Abstract
Background:
Hepatectomy is one potential treatment for intrahepatic cholangiocarcinoma (IHCC). Recurrent rate is high after curative resection and most recurrences occur within residual liver parenchyma. The aim of this study was to elucidate the impact of different treatment modalities on recurrent diseases in patients with IHCC after primary liver resection.
Methods:
Between February 1999 and December 2015, we retrospectively identified patients who received curative resection for IHCC. Patients who experienced recurrences were included. Locoregional therapies included re-hepatectomy, radiofrequent ablation, and transhepatic arterial chemoembolization. These patients were categorized into three groups: intrahepatic recurrence without locoregional therapies (group A), intrahepatic recurrence with locoregional therapies (group B) and extrahepatic metastases (group C).
Results:
Forty-three patients were included and there were 12, 15, and 16 patients in groups A, B, and C, respectively. The median disease-free survival times were 8.3, 9.1, and 8.7 months in groups A, B, and C (p = 0.099). The median after-recurrence overall survival times (period between recurrence and death/censor) were 6.4, 34.0, and 8.3 months in groups A, B, and C (p = 0.001). Locoregional therapies showed favorable benefit in multivariant analysis (hazard ratio: 0.274, confidence interval: 0.083–0.908, p = 0.010).
Conclusion:
Locoregional therapies offered favorable benefits for patients with recurrent intrahepatic cholangiocarcinoma.
Introduction
Intrahepatic cholangiocarcinoma (IHCC) is the second most common primary malignancy of the liver in both eastern and western countries.1,2 Risk factors for IHCC include chronic hepatitis C infection, cholelithiasis, alcoholic liver disease, non-alcoholic liver disease, liver cirrhosis, diabetes, obesity, and smoking. 3 Hepatectomy is the only curative treatment for patients with resectable disease, although most patients present with unresectable disease.4–7 The prognostic factors for IHCC included tumor size, tumor number, surgical margin, periductal infiltrating pattern, vascular invasion, perineural invasion, lymph node metastases, and preoperative carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9) levels.8–13
Tumor recurrence after curative liver resection mostly occurs in the remnant of the liver parenchyma, followed by lymph node metastases.11,14–16 One study reported that only about one quarter of patients underwent curative-intent repeat hepatectomy for intrahepatic recurrence. 16 Radiofrequency ablation (RFA) and transhepatic arterial chemoembolization (TACE) can also offer some benefits of treatment for patients with intrahepatic recurrence.17,18 Systemic chemotherapy has also been used as adjuvant therapy after surgery.19–21 Locoregional therapies (re-hepatectomy, RFA, and TACE) for intrahepatic recurrences prolong the survival of patients with IHCC after primary liver resection.19,22
Few studies to date have evaluated the advantages of different locoregional therapies for recurrent IHCC. We therefore conducted a retrospective study to elucidate the efficacy of locoregional therapies in patients with recurrent IHCC.
Methods and materials
Patients
Data of patients undergoing curative-intent hepatectomy for primary IHCC between February 1999 and December 2015 were retrospectively collected. Patients with perihilar cholangiocarcinoma (Klatskin tumor), combined hepatocellular carcinoma, and those with lymph node or distant metastases proved histologically by surgical sampling were excluded. Treatments for intrahepatic recurrences comprised re-hepatectomy, RFA, TACE, and systemic chemotherapy, while lymph node or distant metastases were treated with systemic chemotherapy. Based on the recurrence patterns and locoregional therapies, patients were categorized into three groups: intrahepatic recurrence without locoregional therapies (group A), intrahepatic recurrence with locoregional therapies (group B), and extrahepatic metastases (group C).
Preoperative assessment
All patients underwent liver computed tomography (CT) or magnetic resonance imaging (MRI) before curative hepatectomy. In cases in which there were multiple intrahepatic tumors on the diagnostic images, the largest tumor size was recorded. Hepatitis B and C viral serology, including hepatitis B virus surface antigen (HBsAg), hepatitis B virus surface antibody (HBsAb), and anti-hepatitis C virus antibody (anti-HCV), were measured. Serum levels of CEA, CA19-9, alkaline phosphate (Alk-P), alanine aminotransferase (ALT), total bilirubin, albumin, creatinine, neutrophil–lymphocyte ratio (NLR), and retention rate of indocyanine green at 15 min (ICGR15) were also assessed. The liver resectability and resection extent were determined according to modified Makuuchi’s criteria. 23
Intraoperative assessment
Tumor margin and major intrahepatic vessels of the liver were evaluated by intraoperative sonography and defined using the Brisbane 2000 terminology. 24 Hepatectomy was performed under intermittent portal triad clamping for inflow control and parenchymal transection using the Kelly crushing method. 25 Meticulous hemostasis was performed, followed by checking the bile leaks on the cut surface. A closed negative pressure drainage tube was placed over the cut surface of the liver after hepatectomy. Curative resection was defined as no residual tumors identified by intraoperative sonography after liver resection. Lymphadenectomy was not routinely performed unless gross lymphadenopathy was noted intraoperatively.
Postoperative assessment
Surgical mortality was defined as death due to any cause within 90 days after liver resection. A diagnosis of IHCC was confirmed with immunohistochemistry showing positive CK-7 and negative CK-19. 26 Hepatocyte antigen was also used to exclude combined hepatocellular carcinoma. 27 Pathological features included tumor size, tumor numbers, vascular invasion, perineural invasion, and histological scoring of liver fibrosis. No postoperative adjuvant treatment was administered to patients without lymph node metastases. Patients received regular outpatient radiological follow-up (CT or MRI) every 3–6 months. Recurrence was defined as radiological evidence of a new tumor (both intrahepatic and extrahepatic). Early recurrence was defined as recurrence that developed within 1 year after the first hepatectomy. Tumor size and number of intrahepatic recurrences were measured by radiological studies. Locoregional therapies for patients with intrahepatic recurrences include re-hepatectomy, RFA, and TACE. Systemic treatments for patients with intrahepatic or extrahepatic recurrences involved fluoropyrimidine-based or gemcitabine-based chemotherapy.
Radiofrequency ablation
Radiofrequency ablation for intrahepatic tumors was carried out under CT guidance in our institution. The exact locations and depths of the lesions were determined on the acquired CT slices. If the lesion could not be clearly located on non-enhanced CT scan, the needle was placed on nearby anatomical landmarks to correlate with pre-ablation CT or MRI for precise localization of the lesion. In some cases, a bolus of contrast medium was administered intravenously to determine the exact location of the lesion. All RFA procedures were performed with a Cool-tip RF system (Medtronic, Covidien). Artificial ascites would be created for protection of thermal injuries if the lesion was within 1 cm of the gastrointestinal tract. Immediately after the procedure, all patients underwent dynamic contrast-enhanced CT with both arterial and venous phases to evaluate the ablative zone and procedure-related complications.
Transhepatic arterial chemoembolization
The femoral artery was approached with placement of the vascular sheath under the Seldinger technique. Angiography was performed through the superior mesenteric artery, common hepatic artery, and feeding branches of the tumor to confirm the location, size, and arterial supply. After super-selection of feeding vessels, TACE was performed using mixed epirubicin–lipiodol (André Guerbet Laboratories; Lipiodol Ultra-Fluide) emulsion and Gelfoam (Pfizer) cubes. The endpoint of TACE was stasis of feeding vessels. Post-treatment angiography was performed to check if there were residual tumor stains. The dosage of anticancer drug–lipiodol emulsion for TACE was calculated before the treatment based on tumor size and feeding vessels of the tumor.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics version 22. Because most clinical data did not meet the normal distribution, non-parametric statistical methods were used. All numerical data are presented as median with range. Among the patient groups, Pearson’s chi-square or Fisher’s exact tests were used for comparison of categorical variants, as appropriate. The Kruskal–Wallis test and Mann–Whitney U test were used for comparison of numerical variants. Both disease-free and overall survival curves were generated by the Kaplan–Meier method and compared using the log-rank test. Cox regression was used for multivariate analysis of survival. The disease-free survival period was defined as the time interval between the first liver resection and disease recurrence. The overall survival period was defined as the time interval between the first liver resection and death. The overall survival period after recurrence was defined as the time interval between disease recurrence and death. A significant difference was considered when the two-sided p value was less than 0.05.
Research ethics
This study was approved by the Institutional Review Board (IRB) of Taichung Veterans General Hospital (IRB approval number CE18063B). The requirement for informed consent was waived due to the retrospective nature of this study.
Results
A total of 71 patients with IHCC underwent curative-intent hepatectomy. Three patients died within 90 days after surgery due to postoperative liver failure (two patients) and postoperative intra-abdominal sepsis (one patient). Eight patients were histologically proved to have lymph node metastasis. Among the remaining 60 patients, 43 experienced disease recurrence with a median disease-free survival time of 8.9 months. The 1-year and 3-year disease-free survival rates were 39.5% and 18.6%, respectively. The median overall survival time was 27.1 months. The 1-year, 3-year, and 5-year overall survival rates were 73.8%, 42.1%, and 38.2%, respectively.
Patients were categorized into three groups: intrahepatic recurrence without locoregional therapies (group A, n = 12), intrahepatic recurrence with locoregional therapies (group B, n = 15), and extrahepatic metastases (group C, n = 16). Ten patients in group B underwent curative treatments (re-resection and/or RFA), and the other five patients underwent TACE only. The details of locoregional therapies of patients in group B are shown in Table 1. Seven patients, four patients, and six patients underwent chemotherapy after recurrence in groups A, B, and C, respectively. As for lymph node metastasis, there were three regional (hepatic hilum), five intra-abdominal (both intraperitoneal and retroperitoneal), one supraclavicular, one mediastinal metastasis, and distant metastasis comprised four bone metastases and two lung metastases in group C.
Patients’ results in group B.
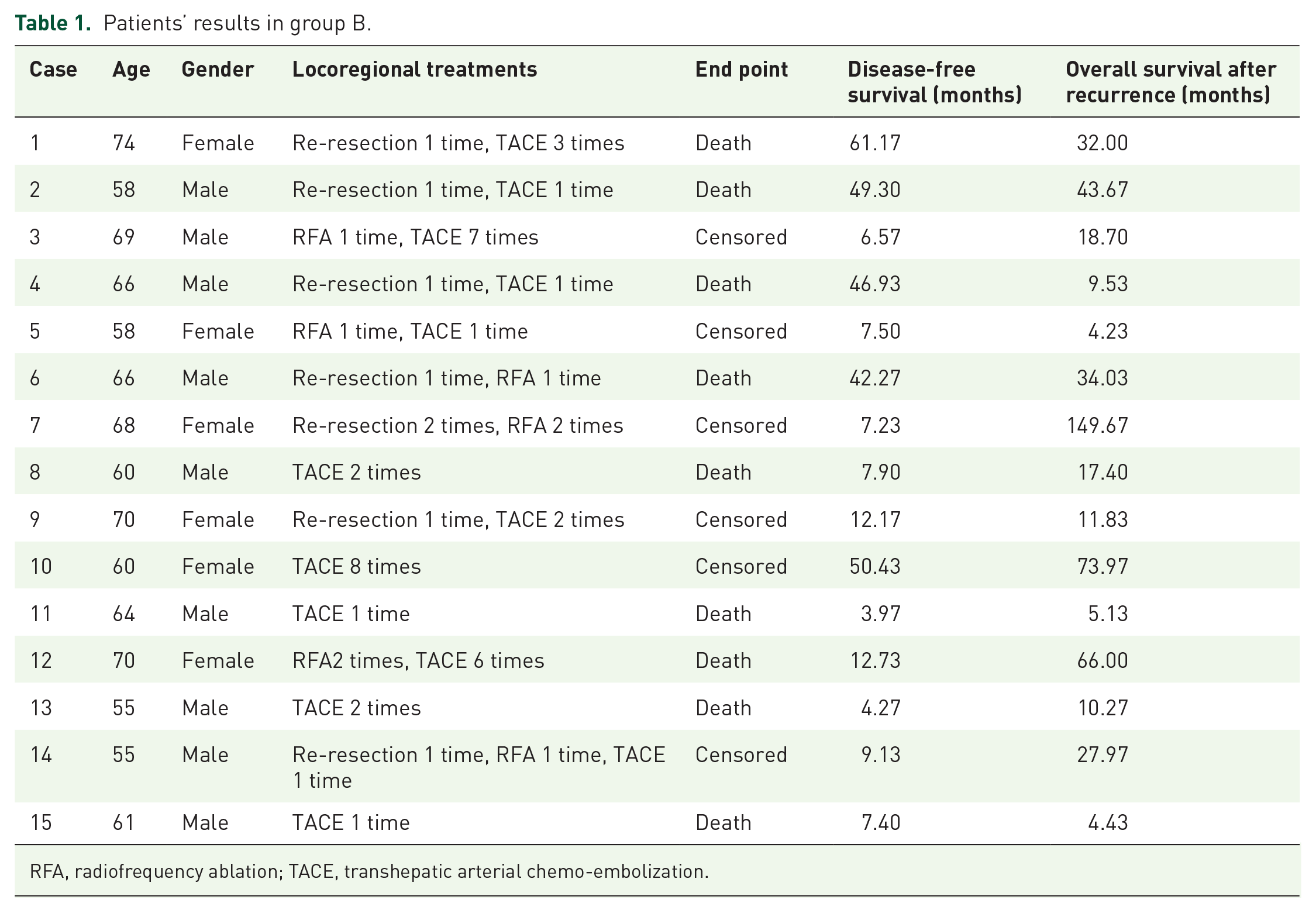
RFA, radiofrequency ablation; TACE, transhepatic arterial chemo-embolization.
The demographic characteristics and comparisons among the three groups are shown in Table 2. There were no significant differences among groups in perioperative factors, including gender, age, serum CA19-9, serum ALT, serum bilirubin, serum Alk-P, serum albumin, serum creatinine, ICGR15, HBsAg, anti-HCV antibody, estimated blood loss, and intraoperative blood transfusion. There were no differences in early recurrences among the three groups. Regarding pathological features, patients in group B had significantly more liver cirrhosis, less periductal infiltration pattern, and less perineural invasion. There were no significant differences in serum albumin, serum bilirubin, recurrent tumor size, and multiple intrahepatic tumors between groups A and B at the time of recurrence.
Demography.
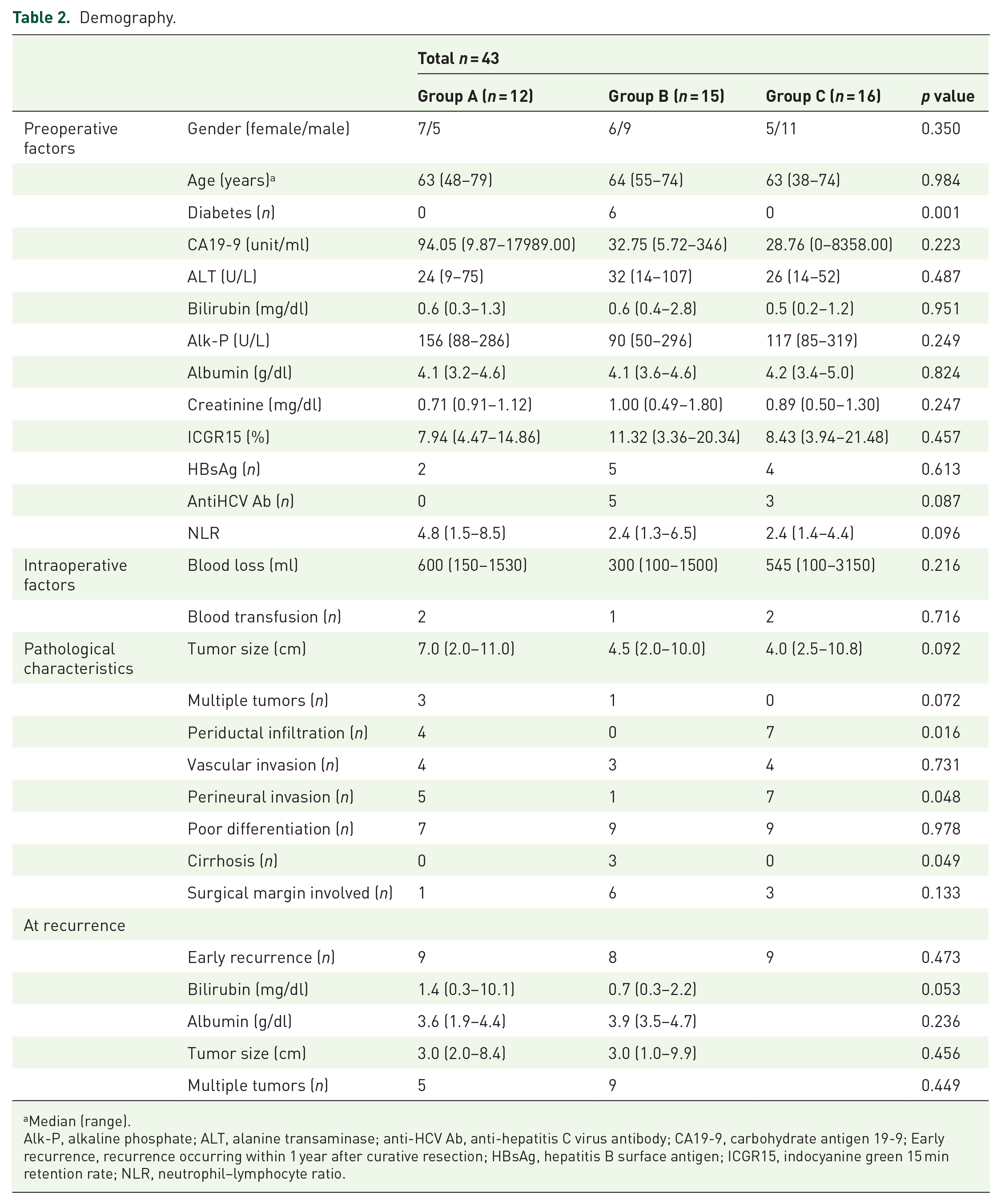
Median (range).
Alk-P, alkaline phosphate; ALT, alanine transaminase; anti-HCV Ab, anti-hepatitis C virus antibody; CA19-9, carbohydrate antigen 19-9; Early recurrence, recurrence occurring within 1 year after curative resection; HBsAg, hepatitis B surface antigen; ICGR15, indocyanine green 15 min retention rate; NLR, neutrophil–lymphocyte ratio.
Disease-free survival curves are shown in Figure 1. The median disease-free survival times in groups A, B, and C were 8.3, 9.1, and 8.7 months, respectively. The 1-year disease-free survival rates in groups A, B, and C were 25.0%, 46.7%, and 43.8%, respectively. The 3-year disease-free survival rates in groups A, B, and C were 0.0%, 33.3%, and 18.8%, respectively. Patients in group B had a slightly longer disease-free survival time, but there were no significant differences among the three groups (p = 0.099). The overall survival curves after recurrence are illustrated in Figure 2. The median post-recurrence survival times in groups A, B, and C were 6.4, 34.0, and 8.3 months, respectively. The 1-year post-recurrence overall survival rates were 12.2%, 77.9%, and 35.2%, respectively. The 3-year post-recurrence overall survival rates were 0.0%, 46.2%, and 0.0%, respectively. Among the three groups, patients in group B had significantly better post-recurrence overall survival (p = 0.001).

Disease-free survival curves.

Overall survival curves after recurrence.
Table 3 shows the analysis of prognostic factors for overall survival. In univariate analysis, high NLR (⩾3), tumor size >5 cm, pathological perineural invasion, and early recurrence were significant adverse factors. Patients who received locoregional therapies had a better overall survival. In multivariate analysis, early recurrence was a significant adverse factor, and receiving locoregional therapies remained a favorable factor.
Analysis of the overall survival prognostic factors.
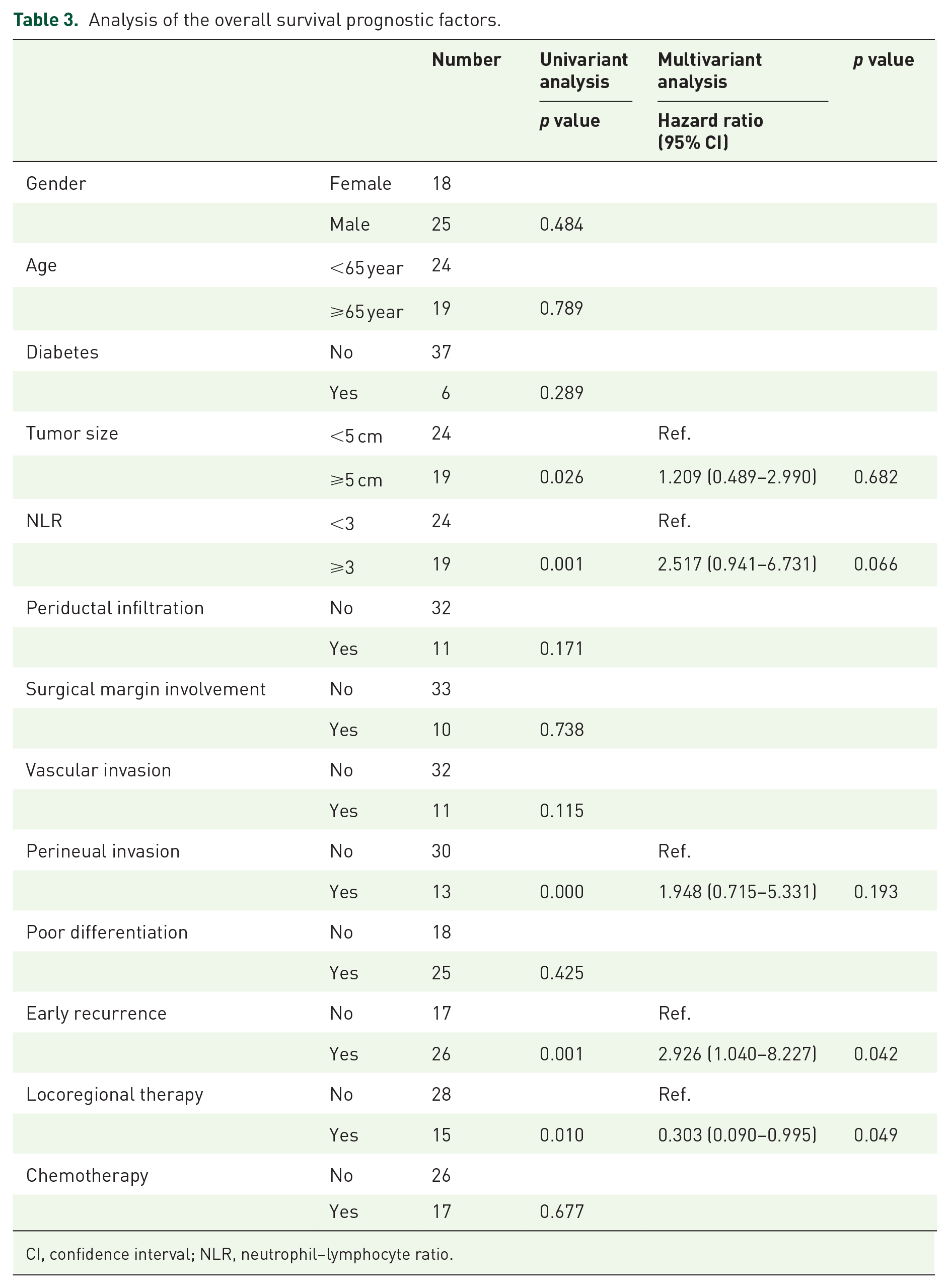
CI, confidence interval; NLR, neutrophil–lymphocyte ratio.
Discussion
In this retrospective study, we elucidated the efficacy of locoregional therapies in patients with recurrent IHCC. The results of this study indicated that locoregional therapies improved the overall prognosis of patients with recurrent IHCC. Among patients with intrahepatic recurrences, those treated with locoregional therapies had a longer median overall survival time after recurrence (34.0 months) than those treated without locoregional therapies (6.4 months, p = 0.001). Multivariate analysis confirmed that locoregional therapy was a favorable prognostic factor (hazard ratio = 0.274; p = 0.034). As intrahepatic recurrence is the major recurrence pattern in patients with IHCC after curative resection,11,14–16 it is expected that the treatments for intrahepatic lesions would have an influence on prognosis. Re-hepatectomy for intrahepatic recurrences was thought to be feasible and produced fair outcomes on overall survival.22,28,29 RFA was also considered an effective treatment for patients with intrahepatic recurrence after hepatectomy. 30 An aggressive approach by both re-hepatectomy and RFA for intrahepatic recurrence could prolong patient survival. 19 Park et al. reported the possible effectiveness of locoregional therapies, including re-hepatectomy, RFA, and TACE. 31 The present study revealed the remarkable benefit of locoregional therapies in patients with intrahepatic recurrence.
In patients with unresectable IHCC, TACE might offer longer survival than palliative modalities. 32 The effectiveness would be better in patients with Child–Pugh class A liver cirrhosis, hypervascular intrahepatic tumors, and tumor size less than 8 cm. 33 In addition, Wright et al. reported that intra-arterial therapies and surgery provided the same survival advantage in patients with multifocal IHCC. 34 In the current study, 13 patients in group B underwent TACE with or without other locoregional therapies. TACE seemed to be a more suitable treatment option for most patients with intrahepatic recurrence with regard to liver function, tumor number, and patient tolerance.
Lymph node metastases are a major unfavorable prognostic factor in patients with IHCC after hepatectomy.35,36 However, routine lymph node dissection did not improve long-term survival.37,38 We did not perform routine lymph node dissection unless lymph node metastasis was suspected intraoperatively. Those with histologically proved lymph node metastases were excluded to simplify the initial tumor stage in this study. Adjuvant systemic chemotherapy had been suggested for patients with resected biliary tract cancer.21,39 Therapeutic chemotherapy has also been used for unresectable IHCC but offers minimal long-term survival benefits.40,41 Programmed death ligand 1 (PD-L1) is mainly expressed in IHCC with a high density of tumor-infiltrating lymphocytes. 42 Immune checkpoint inhibitors may also provide benefits in unresectable and recurrent biliary tract cancer. 43 In our study, no patient received adjuvant chemotherapy after surgery. All chemotherapies were administered as a therapeutic strategy for patients with recurrence. There were no differences in overall survival between patients treated with or without chemotherapy.
The microenvironment and pro-inflammatory state of cancer play an important role in cancer progression and prognosis. The NLR may be an indicator of high inflammation and is associated with poor prognosis of many solid cancers. 44 High NLR has also been demonstrated to be a poor prognostic factor in IHCC after systemic chemotherapy and resection.45,46 In the present study, high NLR was an adverse prognostic factor in the univariate analysis and still had a significant trend (p = 0.066) in multivariate analysis.
There were no differences in early recurrence, serum albumin level, tumor size, and tumor number at recurrence between groups A and B, although there was a significant trend of serum bilirubin level (p = 0.053) at recurrence between groups A and B. As most patients had no cirrhosis on histological examination, hyperbilirubinemia may result from obstructive jaundice, depending on the relationship between recurrent tumors and the main bile ducts. The extent and occurrence time of intrahepatic recurrence did not affect the physicians’ treatment plan, but liver function did.
This study had two main limitations. First, although we excluded lymph node metastases, it is possible that our patients had occult regional lymph node metastases. This might have influenced the extrahepatic recurrence patterns. Second, we also arranged metastatectomy or chemoradiation for patients in group C, but the number was too small to analyze. We expected that metastatectomy and chemoradiation may improve survival, but the overall prognosis was still poor in patients in group C.
Conclusion
Intrahepatic recurrence is the most common pattern of recurrence in patients with intrahepatic choangiocarcinoma after curative resection. Locoregional therapies, including liver re-hepatectomy, radiofrequency ablation, and transhepatic arterial chemoembolization, offered favorable survival benefits.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
