Abstract
Background:
BRCA2 plays a key role in homologous recombination. However, information regarding its mutations in Chinese patients with breast cancer remains limited.
Objectives:
This study aimed to assess the clinicopathological characteristics of BRCA2 mutation breast cancer and explore the mutation’s effect on hormone receptor (HR)-positive/human epidermal growth factor receptor 2 (HER2)-negative breast cancer survival in China.
Design:
This hospital-based cohort study prospectively included 629 women with breast cancer diagnosed from 2008 to 2023 at Zhejiang Cancer Hospital in China.
Methods:
We compared the clinicopathological characteristics and metastatic patterns and analysed the invasive disease-free survival (iDFS), distant relapse-free survival (DRFS) and first-line progression-free survival (PFS1) of patients with HR-positive/HER2-negative breast cancer according to BRCA2 mutations.
Results:
Among the 629 patients, 78 had BRCA2 mutations (12.4%) and 551 did not (87.6%). The mean age at diagnosis was lower in the BRCA2 mutation breast cancer group than in the non-mutation breast cancer group (38.91 versus 41.94 years, p = 0.016). BRCA2 mutation breast cancers were more likely to be lymph node-positive than non-mutation breast cancers (73.0% versus 56.6%, p = 0.037). The pathological grade was higher in 47.1% of BRCA2 mutation breast cancers than in 29.6% of non-mutation breast cancers (p = 0.014). The proportions of patients with BRCA2 mutations who developed contralateral breast cancer (19.2% versus 8.8%, p = 0.004), breast cancer in the family (53.8% versus 38.3%, p = 0.009) and ovarian cancer in the family (7.6% versus 2.4%, p = 0.022) were higher than those of patients without the mutation. The median follow-up time was 92.78 months. Multivariate analysis showed that BRCA2 mutation was not associated with poorer iDFS [hazard ratio = 0.9, 95% confidence interval (CI) = 0.64–1.27, p = 0.56] and poorer distant relapse-free survival (DRFS) (hazard ratio = 1.09, 95% CI = 0.61–1.93, p = 0.76). There was no significant difference between the two groups with regard to metastatic patterns in the advanced disease setting. In the first-line metastatic breast cancer setting, PFS1 expression was broadly similar between the two groups irrespective of chemotherapy or endocrine therapy.
Conclusion:
HR-positive/HER2-negative breast cancer with BRCA2 mutations differs from those without mutations in clinical behaviour and reflects more aggressive tumour behaviour. Our results indicate that BRCA2 mutations have no significant effect on the survival of Chinese women with HR-positive/HER2-negative breast cancer.
Introduction
Hereditary breast cancer accounts for approximately 5–10% of all breast cancer cases. 1 Breast cancer gene 1 (BRCA1) or breast cancer gene 2 (BRCA2) mutations are responsible for 50–60% of hereditary breast cancer cases.2,3 In a large consecutive, unselected sample of 8627 Chinese patients with breast cancer, the BRCA2 pathogenic germline mutation rate was 3.7%. 4
BRCA2 mutation breast cancers are mainly hormone receptor (HR)-positive and human epidermal receptor 2 (HER2)-negative, and they are more likely to present as high histologic grade.5,6 Generally, HR-positive breast cancers are characterized by a low pathological grade, late-onset and favourable prognosis.7,8 However, several previous studies have found that patients with HR positivity with a BRCA2 mutation have a higher rate of lymph node metastasis, an earlier age of onset and an adverse prognosis than do those without.9–12 HR-positive breast cancers are currently treated with endocrine therapy combined with targeted therapies, including cyclin-dependent kinase 4 and 6 (CDK4/6), histone deacetylase and mammalian target of rapamycin (mTOR) inhibitors.13,14 A recent study found that the loss of heterozygosity (LOH) of retinoblastoma 1 (Rb1) is frequent in breast cancers with a BRCA2 germline mutation. LOH of Rb1 correlates with resistance to CDK4/6 inhibitors. 15 Thus, the addition of CDK4/6 inhibitors to endocrine treatment may not be the optimal strategy for treating HR-positive and BRCA2 mutation breast cancers.
Breast cancers exhibit specific patterns of recurrence and metastasis, which are mediated by factors such as molecular subtypes of cancers and host organ microenvironments.16,17 The BRCA2 mutation is related to central nervous system (CNS) metastasis and an increased risk of death. 18 In the pre-CDK4/6 inhibitors era, unless there was a visceral crisis, endocrine therapy was considered the preferred option for the first-line treatment of HR-positive/HER2-negative metastatic breast cancer (MBC). 19 BRCA1/2 mutations are sensitive to cytotoxic chemotherapeutic agents. 20 Therefore, such agents should be considered for HR-positive/HER2-negative and BRCA2 mutation breast cancers. However, data from metastatic settings are lacking.
Additionally, the oestrogen receptor (ER) signalling pathway is intrinsically linked to the BRCA2 protein. BRCA2 activates the ER signalling pathway, which conversely increases the expression of BRCA2.21,22 Sustained DNA double-strand breaks are present in ER-positive breast cancer. Nevertheless, mutant BRCA2 proteins fail to repair broken double-stranded chains. 23 Therefore, breast cancers with BRCA2 mutations may be more responsive to poly-ADP ribose polymerase (PARP) inhibitors, which inhibit DNA repair.
Consequently, HR-positive/HER2-negative and BRCA2 mutation breast cancers may be specialized types that are distinct from sporadic HR-positive/HER2-negative breast cancers at the molecular level. However, studies on HR-positive/HER2-negative and BRCA2 mutation breast cancers are lacking. The relationship between BRCA2 mutation and predictive or prognostic value remains unclear. Whether the metastatic patterns of breast cancer are affected by the BRCA2 mutation status is also uncertain. Therefore, we aimed to describe the clinicopathological features of HR-positive patients with and without a BRCA2 mutation and investigate whether BRCA2 mutation is an independent prognostic factor for HR-positive breast cancer in Chinese women.
Methods
Ethics statements
This study was approved by the Ethics Committee of the Zhejiang Cancer Hospital. All participants provided informed written consent.
Study design and patients
In this single-centre observational prospective study, we enrolled 1636 Chinese breast cancer patients at high genetic risk diagnosed at Zhejiang Cancer Hospital from February 2008 to June 2023. In total, 629 HR positive/HER2 negative were identified. The eligibility criteria were based on the National Comprehensive Cancer Network guidelines for genetic high-risk assessment of breast, ovarian and pancreatic cancers. 24 Patients were eligible for disease diagnosis at any time point. A flow chart of the study population is exhibited in Figure 1. The reporting of this study conforms to the Strengthening The Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material 1). 25

Flow chart of the study population.
Data collection
Patients’ information and tumour characteristics including age at first diagnosis, menopausal status at diagnosis, age of menopause, body mass index (BMI), T stage, lymph nodes status, pathological type, grade, vascular invasion, HER2 status, ER status, progesterone receptor (PR) status, surgical approach for primary tumours and lymph nodes, treatment with (neo)adjuvant chemotherapy and radiotherapy, endocrine therapy, first event in the advanced disease setting, visceral metastases, number of metastatic sites and first-line treatment were retrieved from the medical records and pathology reports of Zhejiang Cancer Hospital. Positive results for ER, PR and HER2 were defined as previously described.26,27 Based on the prognostic impact of different pathological types, 28 we classified them as non-invasive carcinoma with good prognosis, invasive special carcinoma with good prognosis, invasive ductal carcinoma, invasive lobular carcinoma, other types with poor prognosis and others and unknown. De novo stage IV breast cancer was defined as metastasis diagnosed at the time of primary breast cancer diagnosis or within the adjuvant therapy period. The first events in the advanced disease setting were categorized as local, regional, brain, bone, liver and lung events. Local–regional recurrence was defined as the involvement of the ipsilateral breast, chest wall or lymph nodes.
Information on first-degree relatives and family history of tumours was obtained by interviewing the patients and asking them to report whether their family members, including parents, siblings and children, had ever been diagnosed with cancer and if so, to gain further information about which family member, what type of cancer and when the cancer was diagnosed.
Outcomes
The primary outcome was the comparison of invasive disease-free survival (iDFS) between patients with BRCA2 mutation breast cancer and those with non-mutation breast cancer. iDFS was defined as the time from diagnosis to the first occurrence of one of the following events: an ipsilateral invasive breast tumour, local invasive disease, distant recurrence, contralateral invasive breast cancer, second primary invasive cancer or death due to any cause. The secondary outcomes were distant relapse-free survival (DRFS) and first-line progression-free survival (PFS1) between the two groups of patients treated with first-line chemotherapy-based or only endocrine-based treatment. DRFS was calculated from the time of diagnosis to the first occurrence of distant recurrence or death due to any cause. PFS was measured from the time of starting first-line treatment to the first disease progression or death due to any cause. 29 Survival times were obtained from outpatient and telephone follow-ups.
BRCA2 germline mutation screening
Peripheral blood samples were obtained from the patients, and DNA samples were extracted using the QIAamp DNA Blood Mini kit (Qiagen, Hilden, Germany). We analysed the genetic variants in BRCA2 using a 98-gene panel sequencing assay. 30 We focused on the overall exons and the 10-bp regions upstream and downstream of each exon in these genes. Based on ClinVar (http://www.ncbi.nlm.nih.gov/clinvar/), gnomAD (http://www.gnomad-sg.org/) and RefGene (https://annovar.openbioinformatics.org/en/latest/user-guide/download/) annotated by ANNOVA (24 October 2019, https://annovar.openbioinformatics.org/en/latest/), we classified all variants into different grades: pathogenic, likely pathogenic, variant of uncertain significance, likely benign and benign based on the American College of Medical Genetics guidelines. Pathogenic and likely pathogenic variants were classified as deleterious and analysed in this study.
Statistical analysis
The clinicopathological characteristics of patients were compared based on BRCA2 germline mutation status using χ2 tests or Fisher’s exact test for categorical variables, and non-parametric Kruskal–Wallis’ test for continuous variables. The Kaplan–Meier method was used for survival outcomes, and the log-rank test was used to evaluate differences. We calculated the median follow-up using the reverse Kaplan–Meier method. Hazard ratios and 95% confidence intervals (CIs) for univariate and multivariate analyses (for primary and secondary outcomes) were calculated using Cox proportional hazards models to identify independent factors influencing prognosis. The following prognostic variables were evaluated using univariate analyses for iDFS: BRCA2 status (non-mutation/BRCA2), age at first diagnosis (continuous variables), T stage (Tis + T1/T2 + T3 + T4), lymph node status (N0; N1; N2 + N3), pathological type (non-invasive carcinoma with good prognosis + invasive special carcinoma with good prognosis/invasive ductal carcinoma + invasive lobular carcinoma + other types with poor prognosis), grade (I + II/III), vascular invasion (no/yes), HR status (ER+/PR+/ER+/PR− and ER−/PR+), surgical approach to primary tumours (breast-conserving surgery/mastectomy) and lymph nodes (sentinel node biopsy/axillary dissection), treatment with neoadjuvant (no/yes) and radiotherapy (no/yes) and endocrine therapy (no/yes). Variables yielding p values <0.05, determined by univariate analysis and BRCA2 mutation status, were retained for multivariate analysis. The same assessment was performed for DRFS, with the exception that T stage was used with T1 as a reference and pathological type was used with invasive special carcinoma with good prognosis as a reference. Carcinoma in situ is not included in the DRFS analysis and is considered an early stage of breast cancer. In this stage, the cancer cells have not yet penetrated the basement membrane, and there is no lymphatic or vascular supply to the membrane, reducing the likelihood of distant metastasis. We performed a statistical assessment of the proportional hazard hypothesis. All analyses were two-tailed. Statistical significance was set at p < 0.05. All statistical analyses were performed using SPSS software, version 25.0, or R software, version 3.3.1.
Results
Clinicopathological characteristics in BRCA2 mutation breast cancer and non-mutation breast cancer
Among the 629 patients with HR-positive/HER2-negative breast cancer, 78 had pathogenic germline BRCA2 mutations and the remaining 551 did not have any mutations in other known breast cancer predisposition genes (including BRCA1, TP53, ATM, RAD51C, RAD51D, PALB2, CHEK2, NF1, BARD1, PTEN, STK11, CDH1). Among BRCA2 mutation carriers, one had an additional CHEK2 mutation and five had other unidentified breast cancer susceptibility gene mutations.
A comparison of the clinicopathological characteristics between BRCA2 mutation breast cancers and non-mutation breast cancers is shown in Table 1. The mean age of onset of patients with BRCA2 mutation was lower than that of patients without the mutation (38.91 versus 41.94 years, p = 0.016). The proportion of patients with an initial diagnosis within 35 years of age was significantly higher in the BRCA2 mutation group than in the non-mutation group (43.6% versus 28.6%, p = 0.007). Similarly, the mean age at menopause was lower in the BRCA2 mutation group than in the non-mutation group (46.69 versus 50.48 years, p = 0.006). There was also no significant difference in BMI between the two groups. BRCA2 mutation breast cancers were associated with a higher risk of developing contralateral breast cancer (19.2% versus 8.8%, p = 0.004), a higher family history of breast cancer (53.8% versus 38.3%, p = 0.009) and ovarian cancer (7.6% versus 2.4%, p = 0.022) compared with non-mutation breast cancers. There were no significant differences in personal history of ovarian cancer, personal history of ipsilateral breast cancer, personal/family history of gastrointestinal cancer and pancreatic cancer or family history of prostate cancer between the groups.
Clinicopathological characteristics of HR-positive/HER2-negative breast cancer patients according to BRCA2 mutation.

AI, aromatase inhibitors; CIA, chemotherapy-induced amenorrhea; TAM, tamoxifen.
BRCA2 mutation breast cancers were more likely to be lymph node-positive than non-mutation breast cancers (73.0% versus 56.6%, p = 0.037). The percentage of breast cancers with a higher pathological grade was 47.1% among those with BRCA2 mutations, compared to 29.6% among those without mutations (p = 0.014). The tumour size, pathological type and HR status were not significantly different between the two groups. Of the BRCA2 mutation breast cancers, 41.3% were vascular invasion-positive, compared with 23.4% of non-mutation breast cancers (p = 0.001). Patients with BRCA2 mutation were more often treated with mastectomies (88.4% versus 78.9%, p = 0.003) and axillary dissections than those without mutations (91.3% versus 68.9%, p = 0.00). Patients in the BRCA2 mutation group were more likely to receive anthracycline combined with taxane chemotherapy regimens than single adjuvant chemotherapy. The percentage of patients receiving combination chemotherapy was lower in the non-mutation breast cancer group than in the BRCA2 mutation breast cancer group (47.4% versus 62.1%, p = 0.008). There was no notable difference in the choice of radiotherapy or endocrine therapy between the two groups. The proportions of patients with de novo stage IV breast cancer were 11.5% among those with BRCA2 mutation breast cancers and 7.3% among those with non-mutation breast cancers (p = 0.196).
Disease characteristics and treatments according to BRCA2 mutation status in advanced disease settings are shown in Table 2. BRCA2 mutation and non-mutation breast cancers most frequently had metastases to the bone (38.5% and 47.9%, respectively), followed by the lung (20.5% and 27.5%, respectively), liver (15.4% and 9.0%, respectively) and brain (2.6% and 2.8%, respectively). BRCA2 mutation breast cancers showed a more evident trend towards metastasis than did non-mutation breast cancers (46.4% versus 41.7%, p = 0.869), although the p value was not significant. The number of metastatic sites at the initial diagnosis of MBC was not different between the two groups. Both groups mainly received chemotherapy or endocrine therapy alone as first-line treatment. Patients who received endocrine therapy after chemotherapy were included in the chemotherapy group. Only 9.5% of patients with non-mutation breast cancers received endocrine therapy with CDK4/6, mTOR or histone deacetylase inhibitors.
Patient and disease characteristics and treatment in the advanced disease setting according to BRCA2 mutation.

CDK, cyclin-dependent kinase; Chemo, chemotherapy; ET, endocrine therapy; HDAC, histone deacetylase; mTOR, mammalian target of rapamycin.
Prognosis and long-term survival
The median follow-up time was 92.78 months (range: 1–300 months). In all, 49 patients with an advanced initial diagnosis and 6 who were lost to follow-up were excluded from the analysis of iDFS. In total, 569 patients who were operable at the initial diagnosis had complete clinicopathological information for iDFS analysis, including 68 BRCA2 mutation breast cancers and 501 non-mutation breast cancers. The median iDFS durations of BRCA2 mutation breast cancer and non-mutation breast cancer were 91.21 and 87.71 months, respectively, and no significant difference was identified between the groups (p = 0.56) [Figure 2(a)]. HRs and 95% CIs were estimated after adjusting for tumour size, lymph nodes, grade, vascular invasion and BRCA2 mutations. In multivariate analyses, no differences in iDFS were observed between the two groups, although there was a tendency for better iDFS in BRCA2 mutation breast cancers than in non-mutation breast cancers (adjusted hazard ratio = 0.78, 95% CI = 0.46–1.32, p = 0.37) (Table 3).
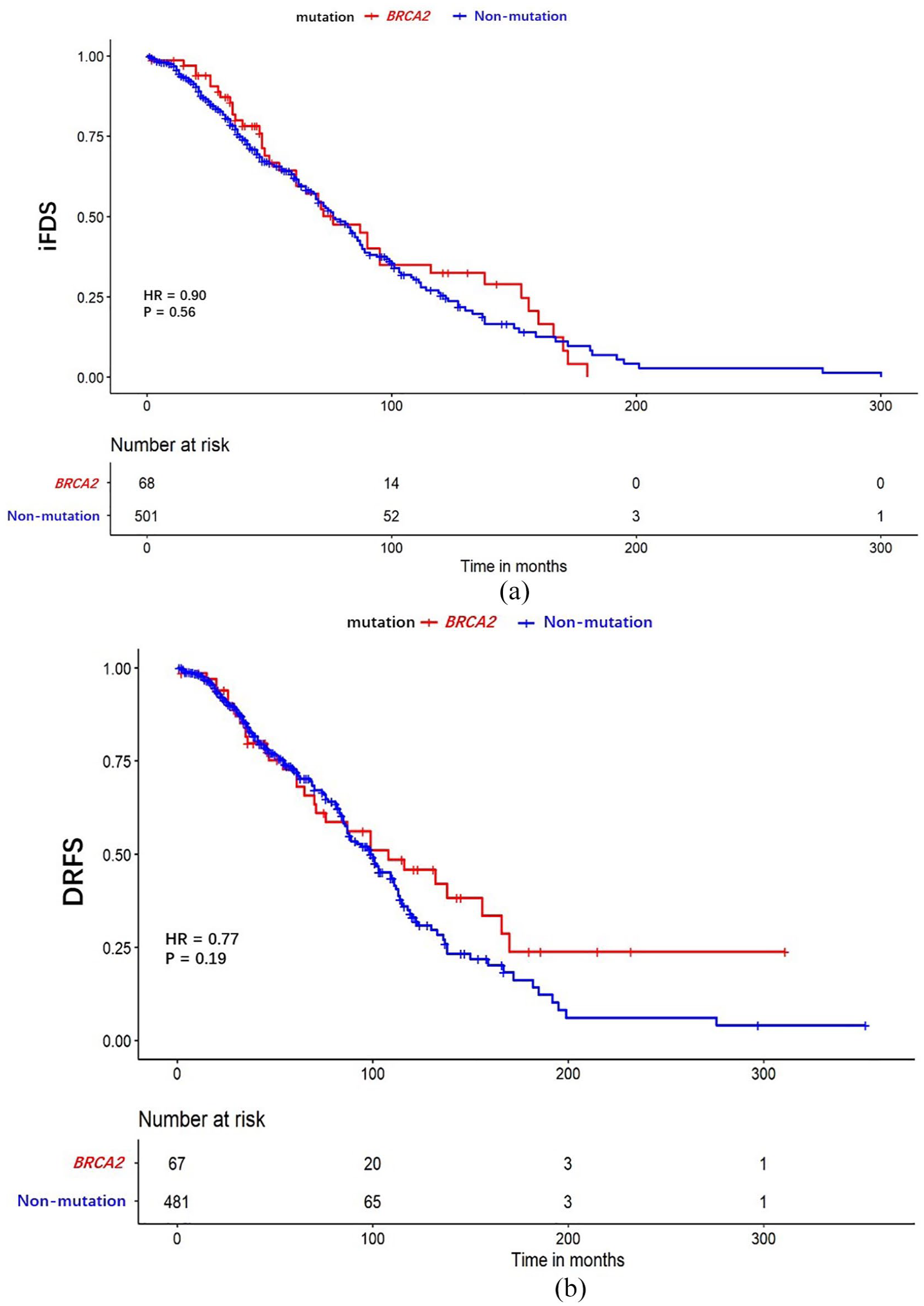
Comparison of iDFS (a) and DRFS (b) between BRCA2 mutation breast cancer and non-mutation breast cancer.
Hazard ratios for iDFS from breast cancer for selected variables.

BMI, body mass index; BRCA2, breast cancer gene 2; CI, confidence interval; ER, oestrogen receptor; HR, hazard ratio; iDFS, invasive disease-free survival; NI, not included; PR, progesterone receptor.
In all, 21 patients with carcinoma in situ were excluded from the analysis of DRFS. In total, 548 patients with invasive breast cancer without metastasis had complete clinicopathological information for DRFS survival analysis (67 BRCA2 mutation breast cancers and 481 mutation-free breast cancers). The median DRFS durations of BRCA2 mutation breast cancers and non-mutation breast cancers were 139.31 and 109.79 months, respectively (p = 0.19) [Figure 2(b)]. In multivariate analyses (adjusted for tumour size, lymph nodes, grade, HR status, vascular invasion, the surgical approach for primary tumours, neoadjuvant chemotherapy and BRCA2 mutation), BRCA2 mutation breast cancers showed a similar DRFS compared to non-mutation breast cancers, although this did not reach statistical significance (hazard ratio = 1.09, 95% CI = 0.61–1.93, p = 0.76) (Table 4).
Hazard ratios for DRFS from breast cancer for selected variables.

BMI, body mass index; CI, confidence interval; DRFS, distant relapse-free survival; ER, oestrogen receptor; HR, hazard ratio; NI, not included; PR, progesterone receptor.
We further compared the differences in PFS1 between the two groups. When treated with chemotherapy as the first-line treatment, no apparent difference was detected between BRCA2 mutation breast cancers and non-mutation breast cancers (median PFS1: 24.07 and 24.28 months, respectively). When treated with endocrine therapy for the first-line treatment, we also found a similar outcome between BRCA2 mutation breast cancers and non-mutation breast cancers (median PFS1: 15.51 and 16.96 months, respectively) (Figure 3).

PFS1 for patients receiving first-line chemotherapy (a) or endocrine therapy (b) according to BRCA2 mutation.
Discussion
In this study, we investigated the differences in clinicopathological characteristics between patients with HR-positive/HER2-negative breast cancer with and without BRCA2 mutation. Our study demonstrated that BRCA2 mutation breast cancers had inferior clinicopathological characteristics compared to non-mutation breast cancers. We found that mutations in BRCA2 were not associated with poorer iDFS and DRFS in the multivariate analysis. In the first-line MBC setting, PFS1 was broadly similar between the two groups irrespective of chemotherapy or endocrine therapy.
Our data showed that the mean age of BRCA2 mutation breast cancer diagnosis was lower than that of non-mutation breast cancer and was consistent with the peculiarity of early onset in women with BRCA2 mutation.31,32 A review indicated that obesity is related to a higher risk of developing breast cancer and adverse breast cancer survival. 33 Our data showed no significant differences in the BMI between the two groups. Patients with BRCA2 mutation breast cancer had a higher proportion of lymph node positivity, grade 3 tumours and vascular invasion than did patients with non-mutation breast cancer. These results are similar to those of several previous studies, indicating that BRCA2 mutation breast cancers have more aggressive tumour features than non-mutation breast cancers do.10,34 We observed that bilateral breast cancers were more frequent in patients with BRCA2 mutation breast cancers than in those with non-mutation breast cancers. Similar findings were reported in a previous study. 35
A recent study demonstrated that BRCA2 pathogenic mutations are associated with an increased risk of seven cancers, including female breast, male breast, gastric, ovarian, pancreatic, prostate and oesophageal cancers. 36 We observed that patients with BRCA2 mutation breast cancer were more likely to have a family history of breast or ovarian cancers. Unfortunately, our results did not demonstrate a relationship between BRCA2 mutations and other cancer types. This may be attributed to the small number of patients with BRCA2 mutation breast cancer, which lowered the statistical power.
In our cohort, patients with BRCA2 mutation preferred mastectomy and axillary dissection to breast-conserving surgery and sentinel node biopsy. This may be because some patients and doctors were aware of their BRCA2 mutation status before surgery. In germline BRCA1/2 mutation breast cancers, breast-conserving surgery (BCT) is associated with a higher probability of local recurrence than mastectomy. 37 However, there is no difference in overall survival between BCT and mastectomy for germline BRCA1/2 mutation breast cancers, and this therapy can improve the quality of life. 38
Several studies have suggested that BRCA2 mutations are associated with adverse prognostic significance in HR-positive breast cancers.11,39–42 Moreover, the prognosis varies between races, and there is still a lack of large-sample research in China. Remarkably, our results suggest that BRCA2 mutations have no significant effect on the survival of Chinese women with HR-positive/HER2-negative breast cancer. After adjusting for several main prognostic factors, a tendency towards prolonged iDFS was observed, although the difference was not statistically significant. Most patients in our study population received anthracycline-based adjuvant chemotherapy. Adjuvant chemotherapy may improve patients’ outcomes. These results imply that BRCA2-deficient tumours are more sensitive to chemotherapy regimens, causing DNA breaks due to homologous recombination defects.43,44
DNA damage drugs such as PARP inhibitors, alkylating agents, topoisomerase II inhibitors and platinum are promising strategies for treating HR-positive/HER2-negative and BRCA2 mutation breast cancer, as they have shown high efficacy in BRCA2 mutation breast cancer, whether alone or in combination with other drugs.20,45,46 A retrospective study indicated that HR-positive and germline BRCA1/2 mutation breast cancers had Oncotype DX recurrence risk scores approximately three times higher than those of non-mutation breast cancers. 47 Therefore, these drugs should be included among the choices for adjuvant treatment. The OlympiA study demonstrated that 1 year of adjuvant intensive therapy with the PARP inhibitor olaparib significantly improved the 3-year iDFS in patients with mutations in the BRCA1 or BRCA2 genes. 48 In HR-positive/HER2-negative breast cancer patients, the 3-year iDFS rates were 83.5% and 77.2% in the olaparib and placebo groups, respectively. Based on the results of this trial, the 2021 American Society of Clinical Oncology guideline updated the recommendation that suggested providing 1 year of adjuvant olaparib to patients with early-stage HER2-negative and BRCA mutation breast cancer who had finished chemotherapy and local therapy. 49 Because of the poor response to CDK4/6i,50,51 PARP inhibitors could be the preferred first-line treatment choice in patients with HR-positive/HER2-negative MBC carrying a BRCA2 mutation, which is an explorable issue that deserves more clinical trials to address. Furthermore, experiments have confirmed that BRCA2-deficient breast cancer cells respond more strongly to immune checkpoint inhibitors (ICIs). 52 Denkert et al. 53 demonstrated that pathological complete response rates increased as tumour-infiltrating lymphocytes increased in luminal-HER2-negative breast cancer, indicating the potential effectiveness of immunotherapy. PARP inhibitors combined with ICIs have also demonstrated favourable performance in the management of HR-positive and BRCA2 mutation breast cancers. 54 Furthermore, well-designed studies are warranted to validate these findings.
Several studies have shown that HR-positive breast cancers are more prone to bone metastasis, which was also reflected in our study population.55,56 Song et al. 18 demonstrated that BRCA2 mutation breast cancers have a higher frequency of CNS metastasis than non-mutation breast cancers do, which was not reflected in our results. The study by Frenel et al. 57 demonstrated that HR-positive/HER2-negative and BRCA2 mutation breast cancers exhibit lower tumour sensitivity to first-line endocrine therapy than non-mutation breast cancers do, but not to first-line chemotherapy. We sought to validate this in our data; however, our results showed no difference in PFS1 between the two groups when receiving first-line endocrine therapy. The discrepancies in results could have been caused by several reasons. First, the enrolled population in the study by Frenel et al. included both BRCA1 and BRCA2 mutation breast cancer. Our study population consisted solely of breast cancer patients with BRCA2 mutation. Because BRCA1 or BRCA2 mutation may differentially affect breast cancer sensitivity to endocrine therapy, 51 it is possible that combining these two groups obscured the distinction. Second, our study was limited by the small number of BRCA2 mutation breast cancers for PFS1 analysis, which may have influenced the results and reduced statistical power. Finally, the number of visceral metastases at first-line treatment was higher in the BRCA1/2 mutation group than in the non-mutation group (67.6% versus 56.7%, p = 0.0003) in the study by Frenel et al., which may have led to a worse PFS1 than that in the non-mutation group; conversely, there was no difference in the number of visceral metastases between the two groups in our study (46.4% versus 41.7%, p = 0.869). Whether BRCA2 mutation reduces sensitivity to endocrine therapy remains unclear.
Our study has several advantages. A multitude of previous studies of Chinese women were limited by their small sample sizes. Our sample size was large, and the follow-up time was long. Moreover, we detected all the coding regions and exon–intron boundaries of the genes and had adequate details on the family history of cancer and clinicopathological characteristics. The limitations of this study include an insufficient number of patients with BRCA2 mutation breast cancer. Another significant limitation of the study is that it was conducted at a single centre, which may have led to selection bias. Most of the patients were from Zhejiang Province, China, and breast cancer may show variation in different geographic populations. Future studies involving larger sample sizes and multi-centre collaborations will yield more comprehensive results. Moreover, we did not obtain the complete overall survival data.
In summary, BRCA2 mutation breast cancers differ from non-mutation breast cancers in terms of their clinical behaviour. Our study may have implications for the genetic counselling and administration of BRCA2 mutation breast cancers. Our evidence does not support a clear effect of BRCA2 status on survival in HR-positive/HER2-negative breast cancers. Nevertheless, routine BRCA2 gene testing is necessary for patients with breast cancer due to the availability of chemotherapeutic and targeted agents resulting in DNA breaks that are therapeutically effective. All conclusions drawn from this study need to be treated with caution and confirmed in a larger population. Given the strides in gene sequencing and personalized medicine, an increasing number of breast cancer patients with a BRCA2 mutation have been identified. In this era of personalized medicine, individuals with HR-positive/HER2-negative breast cancer and a BRCA2 mutation should receive more tailored and precise treatment.
Conclusion
In conclusion, breast cancer patients with a BRCA2 mutation exhibited distinct clinical characteristics compared to those without mutations in our cohort. They showed more aggressive tumour behaviour, including earlier onset, higher lymph node involvement, higher pathological grade and an elevated risk of contralateral breast cancer, familial breast cancer and familial ovarian cancer. The BRCA2 germline mutations do not significantly impact the prognosis of HR-positive/HER2-negative early breast cancer patients or the effectiveness of first-line treatment for MBC. This study offers a justification for the clinical management of BRCA2 mutation breast cancer, and the findings should be validated in a larger sample.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241242613 – Supplemental material for HR-positive/HER2-negative breast cancer arising in patients with or without BRCA2 mutation: different biological phenotype and similar prognosis
Supplemental material, sj-docx-1-tam-10.1177_17588359241242613 for HR-positive/HER2-negative breast cancer arising in patients with or without BRCA2 mutation: different biological phenotype and similar prognosis by Pu-Chun Li, Yi-Fan Zhu, Jia-Ni Pan, Qiao-Yan Zhu, Yu-Yang Liao, Xiao-Wen Ding, Lin-Feng Zheng and Wen-Ming Cao in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
