Abstract
This article has been retracted, and the online PDF replaced with this retraction notice.
Introduction
Globally, gastric cancer is the fourth most common cancer, with a high mortality rate [1]. About 70% of gastric cancer cases and deaths are seen in developing countries, of which 50% is seen in Eastern Asia (China and Japan) [2]. Since there are no early signs of gastric cancer, most of the cases are diagnosed at advanced stages, and treatment failure is very high in such cases. Gastric cancer is a complex disease involving oncogenes or tumor suppressors, which are responsible for the initiation and progression of the disease [3]. Cell invasion and metastasis are the primary reasons for the high mortality among gastric cancer patients. Epithelial-mesenchymal transition (EMT) plays an important role in cancer progression and metastasis [4, 5]. During the EMT process, cancer cells lose their epithelial properties and gain mesenchymal properties, which leads to increase in migration and invasiveness [6]. However, the mechanism underlying these functions is still not clearly understood.
MicroRNAs (miRNAs), a group of highly conserved small non-coding RNAs, regulate gene expression post-transcriptionally by partial binding (complete or incomplete complementarity) to the messenger RNA of the target gene 3’UTR to lead to either mRNA degradation or repression of translation [7]. MiRNAs are involved in the development of several types of cancer, including gastric cancer [8]. Yang et al. reported that miR-214 could regulate the proliferation, migration and invasion of gastric cancer cells [9]. Song et al. reported that Lin-28 could affect the cell cycle and apoptosis of gastric cancer cells [10]. Several studies have reported that a few miRNAs are down-regulated in gastric cancer [11, 12]. Such down-regulated miRNAs included miR-375 [13] and miR-195-5p [14]. In contrast, several studies reported increased circulating miRNAs in gastric cancer, such as miR-21 [15], miR-221 [16], miR-378 [17], miR-451 and miR-486 [18]. In the present study, we investigated the role of miR-937 in gastric cancer and its underlying mechanism.
Forkhead box (FOX) transcription factor is a large evolutionarily conserved family of transcriptional regulators [19]. FOX family is reported to be involved in cancer progression and metastasis. FOX family acts as both oncogenes and tumor suppressors via a variety of mechanisms [20, 21]. Roles of FOX family members, such as FoxM1c [22], FoxC2 [23], FOXA1 [24] and FOXO3a [21], have been reported in gastric cancer, but their role in the disease progression still remains unclear. In the present study, we also investigated the role of FOXL2 in gastric cancer and its potential underlying mechanism.
Materials and methods
Cell culture
Human gastric cancer cell lines, BGC-823, SGC-7901 and HGC-27, and the gastric mucosal cell line GES were purchased from the Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). All the cell lines were maintained in RPMI-1640 medium (Gibco, Garlsbad, CA, USA), supplemented with 10% fetal bovine serum (FBS, Invitrogen, USA), 2 mM glutamine, 100
Tissue specimens
Fifteen human gastric cancer tissues and normal tissues were obtained from the Affiliated Traditional Chinese Medicine Hospital of Xinjiang Medical University, with the multiple patients’ informed consent. The approval was granted by the Ethics Committee of the Affiliated Traditional Chinese Medicine Hospital of Xinjiang Medical University. All tissue stages of cancer were confirmed by pathology and immunohistochemistry, and samples were collected and frozen in liquid nitrogen, and stored at 80
Transfection
The cells were plated in a 48-well or 24-well plate 24 hours before transfection. Then, BGC-823 and SGC-7901 cells were transfected with miR-937 mimic, ASO-miR-937 and their corresponding controls. For the analysis FOXL2 functions, the pcDNA3.1 vector (Sangon Biotech, Shanghai, China) was used to construct a FOXL2 overexpression plasmid and empty pcDNA3.1 plasmid was used as a negative control. Also, small interfering RNA (siRNA) plasmid was used to direct knockdown FOXL2, and its non-targeting sequence (si-NC) as a negative control. After this, Lipofectamine 2000 reagent was used for transfections according to the manufacturer’s protocol (Invitrogen, Carlsbad, CA, USA). The plasmids were used at a concentration of 5 ng/L.
Luciferase reporter assay
Dual-luciferase activity assays were performed as previously described [25]. PCR was used to amplify the full-length 3’-UTR segments of FOXL2 mRNA containing the miR-937 binding site and inserted into the Xba1-site of pGL3 vector (Promega, WI) and named pGL3- FOXL2. The pGL3-FOXL2-mut reporter construct with point mutations in the seed sequence was synthesized using a site-directed mutagenesis kit (Stratagene, CA). Then, 1
qRT-PCR
Large and small RNAs from the tissue were isolated with mirVana miRNA Isolation Kit (Ambion, Austin, TX, USA) according to the manufacturer’s protocol. For RNA integrity assessment, part of an RNA sample was used for concentration and purity measurement (by A260 and A280 spectrophotometry), and another part of the sample was run on a 1.5% denaturing agarose gel stained with ethidium bromide. A ratio of the absorbance at 260 and 280 nm (A260/280) of 1.8–2 was accepted. The sharp, clear 28S and 18S rRNA bands and the 2:1 ratio (28S:18S) were good indicators that the RNA was completely intact.
Five
To detect the mature miR-937 levels, a stem-loop RT-PCR assay was performed using specific RT and PCR primers; U6 snRNA was used as an endogenous control. Five
Western blotting
Cells were washed twice with Hanks’ balanced salt solution and lysed directly in lysis buffer (50 mM Tris-HCl, pH 8.0, 1% NP-40, 10 mM NaCl, 2 mM EDTA, 5 mg/mL leupeptin, 2 mg/mL aprotinin, 2 mg/mL pepstatin, 1 mM DTT, 0.1% SDS and 1 mM phenylmethylsulfonyl fluoride). The protein concentrations of the lysates were measured using a Bradford protein assay kit (Bio-Rad, US). Equivalent amounts of protein were separated by 10% SDS PAGE and then transferred to nitrocellulose membranes by electroblotting. The membranes were blocked with 5% BSA in TBST (10 mM Tris-HCl, pH 8.0, 150 mM NaCl, and 0.05% Tween 20) for 1 hour, and then the membrane was immunoblotted overnight at 4
Cell viability assay
At 24 hours after transfection, cells were seeded in 96-well plates at either 6
MiR-937 expression levels in gastric cancer cell lines. (A) The expression levels of miR-937 in tumor tissue and normal tissue were analyzed by qRT-PCR. (B) Comparison of expression of miR-937 in three gastric cancer cell lines (BGC-823, SGC-7901 and HGC-27) and one human immortalized gastric epithelial cell line (GES-1). 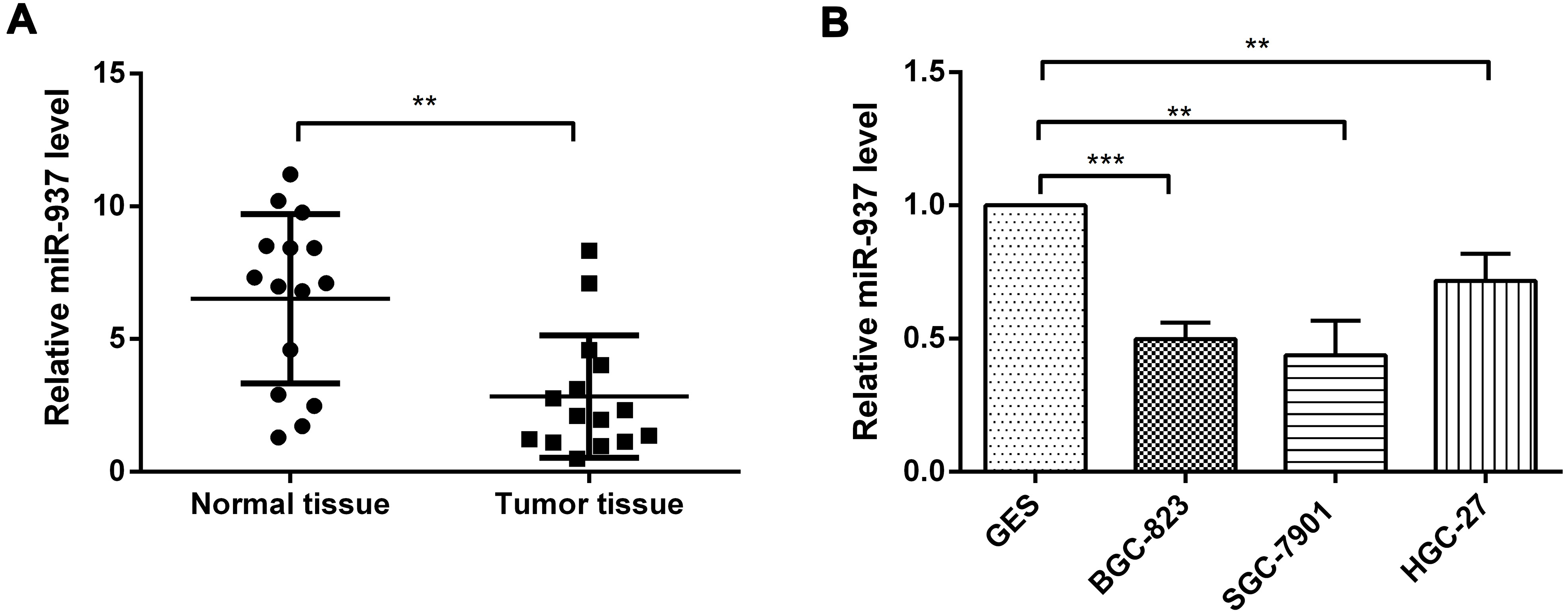
Cells were counted and seeded (300 cells/well) in 12-well plates (in triplicate). Fresh culture medium was replaced every three days. Colonies were counted only if they contained more than 50 cells, and the number of colonies was counted either 12 days after seeding. The cells were stained using crystal violet. Colony formation was calculated by the colony formation number.
Flow cytometry analysis of cell apoptosis
To measure apoptosis, cells were collected, washed with PBS and stained with fluorescein isothiocyanate-labelled annexin V (Invitrogen) and propidium iodide, followed by flow cytometry analysis.
Cell migration and invasion
Cell migration and invasion were assessed using Boyden chambers (Millipore, Billerica, MA, USA). In brief, 1
Statistical analysis
Data are expressed as mean
Results
MiR-937 expression levels in gastric cancer cells
To determine the expression levels of miR-937 in tumor tissue, normal tissue and in the cancer cells, qRT-PCR was performed. As shown in Fig. 1A, the expression of miR-937 was significantly lower in the tumor tissue than that in the normal tissue (
Overexpression of miR-937 inhibits cell proliferation and promotes cell apoptosis. BGC-823 and SGC-7901 cells were transfected with control, miR-937 mimic, ASO-NC or ASO-miR-937. (A) The effect of miR-937 on the cell viability was measured by MTT assay at 24, 48 and 72 hours after transfection (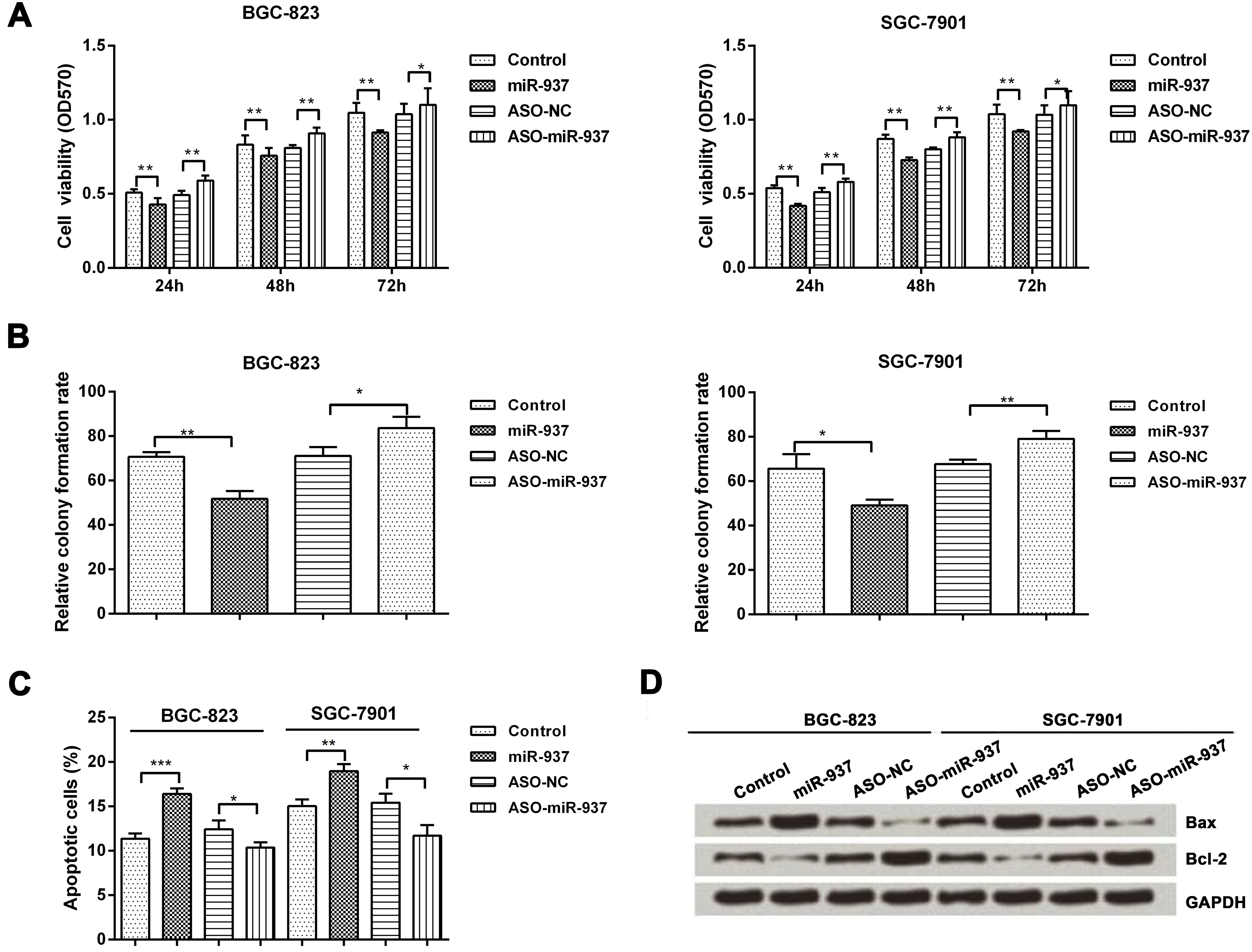
We then determined the effect of the altered expression of miR-937 on the cell viability, proliferation and apoptosis in BGC-823 and SGC-7901 cells. Cell viability was measured by MTT assay at 24, 48 and 72 hours after being seeded and then determined by measuring the optical density at 570 nm. Viable of cells (BGC-823 and SGC-7901) transfected with miR-937 mimic was significantly lower than that of cells transfected with control at 24, 48, and 72 hour (
Next, we determined the effect of miR-937 on the proliferation of BGC-823 and SGC-7901 cells using colony formation assay. The relative colony formation rate was significantly lower in the cells transfected with miR-937 mimic than that in the cells transfected with control (
We then measured cell apoptosis by flow cytometry (FCM) in BGC-823 and SGC-7901 cells. As shown in Fig. 2C, apoptotic cell rate was significantly higher in the cells transfected with miR-937 mimic than that in the cells transfected with control (
MiR-937 decreases cell migration and cell invasion by inhibition of EMT. BGC-823 and SGC-7901 cells were transfected with control, miR-937 mimic, ASO-NC or ASO-miR-937. (A–B) Transwell assay measured cell migration and invasion. (C) qRT-PCR was used to measure the mRNA expressions and (D) western blotting was used to measure the protein expressions of E-cadherin, vimentin, and a-SMA in the BGC-823 and SGC-7901 cells (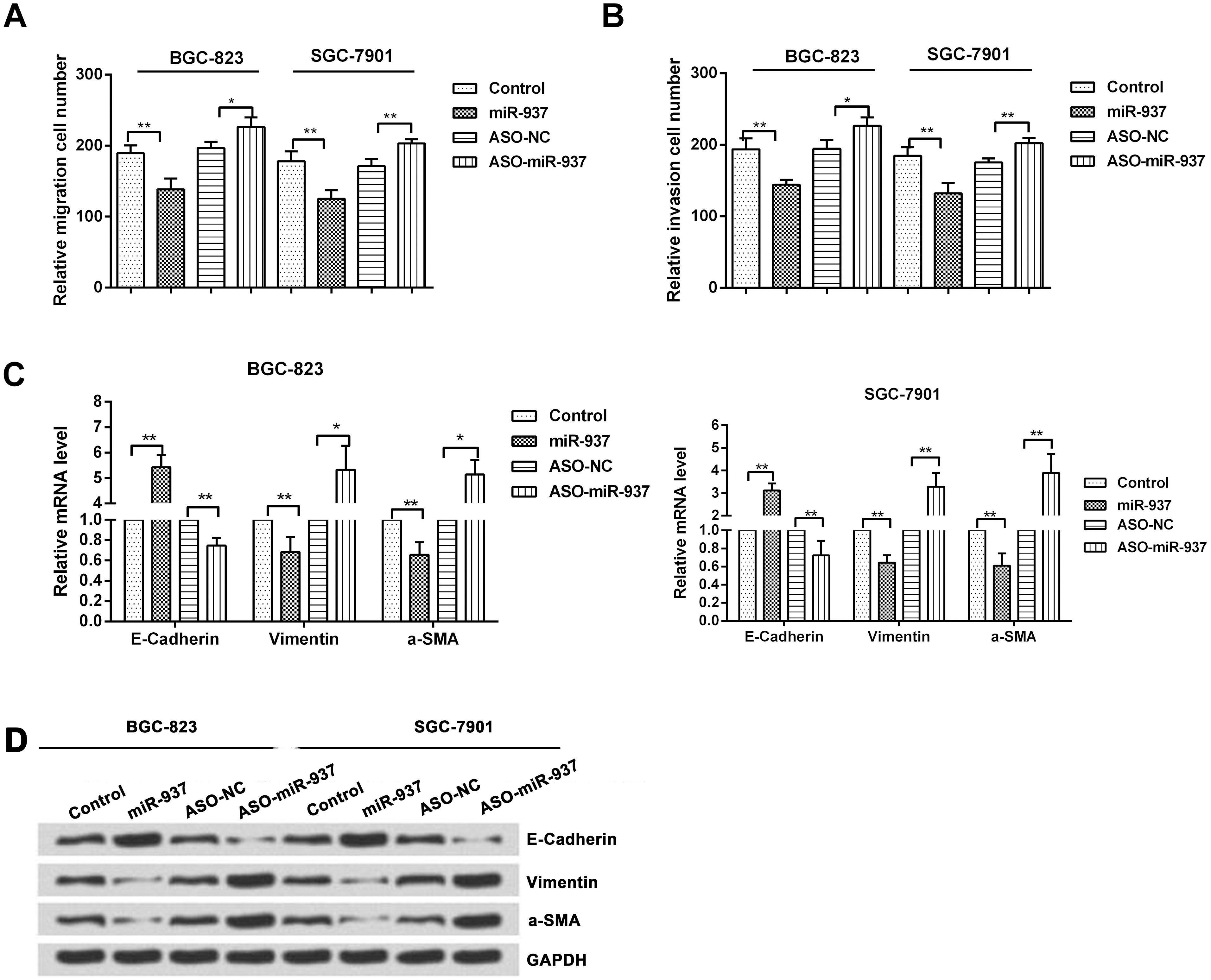
Together, these results indicate that overexpression of miR-937 inhibits cell proliferation, and promotes apoptosis in gastric cancer cells.
Since migration is one of the essential factors for metastasis, we performed a Transwell assay to determine the influence of overexpression of miR-937 on cell migration and invasion. We observed that the migration rate was significantly decreased in miR-937 overexpression group when compared with the rates in the control group in BGC-823 (
Next, we observed that the relative invasion cell number was significantly lower in miR-937 overexpression group when compared with the rates in the control group in BGC-823 (
We then measured the expression of EMT-associated proteins (E-cadherin and vimentin) using qRT-PCR and western blotting analysis. The relative mRNA levels of E-cadherin were significantly increased in BGC-823 (
These findings were further confirmed by western blotting analysis and it was seen that levels of E-cadherin were significantly increased in BGC-823 and SGC-7901 cells with miR-937 overexpression when compared to control cells. And the relative mRNA levels of vimentin and a-SMA were significantly decreased in these cells with miR-937 overexpression when compared to control cells. On the contrary, the levels of E-cadherin were decreased and levels of vimentin and a-SMA were increased in the cells transfected with miR-937 inhibitor when compared to control cells (Fig. 3D).
These results indicate that overexpression of miR-937 decreases cell migration and invasion by inhibition of EMT.
FOXL2 is a target gene of miR-937 and negatively regulated by miR-937
To identify a target gene of miR-937, luciferase assay was performed to validate the target sites in the FOXL2 3’UTR. There are two predicted miR-937 binding sites (323-329 and 649-655) in the 3’UTR of FOXL2 mRNA (Fig. 4A).
FOXL2 is a direct target of miR-937. Cells were transfected with miR-937 mimic and ASO-miR-937 to overexpress or suppress miR-937 expression. (A) The FOXL2 3’UTR has two putative miR-937 binding sites (323-329 and 649-655). (B) Luciferase assay was performed to measure relative luciferase activity of FOXL2 (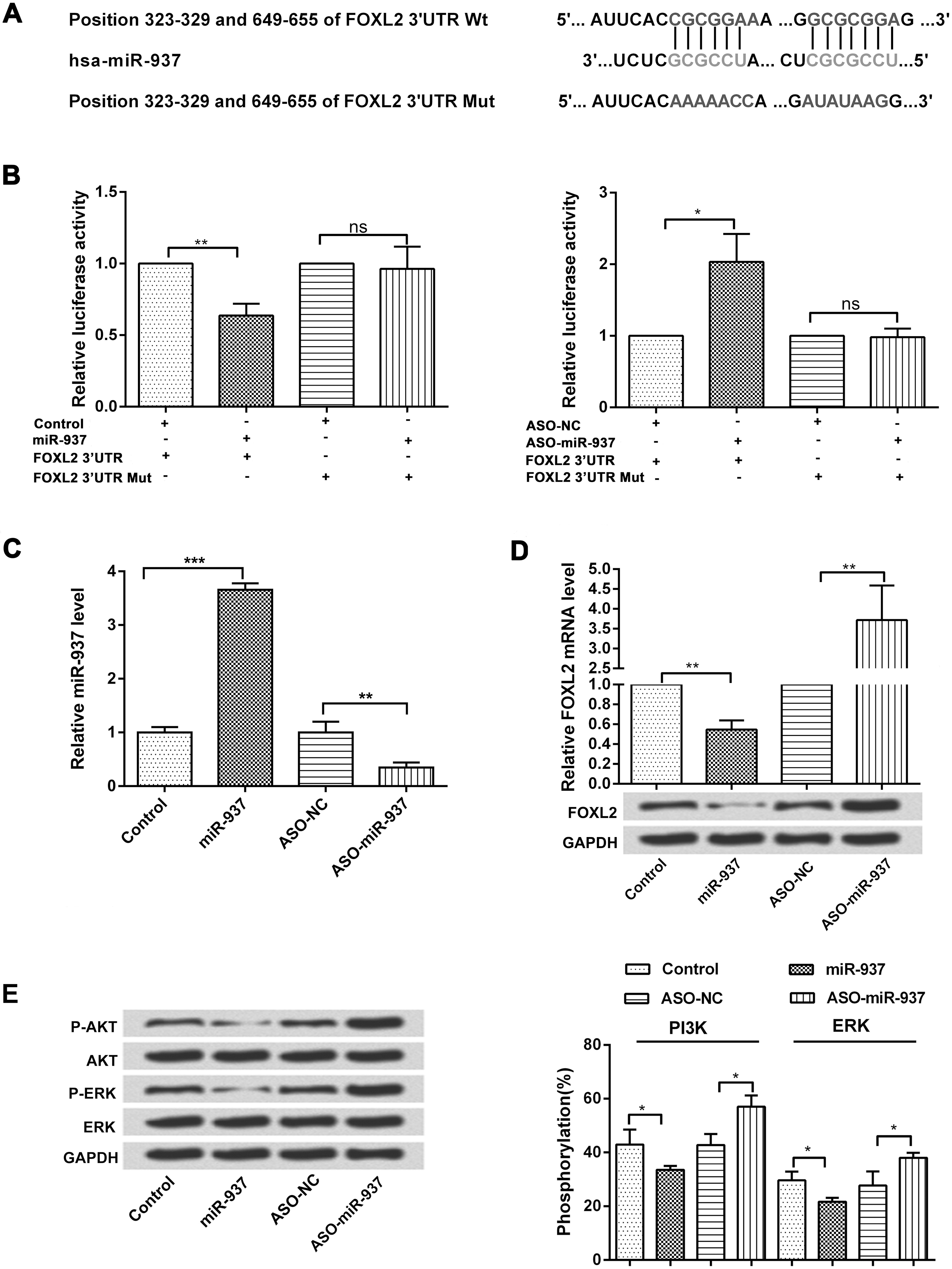
Luciferase reporter assay showed that the relative luciferase activity of FOXL2 was significantly decreased by miR-937 overexpression in BGC-823 (
Subsequently, we further examined the transfection efficiency of miR-937 mimic and ASO-miR-937 in both the BGC-823 and SGC-7901 cell lines by using qRT-PCR. As demonstrated in Fig. 4C, the level of miR-937 was significantly increased by miR-937 overexpression, while reduced by miR-937 inhibitor (
To determine whether miR-937 regulates PI3K/AKT pathway, protein levels of p-AKT, AKT, p-ERK and ERK1/2 were examined by western blotting analysis. As shown in Fig. 4E, overexpression of miR-937 decreased the expression of p-AKT and p-ERK, whereas suppression of miR-937 showed opposite results. To confirm this finding, we measured the rate of phosphorylation of PI3K and AKT in these cells. In the presence of miR-937, there was a significant decrease in the phosphorylation rate of PI3K and ERK compared to the control (
FOXL2 promotes cell proliferation and metastasis. Cells were transfected with FOXL2 overexpression vector, si-FOXL2 and corresponding controls. (A) The protein level of FOXL2 was detected by western blotting analysis. (B) MTT assay was performed 24, 48 and 72 hours after transfection to determine the effect of FOXL2 overexpression on cell viability (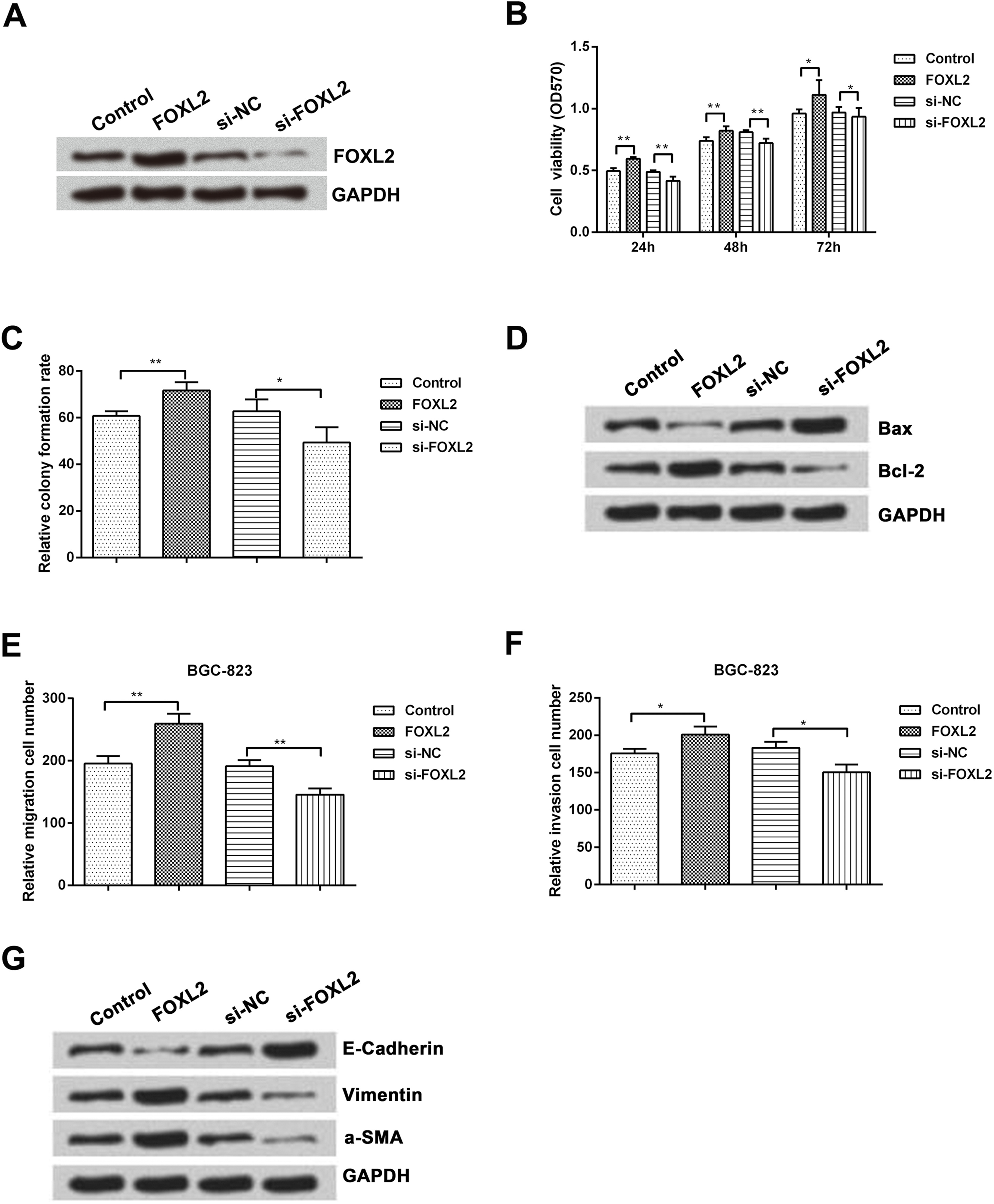
Since miR-937 directly regulates the expression of FOXL2 and suppresses cell growth and migration, we next examined functions of FOXL2 in gastric cancer. BGC-823 and SGC-7901 cells were firstly transfected with FOXL2 and si-FOXL2 to overexpress or suppress FOXL2 expression. As expected, the expression of FOXL2 was obviously up-regulated by overexpression of FOXL2, while was down-regulated by suppression of FOXL2 (Fig. 5A). After transfection, the effects of FOXL2 on cell viability were analyzed by MTT. As shown in Fig. 5B, FOXL2 overexpression significantly increased cell viability at all the time points as compared to the control group (
Next, we performed western blotting analysis to determine the effect of FOXL2 on apoptosis-related proteins. As shown in Fig. 5D, FOXL2 overexpression decreased the level of Bax but increased the level of Bcl-2. Opposite results were seen when FOXL2 was knockdown and it increased the level of Bax but decreased the level of Bcl-2. Furthermore, we also performed transwell assays to determine effect of FOXL2 on cell migration and invasion. The results showed that FOXL2 overexpression significantly increased cell migration and invasion compared to the control (
We then measured the expression of EMT-associated proteins (E-cadherin and vimentin) using qRT-PCR and western blotting analysis. The results showed that FOXL2 overexpression could decrease the expression of E-cadherin but increased the expression of vimentin; suppression of FOXL2 showed opposite results (Fig. 5G).
These results indicate that FOXL2 promotes cell proliferation and metastasis of gastric cancer cells.
Discussion
Recent data suggest that miRNAs are associated with gastric cancer and contribute to carcinogenesis due to abnormality in their expression which in turn affects cell proliferation, apoptosis, motility, and invasion [26]. Therefore, these miRNAs may serve as a biomarker for predicting the prognosis of cancer, including gastric cancer [26]. Even though several studies are conducted, the underlying mechanism of gastric cancer remains unclear. But there are increasing evidences that it is governed by complex interactions between multiple pro- and anti-oncogenic signaling pathways [27]. Therefore, it is necessary to identify specific miRNAs and their targets which can provide valuable insight for the diagnosis and therapy of patients with gastric cancer.
In the present study, we found that the expression of miR-937 was significantly decreased in gastric cancer tumors and cell lines. Moreover, overexpression of miR-937 decreased cell proliferation, migration, and invasion of the gastric cancer cells. These results highlight the significance of miR-937 as a tumor suppressor in gastric cancer.
In the present study, miR-937 inhibited cell invasion by increasing the expression of E-cadherin and decreasing the expression of vimentin. This finding indicates that miR-937 inhibits EMT process. EMT is a key process in cancer metastasis, during which, epithelial cells lose cell-cell junctions and polarity, leading to a more migratory, mesenchymal cell phenotype [28]. EMT is associated with loss of expression of epithelial cell markers (E-cadherin) and increased expression of mesenchymal genes (vimentin) [29, 30]. Loss of E-cadherin expression occurs during the progression of many types of cancer [6, 31]. In a previous study, E-cadherin down-regulation in gastric cancer increased cell survival and metastasis through Wnt/
It is well known that miRNAs regulate gene expression [36] and therefore it is necessary to identify specific miRNA target genes to understand the mechanism involved in the progression of cancer. It has been reported that miRNAs regulate their target genes by binding to the mRNA 3’UTR [37]. Based on functional knowledge, we chose FOXL2 as a target to predict the miR-937 target gene. We identified two sites as the miR-937 binding sites in FOXL2 3’UTR which are responsive to miR-937 overexpression. These results suggested that miR-937 regulates FOXL2 expression by directly binding to the FOXL2 3’UTR. We also observed that the relative luciferase activity of FOXL2 decreased by miR-937 overexpression but increased by miR-937 suppression. We also measured the effects of aberrant expression of miR-937 on expression levels of FOXL2 and found that miR-937 negatively regulated the expression of FOXL2. More importantly, we found that FOXL2 could increase the cell proliferation, migration and invasion but decrease the cell apoptosis. FOX family of transcription factors are reported to be involved in various cancer progression and metastasis [20] and they act as both oncogenes and tumor suppressors via a variety of mechanisms. For example, FoxM1c induces EMT by activating the uPA system/Slug pathway [22]. FoxC2 promotes EMT and colorectal cancer metastasis through the Akt/GSK-3
We have reported several novel findings in this study. We report for the first time that miR-937 overexpression inhibits gastric cancer cell growth and metastasis via inactivation of the PI3K/AKT signaling pathway. Moreover, this is the first study to report the role of FOXL2 in the regulation of cell proliferation, migration and invasion in the progression of gastric cancer. Further studies are required to elucidate the role of FOXL2 in the carcinogenic mechanism by gene amplification, chromosomal translocation, or retroviral integration similar to other FOX members.
In conclusion, our study reports the following findings that miR-937 is expressed at low levels in gastric cancer tissues and cell lines; miR-937 inhibits cell proliferation and promotes apoptosis in gastric cancer cells; miR-937 decreases cell migration and invasion by inhibition of EMT; FOXL2 is a target gene of miR-937; miR-937 inhibits PI3K/AKT signal pathway; and FOXL2 promotes cell proliferation, migration and invasion but decrease the cell apoptosis. These results suggest that miR-937 may be a potential target for the treatment of gastric cancer.
Footnotes
Conflict of interest
The authors have no conflict of interest to report.
