Abstract
BACKGROUND:
Accumulating evidences have shown that long non-coding RNAs (lncRNAs), longer than 200 nucleotides in length, play a crucial role in cancer occurrence and development. However, the relationships between most lncRNAs and gastric carcinogenesis remain poorly understood.
OBJECTIVE:
To explore the diagnostic value of one typical lncRNA, long intergenic non-protein coding RNA 1006 (LINC01006), in gastric cancer.
METHODS:
First, real-time quantitative reverse transcription-polymerase chain reaction (qRT-PCR) was used to determine the expression levels of LINC01006 in various gastric tissues from gastric cancer patients, healthy controls, and gastric dysplasia. Next, the correlation between LINC01006 expression levels and clinicopathological features of patients with gastric cancer was assessed. Finally, the relative levels of LINC01006 in gastric cancer cell lines comparing to normal gastric epithelial cell line were analyzed.
RESULTS:
LINC01006 levels in cancer tissues were significantly lower than those in adjacent normal tissues (
CONCLUSIONS:
All the data implied that LINC01006 may be a novel biomarker for gastric cancer.
Introduction
Among global malignancies, gastric cancer is the second leading cause of cancer death annually [1, 2]. The high morbidity is partially attributed to delayed diagnosis of gastric cancer at a very advanced stage, at which point patients cannot achieve satisfactory outcome from current surgical, chemical or radiological therapies [3, 4]. Thus, accurate and effective diagnosis at an early time will benefit gastric cancer patients. However, existing conventional diagnostic methods are largely limited in sensitivity and specificity. Therefore, discovering novel biomarkers for gastric cancer detection at early period has become a hotspot and gained increasing attention among scholars. Long non-coding RNAs (lncRNAs) is one type of new potential biomarkers [5, 6].
LncRNAs are a new category of transcripts with a length greater than 200 nucleotides [7]. On the basis of genomic localization, they can be classified roughly into six broad categories: sense, antisense, intronic, intergenic, enhancer, and bidirectional lncRNAs [7, 8]. Despite lack of protein-encoding potential, they play essential roles in a variety of biological processes, such as cell cycle, proliferation, apoptosis, and invasion. Also, lncRNAs have multiple regulatory functions in gene expression and chromatin remodeling [9, 10, 11].
Previous studies demonstrated that many lncRNAs were functionally associated with carcinogenesis. Aberrant expression of lncRNAs could promote tumor- genesis-related processes such as apoptotic resistance, metastasis and evasion of tumor suppressors [7, 12]. For instance, Hox transcript antisense intergenic RNA (HOTAIR) and prostate cancer-up-regulated long non-coding RNA 1 (PlncRNA-1) were highly upregulated in primary or metastatic breast tumors and prostate cancer, respectively [13, 14]. At the same time, lncRNA low expression in tumor (LET) and growth arrest specific transcript 5 (GAS5), a tumor suppressive lncRNA, were both proved to be downregulated in gastric cancer [15, 16]. However, the correlation between most lncRNAs and gastric cancer is largely unknown.
In this study, we focused on long intergenic non-protein coding RNA 1006 (LINC01006), whose gene is located at human chromosome 7q36.37. It is one of the dysregulated lncRNAs in gastric cancer firstly identified in the lncRNA expression profile of gastric cancer by lncRNA microarray (GEO No. GSE 47850) [17]. First, we expanded sample size and analyzed LINC01006 expression levels in gastric cancer and their adjacent normal tissues. Then, the relative expression levels of LINC01006 in three types of gastric mucosa were calculated. Next, we evaluated the correlation between their expression levels and clinicopathological characteristics of gastric cancer patients. Finally, we compared LINC01006 levels in gastric cancer cell lines with those in normal gastric epithelial cell line GES-1. Our results revealed that LINC01006 might be a novel valuable biomarker of gastric cancer.
Patients and methods
Specimens and clinical data collection
The 187 samples were collected from The Affiliated Hospital of Ningbo University School of Medicine and Yinzhou Hospital, Ningbo, China, between December 2013 and February 2014. We obtained gastric cancer tissues and their paired adjacent non-cancerous tissues through surgery. Before upper gastrointestinal endoscopic examination or operation, no radiotherapy or chemotherapy was applied to patients. The adjacent non-tumorous tissues were from tissues 5 cm away from the edge of gastric cancer. Meanwhile, no apparent tumor cells were found according to the evaluation of two experienced pathologists. Another 31 samples were obtained from 13 healthy gastric mucosa and 18 gastric precancerous lesions (dysplasia) through biopsy. After being removed from the body, the fresh tissues were immediately stored in RNA fixer (Bioteke, Beijing, China) at
Total RNA extraction
Following the manufacturer’s instructions, total RNA
in tissues was extracted by the TRIzol reagent (Invitrogen, Karlsruhe, Germany) and then dissolved in 10
Reverse transcription and polymerase chain reaction
First, we used random primers and oligo (dT) 15 primers to generate cDNA by reverse transcription with GoScript RT System (Promega, Madison, WI, USA). Two
Next, to measure the relative expression level of LINC01006, real-time polymerase chain reaction (PCR) was performed using GoTaq qPCR master mix (Promega) on the Mx3005P QPCR System (Stratagene, La Jolla, CA, USA) according to the manufacturer’s instructions. Herein, the reference RNA was glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA. The primers for LINC01006 and GAPDH were synthesized by Sangon Biotech Co. (Shanghai, China). Their sequences were as follows: 5
Cell culture
Human normal gastric epithelial cell line (GES-1) and Human gastric cancer cell lines (AGS and MGC-803) were purchased from Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences (Shanghai, China). We cultured cells with RPMI 1640 Medium (Invitrogen, Grand Island, NY, USA) containing 10% fetal bovine serum, 50 U/ml penicillin, and 50 U/ml streptomycin in culture flasks at 37
Serological tumor marker analysis
Carcinoembryonic antigen (CEA) and serum carbohydrate antigen 19-9 (CA19-9) were determined by an Elecsys 2010 machine (Roche Diagnostics, Basel, Switzerland). The cutoff values were 5 ng/ml and 35 U/ml for CEA and CA19-9, respectively.
The relationships of LINC01006 expression levels (
in cancer tissues with clinicopathological factors of gastric cancer patients
The relationships of LINC01006 expression levels (
A smaller
Statistical Product and Service Solutions (SPSS) 19.0 software (SPSS, Chicago, IL, USA) and GPower 3.1 software (Düsseldorf, Nordrhein-Westfalen, Germany) were used to analyze all experimental data. The differences in LINC01006 level among healthy control, dysplastic group, and gastric cancer patients were analyzed by one-way analysis of variance (ANOVA). Besides, ANOVA was recruited to analyze the correlation between LINC01006 levels and clinicopathological factors of gastric cancer patients. All graphs were plotted using OriginPro 8.5.1 (OriginLab Corporation, Northampton, MA, USA).
Results
General characteristics of gastric cancer patients
The majority of the cancer patients were over 60 years old, representing 67%. Patients with gastric cancer were predominantly men (71%). There were 35 patients (52%) whose gastric tissues were moderately differentiated. The positive rates of CEA and CA19-9 were 90% and 58%, respectively (Table 1).
LINC01006 expression level in gastric cancer tissues and adjacent normal tissues (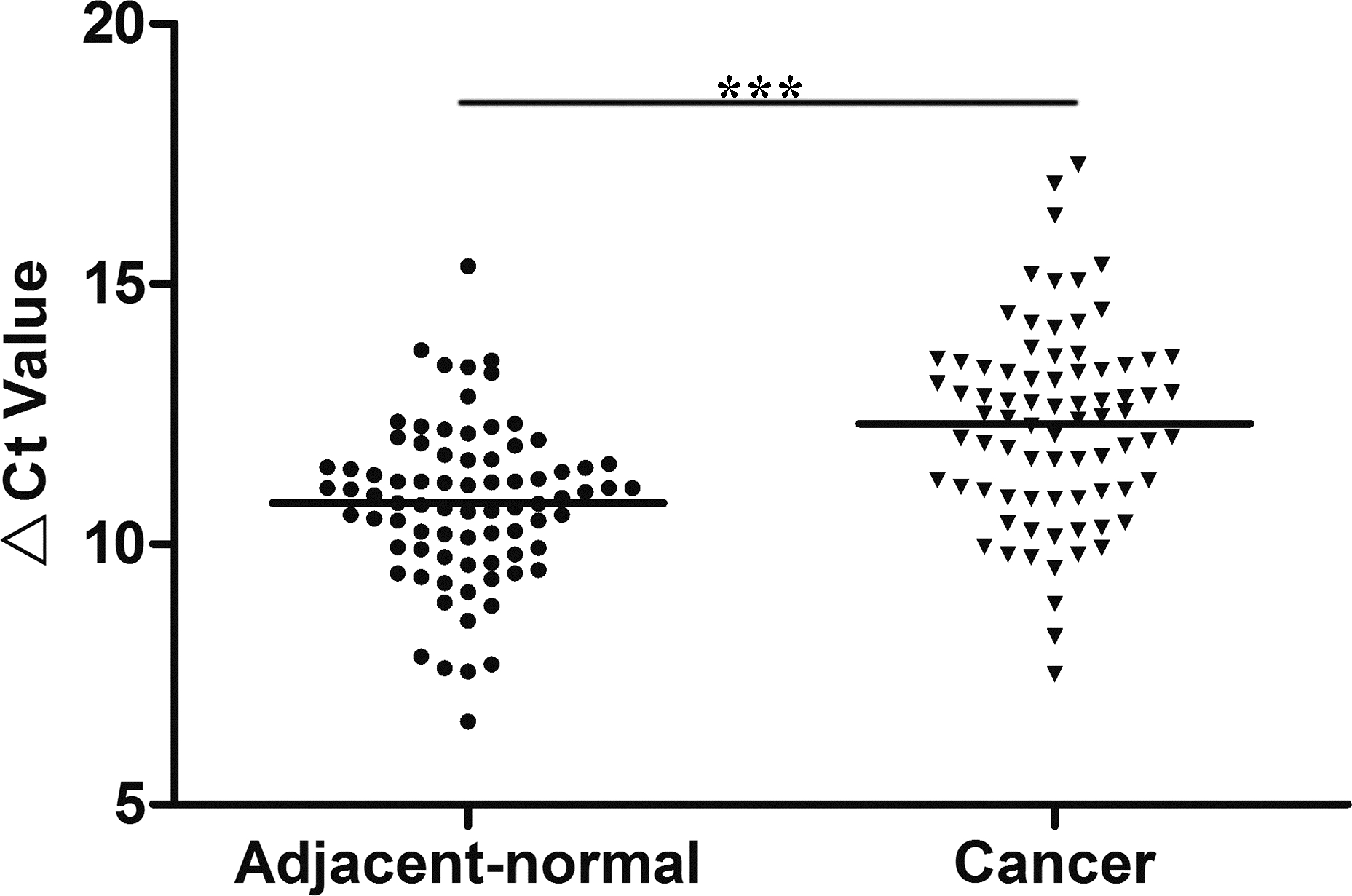
Relative expression of LINC01006 in three types of gastric mucosa. The LINC01006 expression levels in healthy gastric mucosa (
ROC curve for differentiating gastric cancer tissues from healthy controls. ROC, receiver operating characteristic; AUC, area under the curve.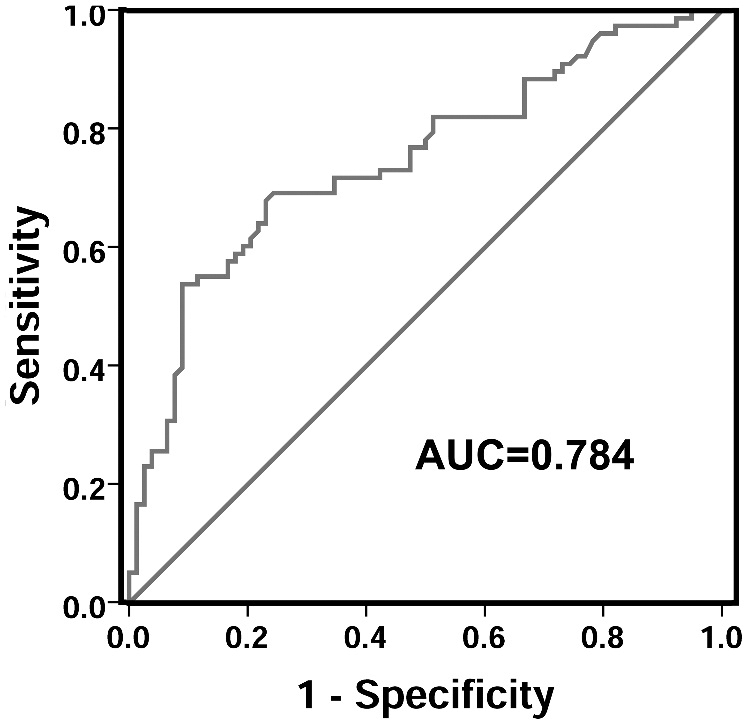
We quantified LINC01006 expression levels in gastric cancer tissues by qRT-PCR. We observed that LINC01006 levels in tumor tissues collected from gastric cancer patients were significantly lower than those in their corresponding adjacent normal tissues (
A receiver operating characteristic (ROC) curve was performed to test whether LINC01006 had value in clinical diagnosis of gastric cancer. The area under the curve (AUC) was up to 0.748 (95% confidence interval 0.671–0.826;
Statistical power of these results was then calculated by GPower 3.1 with
Differences of LINC01006 levels in gastric cancer tissues between genders and ages
Previous studies demonstrated that gastric cancer was distinguishable between males and females as well as between older and younger individuals [21]. In the current study, we observed significant differences under 60 years old and above (
Correlation between LINC01006 levels in cancer tissues and clinicopathological features of gastric cancer patients
Next, we explored whether the expression levels of LINC01006 were correlated to gastric cancer patients’ clinicopathological factors. As shown in Table 1, the LINC01006 expression levels were significantly associated with tumor location (
Expression of LINC01006 was downregulated in gastric cancer cell lines
Finally, we cultured normal gastric epithelial cell line GES-1 and gastric cancer cell lines, MGC-803 and AGS. By using RT-PCR, we found that the expression of LINC01006 in normal gastric epithelial cell line GES-1 was significantly higher than both MGC-803 (
Relative expression of LINC01006 in gastric cancer cell lines, MGC-803 and AGS, and normal gastric epithelial cell lines GES-1. LINC01006 expression levels in gastric cancer cell lines and normal gastric epithelial cell line were determined by RT-PCR. Data are represented as Mean 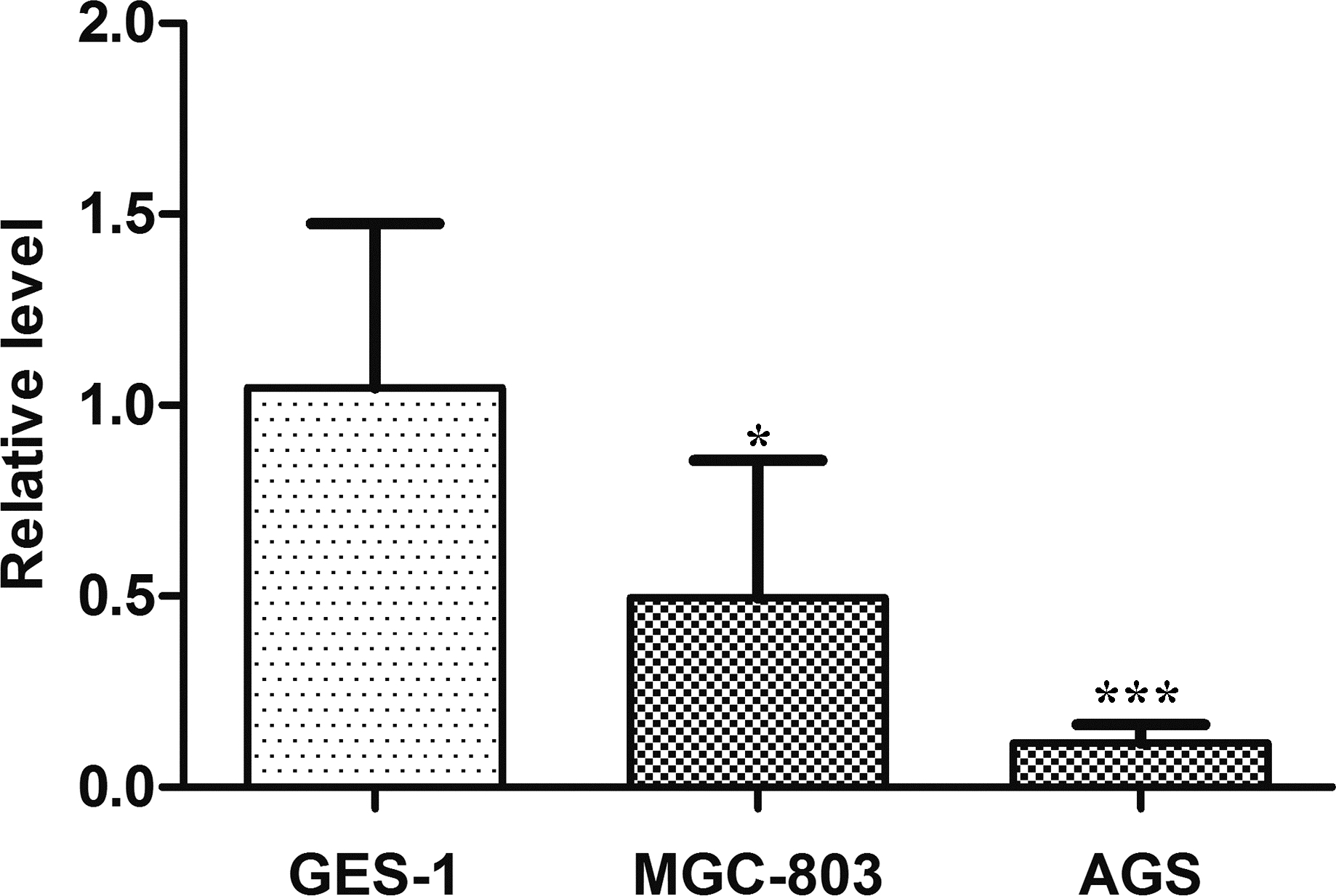
Oncogenes, tumor suppressor genes, growth factors and others function as regulators to balance cell proliferation and apoptosis. Cancers are the ultimate fate of unlimited cell proliferation without appropriate apoptosis, which are a pathological multistep process termed tumorigenesis [4, 6, 7, 11]. Recent studies showed that lncRNAs play a key role in carcinogenesis in several types of cancers [22, 23]. Dysregulated lncRNAs participate on initiation and progression of human tumors as either oncogenes or tumor suppressor genes [7, 24]. For example, HULC plays an oncogenic role in promoting tumorigenesis by regulating multiple pathways [22]. Other lncRNAs including CCAT1, GAPLINC, GAS5, H19, MEG3, and TUSC7 etc. act as oncogenes or tumor suppressors in cancer progression by correlated with tumor suppressor P53 or onco-protein c-Myc, respectively [5, 7, 16, 25, 26].
An increasing amount of studies has shown that through interaction with DNA, RNA and proteins, lncRNAs are closely linked to gastric cancer cell proliferation, apoptosis, invasion, migration, metastasis and tumorigenicity [7, 27, 28]. H19 and HOTAIR are two well-studied oncogenic lncRNAs in gastric cancer progression. H19 was the first reported lncRNA [29]; and Yang et al. first validated its participation on gastric tumorigenesis in 2012 [30]. HOTAIR has been observed in gastric cancer. Its role in gastric cancer resembles H19, promoting cell proliferation, invasiveness, and metastasis but inhibiting apoptosis [31].
The purpose of the current study was to investigate the clinical value of LINC01006 in gastric cancer. We observed significantly decreased LINC01006 expression levels in gastric cancer tissues compared to those of adjacent non-tumorous tissues (Fig. 1), which is in line with lncRNA microarray screening [17]. Besides, we found that LINC01006 levels varied from healthy to gastric cancer group. The LINC01006 levels were adversely associated with tumor stage; the more advanced the tumor was, the lower the expression levels were (Fig. 2). Additionally, compared with normal gastric epithelial cell line (GES-1), LINC01006 was significantly downexpressed in gastric cancer cell lines, MGC-803 and AGS (Fig. 4). The reduction was more remarkable between GES-1 and AGS (Fig. 4). Moreover, the statistical power was high enough for us to make the correct decision. LINC1006 was downregulated in gastric cancer tissues and further decreased in more advanced tumors. These revealed that LINC1006 might function as a tumor suppressor gene in the gastric cancer progression. All of these results implied that LINC01006 possibly played a crucial part in occurrence and development of gastric cancer and may be a good biomarker for gastric cancer.
Biomarkers are predominantly classified into four types, diagnostic, prognostic, predictive and therapeutic biomarkers [32, 33]. An ideal diagnostic biomarker should be a noninvasive marker to detect diseases at early stages. A prognostic biomarker provides assessment or prediction of the possible course of diseases, chances of recovery or survival from the disease, and thus yields valuable advice for further therapies. A predictive biomarker is utilized to identify whether subpopulations of patients respond to an aimed therapy or not. A therapeutic biomarker is usually a potential target protein for cancer therapy [32, 33].
The traditionally used serum gastric cancer markers include CEA and CA19-9. The sensitivity and specificity of CEA were about 70% and 60%, respectively; those for CA19-9 were about 60% and 56%, respectively. However, they had a low positive rate in detecting gastric cancer at early stages [34]. Therefore, novel biomarkers with higher sensitivity and specificity are urgently needed. With features of multiple functions and advantages of high cell-type specificity, tissue and disease specificity, and easy detection in tissues, serum, plasma, urine and saliva, lncRNAs are emerging as suitable minimally invasive makers in gastric cancer diagnosis, prognosis and therapies [7, 9, 32].
Many dysregulated lncRNAs were reported to have diagnostic value in gastric cancer, such as H19, LINC 00152 and FER1L4 [35, 36]. Prostate Cancer Antigen 3 (PCA3), a prostate specific lncRNA that is over-expressed in
Previous research elucidated that independent significant prognostic indicators for gastric cancer included age, gender, tumor location, tumor size, depth of invasion, lymphatic and venous invasion etc. [37, 38]. In the current study, we found that LINC01006 levels were associated with age, tumor location, tumor diameter, and venous invasion (Table 1). Even with similar tumor biology, elderly cancer patients have worse overall survival than younger patients, as they receive less therapeutic effects both systematically and locally [39]. Liang et al. divided gastric cancer patients into three age groups, and they found that elderly group (
However, our results regarding LINC1006 levels associated with tumor location and venous invasion were inconsistent with existing studies. Talamonti et al. suggested that tumor location be an important predictor of overall survival in gastric cancer, as they found that patients with proximally located primary tumors were less likely to survive [44]. Wang et al. also discovered that there was a worse prognosis in patients with proximal gastric cancer and reported tumor location as an independent prognostic predictor [45]. In this study, LINC01006 expression level varied statistically significantly in different locations of gastric cancer (Table 1). However, our results showed that the proximally located gastric cancer (cardia) had higher LINC1006 levels than corpora ventriculi and sinuses ventriculi (Table 1). Venous invasion, associated with invasion depth, metastasis, tumor size, tumor location and growth pattern, was reported to be the independent prognostic factors of gastric cancer. Gastric patients undergoing extramural venous invasion had significantly lower 1-year survival rate than those without [46]. However, in this study, gastric cancer with venous invasion had higher LINC1006 level (Table 1). Those inconsistencies may be partially due to the small number of samples. Thus, more investigations into its prognostic significance are required.
Limitations of our study should be acknowledged. Firstly, compared to studies recruiting thousands of gastric cancer cases and controls [47], our number was smaller. With more cases, our data would be more convincing. Secondly, since that we failed to collect data before and after gastrectomy, we failed to monitor the dynamic changes of gastric cancer. Finally, lncRNAs have been identified not only within cells, but also in biological fluids, including blood, urine and saliva. There are abundant lncRNAs in human blood [9, 10, 32]. However, we only focused on LINC1006 in gastric cancer tissues.
Conclusions
In conclusion, LINC01006 was decreased in gastric cancer tissues and gastric cancer cell lines. Its expression levels were associated with age, tumor location, tumor size, and venous invasion. All our results implied a clear possibility that LINC01006 played an important role in gastric cancer progression and might be a biomarker of gastric cancer.
Footnotes
Acknowledgments
This work was supported by the Applied Research Project on Nonprofit Technology of Zhejiang Province (2016C33177), Zhejiang Provincial High-Education Teaching Reform Project (No. jg2015047), the Scientific Innovation Team Project of Ningbo (No. 2017C 110019), and the K.C. Wong Magna Fund in Ningbo University.
Conflict of interest
The authors declare that they have no conflict of interest.
