Abstract
This paper aimed to probe into the expression of long non-coding RNA (lncRNA) SNHG16 in human gastric cancer (GC) and its potential tumor biological functions. The expression of lncRNA SNHG16 was detected in GC and adjacent tissues and GC cell lines using qRT-PCR. GC MGC-803 cells were transfected with siRNA of lncRNA SNHG16, as well as blank and negative control. A series of experiments including CCK-8, flow cytometry, transwell, and wound healing assay were adopted to evaluate the effects of lncRNA SNHG16 on cell growth and metastasis. Besides, the nude mouse xenograft tumor model was established to draw tumor growth curve and measure tumor volume during treatments. TUNEL staining was used to determine the apoptosis rate of tissues. The expression of lncRNA SNHG16 in GC tissue, significantly associated with invasion depth, lymph node metastasis, TNM stage and histological differentiation (all
Keywords
Introduction
Gastric cancer (GC) is one of the most frequently diagnosed therioma, ranking the second in cancer-causing death in global [1]. The rate of newly diagnosed cases and deaths increases by over 70% annually worldwide [2]. Surgery accompanied with effective adjuvant chemoradiotherapy still remains the primary curative treatment for GC, whereas it left poor clinical efficacy for the patients with GC metastasis [3]. Thus, seeking for a new biomarker for early diagnosis of GC and comprehensively exploring the mechanisms of GC occurrence and progression is required for improving the survival rate and quality of life of GC patients [4, 5].
Long non-coding RNA (lncRNA) is a RNA that comprises a class of transcripts with more than 200 nucleotides in length but do not encode proteins [6], which could not only regulate the normal physiological activities such as organism development, differentiation and metabolism, but also participant in diverse diseases including tumorigeneses [7, 8, 9]. For example, previous studies demonstrated that lncRNA is closely linked to biological activities of multiple tumors, such as breast cancer [10], colorectal cancer [11], lung cancer [12], renal carcinoma [13] and liver cancer [14], and it also plays a potential role in GC incidence and development [15, 16]. Moreover, Yang et al. found that lncRNA H19 expression was upregulated in GC tissue, while tumor cell proliferation got suppressed and apoptosis was enhanced when H19 levels in GC cells were silenced by a small interfering H19 RNA [17]. Saito et al. also revealed that lncRNA ATB activated by TGF-
Therefore, we discussed the expression of lncRNA SNHG16 in GC and the potential molecular mechanism involved in the progression of GC to provide new insights into the pathogenesis and treatment of GC in this paper.
Materials and methods
Ethics statement
According to the declaration of Helsinki, all the subjects were informed and signed the informed consent before entering the investigation, which was approved by the ethics committee of the Beijing Shijitan Hospital of Capital Medical University. And all the animal experiments were carried out in strict accordance with the animal protocols and guidance approved by the ethics committee concerning animal research at our hospital. Every effort designed to minimize impact on the animals under study.
Subjects
A number of 122 cases of patients received GC radical treatment in the General Surgery Department of Beijing Shijitan Hospital of Capital Medical University were involved in the following experiments from June 2014 to June 2016. Inclusion criteria: (a) primary GC cases without recurrence and metastasis; (b) no previous history of other malignancies; (c) not treated with chemotherapy, radiotherapy or biotherapy before surgery; (d) patients’ pathologic biopsy reports after surgery reviewed by a pathologist conformed to GC pathological diagnosis guidelines; (e) with complete clinical and pathological data. The tumor tissue (non-necrotic parts) and the corresponding adjacent tissues (over 5 cm away from the tumor tissue) of each patient were collected, respectively, and immediately stored in frozen tubes in liquid nitrogen. Of the participants, 83 cases were males and 39 cases were females, and the age of the patients ranged from 30 to 81 with an average of 58.14
Cell culture
The GC cell lines (AGS, BGC-823, MGC-803, SGC-7901, and MKN451) and normal gastric epithelial cell line GES-1 were all purchased from the Shanghai Institute of Cell Biology, Chinese Academy of Sciences. Cells were inoculated in RPMI-1640 (Gibico, USA) containing 10% FBS (Gibico, USA), 100 U/mL penicillin and 100
Real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR)
Total RNA from GC, adjacent tissues and all GC cell lines was extracted following the kits instructions (Promega, USA). Reverse transcription PCR was performed on a Reverse Transcription System A3500 (Promega, USA) according to manufacturer’s procedures. PCR was achieved using the ABI PRISM 7500 Sequence Detection System (Applied Biosystems, Foster City, CA). The PCR amplification was carried out with cycling conditions: 95
Cell grouping and transfection
The MGC-803 cells were utilized for transfection and grouped into Blank group (MGC-803 cell without any treatment), negative control (NC) group (MGC-803 cells transfected with negative plasmid), si-SNHG16-1 group (MGC-803 cells transfected with SNHG16 siRNA-1 plasmid), and si-SNHG16-2 group (MGC-803 cells transfected with SNHG16 siRNA-2 plasmid). By the way, two sequences of si-SNHG16 (si-SNHG16-1/-2) were designed according to the previous study [21]. Cells in the logarithmic phase were obtained in each group and inoculated in 6-well plates with a density of 4
Cell proliferation by CCK-8 assay
The harvested cells of each group were washed with PBS and then digested with trypsin. The uniform cell culture was seeded with inoculation of 100
Cell cycle by flow cytometry
After transfection for 48 h, the cells in each group were fixed with ice cold anhydrous ethanol at 4
Apoptosis rate analysis by flow cytometry
Annexin V Kit (C1063, Beijing, China) was adopted to evaluate cell apoptosis. With 0.25% trypsin digestion and gently triturating, the digestive solution was mixed with the previously collected medium after transfection for 48 h. Afterwards, the mixtures were centrifuged at 12000
Transwell assay
The prepared matrigel was mixed with serum-free DMEM medium as a ratio of 1:3, and then was coated onto the upper surface of Transwell chamber for air-drying at room temperature. The trypsin digested cells were diluted into a density of 1
Wound healing assay
The cells in each group were inoculated in 6-well plates with 5
Expression of lncRNA SNHG16 in GC tissues and cell lines detected by qRT-PCR. Note: A, Expression of lncRNA SNHG16 in 122 pairs of GC tissues and matched adjacent tissues; B, Receiver operating characteristic (ROC) curve analysis was used to determine whether SNHG16 is really a good candidate to discriminate tumor tissues from non-tumor tissues; C, Expression of lncRNA SNHG16 in GC cells, 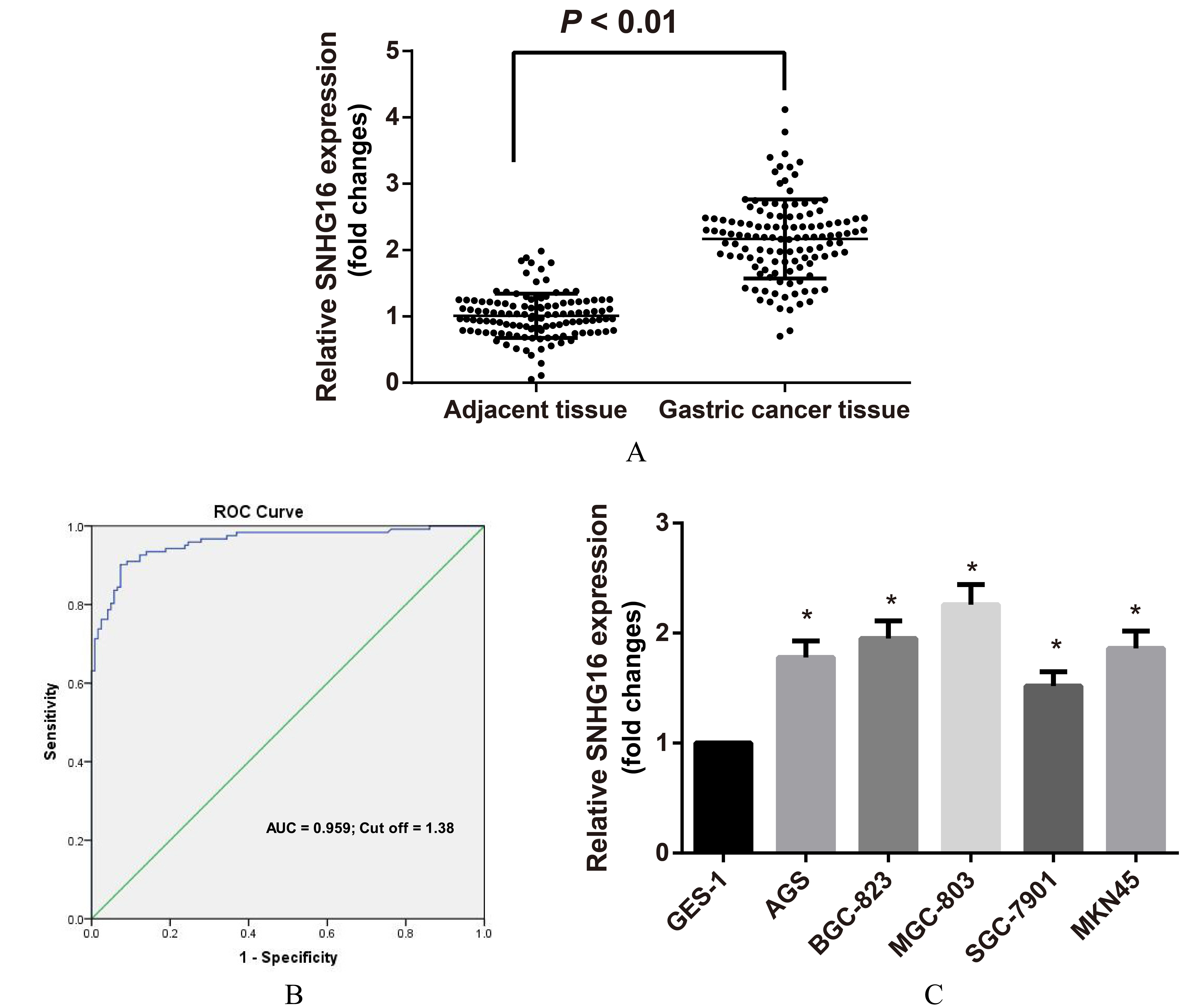
Relationship between lncRNA SNHG16 expression and GC clinicopathological features. Data were presented as 2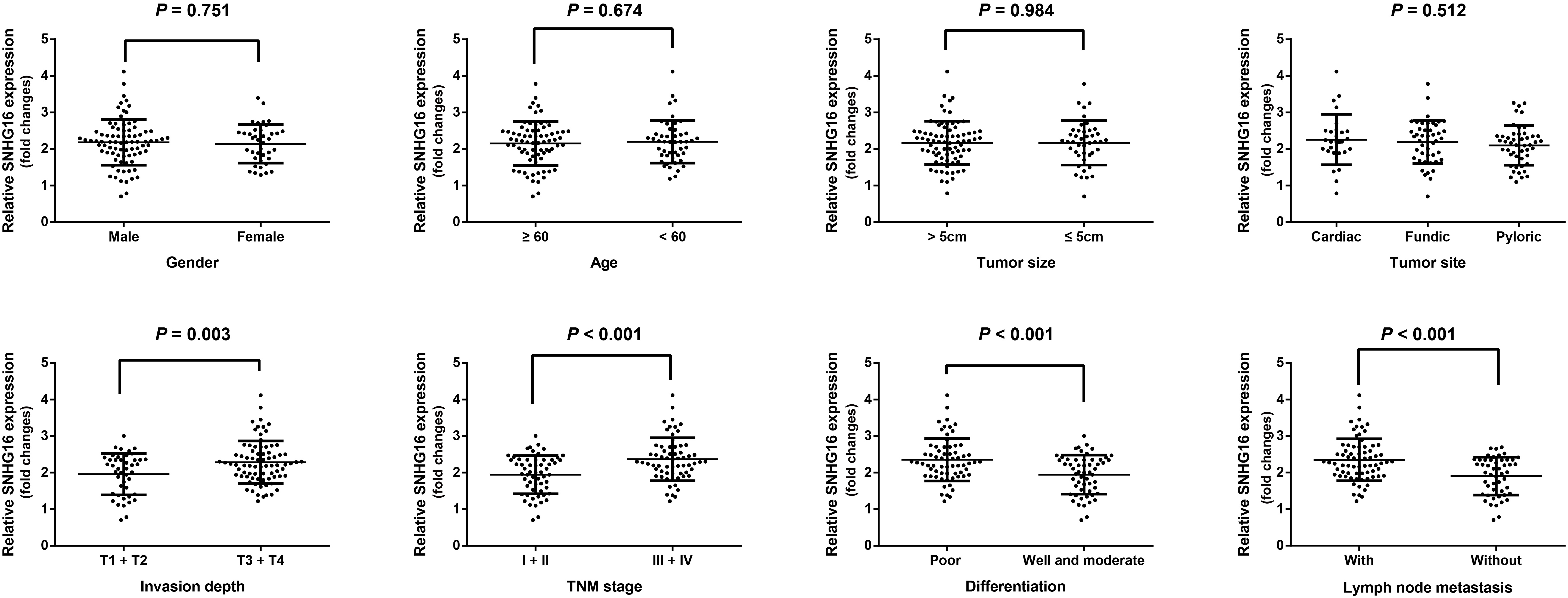
Comparison of SNHG16 expression and cell proliferation in each group when transfected with siRNA of lncRNA SNHG16. Note: A, lncRNA SNHG16 expression in each transfected group detected by qRT-PCR, 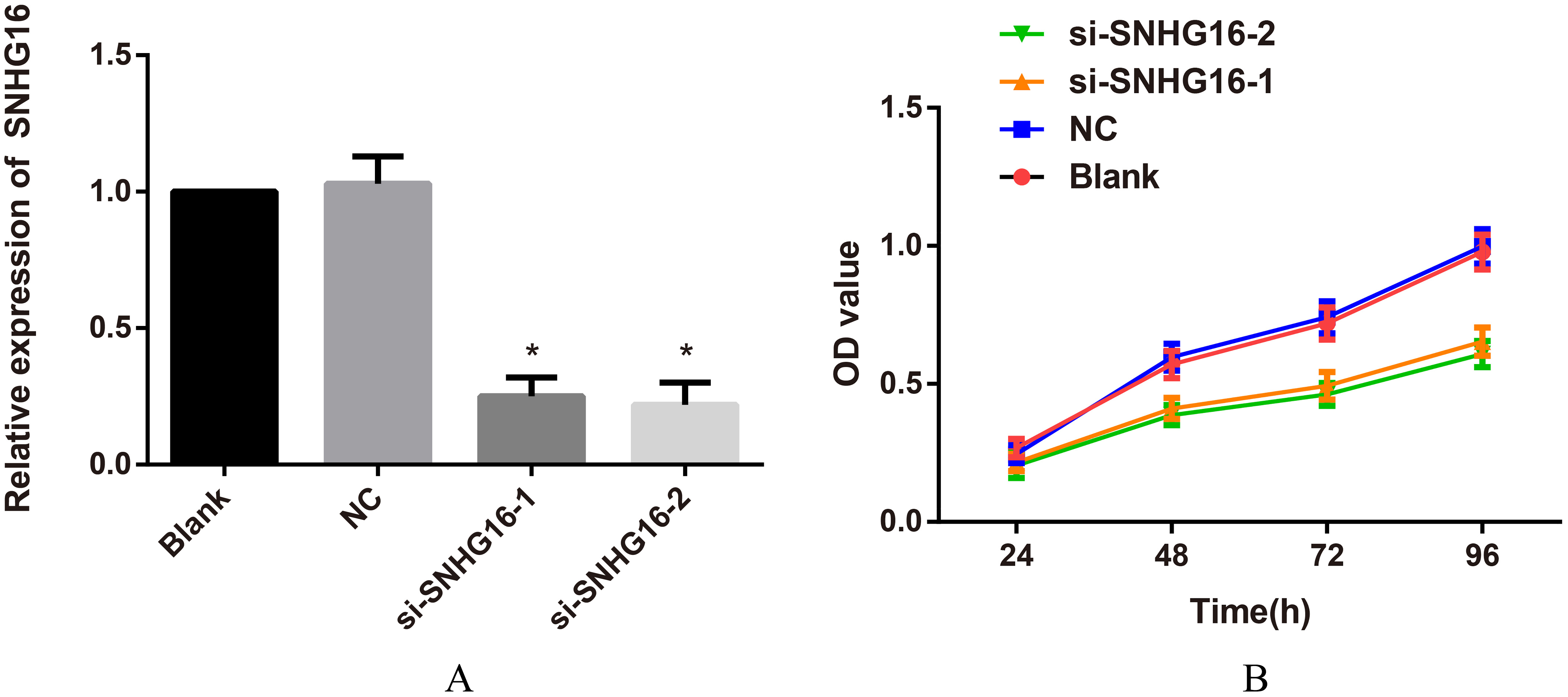
Effects of siRNA of lncRNA SNHG16 on cell cycle and apoptosis of GC cells. Note: A, Cell cycle examined by flow cytometry; B, Apoptosis analyzed using flow cytometry. 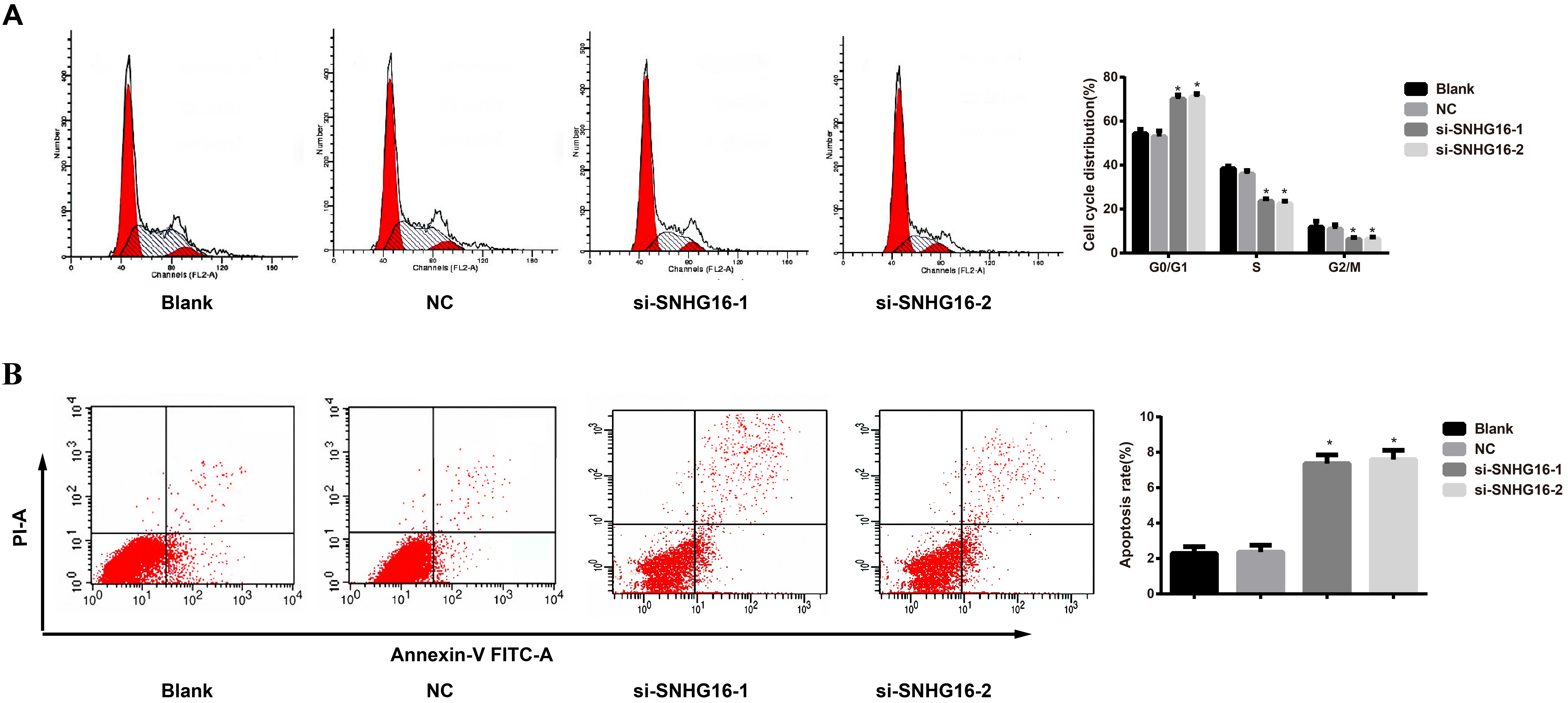
Transwell and wounding healing assay for cell invasion and migration analysis in each transfected group. Note: A, Comparisons of cell invasion in each group; B, Comparisons of cell migration in each group. 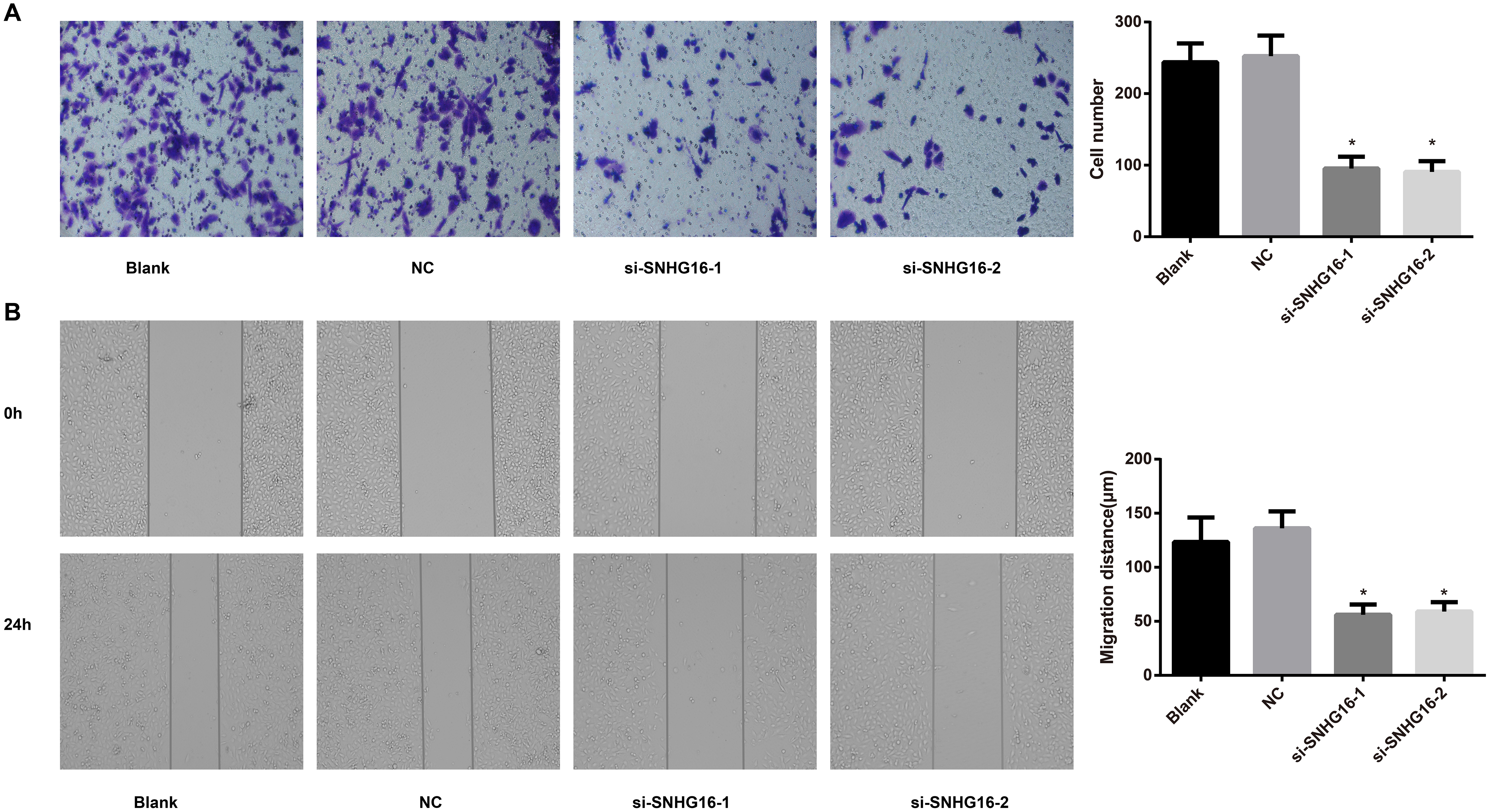
Subcutaneous xenograft trials in nude mice. Note: A, Xenograft growth curve in nude mice; B: Comparison of xenograft weight in nude mice; C: TUNEL staining for xenograft apoptosis analysis. 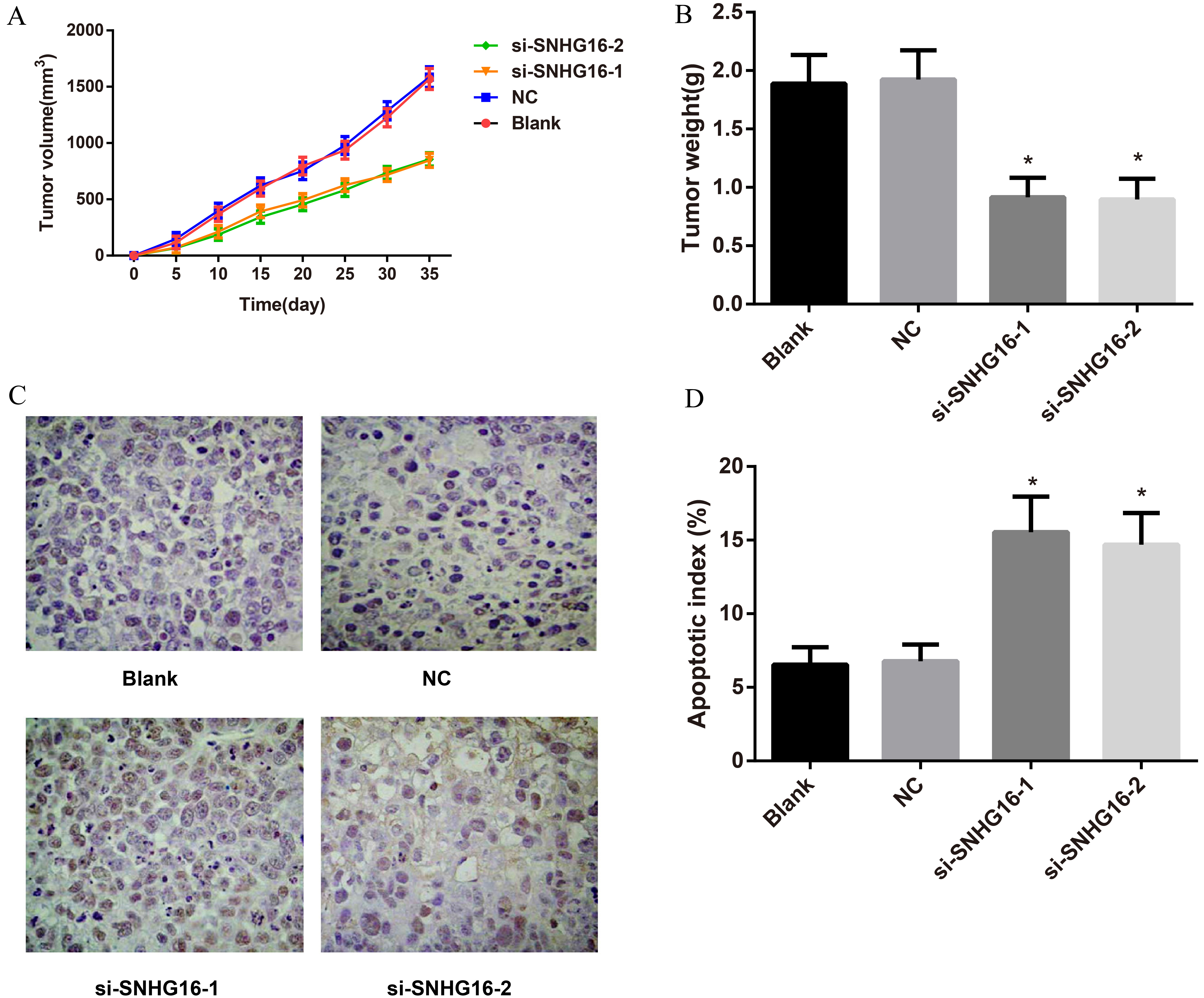
A number of 30 female BALB/C nude mice (4
TUNEL staining
The isolated xenografts were fixed using TUNEL detection kit (Roche, Switzerland) at room temperature 30 min, washing with PBS, and then they were immersed in 0.2% Triton X-100 for 5 min to strengthen the permeability. An addition of 50
Statistical analysis
All statistical data were analyzed by the SPSS 19.0 software (SPSS, Inc, Chicago, IL, USA). The measurement data were expressed as means
Results
Expression of lncRNA SNHG16 in GC tissues and cell lines
QRT-PCR was performed for lncRNA SNHG16 expression analysis in GC tissues and cell lines. The lncRNA SNHG16 expression in GC tissue was higher than in adjacent tissues (
Relationship between lncRNA SNHG16 expression and clinicopathological features of GC
The lncRNA SNHG16 expression was observed to have no clearly relevance to clinical pathological features in age, gender, tumor size and tumor site (all
Effects of siRNA of lncRNA SNHG16 on cell proliferation
In contrast with Blank group and NC group, the expressions of lncRNA SNHG16 in si-SNHG16-1 and si-SNHG16-2 groups were markedly downregulated (
Effects of siRNA of lncRNA SNHG16 on cell cycle and apoptosis in each group
Cell cycle observed by flow cytometry showed that the si-SNHG16-1 and si-SNHG16-2 groups performed elevated number of cells in G0/G1 phase (all
Effects of siRNA of lncRNA SNHG16 on migration and invasion of cells in each group
Transwell assays confirmed that the si-SNHG16-1 and si-SNHG16-2 groups showed markedly reduction in invasive cells in comparison with Blank group and NC group (
Effects of siRNA of lncRNA SNHG16 on the growth of human GC xenografts in nude mice
The xenografts in si-SNHG16-1 and si-SNHG16-2 groups grew slower than that in Blank group and NC group (all
Discussion
Despite being absent of protein-coding capacity, lncRNA performed active roles in regulating gene expression at the epigenetic, transcription and post-transcription level [23]. To our knowledge, lncRNA is not only widely involved in diverse physiological processes including growing development, and its specific expression may also facilitate the occurrence and development of many diseases, particularly the tumorigenesis [24]. In recent years, more and more lncRNAs have been identified in extensive observations to be obviously overexpressed in various tumors. For instance, the LSINCT5 was proved to be highly expressed in both breast and ovary cancer cells and tissues [25], and LncRNA HULC was substantially upregulated in liver and pancreatic cancers, which had a closely relationship with tumor size, lymphatic metastasis and vascular invasion [26, 27]. Moreover, HIF1A-AS2 was validated to be associated with advanced tumor stage, lymph node metastasis, severe invasion, or poorer prognosis as it presented elevated expression, indicative of a novel marker for GC diagnosis and prognosis [28]. Besides, Yang et al. also discovered that c-Myc can directly bind to the E-box element in the promoter region of long non-encoding CCAT1, and c-Myc ectopic expression can activate CCAT1 to be highly upregulated in GC, and thus promoting GC metastasis [29]. Additionally, it is important to emphasize that SNHG16 expression in highly aggressive neuroblastoma specimens and neuroblastoma cell lines was greatly upregulated, implying SNHG16 to be related to cancer progression and nervous system development [19]. Not surprisingly, SNHG16 observed in this study was significantly upregulated in GC tissue and GC cell lines, which was consistent with previous findings, especially under conditions of poorer degree of differentiation, more advanced tumor stage, and increased tumor invasion and metastasis, suggesting that SNHG16 may function as an oncogene in GC incidence and development, more importantly, its overexpression even aggravated cell malignant transformation and tumor progression through expanding GC cell invasion and metastasis.
Another important findings in our experiment demonstrated that migration distance and invasive cell number were clearly decreased in the SNHG16 siRNA-transfected group. In the past studies, evidence revealed that lncRNA HOTAIR appeared high levels in GC tissue and cells, whereas knockdown of HOTAIR could control GC cell proliferation and induce apoptosis, so as to weaken tumor cell invasion and reduce cell migration [30]. As authenticated by Zhao’s group, silencing of HULC, another lncRNA, could effectively reverse the EMT phenotype in GC cells [31]. Similarly, silencing the expression of Linc00152 could suppress GC cell proliferation and metastasis, arrest cell cycle at G1 phase, and then increase apoptosis by regulating the EMT procedure [32]. Therefore, we used siRNA to interfere with SNHG16 expression for further elucidating the associations between SNHG16 expression and GC occurrence, and our results turned out that the expression of SNHG16 was dramatically lowered after transfected with SNHG16 siRNA in GC cells, which could markedly reduce GC cell proliferation and invasion and migration, promoted cells arrest at G0/G1 phase and enhanced apoptosis rate, showing that SNHG16 expression could be served as a strong prognostic indicator of GC cell growth and metastasis. Similar finding was also obtained in bladder cancer that high SNHG16 expression was confirmed to promote cell proliferation, invasion and migration, while silencing of SNHG16 improved the chemosensitivity of bladder cancer [33], supporting the findings we did in this research. As we know, disruption of a proper regulation of cell cycle contributing to cancer development resulting from the oncogenes activation or tumor suppressor genes inactivation, leads to uncontrolled cellular proliferation [34], providing an explanation that silencing of SNHG16 expression mitigated cell growth and suppressed cell invasion and migration, possibly by functioning on the cell cycle control program to affect the progression of tumor cell cycle. Considering the above, SNHG16 was similar to many other lncRNAs in GC, as well as its biological characteristics to influence GC, so as to accelerate tumorigenesis and progression, which may support the notion that SNHG16 may become a potential target for GC therapy.
In addition, we established a model of human GC xenografts in nude mice, which is a good model to offer valuable approaches for further studying its biological and malignant behavior of SNHG16 in GC, because the histological form or tumor biological features of transplant tumor is the same as original tumor [35, 36]. The present study showed that the xenograft in si-SNHG16 group presented inhibited tumor growth, decreased tumor volume and improved apoptosis rate, demonstrating that SNHG16 plays an important role in regulation of the growth of GC xenografts
In conclusion, up-regulation of SNHG16 exhibits a tumorigenic effect in GC, and suppression of SNHG16 could restricted cell proliferation and metastasis, and promoted apoptosis in GC cells in this paper, providing a promising target for GC diagnosis and treatment.
Footnotes
Acknowledgments
The authors appreciate the reviewers for their meaningful comments in the current study.
