Abstract
Neuroblastoma is a brain malignancy of childhood and accounts for 7–10% of childhood cancers, leading to approximately 15% of pediatric cancer deaths. MicroRNAs (miRNAs) are a family of short (about 18–25 nucleotides), noncoding and single stranded endogenous RNAs, which complementarily bind to the 3’ untranslated regions of their target genes. Recently, glutamine metabolism has been recognized as an important nutrition source for tumor cells, and hence targeting glutamine metabolism could benefit to development of anti-cancer agents. In this study, we investigate the roles of miR-513c in human neuroblastoma. We report miR-513c is significantly downregulated in human neuroblastoma tissues compared with their adjacent normal tissues. Moreover, miR-513c is significantly downregulated in neuroblastoma cell lines compared with normal neuroblast cells. Overexpression of miR-513c suppresses neuroblastoma cells’ migration, invasion, and proliferation. We demonstrate the glutaminase (GLS) is a direct target of miR-513c in human neuroblastoma cells. In addition, we found restoration of GLS expression recovered the neuroblastoma cells’ migration, invasion, and proliferation. In summary, this study illustrates a miR-513c mediated neuroblastoma cells suppression, providing a new aspect on the miRNA-based therapeutic approach for the treatments of neuroblastoma.
Abbreviation
Glutaminase: GLS;
MicroRNAs: miRNAs;
MicroRNA-513c: miR-513c;
3’ untranslated regions: 3’UTRs.
Introduction
MicroRNAs (miRNAs) are a family of short (about 18–25 nucleotides), noncoding and single stranded endogenous RNAs [1, 2]. MiRNAs inhibit protein expression through binding to their target mRNA with perfect or imperfect complementary sequences in the 3’ untranslated regions (3’UTRs) [3]. Recent studies have shown that dysregulated miRNAs play important roles in multiple cellular processes in cancers including carcinogenesis, apoptosis, migration, invasion, differentiation and chemoresistance [4, 5, 6]. Moreover, miRNAs specifically suppress their target genes, providing novel therapeutic opportunities for clinical application [7]. Previous studies indicated miR-513c might act as a tumor suppressive in multiple cancers such as prostate cancer [8], breast cancer [9], glioma [10] and esophageal adenocarcinoma [11]. However, to date, there have been only a few studies focusing on the roles of miR-513c in human neuroblastoma.
Neuroblastoma is a brain malignancy derived from precursor cells of the sympathetic nervous system during embryonal development [12]. It is the most common extracranial solid tumor of childhood and accounts for 7–10% of childhood cancers, leading to around 15% of pediatric cancer deaths [13]. The primarily metastasizes site of neuroblastoma is to bone marrow [14]. Currently, the survival rate of children with bone marrow metastasis is only approximately 40% [14]. Moreover, both primary neuroblastoma and metastatic tumor in older children are normally refractory to treatment [15]. Therefore, it is necessary to elucidate the molecular mechanism of the metastatic neuroblastoma for the development of novel therapeutic anti-tumor agents.
Glutamate, catalyzed from glutamine by mitochondrial glutaminase (GLS), is the principal neurotransmitter in brain [16]. Moreover, glutamine metabolism is essential for the maintenance of high levels of glutamine in the blood, providing a source of carbon and nitrogen to support biosynthesis, energetics and cellular homeostasis for tumor growth [17]. In this study, we will investigate the functions of miR-513c in human neuroblastoma. The mechanisms for the miR-513c-mediated glutamine metabolism and its correlation to neuroblastoma cells migration will be studied.
Materials and methods
Cell culture
Human neuroblast cell lines SK-N-SH, SK-N-BE2 and SH-SY5Y compared with normal human neuroblast cells, SK-N-AS and SK-N-DZ were purchased from the American Type Culture Collection (ATCC). Cells were cultured in DMEM Ham’s F12 medium containing 10% (v/v) heat-inactivated fetal bovine serum and 2 mM glutamate (all from Invitrogen, Carlsbad, CA, USA) at 37
Patient tissues
Primary neuroblastoma tissues and matched adjacent non-tumor tissues were obtained from 10 neuroblastoma patients undergoing surgery at the Children’s Hospital of Soochow University. Samples were snap-frozen in liquid nitrogen immediately after surgery and stored at
qRT-PCR
Total RNA was extracted from neuroblastoma cells and patient tissues using Trizol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. Concentrations and purity of the RNA samples were determined by spectrophotometric methods. cDNA was synthesized using the TaqMan™ Advanced miRNA cDNA Synthesis Kit from Thermo fisher Scientific (Waltham, MA, USA). The expression levels of miR-513c were assayed in triplicates by the stem-loop RT-PCR method using the Hairpin-it™ miRNAs qPCR Quantitation Kit (GenePharma, Shanghai, China). Relative miRNA was used as an internal control. Expression of miR-513c was normalized against the endogenous control (RNU6B) using the 2
Luciferase assay
A total of 5
Cell proliferation assay
The cell proliferation rates were measured using the Cell Counting Kit-8 (Sigma-Aldrich, St. Louis, MO, USA) following the manufacturer’s instruction. Briefly, after 24 hours’ post-transfection, cells (2
Cell migration and invasion assay
For cell scratch wound migration assays, Equal numbers of cells were seeded and grown to confluency in a 6 well-plate. A plastic pipette tip was used to produce a scratch wound of approximately 10 mm length and 500 mm width. Medium was then refreshed to remove dead cells. Cells were then incubated at 37
MiR-513c is downregulated in neuroblastoma tissues and cell lines. (A) Expression of miR-513c in 10 pairs of human neuroblastoma and their adjacent non-tumorous tissues. Statistical analysis was performed by paired t-test. (B) Expression of miR-513c in human normal neuroblast cell lines or neuroblastoma cell lines. Expression level of miR-513c was determined by TaqMan real-time PCR and normalized against an endogenous control (U6 RNA). *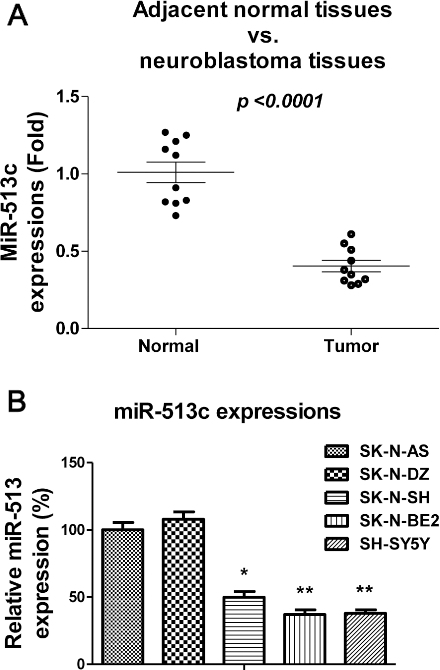
Overexpression of miR-513c suppresses the migration, invasion and proliferation of neuroblastoma cells. (A) SH-SY5Y cells were transiently transfected with control mimic or miR-513c mimic for 48 hours, expression of miR-513c was determined by TaqMan real-time PCR and normalized against an endogenous control (U6 RNA). (B) Wound healing assay, (C) Transwell invasion assay, and (D) Proliferation assays of SH-SY5Y cell with or without miR-513c overexpression. *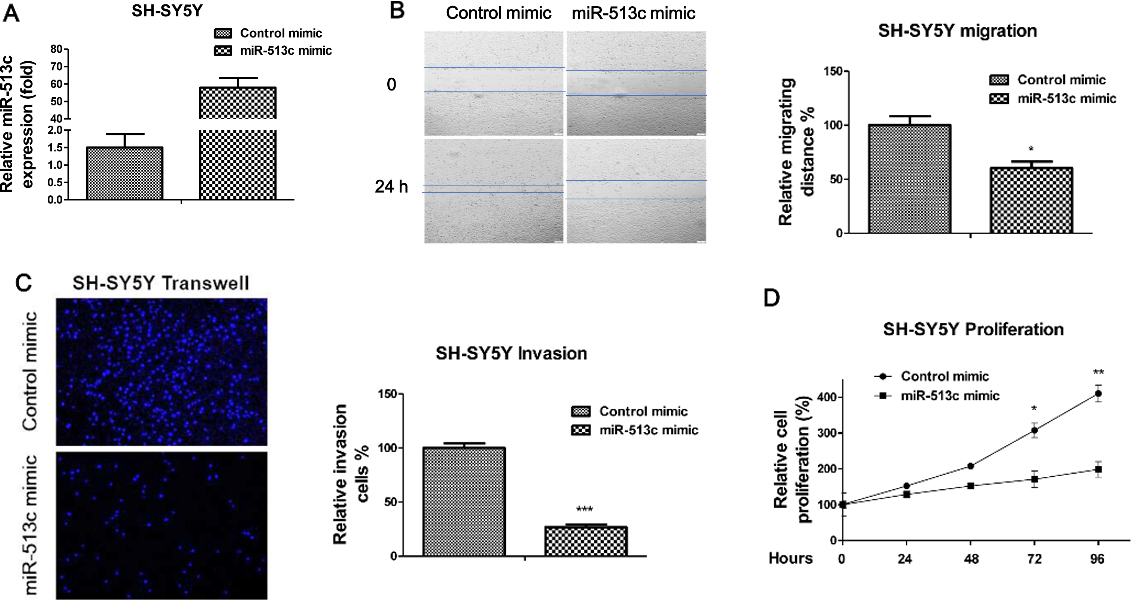
MiR-513c mimics (dsRNA oligonucleotides) and negative control mimics (control mimic) were purchased from GenePharma (Shanghai, China). Control siRNA and siGLS were purchased from GenePharma (Shanghai, China). Cells were seeded into 6-well plates at a density of 3
Detection of glutamine metabolism
The glutamine uptake was measured using the Glutamine and Glutamate Determination Kit (Sigma-Aldrich, St. Louis, MO, USA) according to the manufacturer’s procedure.
Western blot
Proteins from whole cell lysates were extracted using RIPA buffer (Sigma-Aldrich, St. Louis, MO, USA) according to the manufacturer’s protocol. Protein concentrations were determined by Bradford method. Equal amount proteins from each samples were separated on SDS- polyacrylamide gel electrophoresis (PAGE) and transferred to a nitrocellulose membrane (Bio-Rad, Hercules, CA, USA). The membrane was blocked with 5% non-fat milk for 1 hour at room temperature and incubated with rabbit anti-GLS polyclonal antibody (1:1000; Abcam #ab93434, Shanghai, China) and mouse anti-
Statistical analysis
Statistical analysis was performed with Prism5 (GraphPad). Data were presented as mean
MiR-513c targets GLS in neuroblastoma cells. (A) The predicted miR-513c binding sequence in the 3’-UTR of GLS from Targetscan.com. (B) Western blot analysis of the expression of GLS in SH-SY5Y cells transfected with or without miR-513c. 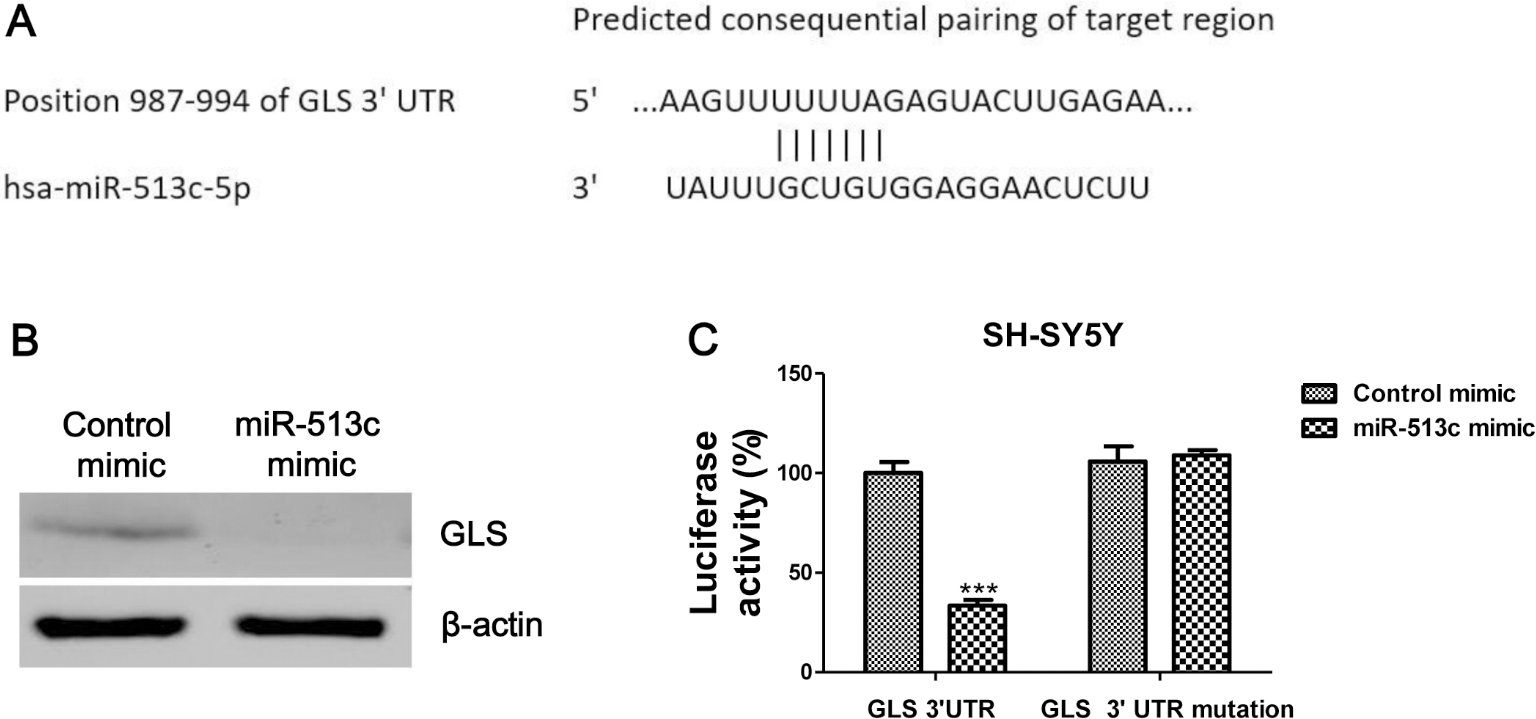
Knockdown GLS suppresses the migration, invasion and proliferation of neuroblastoma cells. (A) SH-SY5Y cells were transfected with control siRNA or siGLS for 48 hours. The expressions of GLS were measured by Western blot. 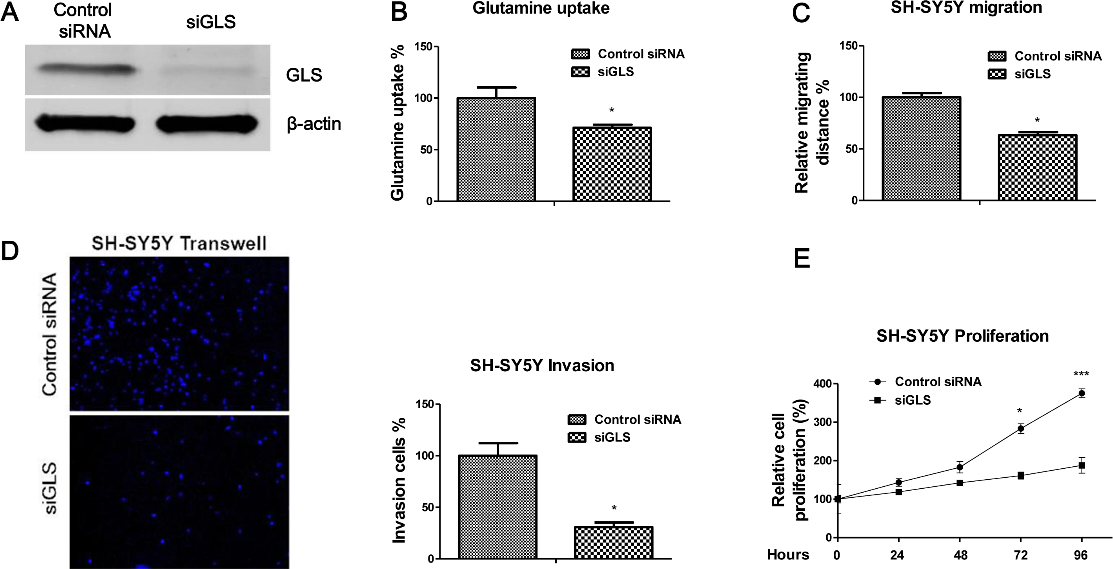
Restoration of GLS recovers the migration, invasion and proliferation of neuroblastoma cells. (A) SH-SY5Y cells were transfected with miR-513c mimic or miR-513c mimic plus GLS overexpression vector for 48 hours. The expressions of GLS were measured by Western blot. 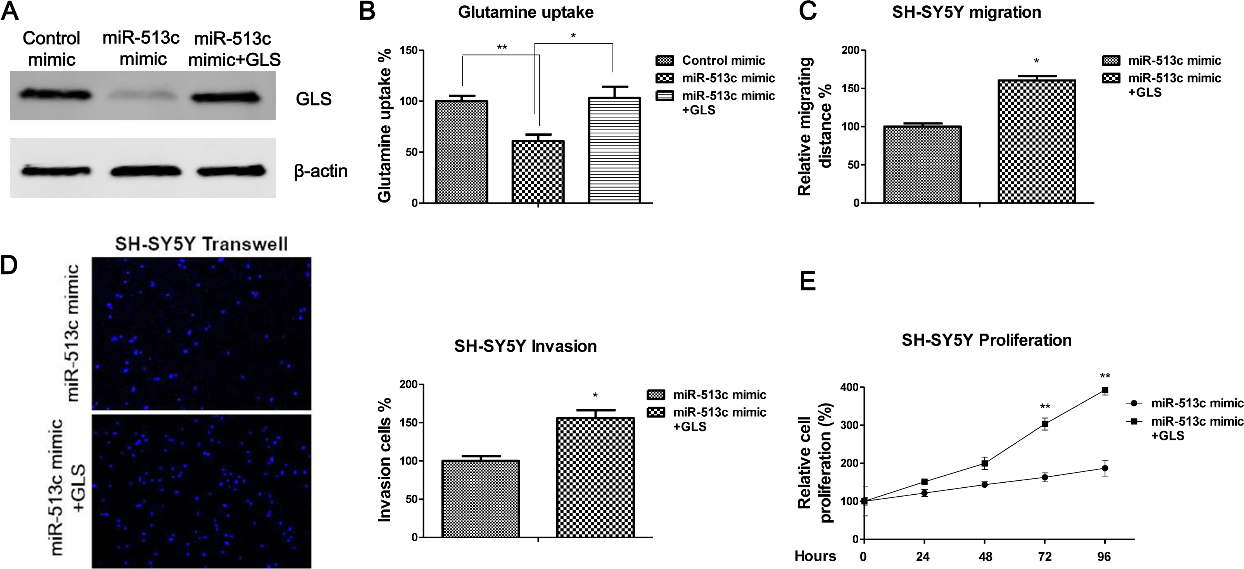
Reverse correlation between miR-513c and GLS in human neuroblastoma tissues. (A) The correlation of miR-513c and GLS mRNA levels in neuroblastoma tissues was shown. (B) Expression of GLS mRNA in 10 pairs of human neuroblastoma and their adjacent non-tumorous tissues by qRT-PCR. Statistical analysis was performed by paired t-test.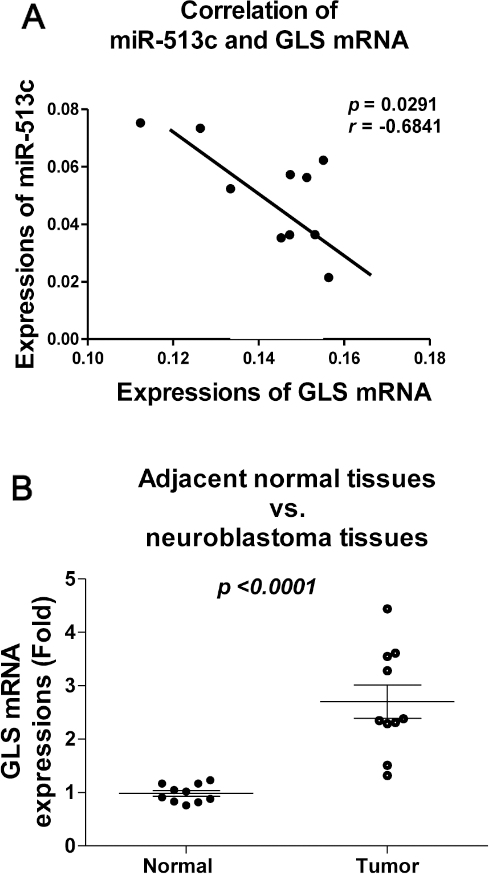
MiR-513c is downregulated in neuroblastoma tissues and cells
Previous reports demonstrated miR-513c possesses tumor suppressive functions in multiple tumors [8, 9, 10, 11]. To investigate the roles of miR-513c in human neuroblastoma, we compared the expressions of miR-513c in human neuroblastoma tissues and their adjacent normal tissues. As we expected, the expressions of miR-513c were significantly downregulated in human neuroblastoma tissues (Fig. 1A). In addition, we observed miR-513c was significantly downregulated in neuroblastoma cell lines, SK-N-SH, SK-N-BE2 and SH-SY5Y compared with normal human neuroblast cells, SK-N-AS and SK-N-DZ (Fig. 1B). Taken together, the above results suggested a tumor suppressive role of miR-513c in neuroblastoma.
Overexpression of miR-513c suppresses neuroblastoma cells migration, invasion, and proliferation
To identify the functions of miR-513c in neuroblastoma cells, we transiently transfected miR-513c mimic into SH-SY5Y cells (Fig. 2A). Since cell migration is a fundamental biological process during the brain development through regulating a highly specific pattern of connections between nerve cells. We next assessed the roles of miR-513c in the regulation of neuroblastoma cells’ migration, invasion and proliferation. In vitro wound healing assay showed overexpression of miR-513c inhibited the migration capacity of SH-SY5Y cells (Fig. 2B). Neuroblastoma cells with overexpression of miR-513c exhibited significantly decreased migration ability after 24 hours’ scratch. Moreover, transwell assay demonstrated overexpression of miR-513c inhibited the cell invasion (Fig. 2C). Consistently, we observed cell proliferation rates were suppressed by overexpression of miR-513c (Fig. 2D). Taken together, the above results suggested overexpression of miR-513c might be selected as a therapeutic target for the treatments of neuroblastoma.
GLS is a direct target of miR-513c in neuroblastoma cells
The above results demonstrated a tumor suppressive role of miR-513c, we continue to explore the mechanisms for the suppression of neuroblastoma cells migration and invasion by miR-513c. It has been reported that growing tumor cells uncommonly exhibit elevated aerobic glycolysis, which is a hallmark known as the “Warburg effect” [18]. Moreover, elevated glutamine metabolism and glycolysis are both required to meet the total metabolic demands of proliferating cancer cells [17]. We next performed bioinformatics analysis to search the potential miR-513c targets which involves in the cellular metabolism. Results from bioinformatics analysis illustrated the GLS, which contains a binding site for miR-513c, is a potentially regulatory target of miR-513c (Fig. 3A). GLS catalyzes the conversion of glutamine to glutamate, the first step in glutaminolysis. To determine whether miR-513c affected the expression of GLS, we transfected miR-513c mimic or control mimic into SH-SY5Y cells, the expression levels of GLS were detected. Western blot analysis demonstrated that the miR-513c mimics markedly suppressed the protein level of GLS (Fig. 3B). To further confirm whether the effect of miR-513c on the inhibition of the expression of GLS was through the direct targeting the 3’UTR region of GLS mRNA, a GLS 3’-UTR wild-type vector or 3’UTR mutant vector was co-transfected into SH-SY5Y cells with either the control mimic or miR-513c mimic, followed by measurement of luciferase activity. As shown in Fig. 3C, a reduction in the luciferase activity was observed in cells transfected with the miR-513c mimic and GLS 3’UTR wild-type vector, whereas the luciferase activity was no significantly change in the co-transfection with miR-513c and GLS 3’UTR mutant vector (Fig. 3C). These results demonstrated that GLS is direct target of miR-513c in neuroblastoma cells.
Knockdown GLS inhibits glutamine metabolism, migration, invasion and proliferation of neuroblastoma cells
To test whether targeting GLS by miR-513c could specifically inhibits glutamine metabolism and migration capacity of neuroblastoma cells, the effects of downregulation of GLS on neuroblastoma cell migration, invasion and proliferation were assessed. We transfected GLS siRNA to specifically knockdown GLS expression in SH-SY5Y cells (Fig. 4A). As expected, the glutamine uptake was decreased in GLS knockdown cells compared with control siRNA transfected SH-SY5Y cells (Fig. 4B). The present functional studies demonstrated knocking down GLS by
Restoration of GLS in miR-513c overexpressing cells recovers migration, invasion and proliferation
To verify the above results that miR-513c suppresses neuroblastoma cells through the inhibition of the GLS-mediated glutamine metabolism, we co-transfected overexpression vector containing wild type GLS or control vector into miR-513c overexpressing SH-SY5Y cells (Fig. 5A). Results showed co-transfection of GLS overexpression vector into miR-513c overexpressing SH-SY5Y cells could recover the original GLS expression and glutamine metabolism (Fig. 5A and B). Next, the cell migration, invasion and proliferation rates were assessed. As we expected, restoration of GLS in miR-513c overexpressing neuroblastoma cells could recover the cell migration (Fig. 5C), invasion (Fig. 5D) and proliferation (Fig. 5E). Taken together, these results demonstrated that direct targeting GLS by miR-513c was specific for the miR-513c-inhibited neuroblastoma cell migration, invasion and proliferation.
Reverse correlation between miR-513c and GLS in neuroblastoma tissues
In order to investigate whether the above in vitro findings are applicable to clinical administration, we compared the expressions of miR-513c and GLS mRNAs in neuroblastoma tumor samples. The correlation between the expressions of miR-513c with GLS mRNA in neuroblastoma tissues demonstrated that GLS mRNA and miR-513c exhibited inverse expression pattern (Fig. 6A). Moreover, the GLS mRNA was significantly upregulated in neuroblastoma tissues compared with their adjacent normal tissues (Fig. 6B). These results support the in vitro results that miR-513c targets GLS in human neuroblastoma cells.
Discussion
Neuroblastoma are often successfully eliminated in children under the age of 18 months, while tumors in older children often become refractory to treatment due to metastasis, which is the major cause of mortality among neuroblastoma patients [12, 13, 14, 15]. The primarily metastasis site of neuroblastoma is bone marrow and the survival rate of children with bone marrow metastasis is very low (approximately 40%). In this study, we described a miRNA based molecular mechanism for the development of anti-neuroblastoma cells migration and invasion. MiR-513c is significantly downregulated in neuroblastoma patient tissues and cell lines, compared with normal tissues and neuroblast, suggesting miR-513c might act as a tumor suppressor in neuroblastoma. Our findings were consistent with previous reports of the roles of miR-513c in other tumor types [8, 9, 10, 11]. Moreover, we found overexpression of miR-513c could significantly decrease the migration, invasion, and proliferation of neuroblastoma cells, suggesting miR-513c is a therapeutic target for the neuroblastoma treatments.
Warburg discovered that animal and human tumors displayed high avidity for glucose, which was largely converted to lactate through aerobic glycolysis [18]. Moreover, the roles and importance of glutamine metabolism in cancer cell biology has slowly been appreciated recently [17]. The maintenance of high levels of glutamine in the blood provides a steady source of carbon and nitrogen to support biosynthesis and cellular homeostasis for cancer cells growth [19]. In addition, targeting glutamine metabolism together with traditional chemotherapy offers a potential treatment strategy particularly in drug resistant cancers [20]. GLS catalyzes the conversion of glutamine to glutamate, the first step of the glutamine metabolism [21]. GLS is more broadly expressed in normal tissue and is thought to have a crucial role in many cancers [21]. Importantly, we report in this study that inhibition of the GLS-mediated glutamine metabolism by miR-513c significantly suppressed neuroblastoma cells’ migration, invasion, and proliferation, suggesting a new approach for the treatment of neuroblastoma. Accordingly, GLS silencing inhibited cell migration in glioma cancer cells. On the other hand, GLS2 had an opposite effect in both cell growth and migration [22]. To our knowledge, this is the first study to examine the roles of miR-513c in regulating glutamine metabolism in human neuroblastoma in vitro model. In our ongoing project, we continue to investigate the roles for the miR-513c regulated neuroblastoma behaviors using an in vivo model. The underlying mechanism of the tumor suppressive function of miR-513 as well as its target genes need to be further elucidated. In summary, this study implies that delivery of miR-513c might be a therapeutic strategy for retarding the process of tumorigenesis of neuroblastoma.
Conflict of interest
No potential conflicts of interest were disclosed.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (no. 81502496), Science Foundation of Jiangsu Provincial Commission of Health and Family Planning (no. Q201503) and Jiangsu Government Scholarship for Overseas Studies.
