Abstract
PURPOSE:
To investigate the association between the HER2 germline mutation Ala270Ser (A270S), located in HER2 extracellular domain, and survival in breast cancer patients.
METHODS:
HER2 germline mutation A270S was identified in 5395 consecutive patients with operable primary breast cancer using direct Sanger sequencing analysis. Survival curves for patients with HER2 A270S mutation were compared using the Kaplan-Meier method with log-rank test.
RESULTS:
We identified that 31 cases carried HER2 germline mutation A270S in 5395 patients (0.6%, 31/5395). The HER2 A270S mutation was significantly associated with recurrence-free survival (RFS) and distant recurrence-free survival (DRFS) in the entire cohort of 5395 patients (RFS, unadjusted hazard ratio [HR]
CONCLUSIONS:
Breast cancer patients with the HER2 germline mutation A270S had a worse survival, especially in HER2-negative patients. Therefore, HER2-negative patients with a HER2 germline mutation A270S might be potential candidates for HER2-targeted therapy.
Introduction
The HER2 (human epidermal growth factor receptor 2) gene (also known as erbB-2 or neu) encodes a transmembrane glycoprotein with tyrosine kinase activity. HER2 gene is a member of the epidermal growth factor receptor (EGFR) family and plays an important role in the regulation of cell growth, differentiation, and invasion [1, 2]. The HER2 protein is composed of four domains: extracellular domain (ECD), transmembrane domain (TMD), tyrosine kinase domain (TKD) and the carboxy tail [3, 4].The cysteine-rich domain (CR1) is a secondary structure element of extracellular region that plays a crucial role in HER2 dimerization [5, 6].
It is well-known that HER2 gene amplification is associated with worse clinical outcome in breast cancer [7, 8, 9, 10]. HER2-targeted therapy, i.e., trastuzumab, significantly improves the survival in HER2-positive breast cancer [11, 12, 13, 14]. Although HER2 amplification and/or overexpression was associated with poor survival, the prognostic value of HER2 gene mutation in breast cancer was unclear. Recently, Yamamoto et al. identified a novel germline mutation (G660D, GGC to GAC) in the transmenbrane domain of HER2 in lung cancer [15]. Mutant HER2G660D protein was more stable than wild-type proteinand activated Akt, and may be oncogenic, causing lung adenocarcinomas [15]. In our previous study, we found that HER2 Ile655Val polymorphism could affect the function of HER2 gene restricted in HER2-positive breast cancers. Patients with Val variant had an aggressive phenotype and were sensitive to trastuzumab treatment [16]. These inspired us to explore HER2 germline mutation in breast cancer and its significance in survival prediction and targeted therapy. We initially screened all exons of the HER2 gene in 1000 patients with breast cancer and found 6 patients with a novel germline mutation A270S (c.808G>T, p.Ala270Ser, located in Chromosome 17: 39710388). Codon 270 is located in CR1, especially in the dimerization and pertuzumab binding interface [17]. So missense mutation A270S of the codon 270 may affect the function of HER2 protein or biological effects of pertuzumab applying to HER2 protein.
Therefore, to identify the clinical effects of the HER2 germline mutation A270S, we identified A270S mutation in a relatively large cohort of 5395 women with operable primary breast cancer, and analyzed the association between A270S mutation and survival in 5395 breast cancer patients, especially in HER2-negative patients.
Patients and methods
Patients
A total of 5681 patients with operable primary breast cancer were treated at the Breast Center, Peking University Cancer Hospital from October 2003 to December 2013. Of these 5681 patients, the result of HER2 mutation status was not obtained due to the poor quality of the DNA samples in 181 patients, and follow up was not available for 95 patients. Thus, 5395 patients with primary breast cancer were included in this study. Ages at diagnosis of the patients ranged from 19 to 90 years, with a median age of 50 years. The stage of the tumors was classified according to the tumor-node-metastasis classification of the Union Internationale Contre Le Cancer. Tumor size was defined as the maximum tumor diameter measured by ultrasound at the time of diagnosis. Tumor grade, tumor size, HER2 status, estrogen receptor (ER) status, progesterone receptor (PR) status and adjuvant therapy were obtained from review of medical records, and all are shown in Table 1. Informed consent was obtained from all individual participants and this study was approved by the Research and Ethics Committee of Peking University Cancer Hospital.
Association between HER2 germline mutation A270S and clinicopathologic characteristics in the entire cohort (
5395)
Association between HER2 germline mutation A270S and clinicopathologic characteristics in the entire cohort (
ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2; C: chemotherapy; E: endocrine therapy; C plus E: chemotherapy plus endocrine therapy.
The blood samples were collected form breast cancer patients treated at the Breast Center, Peking University Cancer Hospital. Genomic DNA was isolated from peripheral blood leukocytes using a phenol-chloroform assay. The HER2 germline mutation A270S was identified by Sanger-sequencing. Amplification of DNA fragments was performed by polymerase chain reaction (PCR) in a thermocycler (Gene CyclerTM, Bio-rad, Hercules, CA, USA) in 20
HER2 Status
The HER2 status was determined by immunostaining according to a standard method: a score of 0 and 1
Statistical analysis
The associations between the HER2 A270S mutation and clinicopathologic characteristics in the entire cohort were evaluated using Pearson Chi-Square test. Survival curves were derived from Kaplan-Meier analysis and the differences between the curves were compared by log-rank tests. Recurrence-free survival (RFS) was defined as the time from the date of pathological diagnosis to the date when locoregional recurrence or metastases, distant metastases or death from breast cancer. Distant recurrence-free survival (DRFS) was defined as the time from diagnosis to the occurrence of distant metastasis or death for which breast cancer was the primary or underlying cause. The Cox proportional hazards model was used to determine the association between HER2 A270S mutation and the risk of local/distant recurrence or death after adjustments for patient and tumor characteristics. All statistical tests were two sided, and
Results
Patient characteristics
A total of 5395 patients with primary breast cancer were included in this study. The clinicopathological characteristics of these patients are presented in Table 1. Totally, HER2 germline mutation A270S was found in 31 (0.6%, 31/5395) patients (Fig. 1). Among these 31 breast cancer patients, 6 patients were HER2-positive, 22 patients were HER2-negative, and 3 were unknown.
Genotyping the HER2 A270S mutation by DNA sequencing assay. (a) Wild type; (b) A270S mutation.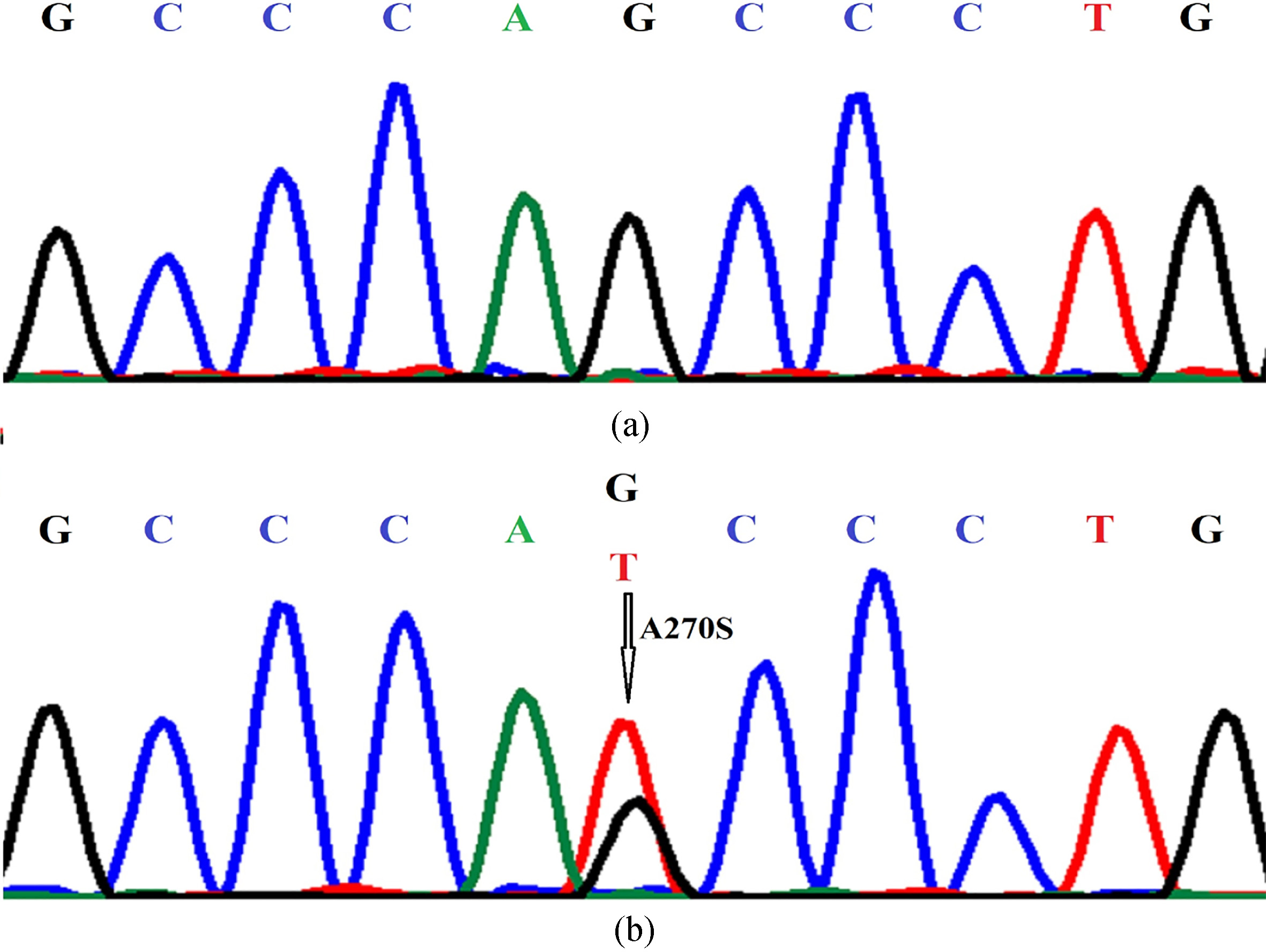
There was no significant difference with respect to age, tumor size, tumor grade, lymph node status, ER status, PR status, HER2 status, adjuvant therapy, and trastuzumab treatment between the breast cancer patients with A270S mutation and non-carreriers in this cohort of 5395 patients (Table 1).
Multivariate analyses of RFS and DRFS in the entire 5395 breast cancer patients
RFS: Recurrence-free survival; DRFS: Distant Relapse-free survival; HR: hazard ratio; CI: confidence interval; ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2; vs: versus.
Kaplan-Meier estimate of (a) recurrence-free survival (RFS) and (b) distant recurrence-free survival (DRFS) in the entire study population of 5395 patients, according to HER2 germline mutation A270S.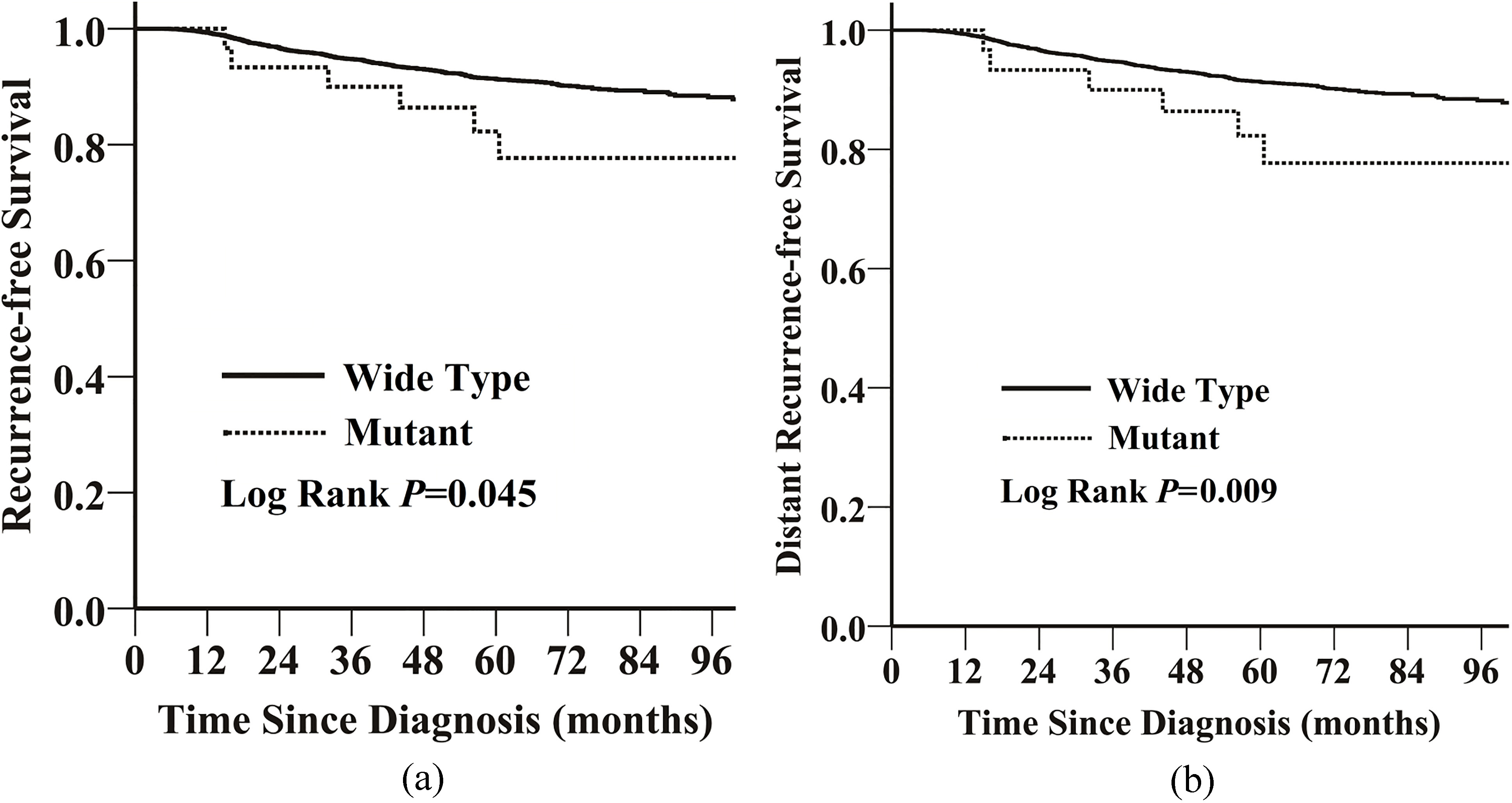
The median follow-up was 51 months (ranged from 1 to 195 months). The estimated 5-year RFS and DRFS in the 5395 patients were 91.3% (95% confidence interval [CI]
Multivariate analyses of RFS and DRFS in the 3825 HER2-negative breast cancer patients
Multivariate analyses of RFS and DRFS in the 3825 HER2-negative breast cancer patients
RFS: recurrence-free survival; DRFS: distant recurrence-free survival; HR: hazard ratio; CI: confidence interval; ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2; vs: versus.
Kaplan-Meier estimate of (a) recurrence-free survival (RFS) and (b) distant recurrence-free survival (DRFS) in the 3825 HER2-negative patients, according to HER2 germline mutation A270S. 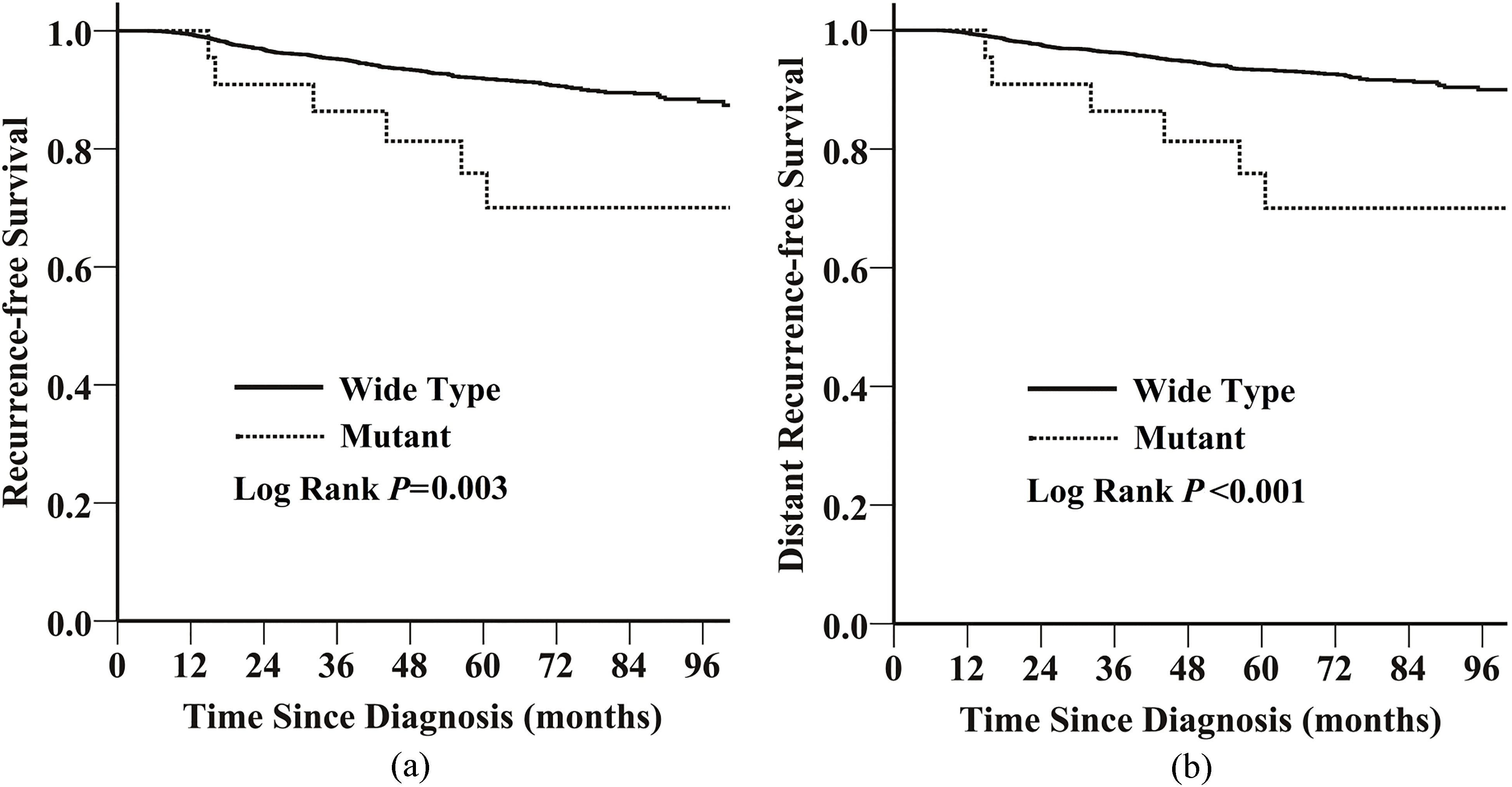
The HER2 expression status was available for 5044 of 5395 patients. Of these, 1219 (24%) were HER2-positive breast cancer patients, 3825 (76%) were HER2-negative patients (Table 1). In the HER2-negative patients, those with the HER2 A270S mutation had worse RFS (unadjusted HR
Among the 1219 HER2-positive breast cancer patients, only 6 patients harbored HER2 A270S mutation, statistical analysis was not conducted as a result of the underlying major bias. In addition, all the 6 patients had not been treated with HER2-targeted therapy, so it is very regrettable that we could not evaluate whether the mutation A270S would influence the biological effects of HER2-targeted therapy.
Discussion
In this study, we found 31 (0.6%) patients with HER2 germline mutation A270S in a relatively large cohort of 5395 unselected breast cancer patients. The mutation A270S was associated with a worse RFS and DRFS than the wild type in the total cohort of 5395 breast cancer patients and especially in the HER2-negative breast cancer patients (
It was well-known that patients with HER2-positive breast cancer had a relatively unfavorable survival compared with the HER2-negative patients [10, 19, 20]. Clinical application of HER2-targeted monoclonal antibody has significantly improved the survival of HER2-positive breast cancer patients [11, 12, 13]. Although HER2-negative breast cancer patients have a relatively more favorable survival than HER2-positive patients, a minority of HER2-negative patients may still suffer from metastases after treatment [19, 21]. Absence of an effective target drug like trastuzumab for HER2-positive breast cancer, is still a major adverse factor in the therapy of HER2-negative breast cancer. Identification of HER2 gene mutation may open up new avenues for treatment of HER2 negative breast cancer precisely.
HER2 mutations might be employed as treatment targets in patients with HER2-negative breast cancer [22]. Recent studies suggested that the HER2-targeted drugs trastuzumab and lapatinib inhibit the growth of cells with HER2 mutation in vitro and in vivo [22, 23, 24]. Oncogenic HER2 kinase domain mutations occur at low frequency in lung and breast cancer, and the HER2 gene mutations were found predominantly in HER2-negative patients [22, 25, 26]. Recently, recurring HER2 extracellular domain mutations in breast and lung cancer have been identified [27]. A phase II study of neratinib (an investigational irreversible oral tyrosine kinase inhibitor) in metastatic HER2 negative breast cancer with HER2 mutation was launched in 2012 (NCT01670877). An open-label, phase II study exploring the efficacy and safety of neratinib therapy in patients with solid tumors (breast, lung bladder colorectal biliary endometrial ovarian cancer) with activating HER2, HER3 or EGFR mutations or with EGFR gene amplification has also been lunched in 2013 (NCT01953926). These clinical trials indicated that HER2 mutations might play an important role in HER2-targeted therapy of HER2 negative breast cancer patients.
This study has several limitations. First, although the cohort in this study is large, the number of individuals with HER2 A270S mutation is relatively small; particularly when the mutation carriers were stratified by HER2 expression status. In addition, the HER2-positive patients with A270S mutation had not been treated with HER2-targeted therapy, therefore, the results are premature and should be interpreted cautiously.
In summary, breast cancer patients with the HER2 germline mutation A270S had a worse survival, especially in HER2-negative breast cancer patients. HER2 germline mutation A270S might be a target for therapy of HER2-negative patients in the future. Further functional and independent studies are warranted to confirm our findings.
Footnotes
Conflict of interest
The authors have declared no conflicts of interest.
Funding
This study was funded by the 973 project (2013CB 911004), and the National Natural Science Foundation of China (30973436 and 81071629).
