Abstract
Background:
There is currently a lack of sufficient evidence on the prognosis of human epidermal factor receptor 2 (HER2)-positive microinvasive breast cancer (MIBC) and whether chemotherapy (CT) and targeted therapy can bring benefits.
Objectives:
To explore the prognosis and treatment of HER2-positive MIBC.
Design:
A retrospective multicenter study.
Methods:
In this multicenter real-world study, we consecutively enrolled patients who received local management and were diagnosed with lymph node-negative HER2-positive MIBC from January 2010 to December 2020.
Results:
A total of 163 patients were included. Thirty-nine patients (23.9%) received adjuvant CT, of which 12 patients (7.4%) received CT combined with trastuzumab. One patient received trastuzumab therapy alone. In total, nine patients (5.5%) experienced recurrence, and the 3-year disease-free survival (DFS) rate and 5-year DFS rate were both 95.1%. Kaplan–Meier analysis showed that patients aged ⩽40 had worse outcomes than those aged >40 (3-year DFS rate 81.8% vs 96.1%, p = 0.046). Using propensity score matching, we matched patients who received trastuzumab or CT ± trastuzumab with those who did not receive treatment based on age, tumor size, estrogen receptor status, and number of infiltration lesions. Patients who received targeted therapy or CT ± targeted therapy did not achieve further improvement in 3-year DFS rate (95.0% vs 95.0%, p = 0.630).
Conclusion:
HER2-positive MIBC has relatively good prognosis, and age ⩽40 years might be a poor prognostic factor. CT or targeted therapy seemed to bring little benefit for MIBC. Further prospective studies are needed to assess potential benefits of management for MIBC patients under the age of 40 years in the future.
Introduction
Approximately 20% of breast cancers exhibit overexpression of human epidermal growth factor receptor 2 (HER2), and HER2 positivity has been linked to a poorer prognosis.1,2 Even for T1a,bN0M0 tumors, patients with HER2-positive tumors experience significantly worse clinical outcomes compared to those with HER2-negative tumors.3–5 A prospective trial demonstrated excellent survival in patients with HER2-positive node-negative small tumors who received trastuzumab-based CT.6,7 According to consensus guidelines, including those from the National Comprehensive Cancer Network (NCCN), 8 adjuvant chemotherapy (CT) with trastuzumab can be considered for HER2-positive tumors that are smaller than 0.5 cm in size.
Microinvasive breast cancer (MIBC), which refers to invasive tumor foci measuring 1.0 mm or smaller, constitutes approximately 0.68%–2.4% of all diagnosed breast cancer cases.9,10 The majority of studies indicate that the clinical outcomes of MIBC fall somewhere between those of ductal carcinoma in situ (DCIS) and T1aN0 stage tumors.11–13 The MIBC T1miN0 stage typically has a favorable prognosis. However, there is ongoing debate about how to accurately identify the small subset of patients with risk of recurrence and metastasis at the time of diagnosis and provide them with appropriate systemic treatment. Treatment for T1miN0 HER2-positive tumors is not recommended in NCCN and ESMO guidelines.8,14 However, the treatment of HER2-positive and hormone receptor-negative multiple foci MIBC remains controversial and requires further research.
With the promotion of screening and the improvement of pathological diagnosis, the number of patients with MIBC has been steadily increasing in China. In light of this trend, we have undertaken a real-world multicenter study to explore the prognosis of HER2-positive MIBC, identify influencing factors, and determine whether CT and anti-HER2 targeted therapy can provide benefits for this population.
Methods
Study design and patient eligibility
This study was a multicenter real-world analysis. Patients with lymph node-negative HER2-positive MIBC who were diagnosed at the National Cancer Center, Cancer Hospital and Shenzhen Hospital, Chinese Academy of Medical Sciences, Liaoning Cancer Hospital, and Cancer Hospital of Huanxing Chaoyang District, Beijing, from January 1, 2010, to December 31, 2020, were enrolled. The flow chart of the detailed patient enrollment process is shown in Figure 1. The inclusion criteria were as follows: (1) female patients; (2) received surgical (radical or conservative) surgery and confirmed by postoperative pathology as MIBC; (3) without metastasis of axillary lymph nodes; and (4) postoperative pathological status of the invasive components confirmed by immunohistochemistry were HER2 3+ or HER2 2+ accompanied by fluorescence in situ hybridization (FISH) amplification. The exclusion criteria were as follows: (1) patients who had already experienced distant metastasis before surgery; (2) the HER2 status of the invasive components was unknown, such as the inability to undergo immunohistochemical testing due to too few cells or the inability to undergo FISH testing in immunohistochemical HER2 2+ cancer; (3) patients who had received neoadjuvant therapy before surgery; and (4) patients who had previously suffered from breast cancer and other malignant tumors. All pathological results were reviewed by experts from the pathology department of our hospital. We recorded the clinicopathologic characteristics and treatment information of the patients and followed up on whether the patients relapsed. The study was approved by the Institutional Review Board of the National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences, and Peking Union Medical College. The reporting of this study conforms to the STROBE statement 15 (Supplemental Files).

The flow chart of the detailed patient enrollment process.
Statistical analysis
Disease-free survival (DFS) was defined as the duration from surgery to the date of first occurrence of tumor recurrence, metastasis, or death. Overall survival (OS) was defined as the duration from surgery to the date of death from any cause. The Kaplan‒Meier method was used to draw survival curves. The log-rank test was used to compare DFS differences between groups. The propensity matching score was used to match age, estrogen receptor (ER) status, tumor size, and number of infiltration lesions factors between patients who received targeted therapy or patients who received CT ± targeted therapy (treatment group) and those who did not receive the treatment (non-treatment group) and to analyze the difference in DFS between the two groups of patients. All statistical analyses were performed using SPSS software, version 23.0 (IBM Corporation, Armonk, NY, USA), and GraphPad Prism, version 9.0 (GraphPad Inc.). The propensity score matching was performed using R software, version 4.1.1 (R Foundation for Statistical Computing, PBC, Boston, MA, USA). p < 0.05 was considered statistically significant.
Results
Patient characteristics
A total of 163 patients who met the criteria were included between January 1, 2010, and December 31, 2020. The median age of the patients was 50 years, and the median follow-up time was 83.5 (78.6–88.3) months. Seventeen patients underwent breast-conserving surgery (including 16 patients who received radiotherapy), while the remaining patients underwent mastectomy. There are 44 ER-positive breast cancer patients, of whom 35 received endocrine therapy. Among the 24 patients with ER-negative/PR-positive, 9 received endocrine therapy. Ninety-eight patients had a single focus of microinvasion, 6 patients had 2 foci, while 59 patients had multiple foci (⩾3 invasive foci). One hundred forty-two patients had a mass ⩽5 cm, while 21 patients had a mass >5 cm. Thirty-nine patients received adjuvant CT, of whom 12 patients received CT combined with trastuzumab. One patient only received targeted therapy. Among the patients receiving CT, two patients were aged ⩽40, and 31 were ER-negative breast cancer patients. Among the patients only receiving CT, 20 patients received 4 cycles of docetaxel or anthracyclines combined with cyclophosphamide (TC or AC). Two patients received six cycles of TC, and another patient received six cycles of docetaxel and carboplatin. In addition, two patients received four cycles of AC followed by four cycles of docetaxel. Two patients received six cycles of docetaxel, anthracyclines, and cyclophosphamide. Among the patients who received trastuzumab targeted therapy, 8 received 4 cycles of TC, 1 received 12 weekly cycles of paclitaxel, and another 1 received 4 cycles of paclitaxel. Besides, one patient received six cycles of docetaxel and carboplatin, and another one received six cycles of a gemcitabine and irinotecan regimen. The clinical pathological characteristics and treatment information were shown in Table 1.
Clinical pathological characteristics and treatment of MIBC patients.
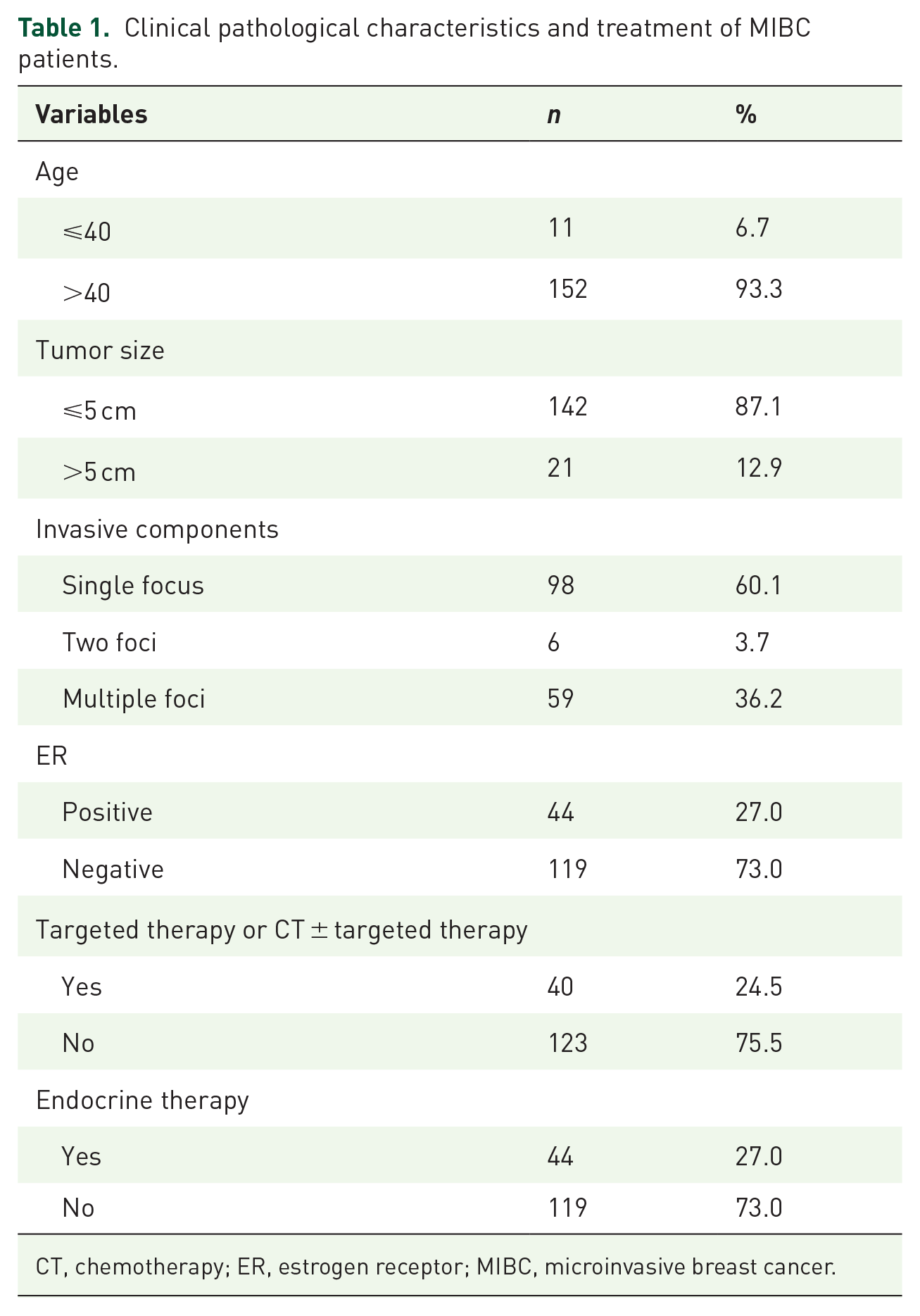
CT, chemotherapy; ER, estrogen receptor; MIBC, microinvasive breast cancer.
Survival outcomes
A total of nine patients experienced recurrence, of which three patients had in situ breast recurrence and six patients had distant metastasis. The initial features and treatments of recurrent patients were shown in Table S1. Eight patients experienced recurrence within 3 years, and one patient experienced recurrence at the 6th year, with a median DFS of 25 months. The 3-year DFS rate and 5-year DFS rate were both 95.1% (Figure 2). Among the nine patients who experienced recurrence, two patients were aged ⩽40. The 3-year DFS rate was 81.8% for those aged ⩽40 years and 95.4% for those aged >40 years. Two of the 40 patients in the treatment group experienced recurrence, while 7 out of 123 patients in the non-treatment group experienced recurrence. The 3-year DFS rates were 95.0% and 94.3%, respectively. Kaplan–Meier curve analysis showed that patients under the age of 40 were more likely to experience recurrence compared to those who were over 40 years old, with the 3-year DFS rate being 81.8% and 96.1%, respectively (p = 0.046, Figure 3). Tumor size, ER status, and number of infiltration lesions were not risk factors for recurrence in MIBC patients (p > 0.05, Table 2). In Cox multivariable regression, we included age, tumor size, ER status, and number of infiltration lesions. However, these variables were excluded from Cox regression models due to the limited number of relapse events.

DFS for all patients. The 3-year DFS rate was 95.1%.

Comparison of DFS between patients under the age of 40 and over 40 years old.
Kaplan–Meier curve analysis of 3-year DFS rate in MIBC patients.

CT, chemotherapy; DFS, disease-free survival; ER, estrogen receptor; MIBC, microinvasive breast cancer.
Using propensity score matching, patients in treatment group were matched with patients in non-treatment group in a 1:1 ratio based on age, tumor size, ER status, and number of infiltration lesions. The baseline characteristics of matched patients were shown in Table 3. There were no statistically significant differences between the two groups in terms of age, tumor size, ER status, or number of infiltrative lesions. Patients who received targeted therapy or CT ± targeted therapy did not achieve further improvement in 3-year DFS rate (95.0% vs 95.0%, p = 0.630, Figure 4).
Characteristics of two groups of patients after using propensity score matching.
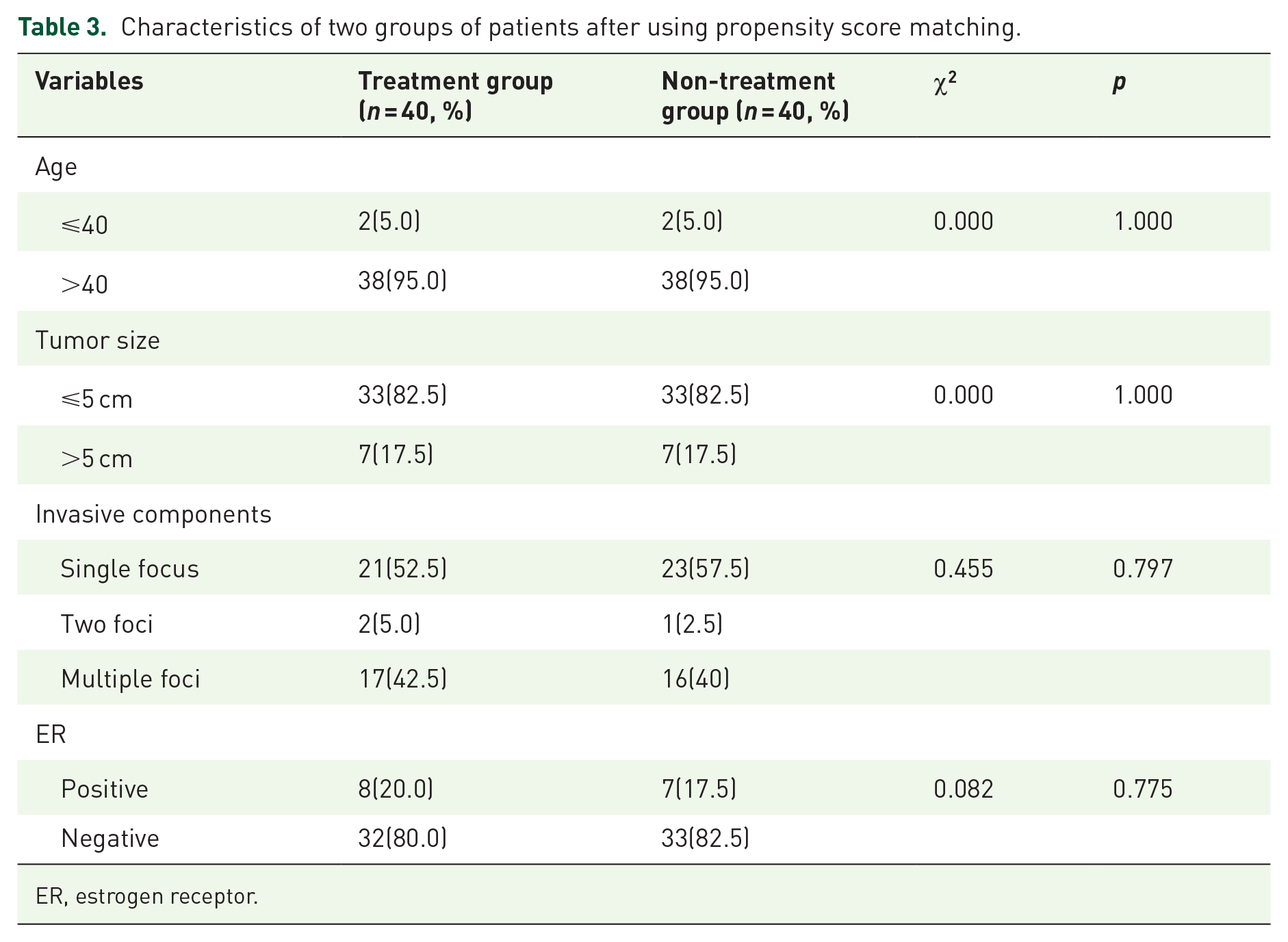
ER, estrogen receptor.
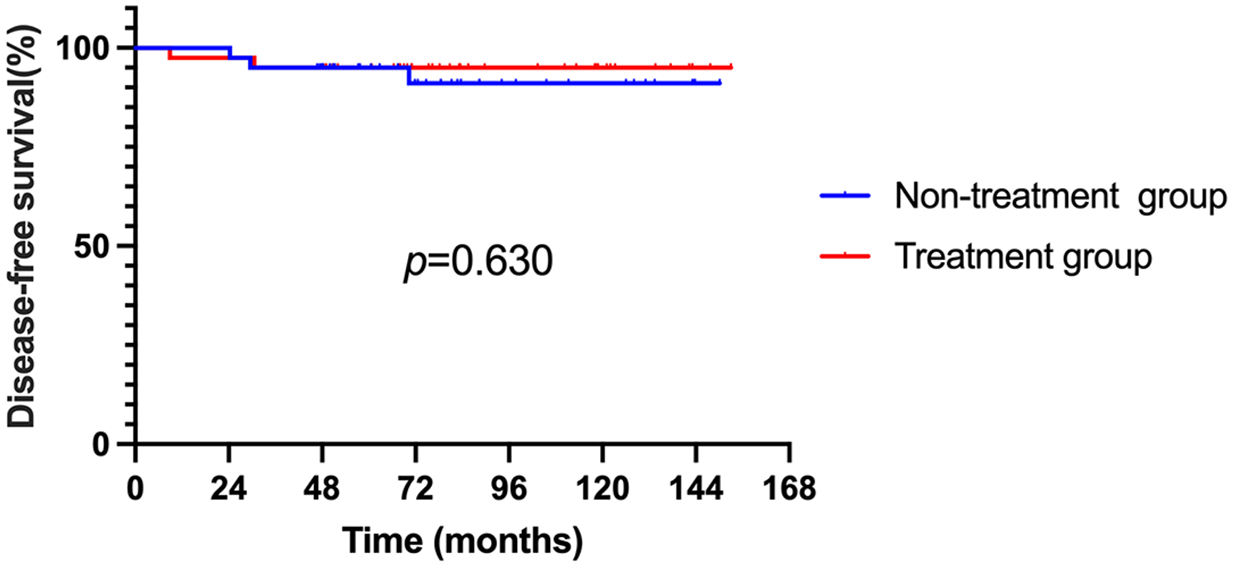
Comparison of DFS between patients who received targeted therapy or CT ± targeted therapy and patients who did not receive treatment. Patients who received targeted therapy or CT ± targeted therapy did not increase the 3-year DFS rate compared to those who did not receive treatment (95.0% vs 95.0%, p = 0.630).
Discussion
Prior to the advancement of anti-HER2 treatment, patients with HER2-positive breast cancer had a high risk of disease recurrence and reduced survival. 16 Due to the notable advantages observed in DFS and OS, CT and 1 year of trastuzumab were recommended as the standard of care in the adjuvant treatment for women with HER2-positive breast cancer.17–20 Numerous retrospective studies have provided evidence that HER2 positivity remains a predictive factor for an unfavorable prognosis among patients with T1a-bN0 tumors.3–5 According to a study conducted by Gonzalez-Angulo et al., 3 which examined 965 cases of T1a,bN0M0 breast cancers, the 5-year recurrence-free survival rates were 77.1% for patients with HER2-positive tumors and 93.7% for those with HER2-negative tumors. A meta-analysis using data from five adjuvant trastuzumab trials demonstrated that women diagnosed with HER2-positive tumors measuring ⩽2 cm could attain substantial improvements in DFS and OS through adjuvant trastuzumab treatment. 21 In the Adjuvant Paclitaxel and Trastuzumab (APT) trial, a prospective investigation was conducted to assess the safety and effectiveness of a treatment regimen consisting of 12 weeks of paclitaxel combined with trastuzumab, followed by 9 months of trastuzumab monotherapy. The study specifically focused on patients with small (⩽3 cm), node-negative, HER2-positive breast cancer. Notably, the trial reported impressive rates of 3-year and 10-year invasive DFS, which stood at 98.7% and 91.3%, respectively. 22 In addition, there were a total of 10 T1mic patients in the APT study, and none of them experienced distant recurrence after treatment with paclitaxel/trastuzumab. Approximately half of the patients included in the study were T1a-bN0 stage. Based on this evidence, the NCCN guidelines have endorsed paclitaxel combined with trastuzumab as the recommended standard treatment regimen for node-negative, small HER2-positive tumors. HER2DX facilitates treatment decision-making in stage I–III HER2-positive breast cancer by integrating genomic profiling with a clinical factor-based model. 23 However, regarding HER2-positive MIBC, there is still a lack of studies exploring the efficacy of anti-HER2 therapy.
Several studies investigating the prognosis of MIBC have consistently revealed that DCIS with microinvasion exhibits more aggressive biological and clinical characteristics than pure DCIS.11–13 A study conducted by our research team demonstrated that individuals diagnosed with MIBC exhibited worse cancer-specific survival and OS than those with DCIS. 13 Recently, a meta-analysis identified 26 studies that described the clinicopathological features of patients in both the DCIS and MIBC groups. The findings revealed that patients with MIBC had considerably shorter DFS and locoregional recurrence-free survival than those with DCIS. Both OS and distant metastasis-free survival tended to be shorter in patients with MIBC than in patients with DCIS, but the difference did not reach statistical significance. 24 Regarding the risk factors associated with relapse, a study including 1299 patients with MIBC reported that patients younger than 40 had poorer local-regional-free survival. 25 ER-negative invasive disease and the presence of lymphovascular invasion have also been identified as potential predictors of lymph node metastasis in MIBC patients.26,27
However, the prognostic value of HER2 in MIBC is still controversial. In a study conducted by Margalit et al., 28 in which 83 consecutive patients with MIBC were analyzed to identify risk factors for recurrence, HER2 overexpression was not found to be associated with an increased risk of recurrence. However, in the study mentioned above, when microinvasive components were unavailable, the HER2 status of carcinoma in situ was used instead. This substitution may have affected the reliability of the study’s results. Another study conducted by Zhang et al. involving 46 patients with MIBC examined the importance of HER2 expression in MIBC. Among the patients with available follow-up, all were alive without recurrence. Based on these findings, the authors concluded that HER2 overexpression in MIBCs does not appear to be linked to a worse outcome. 29 Importantly, the sample size was small, with only 19 cases of HER2-positive MIBC, which limits the generalizability of the findings.
The multicenter study we conducted is one of the largest in the field, involving 163 HER2-positive MIBC patients. All HER2 statuses of the invasive components were confirmed by pathological experts from our center. The rate of breast-conserving surgery in China is significantly lower than in many other countries, such as the United States. It may be that in our study, the inclusion of patients with multiple invasive lesions and in situ cancer made breast-conserving surgery more complex and difficult to perform. We found that HER2-positive MIBC had a favorable prognosis, with a 3-year DFS rate of 95.1%. However, there were eight recurrence events within 3 years after surgery, with five patients experiencing distant metastasis. In addition, one patient experienced distant recurrence 6 years post-surgery. This suggests that a small number of tumors exhibit biological aggressiveness. The type of local surgery treatment does not seem to affect the overall recurrence rate. We found that patients aged ⩽40 might have worse outcomes than those aged >40. Another highlight of this study was the inclusion of 40 HER2-positive MIBC patients who received trastuzumab or CT with or without trastuzumab, which allowed us to initially explore the role of CT and anti-HER2 therapy in this population. Due to a lack of data, the role of CT and anti-HER2 therapy in HER2-positive MIBC patients has been unknown in guidelines, which is increasingly disturbing clinicians. As mentioned earlier, the APT trial included only nine patients with MIBC. 17 In the study by Zhang et al., 29 five patients received adjuvant CT, four of whom were recommended to undergo weekly paclitaxel plus trastuzumab. None of these patients experienced relapse. In this multicenter study, patients under the age of 40 were more likely to experience recurrence. The 3-year DFS rates of patients under the age of 40 years and over 40 years old were 81.8% and 96.1%, respectively. Thirty-nine patients (23.9%) received adjuvant CT, out of which 12 patients received CT combined with trastuzumab. Most of the patients who received CT were young and ER negative. The majority of CT regimens consisted of four cycles of docetaxel or anthracyclines combined with cyclophosphamide. After applying propensity score matching based on age, tumor size, ER status, and number of infiltration lesions, patients who received trastuzumab and patients who received CT with or without trastuzumab did not exhibit a trend toward an increased 3-year DFS rate compared to those who did not receive treatment. These findings suggest that MIBC patients could not benefit from adjuvant CT, with or without trastuzumab. However, further research is needed to determine whether treatment can provide DFS benefits for high-risk MIBC populations under the age of 40 years.
There were several limitations in our study that should be acknowledged. First, it was a real-world study conducted retrospectively, which may impact the generalizability of the findings. Second, although this study constitutes the largest cohort of HER2-positive MIBC patients to date, the sample size is still relatively limited, and the follow-up duration was relatively short, potentially affecting the comprehensive understanding of the outcomes. Therefore, future prospective studies with large sample sizes are still needed.
Conclusion
The 3-year DFS rate of HER2-positive MIBC is 95.1%, indicating a favorable prognosis. Recurrence in these patients mainly occurs within 3 years after surgery. Patients under 40 years old might have a poorer prognosis. MIBC patients seemed to get little benefit from DFS from CT or targeted therapy. However, further prospective studies are needed to determine whether receiving trastuzumab or CT combined with trastuzumab is beneficial for patients under the age of 40 years in the future.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251345716 – Supplemental material for The characteristics of HER2-positive microinvasive breast cancer and the necessity of systemic adjuvant therapy in these patients: a multicenter real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251345716 for The characteristics of HER2-positive microinvasive breast cancer and the necessity of systemic adjuvant therapy in these patients: a multicenter real-world study by Bo Lan, Dan Lv, Changyuan Guo, Zitong Feng, Ying Fan, Tao Sun, Jiayi Huang, Xiaoying Sun, Fei Ma and Binghe Xu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251345716 – Supplemental material for The characteristics of HER2-positive microinvasive breast cancer and the necessity of systemic adjuvant therapy in these patients: a multicenter real-world study
Supplemental material, sj-docx-2-tam-10.1177_17588359251345716 for The characteristics of HER2-positive microinvasive breast cancer and the necessity of systemic adjuvant therapy in these patients: a multicenter real-world study by Bo Lan, Dan Lv, Changyuan Guo, Zitong Feng, Ying Fan, Tao Sun, Jiayi Huang, Xiaoying Sun, Fei Ma and Binghe Xu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
