Abstract
Background
Gastric cancer is the 4th most common and 3rd deadliest cancer worldwide. Research has shown that PDCD2-like (PDCD2L) is elevated in several tumors.
Objective
To explore the relationship between PDCD2L expression and gastric cancer prognosis and its function in gastric cancer.
Methods
Immunohistochemical staining and immunoblotting were performed to examine the expression of PDCD2L. The effect of PDCD2L on gastric cancer cells were evaluated by a series of in vitro cellular function experiments and in vivo proliferation experiments.
Results
PDCD2L was found to be overexpressed in gastric cancer tissues compared to non-tumor tissues, and its higher levels were associated with worse prognosis. In vitro experiments showed that reducing PDCD2L expression in gastric cancer cells led to decreased proliferation, migration, and invasion, with a similar effect observed in animal models. Knockdown of PDCD2L also resulted in lower expression of cell cycle- and motility-related proteins, while upregulation of PDCD2L had the opposite effect. Additionally, NGS analysis revealed that PDCD2L knockdown reduced the expression of RFX1, a gene linked to cell proliferation and migration, suggesting that PDCD2L and RFX1 together promote cancer progression.
Conclusion
PDCD2L could serve as a biomarker for gastric cancer prognosis and a potential therapeutic target.
Introduction
The mortality rate of gastric cancer ranks third among all types of cancer. 1 The treatment of gastric cancer patients remains highly challenging due to limited understanding of the disease's pathological processes and the lack of targeted gene therapies.2,3 However, advancements in molecular biology techniques have made it possible to search for factors related to gastric cancer, including oncogenes and tumor suppressor genes, as potential new biomarkers for gastric cancer. 4 Some molecules with abnormal expression, such as circRNA CDR1as, programmed cell death-1 (PD-1), family with sequence similarity 64, member A (FAM64A), cell division cycle protein 45 (CDC45), and signal transducer and activator of transcription 3 (STAT3), can serve as prognostic biomarkers for gastric cancer.5–9
Since the discovery of programmed cell death domain 1 (PDCD1, also known as PD-1) in 1992, a diverse family of homologous genes has been identified. 10 This homologous gene family is now known as the PDCD family. Until now, in human genome, 16 members of the PDCD gene family have been characterized. The 16 members of the PDCD gene family share some functions, but they also have distinctly different roles. Previous research has shown that PDCD1 is crucial for regulating immune responses. 11 PDCD5, PDCD6 and PDCD10 have been found to be involved in protein synthesis, cell proliferation, cell cycle progression and apoptosis.12–17 Recently, the PDCD family members have gained widespread attention due to their roles in cancer progression. The expression and activity of different PDCD genes are altered in human cancers, with a specific PDCD gene potentially acting as an oncogene or a tumor suppressor gene, often depending on the tissue or tumor type.
PDCD2 is a highly conserved protein expressed in various tissues. 18 The PDCD2 gene was initially identified in rats and is associated with programmed cell death. 19 Initially, PDCD2 was thought to promote stem cell activity and tissue remodeling by inducing. 20 Previous studies have also suggested that PDCD2 may have important functions in cell proliferation and spreading. 21 Moreover, increasing evidence indicates that PDCD2 is involved in cancer development. However, its specific role in cancer progression remains controversial. For instance, some studies suggest that PDCD2 acts as a tumor suppressor in osteosarcoma tumorigenesis. 20 It is also shown that PDCD2 can suppress EMT in hepatoma cells. 22 Conversely, PDCD2 is found to be overexpressed in human acute leukemia cells, indicating that it may promote leukemia progression. 23 PDCD2L (PDCD2-like) shares the same C-terminal domain as PDCD2. However, little is known about PDCD2L, with only seventeen papers published on this protein so far. Data from the Oncomine database show that PDCD2L is overexpressed in various cancers, including colorectal cancer, kidney cancer, lung cancer, ovarian cancer, and gastric cancer. 24 To our knowledge, the role of PDCD2L in gastric cancer are still unknown.
There are few studies on the relationship between PDCD proteins and prognosis. According to research conducted by Aydin et al., 13 genes, including PDCD2, can categorize pituitary adenoma patients into two groups: those with invasive and non-invasive pituitary adenomas, which have different prognoses. 25 PDCD4 was found to have reduced expression in colorectal cancer and can predict poorer patient survival rates. 26 Zhao et al. discovered that PDCD5 inhibits osteosarcoma cell metastasis by affecting the TGF-β1/Smad signaling pathway and is positively associated with good prognosis. 27 Overexpression of PDCD6 in bladder urothelial carcinoma patients has been linked to favorable prognosis. 28 Abo Elwafa and his colleagues demonstrated that the overexpression of PDCD7 is a prognostic marker for acute myeloid leukemia. 29 Although the study by Gao et al. indicated that the overexpression of PDCD2L was not a prognostic marker for gastric cancer. 24 In this study, an independent gastric cancer cohort was enrolled to investigate the amount of PDCD2L in normal and gastric cancer tissues and cells, to explore the role of PDCD2L in the pathological process of gastric cancer, and to assess the possibility of PDCD2L to be a chemotherapeutic target and prognostic biomarker for gastric cancer.
Materials and methods
Cell culture
The human primary stomach epithelial cells (cat. No. H-6039; CellBiologics, Chicago, IL, USA) were cultured in Epithelial Cell Medium (CellBiologics). The gastric cancer cell lines AGS, NCI-N87, TSGH 9201 (cat. No. 60102, 60217, 60146; the Bioresource Collection and Research Center), SK-GT-2, HGC-27 (cat. No. 11012008, 94042256; the European Collection of Cell Cultures, Salisbury, UK), and 23132/87 (cat No. CSC-C0324; Creative Bioarray, Shirley, NY, USA), were maintained as described in our previous publication. 30 All media were supplemented with 10% fetal bovine serum (cat. No. SH30087; Hyclon, Wilmington, DE, USA) and antibiotics (cat. No. 15240; Thermo, Grand Island, NY, USA). Each cell line was authenticated by the respective provider, and we verified their identity using the ICLAC database to ensure no misidentification.
Patients and tissue specimens
One hundred and twenty-seven gastric tissue samples (including tumor and adjacent non-tumor tissues) were acquired from gastric cancer patients who underwent surgical resection between 1998 and 2011 at Taipei Medical University Wan Fang Hospital. This retrospective study adhered to the principles of the Declaration of Helsinki and received approval from the Institutional Review Board of Chi Mei Medical Center (Approval No. 10812-006). Written informed consent was obtained from each patient prior to participation.
Antibodies
The antibodies for PDCD2L (Biorbyt, Cambridge, UK), cyclin B, cyclin E, β-catenin, vimentin, Snail (Cell Signaling, Danvers, MA, USA), RFX1 (Origene, Rockville, MD, USA) and GAPDH (Millipore, Temecula, CA, USA) were purchased.
Protein extraction and immunoblotting
The materials and method for protein extraction and immjunoblotting have been described in detail in our previous publication. 30 Briefly, total tissue and cell lysates were extracted with RIPA Buffer (Thermo). Denatured protein samples were subjected to SDS-PAGE and transferred to nitrocellulose membranes. Blocked blots were incubated at 4°C overnight with primary antibodies. GAPDH was used as an internal control for equal protein loading. After incubation with secondary antibodies conjugated with peroxidase (Sigma), enhanced chemiluminescence reagents (Thermo) were used to visualize the targeted proteins. Image processing was performed using GeneTools software (Syngene, Cambridge, UK).
Immunohistochemistry
Immunohistochemistry, assessment and scoring of PDCD2L immunoreactivity were performed as previously described. 30 Briefly, paraffin-embedded sections were stained with primary antibodies at 4°C overnight. A standard peroxidase-conjugated streptavidin-biotin method was used to detect the immunoreactivity (Dako REAL EnVision Detection System; Dako, Carpinteria, CA, USA). The negative control consisted of the omission of the primary antibody and incubation with 1× phosphate buffer saline (Corning). Images were acquired with BX51 microscope (Olympus, Tokyo, Japan). PDCD2L immunoreactivity was assessed semiquantitatively and scored as follows: 0, no staining; 1, weak and focal staining in < 25% of the tissue; 2, moderate staining in 25%-50% of the tissue; and 3, strong staining in > 50% of the tissue. Sections with a score of 0 or 1 exhibited low expression of KLF16, and those with a score of 2 or 3 were defined as exhibiting high expression or overexpression of PDCD2L. Both clinical data collection and immunohistochemical analysis were independently carried out under blinded conditions.
shRNA treatment and transfection
The procedure for shRNA treatment and transfection have been described in detail in our previous publication. 30 Lentiviral vectors containing two PDCD2L-shRNA constructs (clone IDs: TRCN0000165015, TRCN0000160911) and a control vector (clone ID: pLKO_TRC025), all obtained from the National RNAi Core Facility (Taipei, Taiwan). Human PDCD2L cDNA open reading frame or empty vectors were purchased from OriGene (Rockville, MD, USA). Stable clones resistant to puromycin (Thermo) and G418 (Sigma) were selected. The effects of shRNA treatment and transfection were assessed through immunoblotting.
Cell analyses
Colony formation assay, cell cycle analysis, wound-healing assay, and cell invasion assay were performed, following the procedure outlined in our previous study. 30
Animals
Male athymic 4-week-old BALB/c nude mice were sourced from the National Laboratory Animal Center in Taiwan. All experimental procedures involving animals complied with institutional guidelines, and were approved by the Chi Mei Medical Center Animal Ethics Research Board (Approval No. 108120105).
Tumor cell inoculation for xenograft proliferation
Tumor cell inoculation and xenograft proliferation were conducted to investigate the effect of PDCD2L knockdown, following the method described in our previous study. 30 Briefly, for PDCD2L knockdown, animals were randomly divided into 2 groups (5 mice/group) for tumor cell inoculation. The PDCD2L knockdown and control 23132/87 cells (5 × 106) were mixed in a 200 μL Matrix gel (Discovery Labware, Two Oak Park, MA, USA) and injected subcutaneously on the right hind flank. The xenograft proliferation experiment is expected to last for 6 weeks. However, if tumor growth exceeds 10% of the animal's original body weight, the average tumor diameter exceeds 2 cm, or if the tumor metastasizes or grows rapidly to the point of ulceration, causing infection or necrosis, the animals will be euthanized early to minimize discomfort and distress. The authors confirm that the maximal tumor size/burden is not exceeded.
Statistical analysis
The correlation between PDCD2L levels and clinicopathologic features was assessed using the χ² test. Kaplan-Meier and log-rank tests were utilized to compare disease-free and overall survival, generating survival curves based on high and low PDCD2L immunohistochemical scores. Multivariate survival analysis was conducted for all features identified as significant in univariate survival analysis, using the Cox proportional hazards model. All data analyses were performed with SPSS version 27.0 (IBM, Armonk, NY, USA). The difference in tumor weights between scrambled control and 23132/87 xenografts was analyzed using a paired t-test. All statistical tests were two-sided, and P < 0.05 was considered statistically significant.
Results
PDCD2L is overexpressed in gastric cancer
To explore the potential role of PDCD2L in gastric carcinogenesis, we initially wanted to determine whether PDCD2L expression is abnormal in gastric cancer tissues and cells. TCGA database was used to analyze the PDCD2L expression levels in patients with gastric cancer. The statistical results showed that PDCD2L expression in gastric cancer tissues was significantly higher than in adjacent normal tissues (Figure 1A). Subsequently, we investigated PDCD2L immunostaining in gastric tissues from 127 gastric cancer patients. Immunohistochemistry revealed that PDCD2L expression was higher in tumor tissues compared to non-tumor tissues (Figure 1B). Eight percent of patients had no PDCD2L expression in their tumor tissues (interpretation score of 0), 28% of patients had weak and localized PDCD2L expression in less than 25% of their tumor tissues (interpretation score of 1). Sixty-four percent of patients had higher or overexpression of PDCD2L in their tumor tissues (43% with an interpretation score of 2 and 21% with an interpretation score of 3). The PDCD2L protein level in gastric tissues was also examined and was up-regulated compared with non-tumor tissues (Figure 1C). Additionally, eight gastric cell lines were used to validate the immunohistochemistry results. Immunoblotting showed that compared to normal gastric cells (HPSEC), PDCD2L protein levels were significantly increased in six gastric cancer cell lines (AGS, NCI-N87, TSGH 9201, SK-GT-2, HGC −27, and 23132/87) (Figure 1D).
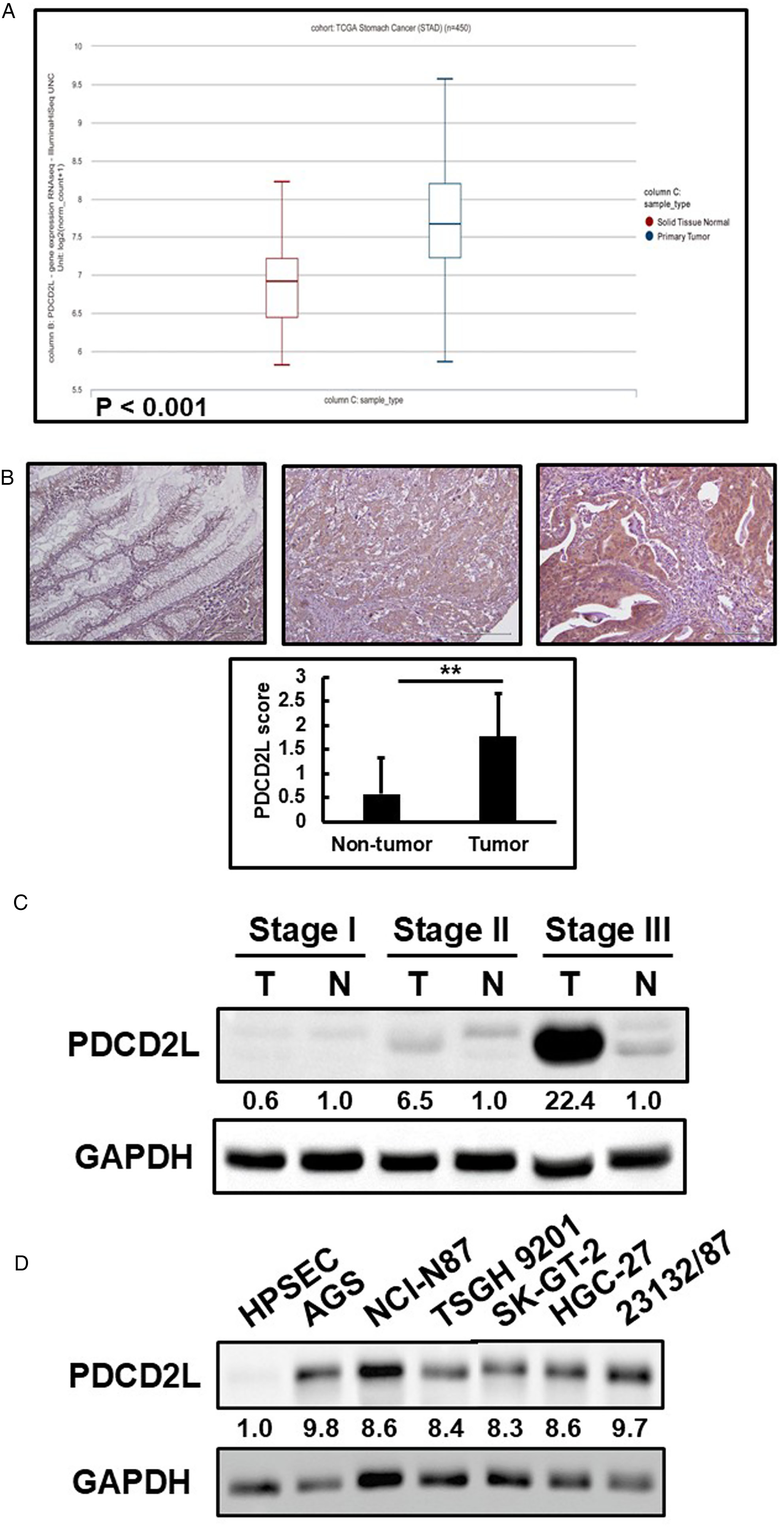
Expression levels of PDCD2L. (A) Expression levels of PDCD2L in normal and tumor tissues from TCGA database. (B) Gastric cancer analyzed by immunohistochemistry with an antibody against PDCD2L. Left panel shows a non-tumor tissue without PDCD2L expression; middle panel shows a tumor tissue with low PDCD2L expression; right panel shows a tumor tissue with high PDCD2L expression. Magnification: 200×. Lower histogram showed the PDCD2L score in non-tumor and tumor tissues (presented as the mean ± SD, **P < 0.05). (C-D) Endogenous PDCD2L protein expression was remarkably increased in gastric cancer tissues and cell lines.
The association between elevated PDCD2L expression and the clinicopathologic characteristics and survival outcomes of gastric cancer patients
The findings of elevated PDCD2L expression in gastric cancer tissues led us to explore the clinical significance of PDCD2L in gastric cancer further. First, we analyzed the correlation between PDCD2L expression and the clinicopathologic characteristics of gastric cancer. As shown in Table 1, PDCD2L expression was significantly positively correlated with patient age. While the expression of PDCD2L did not show a significant correlation with Lauren classification, depth of invasion, or vascular invasion, it was higher in cases of intestinal type gastric cancer, depth of invasion classified as T3 and T4, and in cases with vascular invasion.
Clinical characteristics of gastric cancer patients according to high or low PDCD2L expression
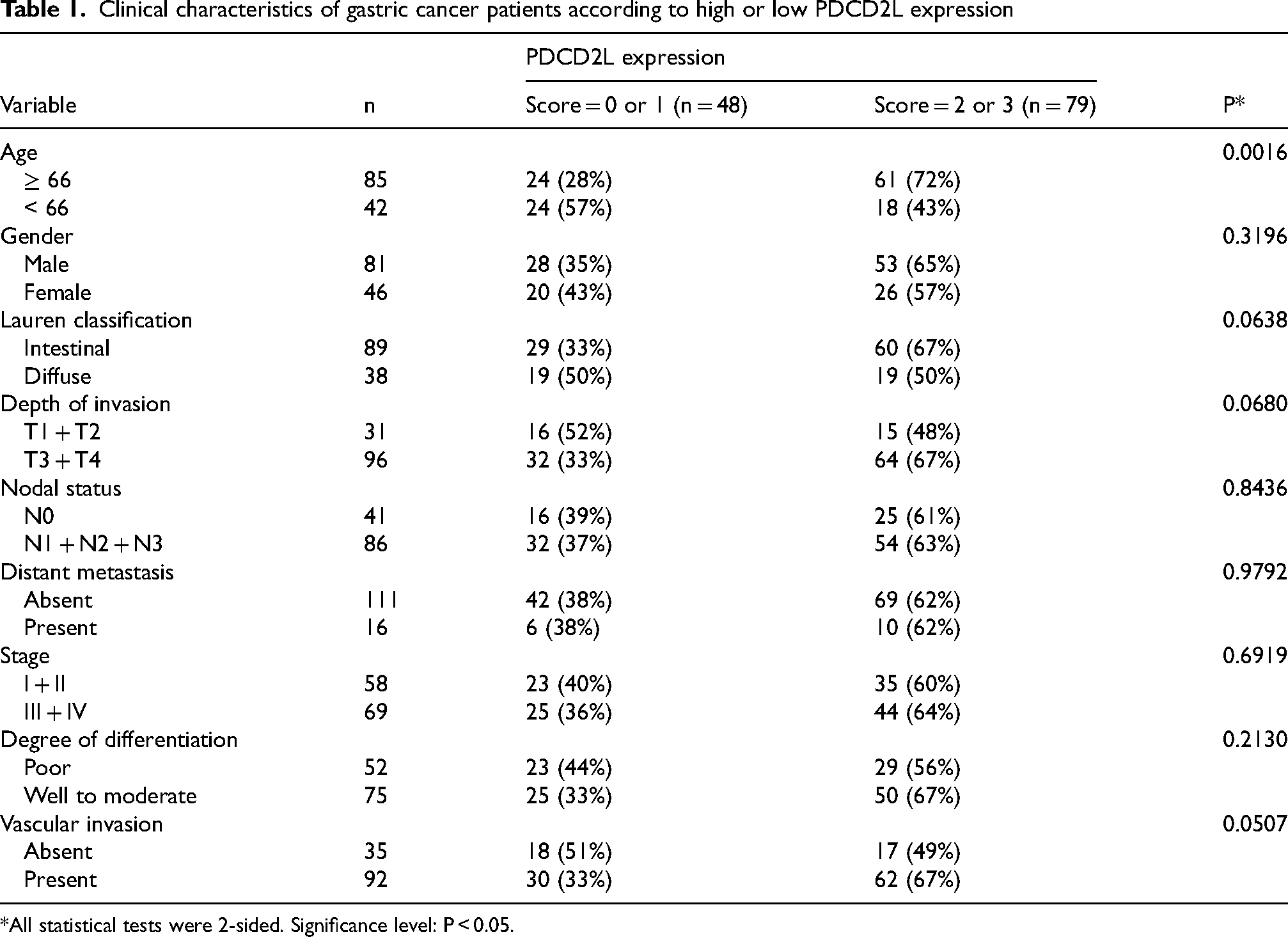
*All statistical tests were 2-sided. Significance level: P < 0.05.
Furthermore, Kaplan-Meier analysis with the log-rank test revealed a significant positive correlation between PDCD2L overexpression and reduced disease-free survival in patients. (Figure 2A, P = 0.032). At the fifth postoperative year, 12 patients with lower PDCD2L expression were at risk, with a disease-free survival rate of 0.659 (95% confidence interval [CI] between 0.586–0.732). In contrast, 5 patients with higher PDCD2L expression were at risk, with a disease-free survival rate of 0.401 (95% CI between 0.336–0.466).
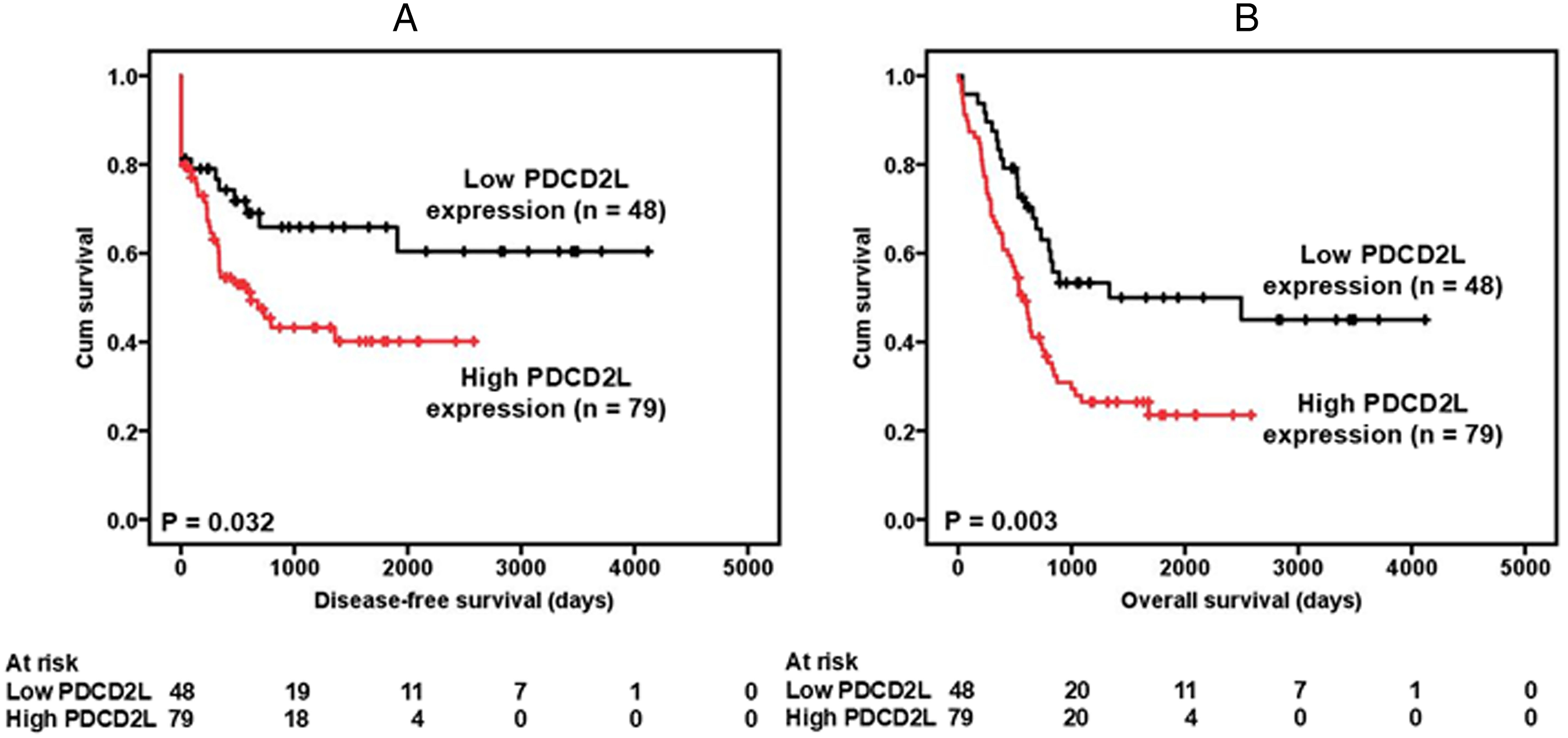
Survival analysis of gastric cancer patients stratified by PDCD2L immunoreactivity. Panel (A) shows the disease-free survival. Patients with high PDCD2L expression had a 5-year disease-free survival rate of 40.1% compared with 65.9% for patients with low PDCD2L expression. Panel (B) shows the overall survival. Patients with high PDCD2L expression had a 5-year disease-free survival rate of 23.5% compared with 50.0% for patients with low PDCD2L expression. All statistical tests were 2-sided. Significance level: P < 0.05.
A significant positive correlation was also observed between PDCD2L overexpression and poorer overall survival. (Figure 2B, P = 0.003). At the fifth postoperative year, 12 patients with lower PDCD2L expression were at risk, with an overall survival rate of 0.500 (95% CI between 0.422–0.578). In contrast, 5 patients with higher PDCD2L expression were at risk, with an overall survival rate of 0.235 (95% CI between 0.181–0.289).
Table 2 summarizes the univariate Cox regression analysis of the prognostic biomarkers and patient survival. PDCD2L overexpression, depth of invasion, nodal status, distant metastasis, stage, degree of differentiation, and vascular invasion were significantly correlated with disease-free survival.
Univariate and multivariate Cox regression analyses of prognostic biomarkers and survival in 127 gastric cancer patients
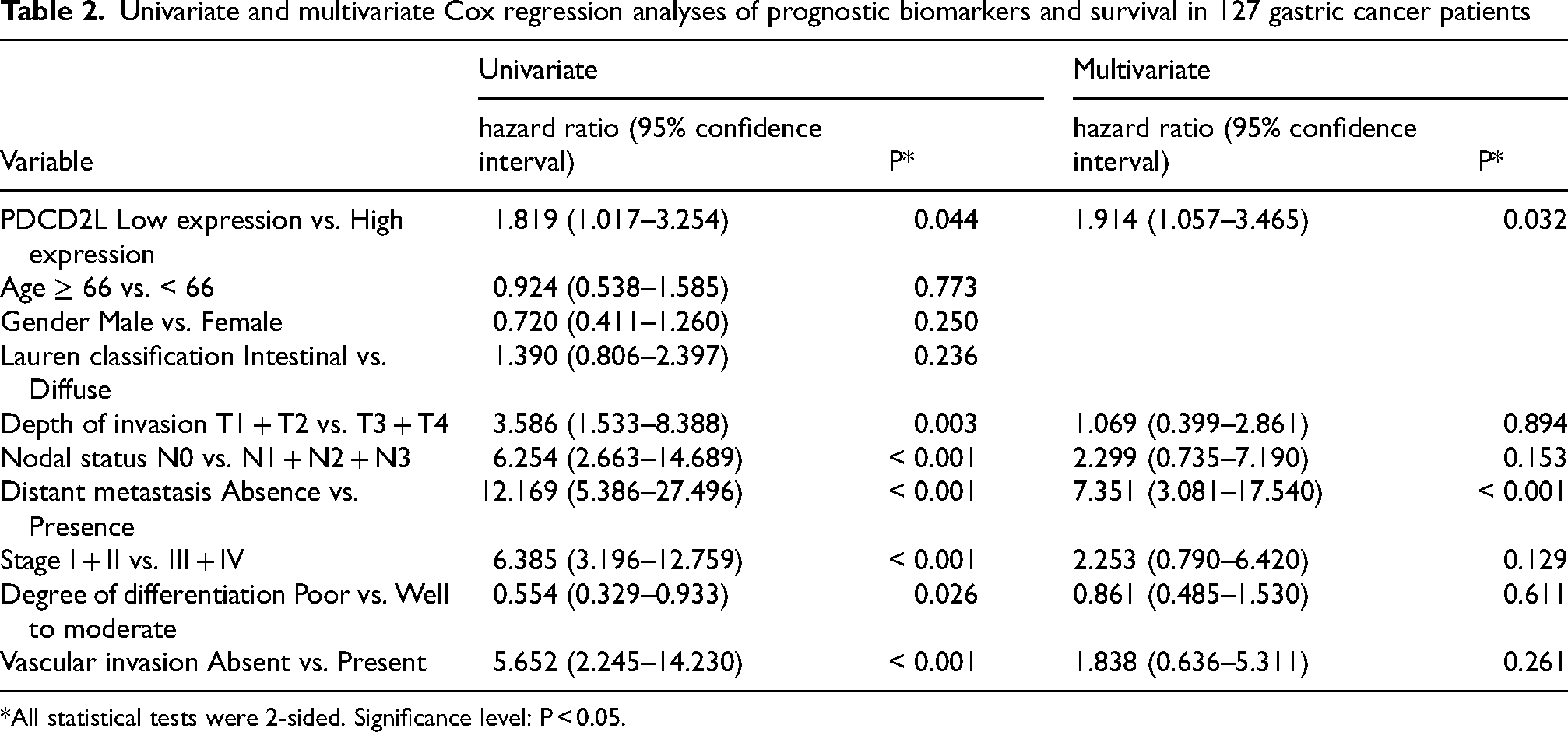
*All statistical tests were 2-sided. Significance level: P < 0.05.
In the multivariate Cox regression analysis, PDCD2L overexpression and distant metastasis were prognostically independent (Table 2).
In summary, PDCD2L overexpression appears to predict poorer prognosis in patients. Increased PDCD2L expression may promote the progression of gastric cancer, and PDCD2L overexpression could serve as a biomarker for gastric cancer.
PDCD2L manipulation affects the proliferation of gastric cancer cells
Based on the expression level of PDCD2L in cells, the 23132/87 gastric cancer cell line, which highly expresses PDCD2L, was utilized to enhance our understanding of the role of endogenous PDCD2L in regulating cell proliferation. After infecting 23132/87 cells with an adenovirus carrying a PDCD2L shRNA vector, we confirmed the suppression of PDCD2L expression in 23132/87 cells using immunoblotting, resulting in PDCD2L knockdown 23132/87 cells (Figure 3A). The 23132/87 cells with PDCD2L shRNA-1 vector was selected for the following experiments. As shown in Figure 3B, compared to control cells, the ability of PDCD2L knockdown 23132/87 cells to form colonies was inhibited. This result indicates that PDCD2L knockdown inhibits the proliferative capacity of 23132/87 cells.
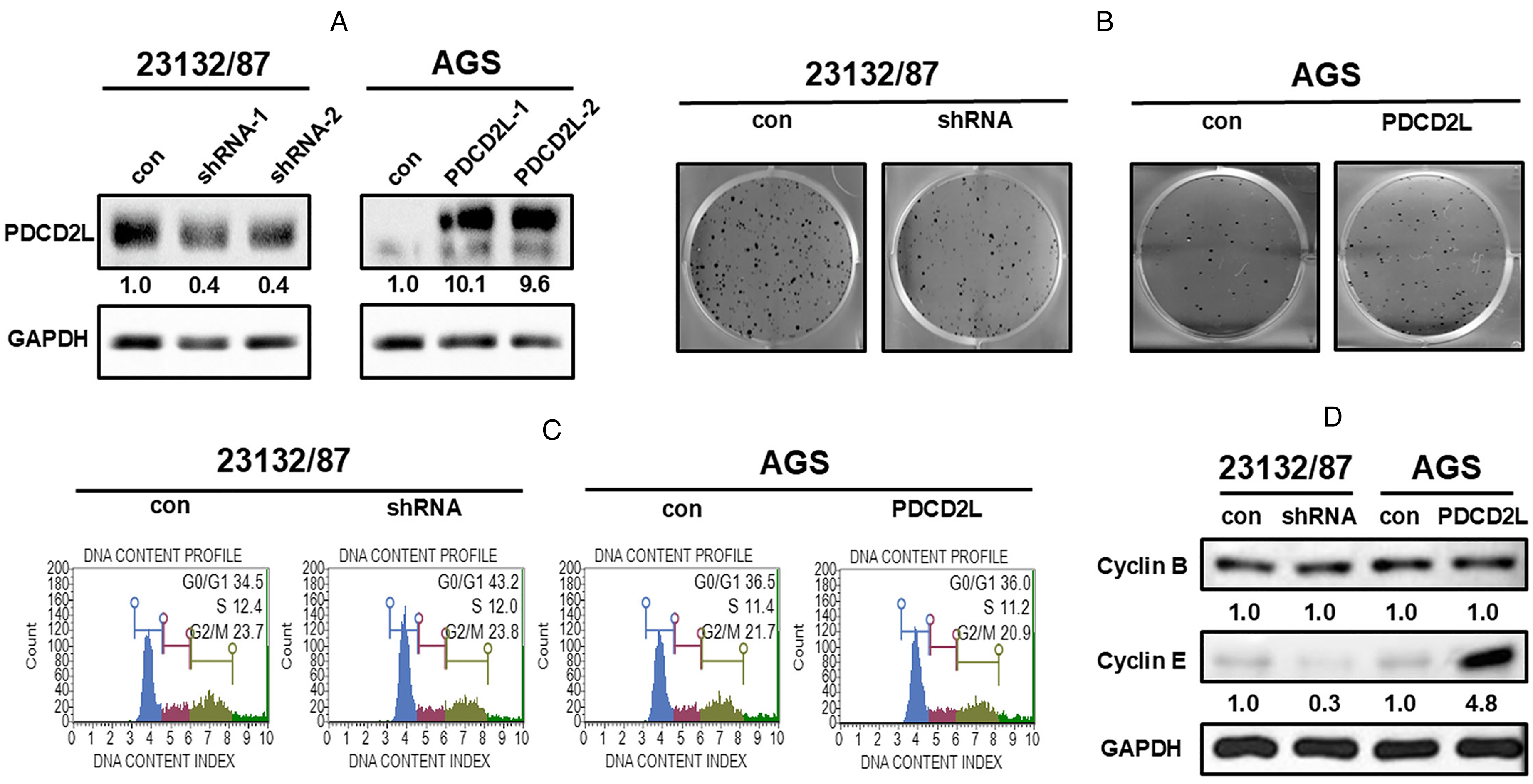
Verification of PDCD2L manipulation in 23132/87 and AGS cells, and the effect of stable PDCD2L manipulation on cell growth, cell cycle distribution, and the expression of cell cycle control molecules in the cells. (A) The immunoblotting results show PDCD2L was knockdown by shRNA treatment and overexpressed by transfection. (B) Stable PDCD2L knockdown and overexpression markedly suppressed and promoted colony formation, respectively. (C) Stable PDCD2L knockdown caused a sustained accumulation of cells in the G0/G1 phase. (D) Stable PDCD2L knockdown and overexpression clearly decreased and increased the expression of cyclin E. con: scrambled control or control vector-transfected cells; shRNA: PDCD2L-knockdown cells; PDCD2L: PDCD2L-overexpressing cells.
The AGS cell line was also selected to further investigate the function of PDCD2L in gastric cancer cell proliferation. PDCD2L overexpression was confirmed through immunoblotting (Figure 3A). The AGS cells transfected with PDCD2L-2 vector was selected for the following experiments. More colonies were formed by PDCD2L-overexpressing AGS cells (Figure 3B).
To clarify the biological events linked to the inhibition of cell proliferation caused by PDCD2L knockdown, we used flow cytometry to analyze changes in DNA content across different phases of the cell cycle. As shown in Figure 3C, PDCD2L knockdown 23132/87 cells exhibited a significant increase in the percentage of cells in the G0/G1 phase. Therefore, our RNA interference experiments demonstrate that PDCD2L knockdown impedes the progression of cell cycle from the G0/G1 phase to the S phase, thereby hindering the proliferation of 23132/87 cells. Quantitative and statistical analysis results were shown in Supplementary Figure 1. As shown in Figure 3C, PDCD2L overexpression did not affect the cell cycle progression.
We measured the cyclin levels to evaluate the underlying mechanism of G0/G1 arrest caused by PDCD2L knockdown. Figure 3D shows that the level of cyclin E, G0/G1 phase regulators, was decreased in PDCD2L-knockdown 23132/87 cells. Upregulation of PDCD2L has the opposite effect. Furthermore, the level of cyclin B, G2 phase regulators, were unchanged in PDCD2L-manipulated gastric cancer cells (Figure 3D).
Manipulation of PDCD2L affects the invasiveness of gastric cancer cells
To confirm the effect of PDCD2L manipulation on the migration of gastric cancer cells, we conducted a wound-healing assay. We noted that compared to control cells, the wound-healing rate of PDCD2L knockdown 23132/87 cells was significantly slower (Figure 4A). Additionally, in the cell invasion assay, compared to control cells, the invasion rate of PDCD2L knockdown 23132/87 cells was also significantly inhibited (Figure 4A). Our shRNA experiments indicate that PDCD2L knockdown disrupts the migration and invasion of 23132/87 cells. By contrast, PDCD2L overexpression promotes the migration and invasion of AGS cells (Figure 4B).
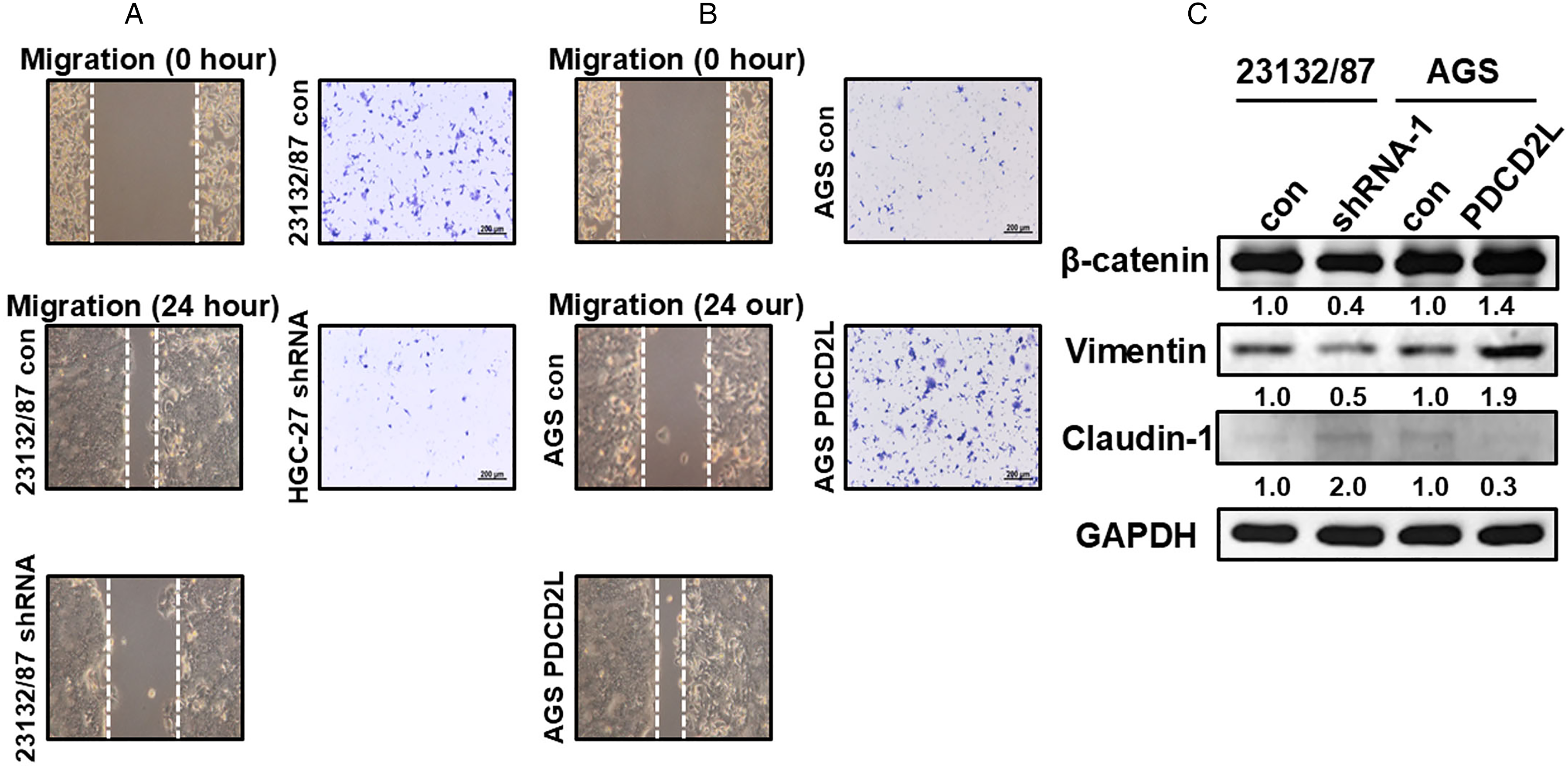
Effect of PDCD2L manipulation in 23132/87 and AGS cells on cell spreading and the expression of EMT-related proteins. (A) Stable PDCD2L knockdown markedly impeded cell migration and invasion. (B) Stable PDCD2L overexpression obviously facilitated cell migration and invasion. (C) Stable PDCD2L manipulation caused the dysregulated expression of EMT-related proteins. con: scrambled control or control vector-transfected cells; shRNA: PDCD2L-knockdown cells; PDCD2L: PDCD2L-overexpressing cells.
We assessed the expression levels of β-catenin, vimentin and claudin-1, involved in the EMT, to understand how PDCD2L regulated EMT. Figure 4C shows that the levels of β-catenin and vimentin decreased, while the level of claudin-1 increased in PDCD2L-knockdown 23132/87 cells. Overexpression of PDCD2L in AGS cells caused the opposite effect (Figure 4C).
Effect of PDCD2L knockdown on xenograft proliferation
The aforementioned in vitro data suggest that PDCD2L has an oncogenic role in vivo. In this study, each experimental mouse bearing scrambled control cells or PDCD2L knockdown 23132/87 cells on the right hind flank began to exhibit conspicuous differences in tumor growth between these 2 groups. Figure 5 shows that six weeks after subcutaneous inoculation, the weight of scrambled control tumors was 3.2 times that of PDCD2L knockdown tumors.
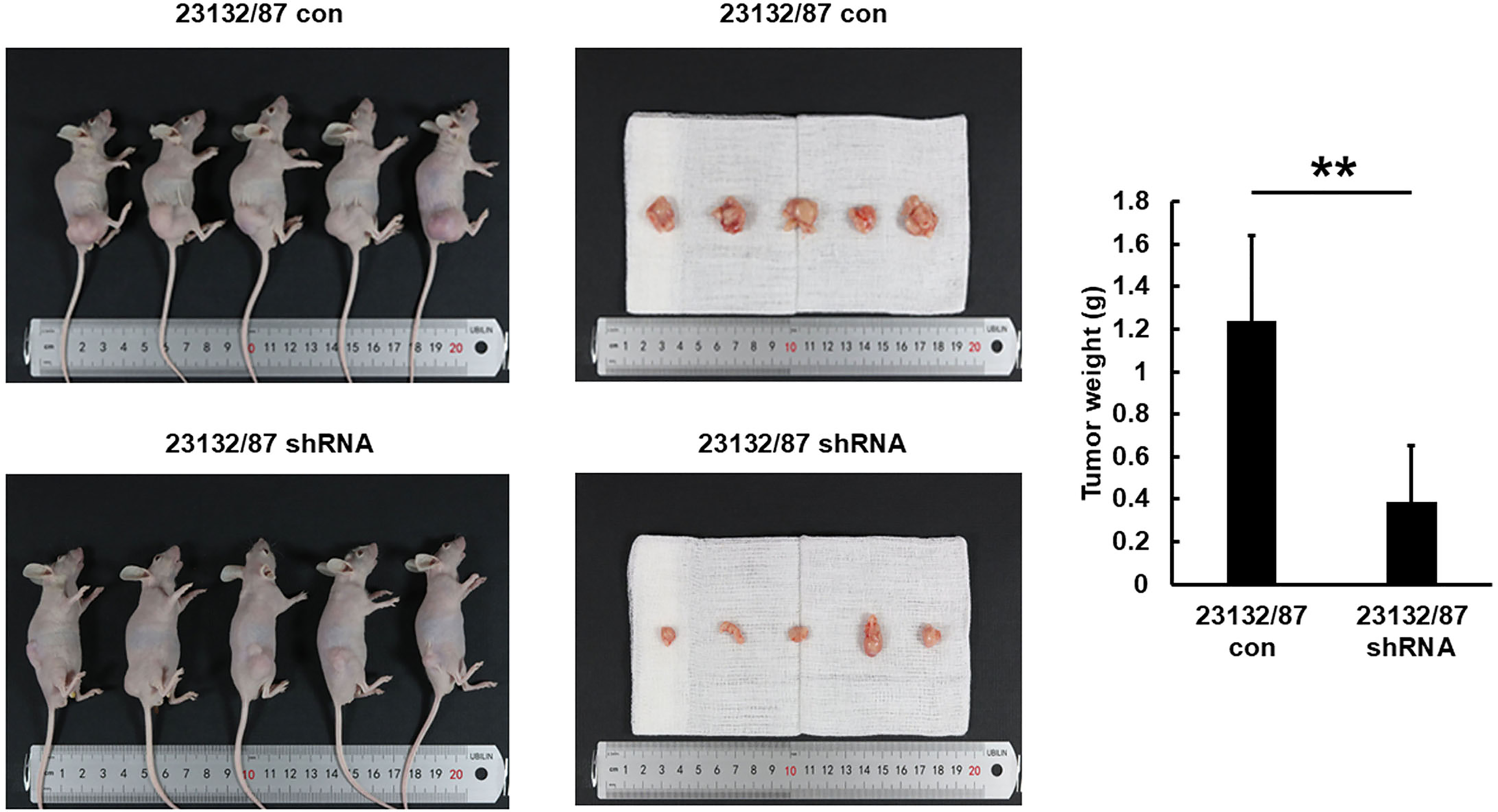
The effect of stable PDCD2L knockdown in 23132/87 cells on in vivo tumor growth in nude mice. Stable PDCD2L knockdown caused the significant suppression of 23132/87 xenograft proliferation. The bar graph represents the results of the average tumor weight (presented as the mean ± SD, **P < 0.05). con: scrambled control or control vector-transfected cells; shRNA: PDCD2L-knockdown cells.
Regulation of RFX1 by PDCD2L
To further understand the underlying mechanism responsible for PDCD2L-regulated cell proliferation, migration, and invasion and to identify the direct targets of PDCD2L, next-generation sequencing was employed in PDCD2L-knockdown 23132/87 cells. Table 3 lists several down-regulated genes in PDCD2L-knockdown 23132/87 cells. Among the differentially expressed genes, RFX1 attracted our attention and interest for its contribution towards increased cell proliferation and spreading, which can facilitate the malignant behavior of cells. The RFX1 level in 8 gastric cell lines was detected. Figure 6A indicates that the RFX1 level was notably increased in six gastric cancer cell lines (AGS, NCI-N87, TSGH 9201, SK-GT-2, HGC-27, and 23132/87). Immunoblotting was also used to measureRFX1 expression in PDCD2L-manipulated gastric cancer cells. Figure 6B shows that RFX1 decreased in PDCD2L-knockdown 23132/87 cells and increased in PDCD2L-overexpressing AGS cells, suggesting a correlation between PDCD2L and RFX1 expression.
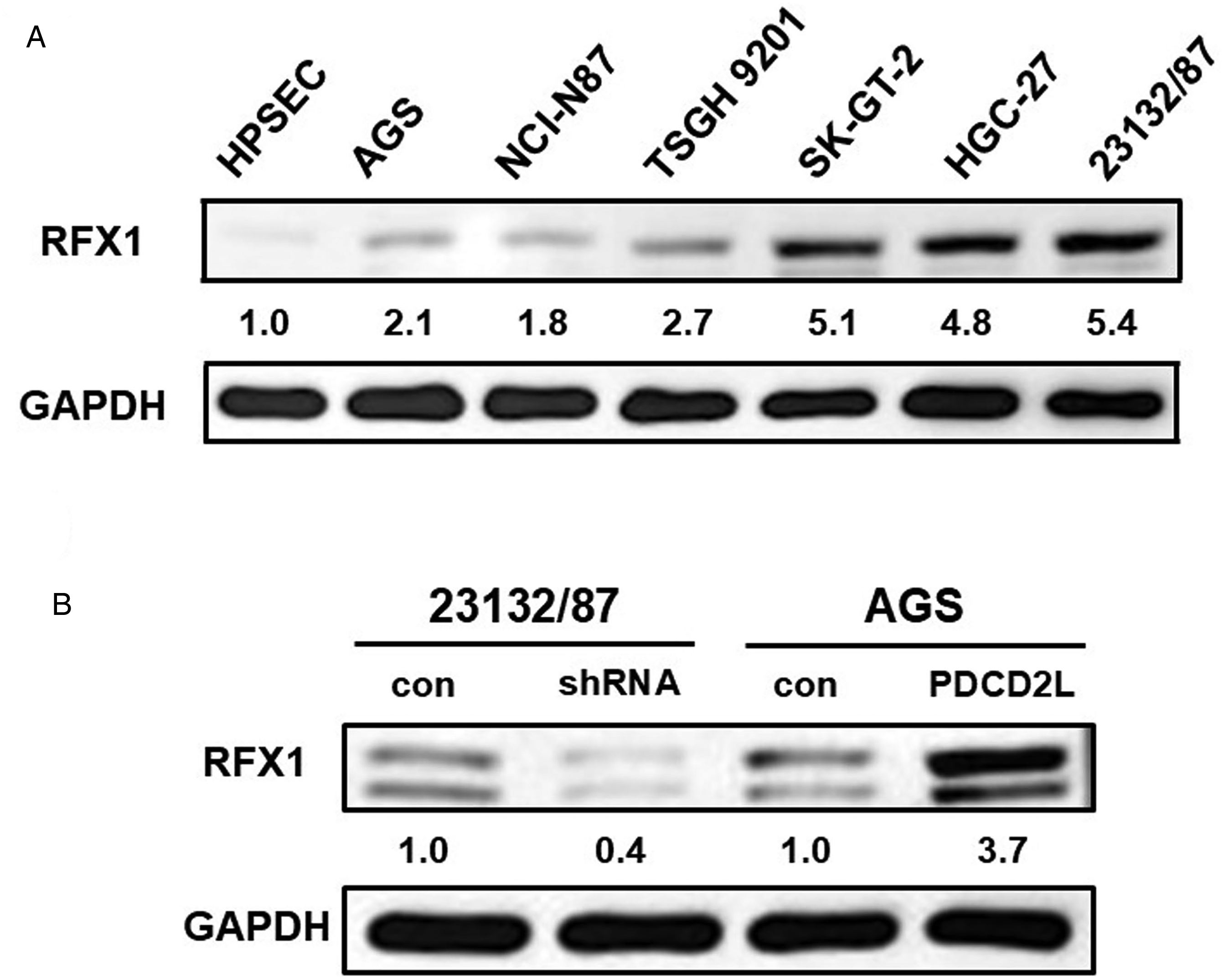
RFX1 expression in gastric cells and the relationship between RFX1 and PDCD2L. (A) RFX1 level was obviously increased in gastric cancer cells as compared with that in HPSEC cells. (B) Stable PDCD2L knockdown and overexpression caused markedly decreased and increased expression of RFX1. con: scrambled control or control vector-transfected cells; shRNA: PDCD2L-knockdown cells; PDCD2L: PDCD2L-overexpressing cells.
Genes down-regulated in PDCD2L-knockdown 23132/87 cells
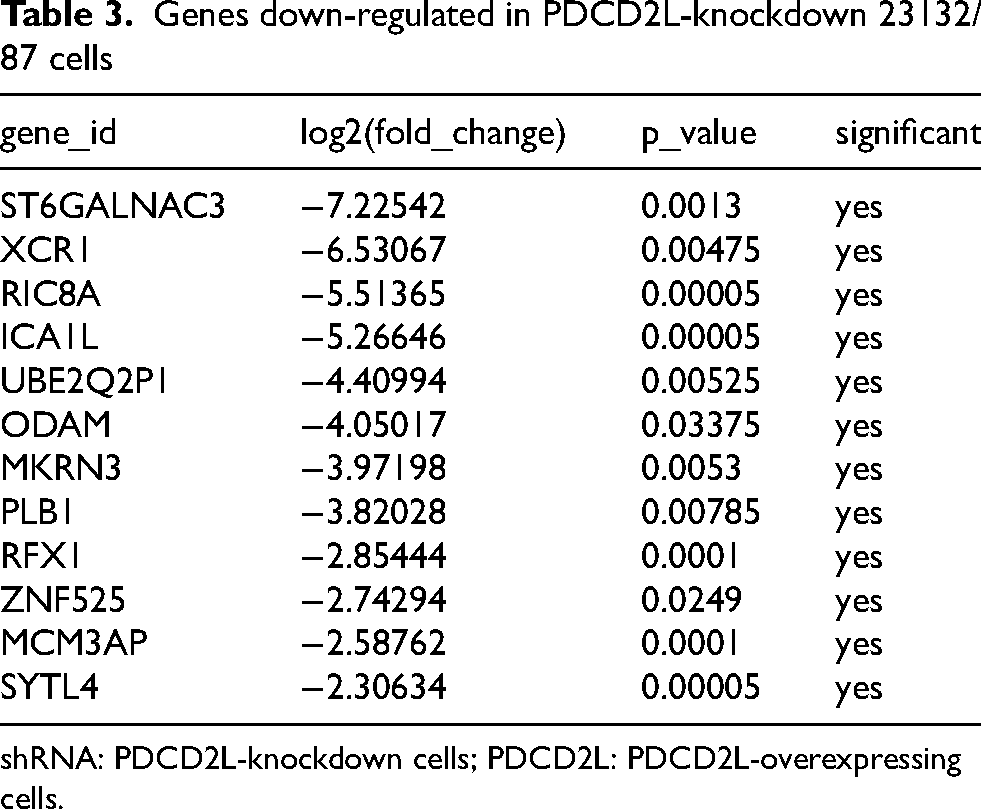
shRNA: PDCD2L-knockdown cells; PDCD2L: PDCD2L-overexpressing cells.
Due to the role RFX1 plays in promoting cell proliferation and spreading, this pilot study implys that the overexpression of PDCD2L may potentially up-regulate RFX1, which in turn promotes cell proliferation and spreading by increasing the levels of cell cycle- and EMT-related molecules. Further experiments are needed to confirm this hypothesis.
Discussion
The PDCD family, as suggested by their name, was initially discovered to be associated with programmed cell death. In addition, different members of the family each have distinct functions. For example, PDCD1 is crucial for regulating immune system responses, while PDCD6 and PDCD10 are both thought to be related to cell proliferation.11,13,15 Previous research has shown that PDCD2 plays an important role in biological functions such as cell proliferation and spreading, processes now known to be closely related to cancer development. 21 Is PDCD2 indeed involved in cancer development? Some studies suggest that PDCD2 acts as a tumor suppressor gene. However, other research indicates that PDCD2 may function as an oncogene.20,22 As for PDCD2L, our understanding of it remains quite limited. It is known that PDCD2L is overexpressed in certain cancers, such as colorectal cancer, kidney cancer, lung cancer, and ovarian cancer. The expression level of PDCD2L in gastric cancer remains unknown to date. Through this study, we have learned that, similar to the aforementioned cancers, PDCD2L is overexpressed in both gastric cancer tissues and gastric cancer cell lines. This result also suggests that PDCD2L may be an oncogene in the development of gastric cancer, which warrants further investigation.
Some molecules are thought to be putative therapeutic targets for gastric cancer. Yun et al. showed that upregulation of FAM64A expression might induce aggressive phenotypes, leading to gastric carcinogenesis and its subsequent progression. FAM64A could be regarded as a prognosis biomarker and a target for gene therapy. 7 Laurino and his colleagues assessed the role of STAT3 in gastric cancer drug efficiency. 31 It was found that combination of STAT3 inhibitor and cisplatin prevented both STAT3 activation and more lethality than induction by cisplatin alone. The data suggest that the inhibition of STAT3 associated with cisplatin treatment may represent a strategy to overcome resistance. Little is known about the function of PDCD2L in cancer, with only two papers published address this issue so far. The study by Gao et al. on the role of PDCD2L in colorectal cancer is, so far, the only research on this topic. 24 Kong et al. found that, in hepatocellular carcinoma, basic transcription factor 3 plays an oncogenic role by regulating the transcriptional expression of PDCD2L, which promotes proliferation and inhibits apoptosis of cancer cells by restraining the p53 pathway. 32 In this paper, the authors not only observed the overexpression of PDCD2L but also found that when PDCD2L expression in colorectal cancer cells was inhibited, the cells began to proliferate and eventually underwent cell death. The authors suggested that PDCD2L may be involved in a process known as apoptosis-induced proliferation. In our study, to understand the significance of PDCD2L overexpression in gastric cancer, we used two gastric cancer cell lines and observed changes in cell proliferation, cell cycle, and cell migration by altering PDCD2L expression levels. We found that when PDCD2L was inhibited, cell proliferation slowed, the cell cycle was arrested, and cell migration decreased. Conversely, when PDCD2L was overexpressed, cell proliferation accelerated, the cell cycle was not arrested, and cell migration increased. Through immunoblotting, we also observed that the expression levels of proteins related to the regulation of cell proliferation (cyclin E, shown in Figure 3D), cell cycle (cyclin E, shown in Figure 3D), and cell spreading (β-catenin, vimentin, claudin-1, shown in Figure 4C) changed accordingly. When PDCD2L is downregulated, cyclin E, a regulator of the G0/G1 phase, is also reduced, leading to cell cycle arrest. Similarly, when PDCD2L is downregulated, the expression of β-catenin and vimentin-two molecules that promote EMT-decreases, while the expression of claudin-1, an EMT-suppressing molecule, increases, leading to inhibited cell migration. These results suggest that PDCD2L may promote processes such as cell proliferation, cell cycle progression, and cell spreading. These data also indicate that PDCD2L appears to have different functions in different types of cancers. Whether there is any connection between these studies or, as the results suggest, if PDCD2L's functions vary depending on the organs or tissues in which it is expressed, remains a question that requires further research for a better understanding.
So far, there have been no studies on the correlation between PDCD2L and cancer prognosis. However, some findings have been made regarding other members of the PDCD family, though these studies have not reached consistent conclusions. PDCD4 is underexpressed in colorectal cancer and can predict a poorer prognosis in colorectal cancer patients. 26 PDCD5 inhibits osteosarcoma metastasis and can predict a better prognosis for osteosarcoma patients. 27 In contrast to PDCD4 and PDCD5, PDCD6 and PDCD7 are overexpressed in ovarian cancer and acute myeloid leukemia, respectively, and predict poorer prognosis in patients.28,29 These findings suggest that different members of the PDCD family predict various prognostic outcomes for cancer patients, depending on their expression levels in the cancers. According to the statistical results of our study, similar to PDCD6 and PDCD7, PDCD2L is overexpressed in gastric cancer and can predict a poorer prognosis for gastric cancer patients. Furthermore, PDCD2L can serve as an independent prognostic marker for gastric cancer. This is the first study to report that PDCD2L overexpression can serve as a biomarker for predicting the prognosis of gastric cancer patients.
This study has certain limitations, such as not delving deeply into the underlying mechanisms and signaling pathways by which PDCD2L promotes gastric cancer development. In this study, we found that the expression of RFX1 was down-regulated in PDCD2L-knockdown gastric cancer cells. RFX1 is a transcriptional factor involved in a wide range of cellular processes such as cell cycle, cell proliferation, differentiation, and apoptosis. 33 The expression pattern of RFX1 in cancer tissues differs from that in normal tissues. 34 A significant reduction in RFX1 expression has been observed in cancers such as breast cancer. In these cancers, RFX1 expression is notably decreased, especially in advanced stages, higher grades, or subtypes associated with poor prognosis, such as triple-negative breast cancer. Conversely, increased RFX1 expression has been observed in cancers such as liver cancer. In these cases, RFX1 expression is significantly elevated, although its specific impact requires further investigation. Overall, RFX1 expression is closely associated with cancer type, stage, and prognosis, and its dual role in tumor suppression and oncogenesis warrants further in-depth study. Only one study examined the expression of RFX1 in gastric cancer. 35 Guo et al. found that the mRNA level of RFX1 was significantly elevated in gastric cancer tissue versus adjacent normal tissue. The regulation and function of RFX1 in gastric cancer were not explored in this study. Our results imply that the overexpression of PDCD2L may potentially up-regulate RFX1, which in turn promotes cell proliferation and spreading. Manipulation of RFX1 can be used to further explore the relationship between PDCD2L and RFX1. Therefore, further research is needed in the future to address these questions.
To date, this is the first study to investigate PDCD2L expression in gastric cancer, the correlation between PDCD2L overexpression, clinicopathologic characteristics, and patient prognosis, and the effects of PDCD2L manipulation on gastric cancer cells. Our conclusion is that PDCD2L is overexpressed in gastric cancer and can serve as a prognostic biomarker, playing a role in promoting gastric cancer cell proliferation and spreading during gastric cancer development.
Conclusion
We investigated the clinical significance of PDCD2L in gastric cancer and the function of PDCD2L in the proliferation, migration, and invasion of gastric cancer cells. Our findings suggested that PDCD2L overexpression may serve as a biomarker in predicting the prognosis of patients with gastric cancer and that PDCD2L may be a target for gastric cancer treatment.
Supplemental Material
sj-docx-1-cbm-10.1177_18758592251374857 - Supplemental material for PDCD2L overexpression represents an unfavorable prognostic marker and its inhibition shows promising therapeutic potential for gastric cancer
Supplemental material, sj-docx-1-cbm-10.1177_18758592251374857 for PDCD2L overexpression represents an unfavorable prognostic marker and its inhibition shows promising therapeutic potential for gastric cancer by Ding-Ping Sun, Chun-Chao Chang, Hsuan-Yi Huang, Nai-Weng Kang, You-Cheng Hseu, Yu-Feng Tian, Chia-Lang Fang and Kai-Yuan Lin in Cancer Biomarkers
Footnotes
Acknowledgments
Not applicable.
Ethical approval and informed consent statements
The study adhered to the principles of the Declaration of Helsinki and received approval from the Institutional Review Board of Chi Mei Medical Center (Approval No. 10812–006). Written informed consent was obtained from each patient prior to participation. All experimental procedures involving animals complied with institutional guidelines, and were approved by the Chi Mei Medical Center Animal Ethics Research Board (Approval No. 108120105).
Consent to participate
Written informed consent was obtained from each patient prior to participation.
Consent for publication
Not applicable.
Author contributions
Ding-Ping Sun: Methodology, Validation, Investigation, Resources, Writing-review and editing. Chun-Chao Chang: Methodology, Validation, Investigation. Hsuan-Yi Huang: Methodology, Investigation. Nai-Wen Kang: Methodology, Investigation. You-Cheng Hseu: Investigation. Yu-Feng Tian: Investigation. Chia-Lang Fang: Conceptualization, Resources, Writing-original draft, Writing-review and editing, Supervision, Project administration, Funding acquisition. Kai-Yuan Lin: Conceptualization, Writing-original draft, Writing-review and editing, Supervision, Project administration, Funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chi Mei Medical Center (grant number 113CM-TMU-01).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data generated and/or analyzed during the current study are available from the corresponding author on reasonable requests.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
