for the cell viability assay.
IC50 Value of PS2ODN 1-5 (µM).

Abbreviations: PS0ODN, phosphorothioate oligonucleotides; IC50, half maximal inhibitory concentration.
Phosphorothioate Oligonucleotide 4 Induces H1650 Cell Apoptosis
Apoptosis is the most common cell death pathway induced by anticancer drugs, and the percentage of H1650 apoptotic cancer cells was detected by Annexin V/PI staining methods by using flow cytometry. H1650 cells were transfected with PS2ODN 4 as well as the mismatched PS2ODN used as the negative control. As shown in Figure 4, PS2ODN 4 significantly induced H1650 apoptotic cell death up to 41.35% at concentrations of 2.1 µM and 79.14% at 6.3 µM.

The effect of PS2ODN 1-5 on H1650 cell apoptosis and cell cycle distribution. H1650 cancer cells were transfected with PS2ODN 1-5 and mismatched PS2ODN. A, Flow cytometry detection was performed to evaluate the effect of PS2ODN 1-5 on H1650 cancer cell apoptosis. Each experiment was repeated at least 3 times. x-axis: cells stained with Annexin V dye; y-axis: PI-labeled cells.
Phosphorothioate oligonucleotide 4 Inhibits the Expression of Apoptotic Protein
Caspase-3 and caspase-8 are key indicators of cell apoptosis. During cell apoptosis, pro-caspase-3 and pro-caspase-8 are cleaved into active forms. Hence, the protein levels of cleaved caspase-3 and caspase-8 were detected by western blot. Compared with the control group, PS2ODN 4 significantly induced the activation of this apoptotic pathway in a time-dependent manner (Figure 5A). The relative expression levels are shown in Figure 5B.

Phosphorothioate oligonucleotide 4 induces the activation of caspase-3 and caspase-8. H1650 cells at 40% to 50% confluence were transfected with PS2ODN 4. A, The expression levels of caspase-3 and caspase-8 were detected by western blot. B, The statistical results of Figure A.
Conclusion
MicroRNA 21 is highly expressed in many cancer cells and participates in many important physiological processes, such as cancer cell differentiation, proliferation, and apoptosis. All these processes are closely related to cancer development. Previous studies have revealed abnormally high expression of miR-21 in lung cancer cells, affecting important cell physiological functions, such as apoptosis, invasion, and metastasis, by regulating the activity of the target gene PTEN. Our research confirmed the high expression of miR-21 in H1650 cancer cells, and we designed 5 phosphorothioate oligonucleotides, PS2ODN 1-5, targeting miR-21 using RNAstructure software.
Among these 5 designed and synthesized PS2ODNs, PS2ODN 4 exhibited the most excellent anticancer activity against H1650 cancer cells after transfection, as revealed by the CCK-8 assay and IC50 values. Phosphorothioate oligonucleotide 4 transfection significantly reduced cancer cell proliferation rate, and PS2ODN 4 had an IC50 of 2.13 ± 0.07 µM, much lower than the values in the other groups. However, PS2ODN 4 failed to inhibit the viability of the normal human cells BEAS-2B, indicating that PS2ODN 4 has high selectivity for cancer cell inhibition and high safety during clinical treatment.
Although initial studies indicated that PS2ODN 4 exhibited excellent antiproliferation activity on H1650 cancer cells, the underlying mechanisms remain unclear. Therefore, preliminary exploration was carried out to confirm the antitumor activity of PS2ODN 4. Many studies have reported that miR-21 is overexpressed in many cancers, such as breast, colon, lung, and gastric cancer, especially in human NSCLC cells. Therefore, in this experiment, we first investigated whether the anticancer activity of PS2ODN 4 was due to changes in the expression and function of miR-21. First, RT-PCR assay was performed to detect the effect of PS2ODN 4 on miR-21 expression. The results revealed that PS2ODN 4 significantly reduced the expression of miR-21, and then the expression of miR-21 target genes increased through the antisense mechanism. Then, the apoptotic rate of H1650 cells indicated that PS2ODN 4 exerted anticancer activity by inducing cell apoptosis. In addition, the activation of caspase-3 and caspase-8 measured by western blotting also confirmed the apoptosis induced by PS2ODN 4. Overall, the synthesized PS2ODN 4 was an excellent strategy for NSCLC treatment.
In the present study, we designed and synthesized a series of PS2ODNs and confirmed that PS2ODN 4 had the highest inhibitory effect against H1650 cancer cell proliferation, and this favorable anticancer activity was attributed to the induction of cell apoptosis through caspase activation. Thus, miR-21 is a potential target for antisense oligonucleotide drugs, and PS2ODN 4 may provide a novel strategy for NSCLC treatment.
Footnotes
Authors’ Note
Jian-Hua Ge and Jing-Wei Zhu have contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
