Abstract
OBJECTIVE:
Hepatocellular carcinoma (HCC) is a highly aggressive malignancy that has a poor prognosis. Through the literatures, TINAG significantly participated in the processes of the renal-associated diseases, but there were no studies about the roles of TINAG in the HCC development. Hence, we attempted to use the HCC samples collected by ourselves to reveal the clinical significance and prognostic impact of TINAG in HCC.
METHODS:
We first measured the expression level of TINAG in HCC on the basis of TCGA database. Then, real time quantitative reverse transcription PCR (RT-qPCR) was used to examine the expression level of TINAG in 100 pairs of HCC tissues and corresponding adjacent non-tumor tissues, as well as HCC cell lines (HepG2, HB611, HHCC, and Hep3B). Moreover, Kaplan-Meier method and COX’s proportional hazards model were utilized to perform the survival and prognosis analyses using the clinical data collected by ourselves. After knockdown of TINAG, the cell proliferation, invasion and migration capacities of HepG2 and Hep3B cells were evaluate by counting kit-8 (CCK-8) assay (24 h, 48 h, 72 h, and 96 h post-cultivation), clone formation experiment, would-healing, and invasion as well as migration assays. To further explore whether the dys-regulated TINAG expression regulates the HCC progression and prognosis, protein biomarkers of PI3K signaling pathway, including AKT, p-AKT, PI3K, p-PI3K, p70S6K, and p-p70S6K were measured based on western blotting analysis.
RESULTS:
According to the data of TCGA database, clinical patients, and HCC cell lines, TINAG was highly expressed in HCC compared with normal. Relationship of TINAG expression level with the clinicopathological factors implicated that the high expression of TINAG was significantly associated with pathologic stage, pathologic-node, and pathologic-metastasis. Univariate as well as multivariate COX analysis indicated that TINAG expression and pathologic metastasis can serve as the independent prognostic factor for overall survival of HCC. After TINAG knockdown in HepG2 and Hep3B cells, cell proliferation rate, the colony numbers, and the invasive and migratory capacity were found to be suppressed. Remarkably, western blot results showed that reduction of TINAG remarkably decreased p-AKT, p-PI3K, and p-p70S6K expression level in HepG2 and Hep3B cells.
CONCLUSION:
Collectively, our results underscore the significance of TINAG in HCC progression and prognosis, and TINAG might be a novel candidate oncogene in HCC. These results propose that targeting TINAG might offer future clinical utility in HCC.
Introduction
Hepatocellular carcinoma (HCC), accounting for 90% of all primary liver neoplasias, is the third highest cause of cancer-related mortality in the world [1]. Estimatedly, half of the deaths in China is related to HCC [2]. Although there are the available treatment strategies including surgical resection, and liver transplantation, the incidence rate of HCC nearly equals the mortality rate. Therefore, new therapeutic modalities are urgently needed. Currently, molecular therapies precisely targeting genes or signaling pathways that are involved in the HCC progression and metastasis are promising options in the improvement of prognosis.
Many biologic functions have been ascribed to basement membranes. Of note, the precise expression of basement membrane proteins helps to induce tissue differentiation and to determine cellular phenotype [3, 4]. Additionally, the basement membrane proteins are involved in cell adhesion, invasion, migration, and serve as selective barriers in the filtration of macromolecules [5, 6, 7]. Significantly, some basement membrane macromolecules have been suggested to be connected with autoimmune disorder [8], and recently, tubulointerstitial nephritis antigen (TINAG) is a described basement membrane macromolecule. Initially, TINAG was detected as a nephritogenic antigen participated in tubulointerstitial nephritis regulated by the anti-tubular basement membrane (TBM) autoantibody present in sera of renal transplant recipients [9]. In humans, it locates in chromosome 6p11-6p12 [10]. A former report has demonstrated that TINAG can promote cell adhesion by interacting with type IV collagen and laminin [11]. Through the literatures, we found that TINAG significantly participated in the processes of the renal-associated diseases, but there were no studies about the roles of TINAG in the cancer development. Tubulointerstitial nephritis antigen-like 1 (TINAGL1) is an important paralog of TINAG. TINAGL1 is a novel matricellular protein in the extracellular matrix (ECM). It is known that TINAGL1 is a positive regulator of angiogenesis and by enhancing invasion in mice [12]. Naba et al. [13] have demonstrated that TINAGL1 is over-expressed in highly metastatic tumors. Further, Umeyama et al. [14] have suggested that TINAGL1 might be a potential candidate for drug compounds in non-small cell lung cancer, by inhibiting its gene expression. Accordingly, we put forward a hypothesis in the current study that TINAG could also affect the progression and metastasis of HCC. Thus, we attempted to unveil the clinical significance of TINAG in HCC.
To verify our aforementioned assumption, we measured the TINAG expression levels in human HCC cell lines and HCC tissues, and then revealed the potential correlations between the expression of TINAG and clinicopathological features, and clinical prognosis in patients with HCC. Additionally, colony formation, cell viability, and cell migration assay were implemented in HepG2, and Hep3B cell lines to investigate the in vitro effect of TINAG in HCC cells. In the current work, we observed that the expression of TINAG was elevated in HCC cell lines and HCC tissues. TINAG overexpression was related to tumor invasion. In in vitro study we suggested that TINAG-dependent cell migration and invasion found in HCC, which was at least partially relevant to the abnormal activation of the PI3K/AKT signaling pathway.
Materials and methods
Data mining in the TCGA database
The data of the gene TINAG were recruited from the database of The Cancer Genome Atlas (TCGA) covering the clinical data from 374 samples of HCC and 50 normal tissue samples. LIMMA package was used to measure the difference of TINAG expression level between normal tissues and HCC samples [15].
Tissue samples
One hundred pairs of HCC and corresponding adjacent normal tissues were randomly collected with written consent between July 2009 and December 2011 at the Department of Hepatobiliary, Shandong Provincial Hospital Affiliated to Shandong University. All sample collections were implemented after the approval of the ethics committee of Shandong Provincial Hospital Affiliated to Shandong University. All collected samples were immediately frozen and stored at
Cell culture
Four human HCC cell lines including HepG2,HB611, HHCC, and Hep3B, and normal human liver cell line HL-7702 were obtained from the Shanghai Cell Bank at the Chinese Academy of Sciences (Shanghai, China). These cells were cultured in Roswell Park Memorial Institute (RPMI)-1640 medium supplemented with 10% fetal bovine serum, 100 U/ml penicillin, and 0.1 mg/ml streptomycin at 37
Small interfering RNA
Small interfering RNA (siRNA) against TINAG (si-TINAG) and one negative control (si-NC) having no definite target were designed and synthesized by GenePharma (Shanghai, China). Cells were seeded on six-well plates at a density of 3
Sequences of siRNA for TINAG was F: 5’-UUCAG GUUCCAAGGAGAAG-3’.
RNA extraction and RT-qPCR
Total RNA of cell lines HepG2, HB611, HHCC, and Hep3B were extracted using Trizol reagent (Invitrogen, Carlsbad, CA, USA). The primers sequences were: TINAG: F: 5’-GTTCCAAGGAGAAGC CCACA-3’, R: 5’-CCGGTCCACATTCTCTGGAA-3’. GAPDH: F: 5’-GGAGCGAGATCCCTCCAAAAT-3’, R: 5’-GGCTGTTGTCATACTTCTCATGG-3’. The PCR procedure was performed using real time PCR detection system based on the following conditions: 95
Cell biological assays (proliferation, colony formation, wound-healing assays, invasion and migration)
Assessment of cell viability
A cell counting kit-8 (CCK-8) assay was used to evaluate cell viability [16]. After 72 h of transfection, HepG2 and Hep3B Cells were seeded into 96-well plate at a density of 2
Colony formation
For colony formation, it was conducted as described previously [17, 18]. Specifically, cells were seeded in six-well plate 48 h after transfection and incubated at 37
Wound-healing assay, cell migration and invasion assay
Cells were seeded in duplicates in 6-well plates. Once confluent, the cultures were pre-treated with serum-free media for 12 h. The following day, the monolayers were scratched using a sterile micropipette tip with size of 10
TINAG level in HCC was significantly over-expressed. (A) TINAG expression level between the patients with HCC (
Cell invasion and migration experiments were conducted by means of chambers containing transwells with a pore size of 8
Cells were harvested and lysed in a lysis buffer. The protein concentrations were examined using the BCA Protein Assay Kit. Cell lysates (20
Statistical analysis
In the current study, all statistical analyses were implemented using SPSS22.0 software. Data were presented as the mean
Results
TINAG expression is up-regulated predominantly in HCC tissues and cell lines
Firstly, the TINAG expression level in HCC samples (HCC samples
The expression of TINAG was also measured in our collected human HCC tissues using RT-qPCR. Results exhibited that TINAG expression level was significantly higher in HCC tissues than that in the corresponding adjacent normal tissues (Fig. 1B,
TINAG expression was also confirmed in HCC cell lines, including (HepG2, HB611, HHCC, and Hep3B), and the normal HCC cell line HL-7702 (Fig. 1C). No difference was observed in the expression level of TINAG in HB611 cells, relative to HL-7702. However, TINAG expression level in HepG2, HHCC, and Hep3B cells was significantly higher than HL-7702 cells (
Correlation between TINAG expression and clinicopathological parameters of HCC
Correlation between TINAG expression and clinicopathological parameters of HCC
Note: T: tumor, N: node, M: metastasis.
Next, we classified the 100 HCC patients into two groups according to the median score of TINAG expression level in all samples as the threshold (high-expression or low-expression). Clinical factors (age, gender, grade, pathologic stage, pathologic-tumor, pathologic-node, pathologic-metastasis) were compared between the two groups (Table 1). From this table, we observed that the TINAG expression level was significantly associated with pathologic stage (
Kaplan-Meier overall survival (OS) curves based on TINAG expression level.
Univariate and multivariate analysis of clinical prognostic factors of HCC
qRT-PCR and western blotting analysis of TINAG expression levels in HepG2, and Hep3B cells infected with siRNA. **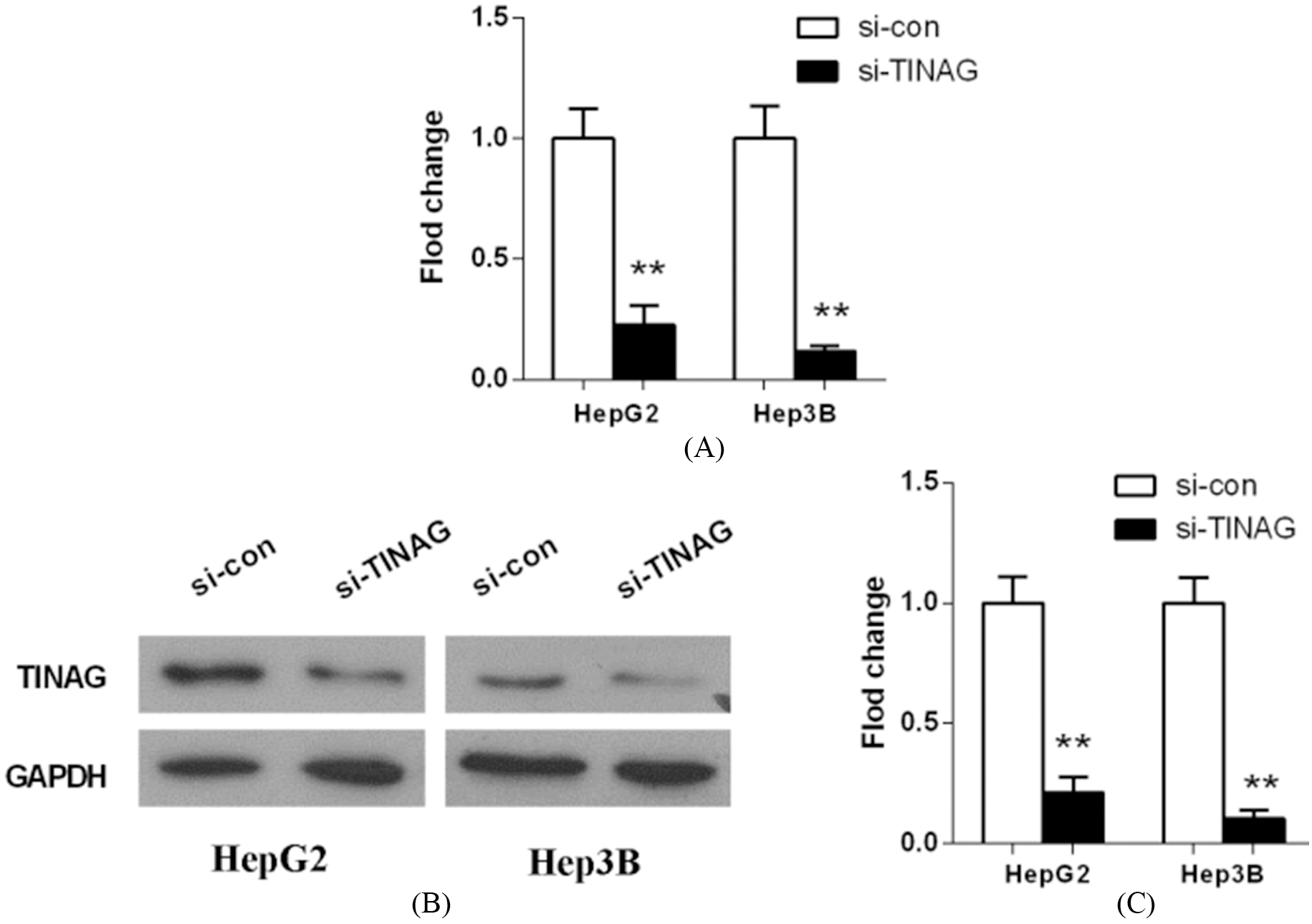
CCK-8 assays were conducted to determine HepG2 (A), and Hep3B (B) cell proliferation after 24 h, 48 h, 72 h, and 96 h of cultivation. **
Colony formation assay was conducted to determine the proliferation of HepG2, and Hep3B cells infected with siRNA. The colonies were imaged and counted. Colony formation assay results are expressed as histograms. Data are presented as the means 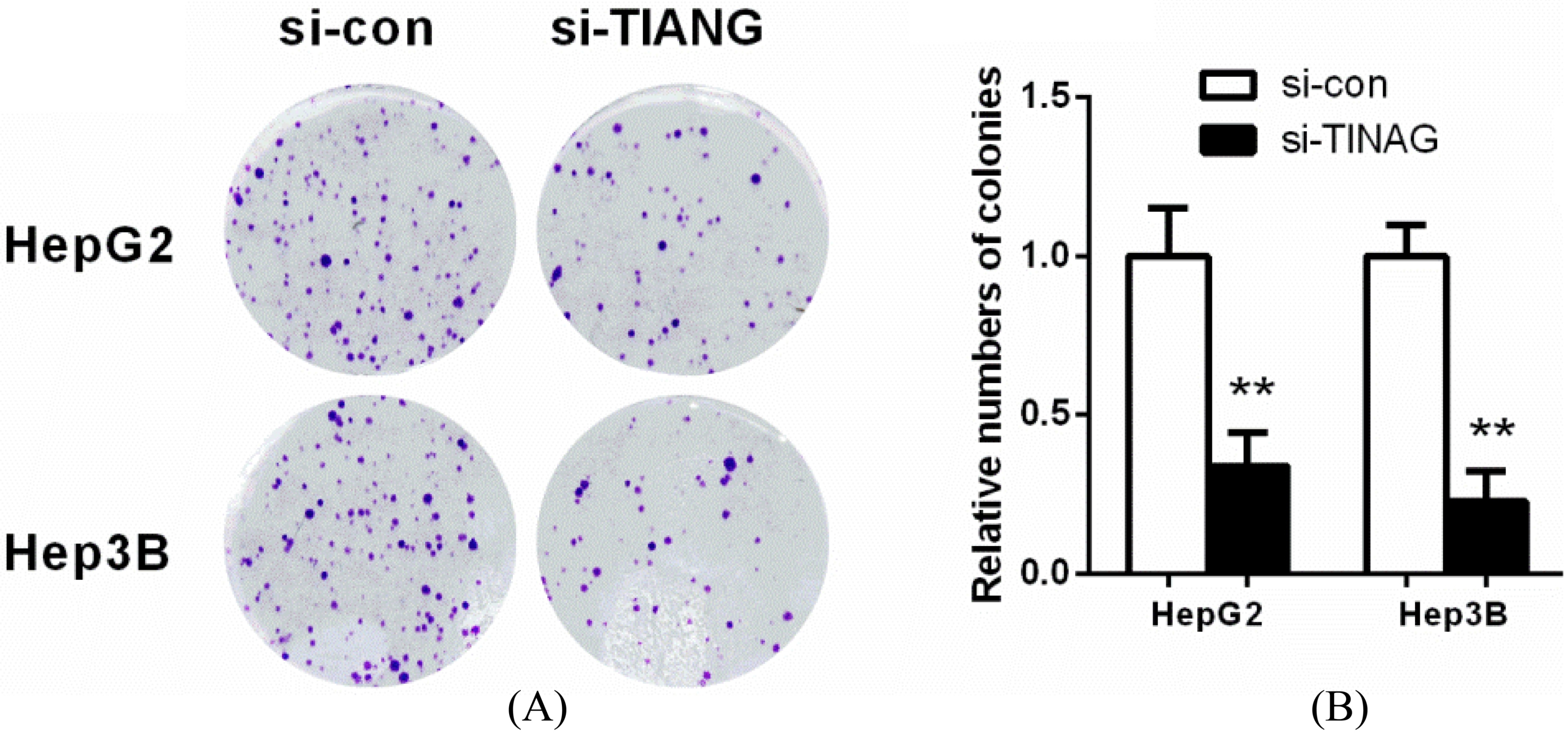
TINAG knockdown inhibits the invasion and migration of HepG2 cells. A. Wound healing assay utilized to determine the migration ability of HepG2 cells with or without TINAG knockdown. Representative images of 0 h and 24 h of three repeated experiments are presented. B and C. Transwell assay was carried out to determine the invasion and migration ability of HepG2 cells with or without TINAG knockdown. **
TINAG knockdown inhibits the invasion and migration of Hep3B cells. A. Wound healing assay utilized to determine the migration ability of Hep3B cells with or without TINAG knockdown. Representative images of 0 h and 24 h of three repeated experiments are presented. B and C. Transwell assay was carried out to determine the invasion and migration ability of Hep3B cells with or without TINAG knockdown. **
The expressions of AKT, p-AKT, PI3K, p-PI3K, p70S6K in HCC cell lines (HepG2, and Hep3B) after transfection analyzed using western blotting. Bars present means 
Figure 2 shows the result of survival analysis. We observed that HCC patients with high TINAG expression had a worse prognosis, relative to those with low level of TINAG (
Subsequently, univariate and multivariate analyses were performed to prove the prognostic role of TINAG for survival in HCC patients (Table 2). In univariate analysis, TINAG expression (
Knockdown of TINAG decreases cell growth in vitro
Because of the abnormal expression of TINAG in HCC cell lines and tissues, we inferred that it might play some key roles in HCC progression. To evaluate the role of TINAG in HCC growth, we first silenced TINAG expression in HepG2 and Hep3B cells by siRNA. The silencing efficacy of decreasing TINAG expression level was more than 90%. Figure 3 showed that si-TINAG transfected cells had a significant decrease both in mRNA and protein level of TINAG, when comparing with those in non-transfected cells (
CCK8 assays revealed that cell growth was inhibited in both HepG2 and Hep3B cell lines transfected with siRNAs, relative to negative control (Fig. 4,
TINAG knockdown impairs the invasion and migration in HCC cells
Scratch experiment as well as transwell assay were used to assess the affect of TINAG on HCC cell migration and invasion. The wound-healing array exhibited that TINAG knockdown remarkably inhibited HepG2 and Hep3B cell migration (Figs 6A and 7A), which was quantified by the percentage of the wound area (Figs 6A and 7A).
Results showed that both HepG2 and Hep3B cells after TINAG knockdown had significantly smaller populations of invasive and migrant cells compared to the corresponding negative controls (Figs 6B, 6C and 7B, 7C). Taken together, these results suggested that TINAG regulated HCC cell mobility and was involved in HCC metastasis in vitro.
TINAG knockdown inhibits PI3K/AKT pathway activation in HepG2 and Hep3B cells
PI3K/AKT signaling pathway may play a key role in the proliferation, and invasion of HCC cells. Thus, we measured the effects of TINAG on the star protein of PI3K/AKT pathway (AKT, p-AKT, PI3K, p-PI3K, p70S6K, and p-p70S6K) using western blotting. Figure 8 exhibited that TINAG knockdown significantly decreased the expression level of p-AKT, p-PI3K, p70S6K, and p-p70S6K in HepG2 and Hep3B cells (
Discussion
Few studies have investigated the roles of TINAG in HCC. To the best of our knowledge, this is the first report to explore the function of TINAG in HCC. Our results indicated that over-expressed TINAG is frequent in human HCC cells as well as tissues. We constructed stable TINAG knockdown cell lines and observed that TINAG regulates cell invasion and migration in HCC cell lines (HepG2 and Hep3B) in vitro. Mounting evidence has suggested that cell migration plays important roles in advanced tumors and might be related to the invasion into adjacent tissues and the formation of metastases [19, 20]. Previous studies have demonstrated that TINAG is associated with cell adhesion and the formation of ECM [11, 21, 22]. One of the first steps in cancer development and metastasis is loss of cell-to-cell adhesion [23]. Based on these results, we confirmed that TINAG-dependent migration occurs in HCC and demonstrate that TINAG is a crucial potential molecule fostering HCC progression and driving metastasis in liver cancer.
The PI3K/AKT is a well known signaling pathway which regulates cellular processes, such as cell proliferation, survival, apoptosis, and migration. In addition, activation of PI3K/AKT signaling exert a key function in maintaining the biological features of malignant cells, including EMT [24]. Persistent AKT activation was associated with low E-cadherin, which could be reversed by the AKT inhibitor [25]. Significantly, Xie et al. [26] have found that cells maintained on the TINAG substratum increases the expression of p-AKT, which was remarkably inhibited with the siRNA transfection in the culture medium. Once the PI3K/AKT signaling pathway is activated, p70S6K directly phosphorylates S6 ribosomal protein [27], leading to enhanced proliferation ability of tumor cells. In the present study, the TINAG-induced suppression of proliferation and migration accompanied by the inhibition of the phosphorylation and activation of PI3K/AKT. In line with our results, several lines of evidence have implicated the roles of PI3K/AKT signaling pathway in the proliferation and invasive potential of HCC cells [28, 29, 30, 31]. Demonstrated here, we speculate that the suppression of PI3K/AKT pathway could account for the anti-tumor effects of TINAG in HCC cell lines HepG2 and Hep3B.
Taken together, the results of our study clearly demonstrate that TINAG is an available factor for HCC prognosis, and this study provides evidence that TINAG suppresses the in vitro cell proliferation, invasion, and migration of HCC cells, in part by inactivating PI3K/AKT signaling pathway. Over-expression of TINAG was not only significantly associated with aggressive clinicopathological factors, but also was an independent poor prognostic biomarker predicting shorter OS in HCC patients. As evidenced by our results, TINAG could be a diagnostic and prognostic biomaker in HCC patients and might be a potential novel target for developing a preventive agent for HCC prognosis. In future studies, we will use animal models for deeper investigation and attempt to explore the molecular mechanisms underlying the altered expression of TINAG in HCC.
Footnotes
Acknowledgments
The work was supported by the National Natural Science Foundation of China (No. 81373172).
