Abstract
Background:
Previous studies have shown that berberine can inhibit glioma progression, although the underlying molecular mechanisms needed to be explored further. The aim of this study was to evaluate the suppressive effects of berberine on human glioma cells, and identify the underlying signaling pathways.
Material and Methods:
The cytotoxic effect of different concentrations of berberine against normal human glial cells (HEB) and 4 glioma cell lines was evaluated by the CCK-8 assay. Apoptosis was assayed by flow cytometry. In vitro migration and invasion were analyzed by the wound healing and transwell assays. The expression levels of specific proteins were measured by western blotting and ELISA.
Results:
Berberine significantly inhibited the proliferation of human glioma U-87 cells, and induced apoptosis in the U-87 and LN229 cells by downregulating Bcl-2, and upregulating Bax and caspase-3. In addition, berberine also inhibited migration and invasion of the glioma cells. Furthermore, berberine exerted its effects on the proliferation, migration, invasion, and apoptosis of glioma cells by inhibiting the TGF-β1/SMAD2/3 signaling pathway, and exogenous TGF-β abrogated the pro-apoptotic and anti-oncogenic effects of berberine.
Conclusions:
Berberine inhibits glioma progression by targeting the TGF-β1/SMAD2/3 signaling pathway.
Introduction
Glioblastoma (GBM) is the most common primary tumor of the brain that can originate from astrocytes, oligodendrocytes, and mixed cell types, which determine its histological subtype. 1 Epidemiological studies show that gliomas account for 45% of primary brain tumors. 2 Despite treatment strategies such as surgical resection and maximum radiotherapy, the prognosis of glioma patients is poor, with a median survival of only 14 months. 3 The dismal prognosis of gliomas can be attributed to their high invasiveness and recurrence. In fact, the invasion of glioma cells into the normal brain tissue and extracranial metastasis are major clinical challenges. 4 Therefore, it is crucial to explore novel drug candidates that can control the aggressive malignant behavior of glioma cells and improve patient prognosis.
Berberine is a natural alkaloid present in various plant families and genera including Annonaceae, Berberidaceae, Menispermaceae, and Rutaceae. 5 It is known to exert anti-cancer, antibacterial, cardioprotective, and antioxidant effects. 6 Multiple studies have shown that berberine can inhibit different tumor cells. For example, berberine inhibited the proliferation of human non-small cell lung cancer cells and induced apoptosis by suppressing the Bcl-2/Bax signaling pathway and MMP-2 production. 7 In addition, berberine can inhibit esophageal cancer cell growth by promoting cell cycle arrest at the G2/M phase and inducing apoptosis, which are mediated by its effects on Akt, mTOR, and AMPK activity. 8 It suppressed metastasis of triple-negative breast cancer cells by decreasing fibronectin production via inhibition of c-Jun phosphorylation, 9 as well as the migration and invasion of A375.S2 cells by inhibiting the metastasis-related FAK, uPA, and NF-κB signaling pathways. 10 Moreover, berberine can also inhibit glioma cells by inducing endoplasmic reticulum stress, disrupting mitochondrial membrane potential, down-regulating ERK1/2 activity, and consequently triggering apoptosis. 11 In addition, studies show that berberine also protects against blood-brain barrier injury. For example, Wu et al 12 demonstrated that berberine mitigated nerve injury by restoring the blood-brain barrier in a mouse model of cerebral hemorrhage. Wang et al 13 further showed that berberine derivatives were able to cross the blood-brain barrier and target mitochondria in a zebrafish model of Parkinson’s disease.
TGF-β1 belongs to the TGF-β super family of growth factors that regulate cell growth and differentiation, 14 and play a dual role in tumorigenesis and development. In the early stage of tumorigenesis, TGF-β inhibits the proliferation of tumor cells and promotes apoptosis. In the advanced stages however, TGF-β exerts a pro-metastatic role. 15 The effects of TGF-β are mediated via SMAD-dependent and SMAD-independent signaling pathways. 14 Following ligand-dependent assembly of the TGF-β receptor complex, the SMAD proteins are phosphorylated and translocated to the nucleus, wherein they bind to the promoters/enhancers of target genes and directly regulate their transcription. The downstream effectors of SMADs include cell cycle-related factors (Ki67, Cyclins) and apoptosis-related factors (Bcl-2, Bax). 16
The TGFβ/SMAD signaling pathway is hyperactivated in gliomas, and promotes tumor cell proliferation, migration, and invasion, which leads to poor prognosis. 17 Studies have shown that the TGF-β1/SMAD2/3 signaling pathway in particular plays a key role in tumor cell proliferation and apoptosis. 18 In addition, a functional link between berberine and TGF-β/SMADs signaling pathway has been observed in various diseases. For instance, berberine can attenuate airway inflammation by down-regulating the TGF-β1/SMADs signaling pathway. 19 Furthermore, berberine inhibited epithelial-mesenchymal transition (EMT)-like changes in tumor-associated fibroblast-induced colonic epithelial cells, and promoted apoptosis by regulating the TGF-β/SMADs pathway. 20 Berberine also suppressed the EMT in hepatocellular carcinoma and gastric carcinoma cell lines by upregulating p-SMAD2 and downregulating p-SMAD3. 21 However, the relationship between berberine and the TGF-β/SMAD signaling pathway in glioma is still unclear.
Based on previous reports, we hypothesized that berberine modulates apoptosis in glioma cells by targeting the TGFβ/SMAD signaling pathway. To this end, we analyzed the effects of berberine on human glioma cells and explored the underlying molecular mechanisms. Berberine significantly inhibited cell proliferation, migration and invasion, induced apoptosis, downregulated Ki67, PCNA and Bcl-2, and upregulated the pro-apoptotic Bax and caspase-3 proteins. The anti-tumor effects of berberine were mediated via inhibition of the TGF-β1/SMAD2/3 signaling pathway.
Materials and Methods
Materials
Berberine (purity up to 98%) was obtained from Sigma-Aldrich (St. Louis, MO), and dissolved in dimethyl sulfoxide (DMSO). Recombinant human TGF-β1 was purchased from Proteintech Co. Ltd., Wuhan, China. CCK8 kit and all other chemicals were from Beijing Solarbio Science & Technology, Beijing, China. The annexin V-FITC/PI apoptosis detection kit was from Shanghai YEASEN Biotechnology Co., Ltd. (Shanghai, China). Rabbit anti-human Ki67, PCNA, Bax, Bcl-2, cleaved caspase, p-SMAD2, p-SMAD3, SMAD2, and SMAD3 antibodies were purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA). Antibodies against β-actin and secondary antibodies were purchased from Sigma-Aldrich. All other chemicals were of analytical grade.
Cell Culture
The normal human glial cell line HEB was purchased from GuangZhou Jennio Biotech Co. Ltd. Human glioma cells lines (U-251, U-138, U-87, and LN229) were purchased from the Cell Bank of Chinese Academy of Sciences (Shanghai). The cells were cultured in high glucose DMEM supplemented with 10% (v/v) FBS and antibiotics (100 mg/ml streptomycin and 100 units/ml penicillin) in a humidified incubator at 37°C with 5% CO2 and 95% air.
Cell Viability Assay
The cells were seeded in 96-well plates at the density of 1 × 104 cells/well and cultured for 24 hours. The medium was replaced with fresh DMEM supplemented with different concentrations of berberine (0, 1, 2, 5, 10, 20, and 40 μM). After culturing for 24 hours, 10 µl CCK-8 reagent was added to each well, and the cells were incubated for 2 hours. The absorbance at 490 nm was measured using a microplate reader, and the viability rates were calculated on the basis of a standard curve.
Western Blotting
The suitably treated cells were washed once with ice-cold PBS and treated with RIPA buffer (Thermo Scientific Pierce, MD, USA) supplemented with a protease inhibitor cocktail and 1% phenylmethyl sulfonyl fluoride PMSF (ST506, Beyotime, Shanghai, China). The lysates were centrifuged, and the protein content in the supernatants was measured using a BCA protein assay kit (P0010S, Beyotime, Shanghai, China). Subsequently, 40 μg protein from each sample was mixed with 5× SDS loading buffer and denatured at 97°C for 10 minutes. The samples were separated by 10% or 15% SDS-PAGE (P0015A, SDS-PAGE) and transferred to a PVDF membrane (FFP24, Beyotime, Shanghai, China). After blocking with 5% skim milk for 2 hours at room temperature, the membrane was incubated overnight with primary antibodies (all from Abcam and diluted 1:1000) specific for GAPDH (ab8245), Ki67 (ab15580), PCNA (ab92552), Bax (ab32503), Bcl-2 (ab32124), cleaved-caspase3 (ab32042), TGFβ (ab215715), SMAD2 (ab40855), p-SMAD2 (ab188334), smad3 (ab40854), and p-smad3 (ab152903) at 4°C. The membrane was then washed thrice with TBS-Tween 20 (0.1%, v/v) and incubated with the corresponding HRP-conjugated secondary antibody for 1 hour at room temperature. After the final 3 washes with TBST (ST677, Beyotime, Shanghai, China), the protein bands were visualized using SuperSignal™ West Pico Chemiluminescent Substrate (Thermo, CA, USA). The density of the bands were calculated using ImageJ software and the value was normalized to GAPDH.
Flow Cytometry
The cells were seeded in a 6-well plate at the density of 5 × 104 cells per well, and cultured in the presence of different concentrations of berberine (5, 10, and 20 μM) for 48 hours. After harvesting with 0.25% trypsin, the cells were washed twice with chilled PBS at 800 rpm, and the viability was determined using trypan blue exclusion method. The cells were resuspended in 1× binding buffer provided with the Annexin V-FITC apoptosis detection kit at the density of 1 × 106 cells/ml, and then stained with 5 μl annexin V-FITC and 10 μl propidium iodide (PI) in the dark for 20 minutes at room temperature. The stained cells were analyzed by flow cytometry, and the dead cells were excluded using forward and side scatter gating. The experiment was repeated at least 3 times.
Wound Healing Assay
The cells were seeded in a 6-well plate at the density of 4 × 105 cells/well and grown till 90% confluent. The monolayer was scratched longitudinally with a sterile pipette tip, and the dislodged cells were rinsed off. Fresh serum-free medium with or without berberine (5, 10, and 20 μM) was added. The wound region was observed under an inverted microscope (Optika, Italy) and phase-contrast images were captured at 0 and 24 hours. The width of the wound region was measured in 3 random fields of view for each well. The migration of the cells was evaluated in terms of the wound healing rate (%) calculated as follows: (0 hour scratch width-24 hour scratch width)/0 hour scratch width × 100%.
Transwell Invasion Assay
The suitably treated cells were seeded in the Matrigel-coated upper compartment of Transwell chambers at the density of 1 × 105 cells/ml in serum-free DMEM, and the lower compartments were filled with 600 μl DMEM containing 10% FBS. After 24 hours, the cells that migrated through Matrigel and 8 μm pore membrane were fixed, stained with crystal violet and counted under a microscope. The number of cells was counted in independent fields and the average was calculated for each chamber.
ELISA
The TGF-β levels in the supernatants of cultured glioma cells were measured using a specific kit (PGE2ELISA, China) as per the manufacturer’s instructions. The sensitivity of the TGF-β kit is 16 pg/ml, with an intra-batch coefficient of variation of <10% and inter-batch coefficient of variation of <15%. The linear range of the standard curve was 0 to 2000 pg/ml. The absorbance (OD) of the wells was measured at 450 nm on an enzyme calibrator, and the standard curve was plotted against the OD of the standard. The experiment was repeated 3 times for each specimen and the mean value was calculated.
Bioinformatics Analysis
The correlation between expression levels of TGF-β1, SMAD2, and SMAD3 and the prognosis of glioma patients was analyzed based on the UALCAN online database.
Statistical Analysis
The data are expressed as mean ± standard deviation of 3 or 4 independent experiments. The Student’s t test or one-way analysis of variance (ANOVA) was performed using SPSS statistical package (USA). Survival duration was estimated using Kaplan-Meier curves and compared by the log-rank test. P < .05 was considered statistically significant.
Results
Berberine Inhibited the Malignant Behavior of Glioma Cells and Induced Apoptosis
To assess the effect of berberine in different human glioma models, we first measured the viability of normal human glial cells (HEB) and the 4 glioma cell lines treated with different doses of berberine (0, 1, 2, 5, 10, 20, and 40 μM). As shown in Figure 1, berberine significantly decreased the viability of the U-87 and LN229 cells in a dose-dependent manner compared to the other cell lines. This indicated that berberine likely induced apoptosis in these 2 cell lines, which were therefore selected for subsequent experiments. In the HEB cells however, only 40 µM berberine showed obvious cytotoxicity. Based on these results, we used 5, 10, and 20 μM berberine for the relevant assays.
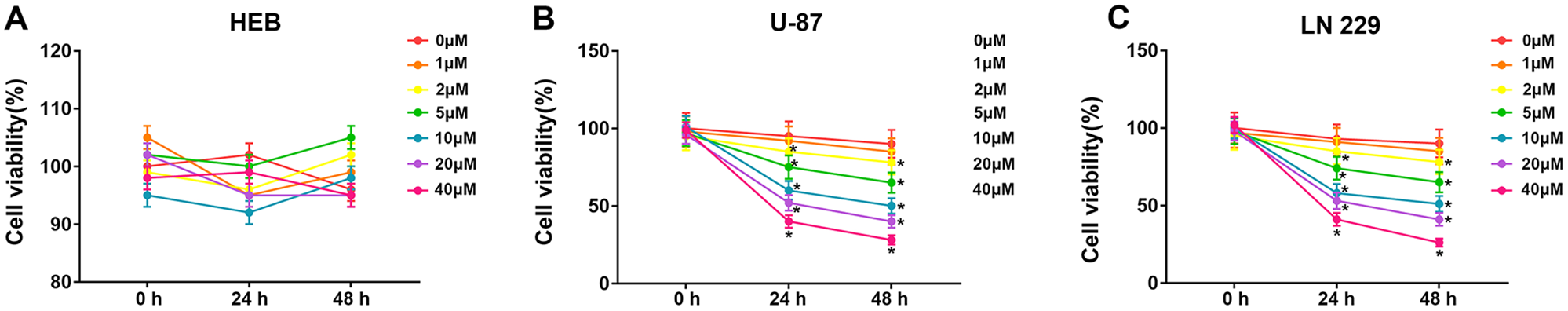
Cytotoxic effects of berberine on normal glial cells and glioma cells. (A–C) The percentage of viable HEB, U-87 and LN229 cells following treatment with the indicated doses of berberine.
Consistent with the above, berberine markedly decreased the levels of the pro-proliferative Ki67 and PCNA proteins in the glioma cells in a dose-dependent manner (Figure 2A and D). In addition, the pro-apoptotic proteins Bax and caspase-3 were upregulated, whereas the anti-apoptotic Bcl-2 was downregulated in the berberine-treated glioma cells (Figure 2B and E), indicating that berberine inhibited the proliferation of human glioma cells by inducing apoptosis. Annexin V/PI staining further showed an increase in the percentage of apoptotic glioma cells in response to berberine treatment (Figure 2C and F); compared to 3.39% in the untreated cells, the apoptosis rates in cells treated with 5, 10, and 20 µM berberine were 12.2%, 20.56%, and 31.14% respectively.

Berberine suppressed the proliferation of glioma cells and promoted apoptosis. (A, B, D, and E) Immunoblots showing expression of Ki67 and PCNA in (A) U87 cells and (D) LN229 cells, and of Bcl-2, Bax, and caspase-3 in (B) U87 cells and (E) LN229 cells. The grayscale ratio of target protein/GAPDH was calculated. (C and F) Flow cytometry plots showing apoptotic (C) U87 cells and (F) LN229 cells stained with Annexin V-FITC and PI. The percentage of early apoptotic cells is indicated.
To determine the effect of berberine on the metastatic potential of glioma cells, we evaluated its migration and invasion capacity in vitro through the wound healing and transwell assays. Berberine inhibited the migration of cells across the wound region in a dose-dependent manner (Figure 3A and C), and decreased the migration and invasion of the glioma cells in the transwell assay (Figure 3B and D). Taken together, these results suggested that berberine has an inhibitory effect on the malignant potential of glioma cells.

Berberine inhibited migration and invasion of glioma cells. (A and C) Percentage of migrating (A) U-87 cells and (C) LN229 cells in the wound healing assay after 24 hours treatment with berberine. (B and D) Percentage of invading (B) U-87 cells and (D) LN229 cells in the transwell assay after 24 hours treatment with berberine.
Berberine Inhibited TGF-β1/SMAD2/3 Signaling Pathway in Human Glioma Cells
To ascertain whether the TGF-β1/SMAD2/3 signaling pathway is involved in the anti-oncogenic effects of berberine on glioma cells, we analyzed the expression levels of TGF-β in the control and berberine-treated cells. Not surprisingly, berberine downregulated the expression of TGF-β protein in a dose-dependent manner (Figure 4A and C). Furthermore, the p-SMAD2 and p-SMAD3 levels were positively correlated with that of TGF-β, and decreased gradually with increasing concentration of berberine (Figure 4A and C). Likewise, the levels of secreted TGF-β level in the supernatants of berberine-treated U-87 and LN229 cells also decreased in a dose-dependent manner (Figure 4B and D). Altogether, these results indicated that berberine targets the TGF-β1/SMAD2/3 signaling pathway in human glioma cells. Based on these findings, we next analyzed the association between the expression level of TGF-β1, SMAD2, and SMAD3 and the prognosis of glioma patients using UALCAN data. As shown in Figure 5A, lower expression level of TGF-β1 correlated with higher survival probability regardless of the tumor grade. In addition, the median overall survival of glioma patients with different tumor grades improved with the lower expression level of SMAD2 and SMAD3 (Figure 5B and C). To summarize, the TGF-β1/SMAD2/3 axis is crucial for glioma progression and is targeted by berberine.

Berberine inhibited the TGF-β1/SMAD2/3 signaling pathway in glioma cells. (A and C) Immunoblot showing expression levels of TGF-β, SMAD2, SMAD3, p-SMAD2, and p-SMAD3 proteins in (A) U-87 cells and (C) LN229 cells after 24 hours treatment with berberine. The grayscale ratio of the target protein/GAPDH was calculated. (B and D) TGF-β levels in the supernatant of (B) U-87 cells and (D) LN229 cells treated with berberine for 24 hours.

TGF-β1, SMAD2, and SMAD3 are prognostically relevant in glioma. Correlation between the expression levels of (A) TGF-β1, (B) SMAD2, (C) SMAD3, and the survival probability of glioma patients with different tumor grades. Survival time was estimated using Kaplan-Meier curves and log-rank tests.
TGF-β Weakened the Inhibitory Effect of Berberine on Glioma Cells by Activating the TGF-β1/SMAD2/3 Signaling Pathway
To further verify that berberine inhibits the TGF-1/SMAD2/3 signaling pathway in glioma, the U-87 and LN229 cells were treated with recombinant TGF-β (10 ng/ml) and/or berberine (20 μM). While berberine inhibited the proliferation of both cell lines and decreased their viability, TGF-β increased the percentage of viable cells. However, the addition of berberine attenuated the proliferative effect of TGF-β (Figure 6A and E). Furthermore, the inhibitory effect of berberine on the expression levels of Ki67 and PCNA proteins was reversed by the addition of TGF-β (Figure 6B and F). Recombinant TGF-β also abrogated the pro-apoptotic effect of berberine on U-87 cells (Figure 6C and G) by upregulating Bcl-2, and downregulating Bax and cleaved caspase-3 expression (Figure 6D and H). Consistent with these findings, recombinant TGF-β enhanced wound healing and also reversed the inhibitory effect of berberine on the migration of glioma cells (Figure 7A and C). Similarly, TGF-β also promoted cell invasion in the transwell assay, and restored the invasiveness of berberine-treated glioma cells (Figure 7B and D). Thus, TGF-β attenuated the inhibitory effects of berberine on glioma cells and protected the cells against apoptosis. Furthermore, the expression levels of p-SMAD2, p-SMAD3, and TGF-β were upregulated in the cells treated with TGF-β alone but markedly downregulated following berberine treatment (Figure 8A and B). On the other hand, co-treatment with TGF-β and berberine led to a significant increase in TGF-β, p-SMAD2, and p-SMAD3 levels compared to that in the berberine group. Taken together, these results indicated that TGF-β might weaken the inhibitory effect of berberine on glioma cells by activating the TGF-β1/SMAD2/3 signaling pathway.
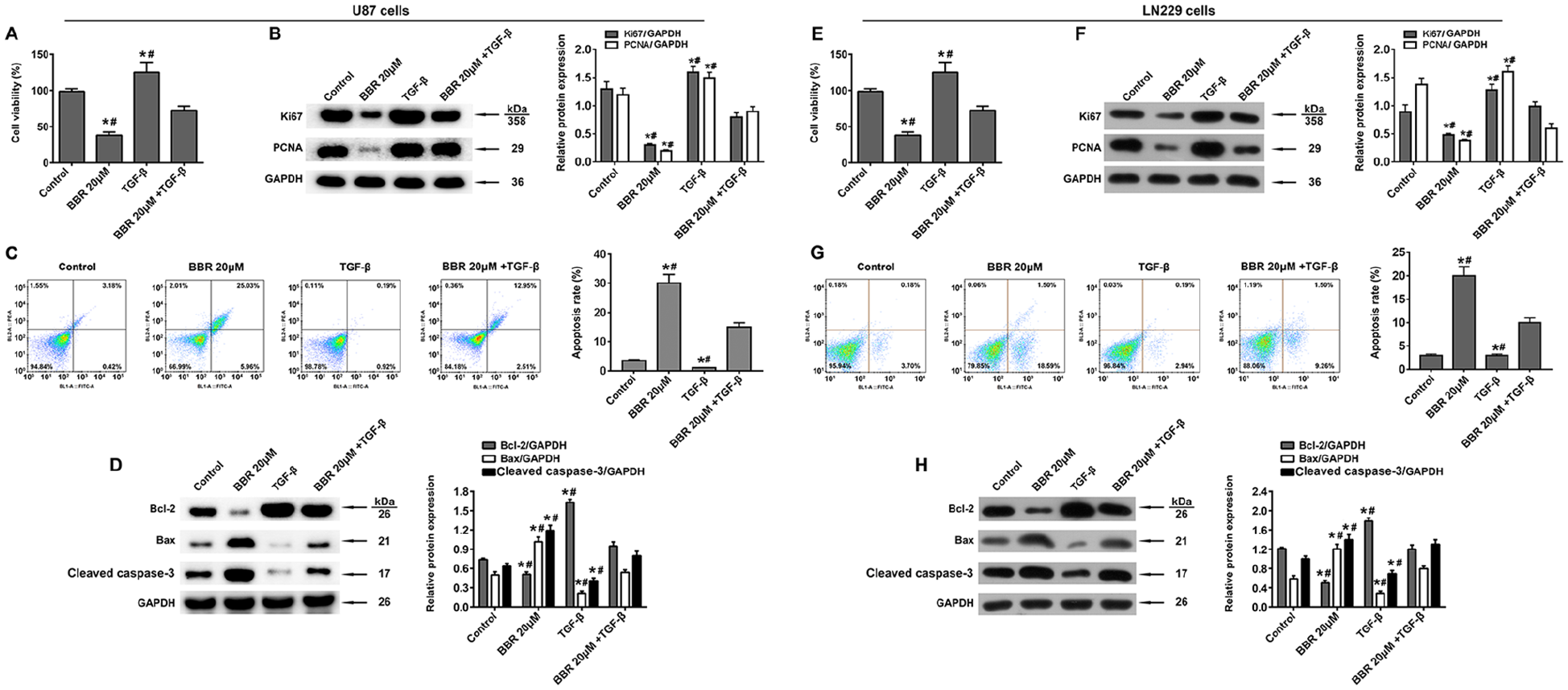
TGF-β reversed the effect of berberine on glioma cell proliferation and apoptosis. (A and E) Viability of (A) U-87 cells and (E) LN229 cells treated with TGF-β and/or berberine. (B and F) Immunoblot showing expression of Ki 67 and PCNA in (B) U-87 cells and (F) LN229 cells treated with TGF-β and/or berberine. (C and G) Percentage of early apoptotic (C) U-87 cells and (G) LN229 cells after treatment with berberine and TGF-β for 24 hours. (D and H) Immunoblot showing of Bcl-2, Bax, and activated caspase-3 in (D) U-87 cells and (H) LN229 cells treated with TGF-β and/or berberine. The grayscale ratio of target protein/GAPDH was calculated.

TGF-β1 reversed the effects of berberine on glioma cell migration and invasion. (A and C) Percentage of migrating (A) U-87 cells and (C) LN229 cells in the wound healing assay after treatment with berberine and TGF-β for 24 hours. (B and D) Percentage of invading (B) U-87 cells and (D) LN229 cells in the Transwell assay after treatment with berberine and TGF-β for 24 hours.
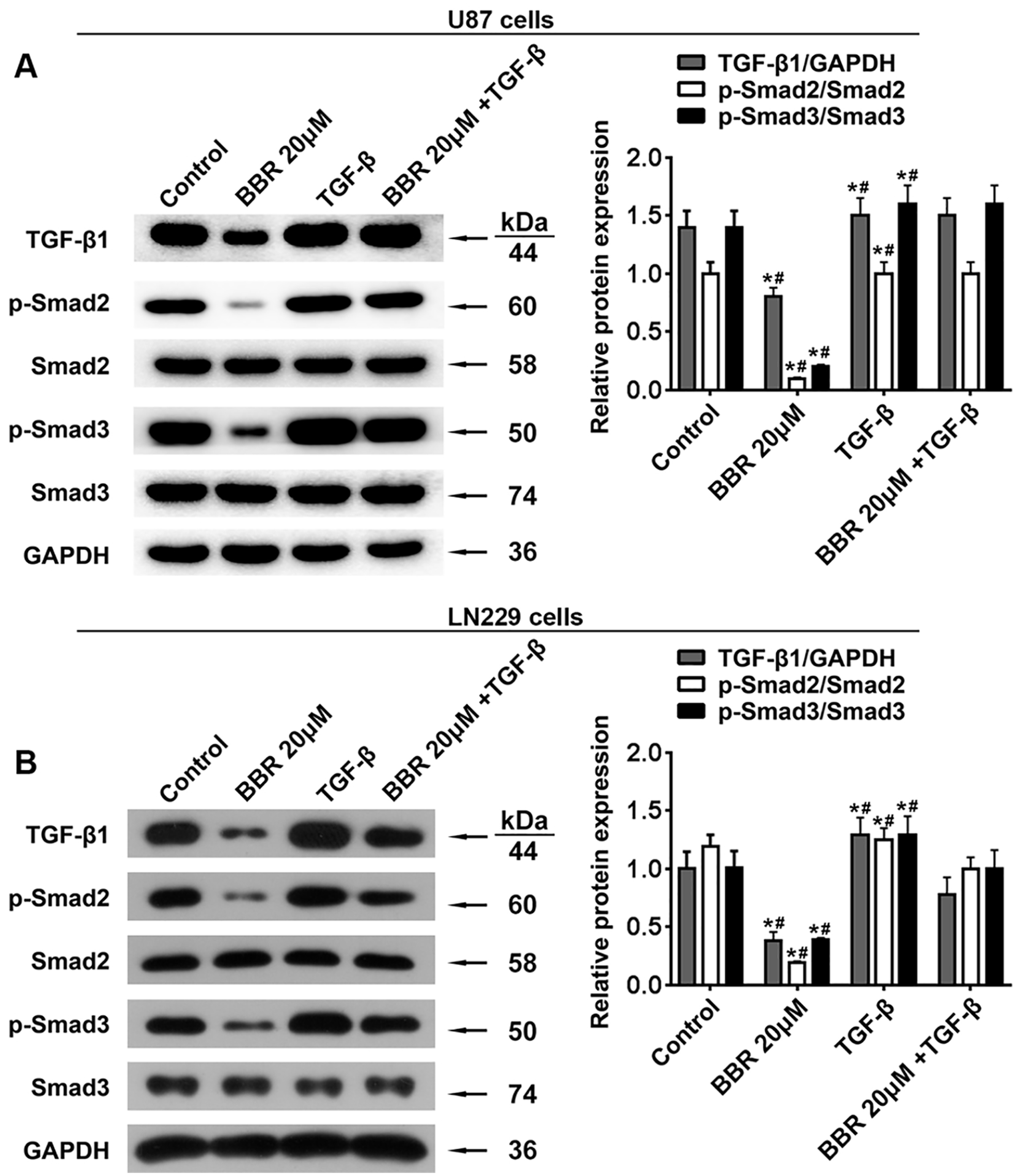
TGF-β1 neutralized the effect of berberine on TGF-β1/SMAD2/3 signaling pathway in glioma cells. Immunoblot showing the expression of TGF-β, SMAD2, SMAD3, p-SMAD2, and p-SMAD3 in (A) U-87 cells and (B) LN229 cells after 24 hours of berberine and TGF-β treatment. The grayscale ratio of target protein/GAPDH was calculated.
Discussion
Glioma is the most common primary tumor of the brain, as well as one of the most malignant and invasive tumors known in humans. 22 The incidence of glioma has increased over the last decades, and despite improvement in treatment strategies, patient survival remains dismal. 23 The progression, recurrence, and therapeutic resistance of tumors are closely related to the migration and invasion of tumor cells.24,25 Studies show that berberine not only inhibits the proliferation of tumor cells by inducing cell cycle arrest, but also promotes apoptosis and autophagy. 26 In addition, berberine can mediate TRAIL-mediated apoptosis recovery, 27 inhibit the NF-κB 28 and JAK-STAT pathways, 29 and regulate epigenetics. 30 A recent study showed that berberine induced apoptosis in glioblastoma U-87 MG cells via oxidative stress. 31 In addition, CYP2J2 expression was induced in berberine-treated human U251 glioma cells via peroxisome proliferator-activated receptor-α. 32 Berberine-induced apoptosis in the glioblastoma T98G cells is mediated by endoplasmic reticulum stress and the accompanying mitochondrial dysfunction. 33 Thus, berberine is a promising therapeutic agent that can target glioma cells via multiple pathways.
Glioma cell lines can be broadly classified into 3 major categories: promotion (U-87 MG), inhibition (U373 MG), or no obvious effect on proliferation. 34 Previous studies have shown that the activated TGFβ signaling pathway promotes tumor growth, including that of glioma stem cells. 35 In view of the etiology and pathogenesis of glioma and the TGF-β sensitivity of glioma cells are really complex and flexible. There is also evidence that berberine targets TGF-β1/SMAD signaling in cancer cells. For instance, berberine inhibited the invasion of nasopharyngeal carcinoma cells in response to TGF-β1 by suppressing EMT. 36 In addition, berberine is known to attenuate the severity of chronic pancreatitis and fibrosis by AMPK-mediated inhibition of TGF-β1/SMAD signaling and M2 macrophage polarization. 37 However, little is known regarding the effect of berberine on the TGF-β/SMAD pathway in glioma.
We found that berberine effectively inhibited the migration, invasion, and proliferation of different human glioma cell lines, and increased the apoptosis rates. At the molecular level, berberine decreased the expression of Ki67 and PCNA, which was the likely basis of its anti-proliferative effect. While Ki67 is a reliable indicator of the proliferative capacity of tumor cells, 38 PCNA is a marker of DNA synthesis and the S phase of cell cycle. 39 In addition, berberine upregulated cleaved caspase-3 and Bax, and downregulated Bcl-2 in a dose-dependent manner. Bcl-2 overexpression inhibits apoptosis via formation of Bcl-2/Bax heterodimer. On the other hand, Bax can form homodimers when expressed at high levels and trigger apoptosis. 40 Caspase-3 is activated in most apoptotic signaling pathways by cleaved caspase-9, and in turn activates caspase-6 and caspase-7 to initiate the apoptotic cascade. 41 Thus, berberine inhibits the proliferation of human glioma cells and induces apoptosis.
EMT is the underlying factor in the initiation and development of tumor metastasis. During EMT, the tumor cells lose their epithelial characteristics such as polarity and contact inhibition with adjacent cells, and become motile, which increases their migration and invasion into healthy tissues. 42 The high invasiveness and metastatic potential of tumor cells is the key cause of their recalcitrance to therapies and eventual recurrence. 43 Studies show that the TGF-β/SMADs signaling pathway facilitates metastasis and invasion by inducing EMT of tumor cells in response to multiple cytokines. 44 Huang et al 45 found that berberine suppressed EMT and growth of tumor cells by inhibiting TGF-β signaling, which promoted differentiation of adjacent stromal cells. Therefore, TGF-β is a potential target for tumor therapy. 46 We found that berberine inhibited the migration and invasion of human glioma cells, and downregulated p-SMAD2 and p-SMAD3. According to UALCAN data, lower expression level of TGF-β1, SMAD2, and SMAD3 correlated significantly with improved median overall survival of glioma patients with different tumor grades. Subsequently, we found that recombinant TGF-β promoted the proliferation, migration, and invasion of glioma cells, inhibited apoptosis, and upregulated p-SMAD2 and p-SMAD3. In addition, TGF-β also reversed the effects of berberine of the malignant behavior of glioma cells, and restored the levels of p-SMAD2 and p-SMAD3. Taken together, berberine restrained the progression of human glioma cells by inhibiting the TGF-β1/SMAD2/3 signaling pathway.
There are certain limitations in our study that ought to be considered. First, the protective effects and mechanisms of berberine in glioma cells were assessed in in vitro models, and will have to be validated in in vivo experiments and clinical trials. Secondly, the other potential mechanisms and pathways associated with berberine in glioma need to be further explored. In conclusion, berberine significantly inhibited the proliferation, migration, and invasion of human glioma cells and promoted apoptosis by blocking the TGF-β1/SMAD2/3 pathway. Our findings provide new insights into the mechanisms underlying glioma progression, and the possibility of using berberine as a therapeutic agent.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
Y.J. and W.Z.S. were responsible for the concept and study design, and provided critical input; J.W.Z. and Y.F.P. were responsible for laboratory experiments, data collection, data analysis, and interpretation; W.Z.S. was responsible for drafting the manuscript; Y.J. provided critical review of the manuscript.
Availability of Data and Material
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our study did not require an ethical board approval because it did not contain human or animal trials.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
