Abstract
BACKGROUND:
Long non-coding RNAs (lncRNAs) exert important functions involved in tumorigenesis and cancer progression including esophageal squamous cell cancer (ESCC), however, the clinical role and underlying biological function of Small Nucleolar RNA Host Gene 1 (SNHG1) in ESCC is not well known.
METHODS:
Quantitative Real-time polymerase chain reaction (QRT-PCR) was used to detect the SNHG1 expression levels in ESCC tissues and adjacent non-cancerous tissues. Chi-square test was used to evaluate the association between clinicopathological features and SNHG1 expression in ESCC patients, Kaplan-Meier curve and log rank test was performed to analyze the association between overall survival and SNHG1 expression. Cell proliferation and invasion was assessed by MTT assay, colony formation, and transwell cell invasion assays. Western blot were also performed to examine protein expression levels of E-cadherin, Vimentin and N-cadherin, Notch 1 and Hes-1.
RESULTS:
We demonstrated that lncRNA SNHG1 was significantly up-regulated in ESCC tissues compared with adjacent non-cancerous tissues in ESCC patients. Meanwhile, increased lncRNA SNHG1 expression levels markedly correlated with lymph node metastasis, depth of invasion, TNM stage and reduced over survival time in ESCC patients. Furthermore, MTT assay, colony formation, transwell cell invasion, qRT-PCR and Western-blot assays demonstrated that knockdown of lncRNA SNHG1 could inhibit cell proliferation and cell invasion capacity and cell Epithelial-Mesenchymal Transition (EMT) phenomenon by up-regulation E-cadherin and down-regulating Vimentin and N-cadherin in ESCC cells. Besides, we demonstrated that knockdown of SNHG1 suppressed the Notch signaling pathway by reducing the Notch1 and Hes-1 expression levels in ESCC cells.
CONCLUSIONS:
These results indicated that lncRNA SNHG1 may be a potential predictor of prognosis in ESCC patients and a novel target for ESCC treatment.
Introduction
Esophageal cancer is the eighth most common cancer worldwide with a highly lethal malignancy [1]. In China, Esophageal squamous cell carcinoma (ESCC) is listed as the predominant histological type [2]. Although great advances have improved in the diagnosis and treatment of ESCC in recent years, due to local or distant recurrences, the over survival rate of ESCC patients is still poor [3, 4]. Thus, exploring novel potential biological biomarkers and potential targets of treatment for ESCC patients is important.
LncRNAs are one of major members in non-coding RNAs family and more than 200 nucleotides in length [5]. Studies had verified lncRNAs function as key regulators in some aspects of biology by regulating chromatin organization, transcriptional and post-transcriptional levels [6, 7]. LncRNAs are found to be involved in the initiation and progression of various cancers including ESCC. Such as, up-regulation of long non-coding RNA HOTTIP promotes metastasis of esophageal squamous cell carcinoma via induction of EMT phenomenon [8]. Long noncoding RNA AFAP1-AS1 is significantly up-regulated in esophageal squamous cell carcinoma and promotes cell proliferation and inhibits cell apoptosis [9]. LncRNA ZEB1-AS1 expression was significantly higher in ESCC patients and high ZEB1-AS1 expression predicts a poorer overall survival and disease-free survival rate [10]. Up-regulation of the long non-coding RNA NEAT1 promotes esophageal squamous cell carcinoma cell progression and correlates with poor prognosis [11]. However, the clinical role and underlying function of Small Nucleolar RNA Host Gene 1 (SNHG1) in ESCC is unknown.
SNHG1 displays higher expression in multiple tumor types and promotes tumor growth through diverse mechanisms, such as, SNHG1 expression was significantly upregulated in hepatocellular carcinoma and lung cancer and functions as oncogene in tumor progression [12, 13]. Down-regulated long non-coding RNA SNHG1 inhibits tumor genesis of colorectal carcinoma [14]. In the study, we demonstrated that SNHG1 was up-regulation in ESCC tissues. Increased SNHG1 expression levels were correlated with lymph node metastasis, depth of invasion, TNM stage and over survival time. Furthermore, knockdown of SNHG1 inhibited cell proliferation and cell invasion and cell EMT process. Moreover, we demonstrated that knockdown of SNHG1 suppressed the Notch signaling way by reducing the Notch1 and Hes-1 expression levels in ESCC cells. These results showed that SNHG1 may be a potential therapeutic target for ESCC.
Materials and methods
Patients and tissue samples
A total of 72 cases of fresh ESCC tissue samples and adjacent normal tissue samples were collected from patients who underwent radical resection at Department of Thoracic in Jilin Cancer Hospital from January 2010 to March 2015. The ESCC patients were diagnosed by histopathological confirmation. None of the patients received preoperative therapy. Tissues samples were snap-frozen in liquid nitrogen immediately after resection and stored at
Cell culture and cell transfection
Two ESCC cell lines (Eca109 and TE-1) were purchased from the Shanghai Institute of Biochemistry and Cell Biology (Shanghai, China). Cells were cultured in RPMI 1640 (Invitrogen) supplemented with 10% fetal bovine serum (Invitrogen), 100 U/ml penicillin and 100 g/ml streptomycin (Gibco; Thermo Fisher Scientific, Inc.), at 37
MTT assay
Three thousand cells/well were seeded into 96-well plates and transfected with si-NC and si-SNHG1. MTT (20
Cell colony formation assay
For cell colony assay, 200 cell/well were transfected with si-NC or si-SNHG1 and seeded in the 12-well plates. Cells were cultured for 2 weeks, and then fixed with methanol and stained using 0.1% crystal violet, and calculated cell colonies number.
The relative SNHG1 in ESCC tissues was higher and associated with prognosis in ESCC patients. (A) The relative expression of SNHG1 was determined by qRT-PCR assays in 72 cases ESCC tissues and adjacent normal tissues. (B) Survival curve was analyzed by Kaplan-Meier method and the log-rank test in higher SNHG1 expression and lower SNHG1 expression group in ESCC patients. 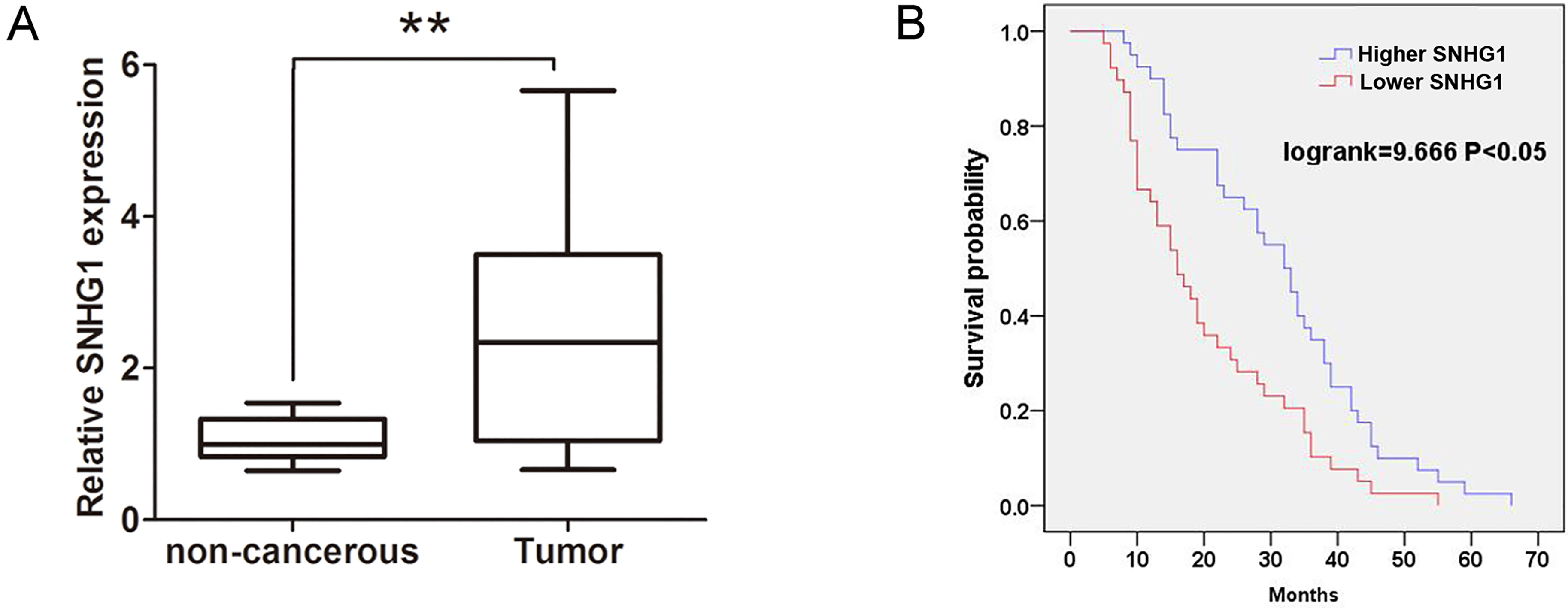
For cell invasion assay, 1
RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA derived from frozen tissue samples and cells was extracted using TRIzol
Western-blot assay
The protein derived from cells were lysed by using mammalian protein extraction reagent RIPA (Sigma) supplemented with protease inhibitors cocktail (Sigma). Protein concentration was determined using BCA assay kit (Millipore). The protein extractions were separated by 10% SDS-PAGE transferred to 0.45 mm nitrocellulose (NC) membranes (Millipore). The membranes were blocked with 5% nonfat milk for 1 h and then incubated with specific antibodies with E-cadherin (1:1000, CST, USA), Vimentin (1:1000, CST, USA), N-cadherin (1:1000, CST, USA), Notch1 (1:2000, Abcam, USA), Hes-1 (1:1000, CST, USA) and GAPDH (1:1000, CST, USA) overnight at 4
Statistical analysis
Statistical analyses were performed using SPSS version 19.0. All data are showed as the mean
The correlation of ESCC clinicopathologic variables with SNHG1 in cancerous tissue samples
The correlation of ESCC clinicopathologic variables with SNHG1 in cancerous tissue samples
SNHG1 is overexpressed in ESCC tissues and correlates with prognosis in patients
To determine the clinical role of SNHG1 in ESCC, we performed qRT-PCR assays to detect the mRNA expression levels of SNHG1 in 72 cases of ESCC tissues and adjacent normal tissues. The results uncovered that SNHG1 expression levels in ESCC tissues were dramatically higher, compared with adjacent normal tissues (Fig. 1A). Furthermore, we investigated the association between clinicopathological feathers and SNHG1 mRNA expression levels in ESCC patients. As shown in Table 1, higher SNHG1 expression levels in ESCC patients were associated with lymph node metastasis (
Downregulation of SNHG1 suppresses cell proliferation and invasion in ESCC cells
To explore the effects of SNHG1 expression on cell proliferation and invasion capacities in ESCC cells, we performed the MTT cell proliferation, colony formation, and transwell cell invasion assays. Firstly, we knocked down the SNHG1 in Eca109 and TE-1 cells using two si-SNHG1 oligos, the results showed dramatically reduced SNHG1 expression were observed in both cells (Fig. 2A–B). The si-SNHG1-1 had a better knockdown efficiency and was used in the following experiment. Furthermore, we transfected with si-SNHG1 or si-NC into TE-1 and Eca109 cells, the MTT assays confirmed that cell proliferation capacity was significantly inhibited after knockdown of SNHG1 compared with that in si-NC group in TE-1 and Eca109 (Fig. 2C–D). The colonies number was significantly reduced in si-SNHG1 group compared with that in si-NC group in TE-1 and Eca109 cells (Fig. 2E–F). The transwell cell invasion showed that cell invasive capacity was also significantly suppressed after knockdown of SNHG1 in TE-1 and Eca109 cells (Fig. 3A–B). Thus, these results showed that knockdown of SNHG1 inhibited cell proliferation and invasion in ESCC cells.
Downregulation of SNHG1 inhibited cell proliferation and invasion ability in ESCC cells. (A)–(B) The relative expression of SNHG1 was determined by qRT-PCR assays after knockdown of SNHG1 using si-SNHG1-1 and si-SNHG1-2 in TE-1 or Eca109 cells. (C)–(D) MTT cell proliferation was performed to determine cell proliferation when transfected with si-NC or si-SNHG1 into TE-1 or Eca109 cells. (E)–(F) cell colony formation was performed to determine cell proliferation when transfected with si-NC or si-SNHG1 into TE-1 or Eca109 cells. Data are represented as mean 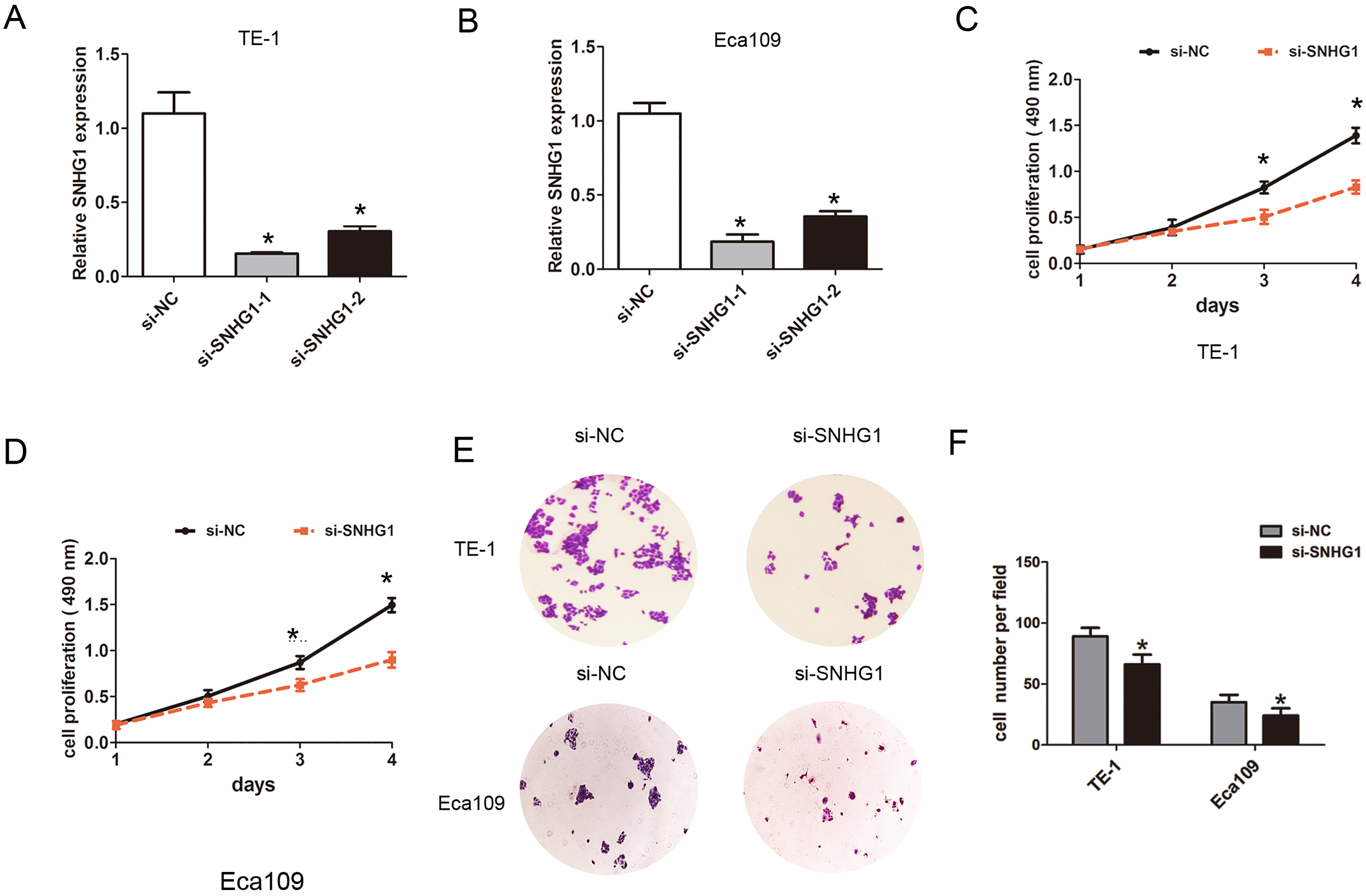
Downregulation of SNHG1 inhibited cell invasion and EMT in ESCC cells. (A)–(B) Transwell cell invasion assays was performed to determine cell invasion capacity when transfected with si-NC or si-SNHG1 into TE-1 or Eca109 cells. (C)–(D) qRT-PCR analysis was performed to assess the expression of E-cadherin, Vimentin and N-cadherin when transfected with si-NC or si-SNHG1 into TE-1 or Eca109 cells. (E)–(F) Western blot was used to assess the expression of E-cadherin, Vimentin and N-cadherin when transfected with si-NC or si-SNHG1 into TE-1 or Eca109 cells. Data are represented as mean 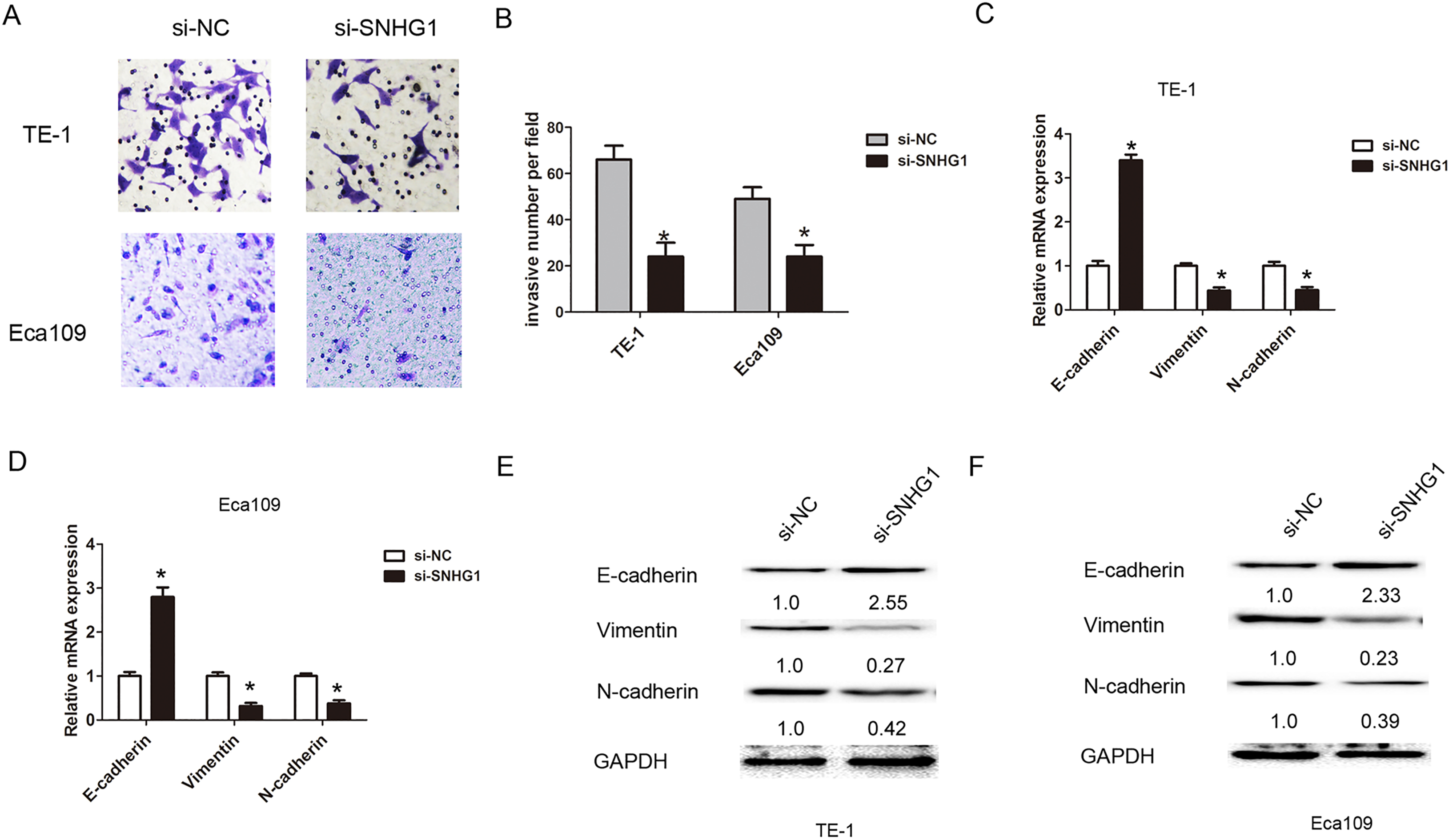
Downregulation of SNHG1 inhibited Notch signaling pathway in ESCC cells. (A) Western blot was used to assess the expression of Notch 1 and Hes-1 when transfected with si-NC or si-SNHG1 into TE-1 cells. (B) Western blot was used to assess the expression of Notch 1 and Hes-1 when transfected with si-NC or si-SNHG1 into Eca109 cells.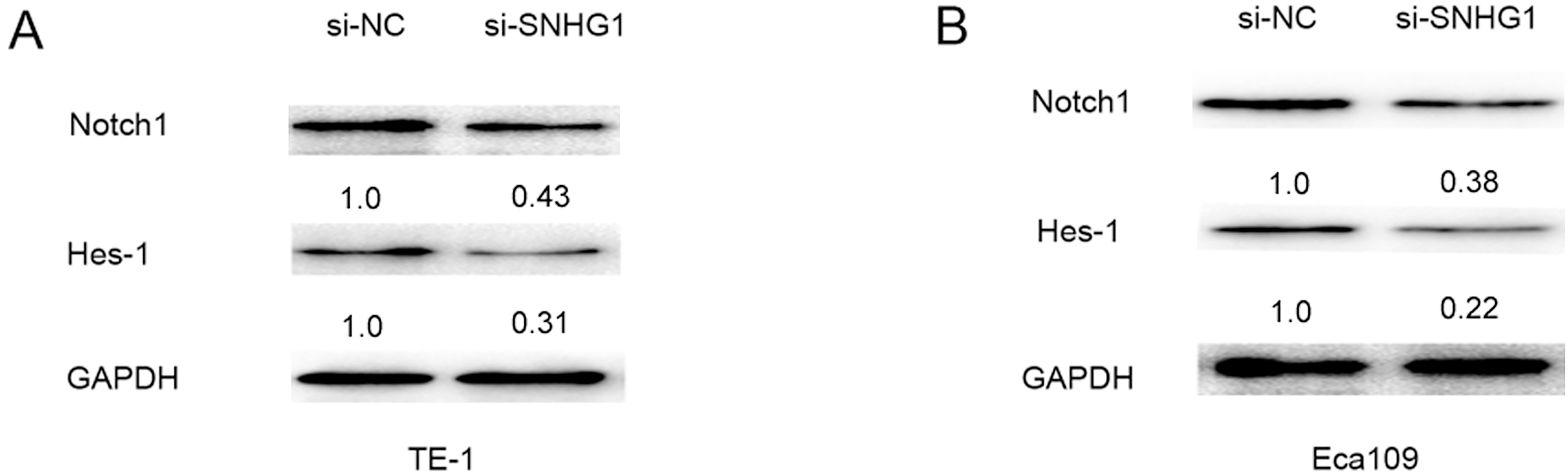
As we known, EMT process was association with tumor invasion and metastasis. We further detected the effects of SNHG1 on cell EMT process in ESCC. QRT-PCR and Western blot assays results showed that mRNA and protein levels of EMT epithelial marker E-cadherin expression was significantly up-regulated, but mesenchymal marker N-cadherin and Vimentin expression levels were dramatically downregulated in TE-1 cells (Fig. 3C and E). The results also showed that knockdown of SNHG1 increased the expression level of E-cadherin and decreased the N-cadherin and Vimentin expression levels in Eca109 cells (Fig. 3D and F). Thus, these results showed that downregulation of SNHG1 suppressed cell EMT in ESCC.
Downregulation of SNHG1 suppresses Notch signaling pathway in ESCC cells
Epithelial-mesenchymal transition (EMT) and Notch signaling are important for some tumors development and progression. EMT is induced by several signaling pathways, including transforming growth factor-
Discussion
Increasing evidence indicates that some LncRNAs are widely involved in the development and progression of ESCC [15]. Up-regulation of LOC100130476 led to the suppression of cell proliferation and invasiveness in esophageal squamous cell carcinoma [16]. High TUG1 expression levels are associated with chemotherapy resistance and had a poor prognosis in esophageal squamous cell carcinoma [17]. Long non-coding RNA SPRY4-IT1 is significantly up-regulated in esophageal squamous cell carcinoma and associated with poor prognosis [18].
In the study, our results showed that SNGH1 was up-regulated in ESCC tissues compared with adjacent normal tissues. To assess the association between higher SNGH1 expression or lower SNHG1 expression and overall survival time, we applied the Kaplan-Meier method and log-rank test according to clinical follow-up analysis. The results demonstrated that higher SNHG1 expression levels group in patients had a poor overall survival time, compared with lower SNHG1 expression levels group. MTT cell proliferation and transwell cell invasion assays showed that knockdown of SNHG1 inhibited cell proliferation and invasion ability. LncRNA SNHG1 had been found to participate in several tumor development, such as, SNHG1 expression was significantly up-regulated in lung cancer cells and promotes cell proliferation in non small cell lung cancer [19]. LncRNA SNHG1 exacerbated HCC cell proliferation, invasion, and migration in vitro through the inhibition of miR-195 [20]. Up-regulated lncRNA SNHG1 contributes to progression of non-small cell lung cancer through inhibition of miR-101-3p and activation of Wnt/
Epithelial-mesenchymal transition (EMT) plays a key role in ESCC invasion and metastasis. In the study, we examined the expression of the EMT-related proteins E-cadherin, N-cadherin and vimentin in ESCC cells. The results showed that silencing SNHG1 up-regulated the E-cadherin expression level and down-regulated the N-cadherin and Vimentin expression levels. In previous report, some lncRNAs had been found to be involved in EMT process. Up-regulation of long non-coding RNA SPRY4-IT1 promotes metastasis of esophageal squamous cell carcinoma via induction of epithelial-mesenchymal transition [22]. Long noncoding RNA RP11-766N7.4 functions as a tumor suppressor by regulating epithelial-mesenchymal transition in esophageal squamous cell carcinoma [23]. Our results showed that knockdown of lncRNA SNHG1 inhibited cell EMT process, which suggested that SNHG1 was significantly associated with ESCC cell invasion.
Notch signaling regulates cell-cell connection, cell polarity and motility during organ development. Recent studies had demonstrated that Notch signaling plays an important role in tumor initiation and cross-talks with some transcriptional factors to promote cell proliferation, invasion and cell EMT process [24]. NOTCH pathway activating associated with promoting progression of esophageal squamous cell carcinoma [25]. In the study, our results showed that Notch signaling pathway core protein Notch1 and Hes-1 were down-regulated after knockdown of SNHG1 in ESCC cells. The results suggested SNHG1 knockdown inactivated the Notch signaling pathway via affecting Notch1 and Hes-1 expression.
In summary, our results showed that SNHG1 was higher expression in ESCC and increased SNHG1 expression levels showed a poor prognosis. Furthermore, SNHG1 knockdown inhibited cell proliferation, invasion and EMT process. Moreover, SNHG1 knockdown inactivated the Notch signaling pathway. Therefore, these results indicated that lncRNA SNHG1 may be a potential predictor of prognosis in ESCC patients and a novel target for ESCC treatment.
Footnotes
Acknowledgments
Not applicable.
