Abstract
OBJECTIVES:
The goal of our study was to assess the prognostic impact of the necroptosis relative protein RIPK1 genetic polymorphism in ischemia-reperfusion injury and survival after hepatectomy in hepatocellular carcinoma (HCC) patients.
METHODS:
In this study, expression of RIPK1 and its genetic polymorphism(rs2272990) were examined in plasma of 44 HCC patients. All these patients were undergoing partial hepatectomy. The prognostic values of RIPK1 genetic polymorphism for tumor development and survival, and ischemia-reperfusion injury after hepatectomy were further determined.
RESULTS:
Plasma RIPK1 expressions were significantly increased in HCC patients, compared to the healthy control group. Totally 19 patients have the GA
CONCLUSION:
In conclusion, the RIPK1 polymorphism is an indicator of hepatic injury and a novel prognostic biomarker for tumor development and survival of HCC recipients after hepatectomy.
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer worldwide and the second leading cause of cancer death in China [1]. Currently, partial hepatectomy and orthotopic liver transplantation (OLT) have been considered as the most effective treatment methods for HCC [2]. During major hepatectomy, intermittent clamping of the hepatoduodenal ligament is commonly performed to reduce intraoperative blood loss. However, this procedure may lead to hepatic ischaemia-reperfusion injury [3]. The common pathology of ischaemia-reperfusion injury is that the transient disruption of the normal blood supply to target organs followed by reperfusion induces an acute generation of reactive oxygen and nitrogen species subsequent to reoxygenation upon vascular reopening. These changes initiate a chain of deleterious cellular responses leading to inflammation, cell death and eventually causing post-hepatectomy liver failure [4]. Previous studies have demonstrated that severe ischemia-reperfusion injury (IRI) at early-phase after hepatectomy or live transplant not only promotes tumor growth but also provides a favorable environment for tumor progression and invasion [5]. Therefore, it was considered that biomarkers for the early-phase IRI might also play an prognostic role on the late late-phase tumor recurrence of HCC patients.
Animal experiments and clinical studies have indicated that cell death is closely related to ischaemia-reperfusion injury. Apoptosis, autophagy-related death and necrosis are the major processes of mammalian cell death. Anti-apoptosis has been largely reported to protect the organs from ischaemia-reperfusion induced injury [6, 7, 8], and increasing evidence indicates that modulation of autophagy is a novel therapeutic strategy for this disorder [8, 9]. Recently, the programmed necrosis or ’necroptosis’ cell death pathway has attracted much interest because of its implication in multiple pathologies, including inflammatory diseases and the cell death arising from ischaemia-reperfusion injuries [10]. The most terminal-known obligate effectors in the necroptosis pathway, receptor (TNFRSF)-interacting protein kinase 1 (RIPK1), RIPK3 and mixed lineage kinase domain – like protein (MLKL), hold particular appeal because of their known function as a mediator of necroptotic cell death [11]. RIPK1 rs2272990 SNP site was located in the coding region in 6 Chromosome. The major allele of this site was G, and the mutant allele was A. This SNP site has been identified recently as a potential indicator for survival after curative resection in patients with colorectal cancer [12]. However, the SNPs with significance should be confirmed in a larger study and need to be validated in more kinds of cancers. The goal of our study was to assess the prognostic impact of the necroptosis relative protein RIPK1 expression and genetic polymorphism in ischemia-reperfusion injury and tumor development and survival after hepatectomy in HCC patients.
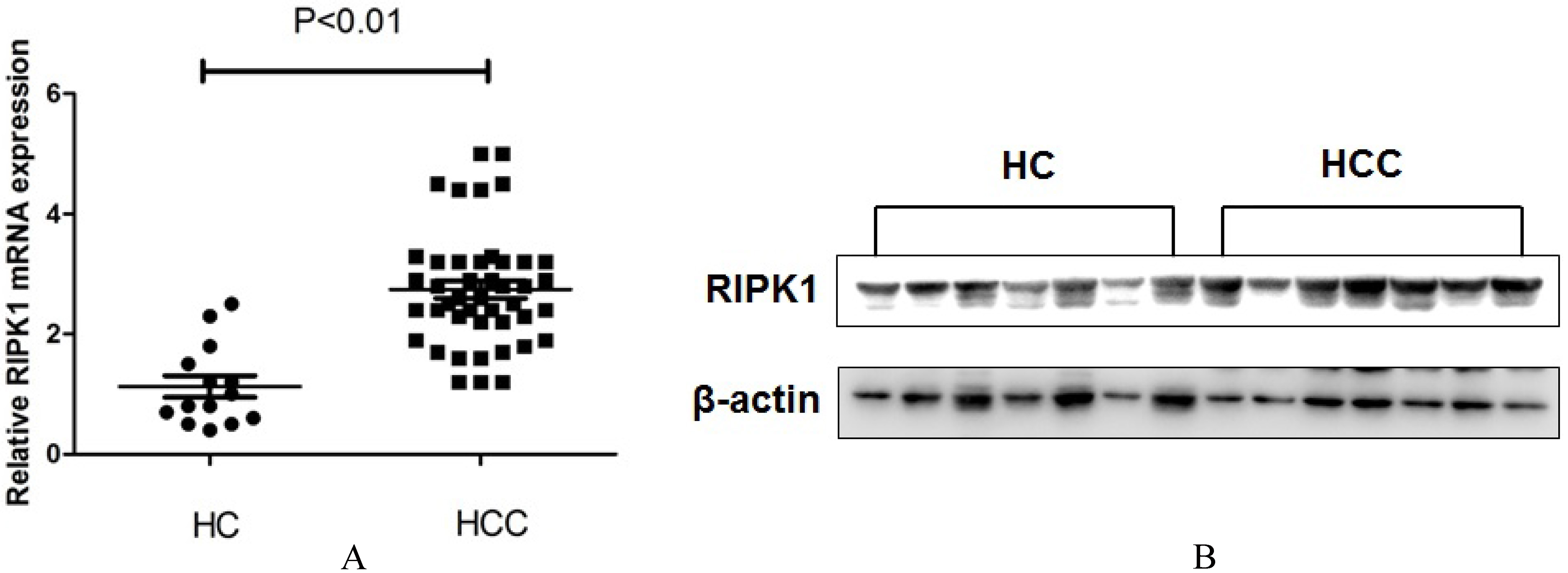
Comparing the (A) mRNA and (B) protein level between HCC patients and healthy controls.
Clinical subjects
HCC patients in Chinese People’s Liberation Army (PLA) 309 hospital and normal volunteers who did not differ from the HCC patients by age, gender, or BMI, were included in this subject. The study was approved by the Ethics Committee of the 309th Hospital of PLA and informed consent was obtained from all patients before participation. The 7th edition TNM staging system for hepatocellular carcinoma was used in this work in accordance with previous report [13]. All the experiments were carried out according to principles of Helsinki Declaration [14]. Blood biochemistry, coagulation function, chest X-ray, and electrocardiogram were performed to assess the patient’s bodily functions. Transabdominal ultrasound, CT angiography, multislice spiral computed tomography and three-dimensional reconstruction system were used to ascertain the size and location of the lesion. ICG retention rate at 15 min (ICGR15), which has been reported to be effective for preoperative evaluation of hepatic functional reserve [15], was used here to evaluate liver function.
During the liver parenchyma resection surgery, the portal triad was clamped using a silastic catheter. Blood samples were collected at the following time point: A) before surgery (PRE), B) after surgery (AS), C) six hours after surgery (AS 6), D) postoperative days 1 (Day 1), 3 (Day 3), 5 (Day 5). Then liver functions was assessed by monitoring ALT, AST, total bilirubin (TBIL), and direct bilirubin (DBIL).
Blood total protein extraction
Blood samples were extracted according to previous description [3]. Briefly, blood samples were suspended in ice-cold lysis buffer (50 mM Tris (pH 7.4), 5 mM EDTA, 250 mM NaCl, 0.1% Triton X-100, 1 mM phenylmethylsulfonyl fluoride, 5
Western blotting assay
Fifty microgram aliquots of lysates were loaded on a sodium dodecylsulfate (SDS) polyacrylamide 10% gradient gel and transferred to a polyvinylidene difluoride membrane. The membranes were blocked with 5% nonfat dry milk in Tris-buffered saline, pH 7.4, containing 0.05% Tween 20, and were incubated with primary antibodies (1:200; Santa Cruz, Delaware Avenue, CA, USA) and horseradish peroxidase-conjugated secondary antibodies (1:5000; Santa Cruz) according to the manufacturer’s instructions. The protein of interest was visualized using an enhanced chemiluminescence (ECL) Western blotting substrate (Pierce) and the Chemidoc XRS Gel Documentation System (BioRad). Antibodies against RIPK1(#3493) was obtained from cell signal.
Relationship between Plasma RIPK1 expression and clinicopathologic characteristics in HCC patients (
44)
Relationship between Plasma RIPK1 expression and clinicopathologic characteristics in HCC patients (
Total RNA extraction of blood samples from human was performed using TRIzol (Invitrogen) according to the manufacturer’s instructions. Subsequently, RT-qPCR was carried out with the PrimeScript RT-PCR kit (Takara) using
RIPK1 forward 5
The following PCR cycle was used: 95
Genotyping of RIPK1 rs2272990 SNP polymorphism
Genomic DNA was extracted from blood samples of patients using DNeasy Blood & Tissue Kit (QIAGEN; Germany) according to the manufacturer’s guidelines. The rs2272990 genotypes for the patients were determined using Sequenom MassARRAY according to the previous report [12]. Briefly, the biotin-labeled PCR product was captured with streptavidin-coated paramagnetic beads (Dynal Biotech Inc., Lake Success, NY, USA), and the single-strand DNA was isolated by denaturing the bound PCR product with NaOH and washing with a buffer. After removing the wash solutions, a PROBE reaction mixture was added [1 U Thermosequenase (Amersham Pharmacia Biotech, Inc., Piscataway, NJ, USA), 26 mM Tris–HCl pH 9.5, 6.5 mMMgCl2, 50 Meach assay-specific dNTP/ddNTP, and 10 M PROBE primer]. The incubation temperatures were identical for all the tests (80
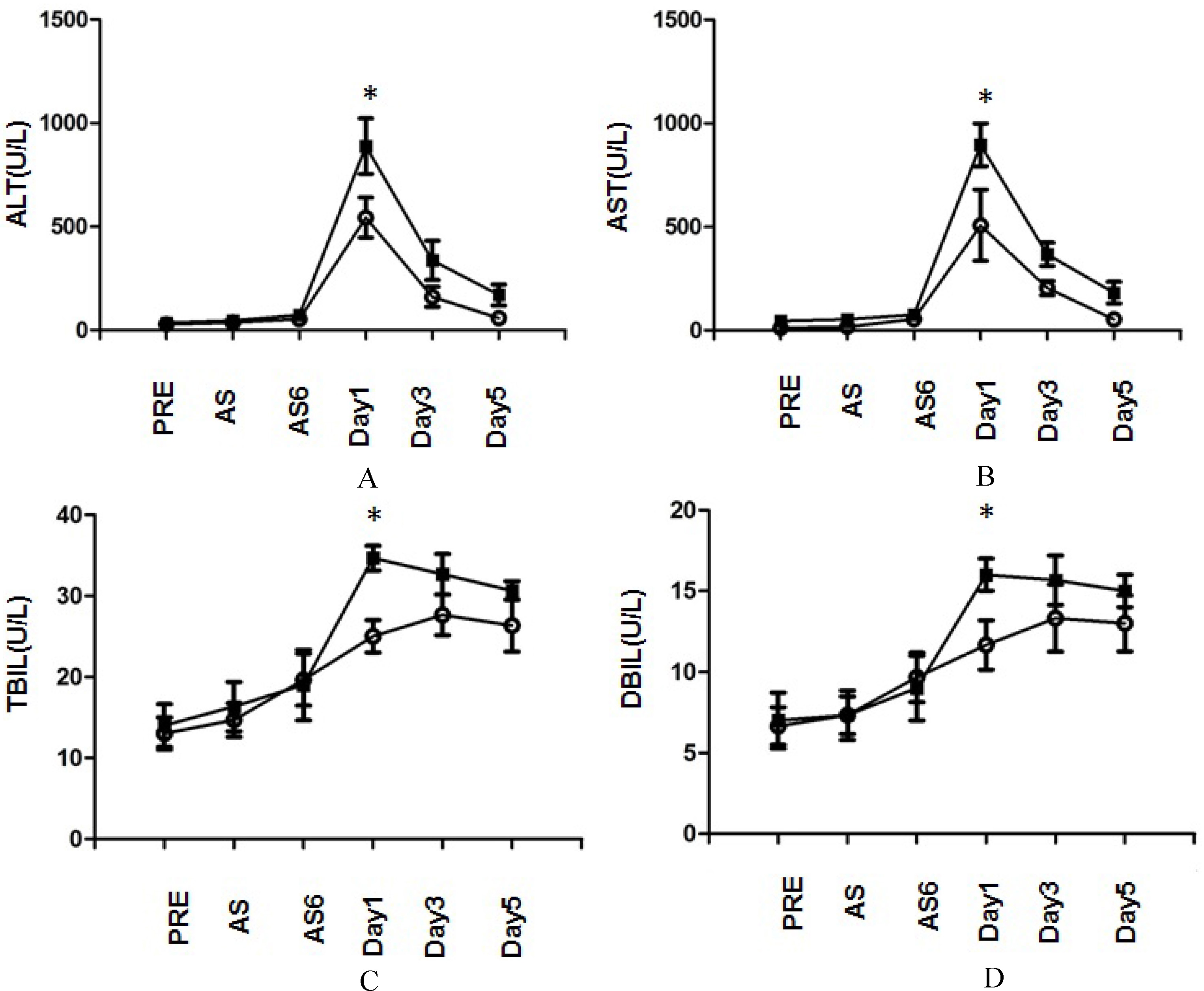
Plasma (A) ALT, (B) AST, (C) TBIL and (D) DBIL levels after partial hepatectomy under continuous inflow occlusion of HCC patients with different genetic polymorphism at RIPK1 rs2272990 SNP site.
Normality test indicated that all the results was are normally distributed, so the results are expressed as means
Results
Plasma RIPK1 expression was increased in HCC patients
The clinical characteristics of HCC patients were shown in Table 1. The HCC group consisted of 44 subjects (13 women). Totally 14 healthy volunteers (4 women) were included in this work. There were no significant differences between the groups regarding the age, gender and body mass index (BMI).
RT-PCR (Fig. 1(A)) and western blotting (Fig. 1(B)) assay demonstrated that the mRNA and protein expressions of plasma RIPK1 were significantly increased in HCC patients, compared to the healthy control group. To evaluate the relationship between RIPK1 expression and the clinical features of HCC, we divided the HCC patients into the high (25 cases) and low (19 cases) RIPK1 expression groups according with the optimal cut-off value (Relative mRNA value
Genetic polymorphism of RIPK1 was correlated with liver injury after hepatectomy
Totally 17 patients have the GA
Genetic polymorphism of RIPK1 predicted survival of HCC recipients after hepatectomy
To investigate the relationship between genetic polymorphism of RIPK1 and clinical prognosis, the above 44 HCC patients were followed up. The 1-year, 3-year and 5-year overall survival rate (OS) were 63.6% (28/44), 31.8% (14/44) and 13.6% (6/44), respectively, for all of the patients in this study. Kaplan-Meier survival analyses demonstrated that GG genotype had an unfavorable overall survival prognosis (Fig. 3(A),

Kaplan-Meier curves for time to (A)recurrence and (B)overall survival of patients with different genetic polymorphism at RIPK1 rs2272990 SNP site. Blue line: GG genotype; Red line: GA+AA genotype.
Cancer development is tightly associated with various cell deaths [16]. While the mechanisms underlying apoptosis have been well characterized over recent decades, another regulated cell death event, necroptosis, remains poorly understood. Accumulating evidence indicates that necroptosis is involved in the regulation of cancer through pro-inflammatory cytokine production and anti-tumor immune response, and characterization of the signal transduction pathways underlying necroptosis was therefore thought to highlight therapeutic targets [17]. RIPK1 was recently identified as a key effector molecule of necroptosis, and the role has been associated to the TNF
As we know, hepatic IRI is the major complication at early-phase after hepatectomy [20], and the main mechanism is the condition of oxidative stress that can induce cell and organ dysfunction. Among the different mechanisms of oxidative stress involved in the IRI damage, cell death is an important biological process that is believed to have a central role in intestinal IRI. Necroptosis represents a newly discovered form of programmed cell death that combines the features of both apoptosis and necrosis, and it has been implicated in the development of a range of inflammatory diseases [21]. Dvoriantchikova et al. reported that treatment with necrostatin-1 (Nec-1), a selective RIPK1 inhibitor, suppresses induction of pro-inflammatory responses in retinal IRI [22]. Hong et al.’s report indicated that necroptosis regulates hepatic damage during IRI, which was involved in the autophagy process [23]. Shashaty et al. reported that plasma RIPK3, an essential mediator of necroptosis, was associated with acute kidney injury in critically ill trauma patients [24]. Interestingly, our results also demonstrated that GG genotype of RIPK1 rs2272990 SNP site was associated with poor outcome in IRI after hepatectomy. The principal biochemical parameters used as indicators of hepatocyte damage after liver resection are ALT and AST. In this study, ALT and AST in the GA
Footnotes
Acknowledgments
This work was financially supported by the China postdoctoral science foundation (2015M572724) and the Youth cultivation project of PLA (15QNP048).
Conflict of interest
None declared.
