Abstract
BACKGROUND:
Epithelial-mesenchymal transition (EMT) is a complicated process that has been implicated in cancer progression and metastasis as well as the formation of many tissues and organs. BTB/POZ domain-containing protein 7 (BTBD7) is reported to regulate transcriptional factors and involved in the process of invasion and metastasis of some malignant tumors. Additionally, our preliminary studies have confirmed that BTBD7 expression was significantly correlated with Slug expression and poor prognosis of primary salivary adenoid cystic carcinoma (SACC). On this basis, this study further investigated function of BTBD7 in the invasion and metastasis of SACC in vitro, which may be a possible target of gene therapy in the future.
METHODS:
The expression of BTBD7 and Slug were both examined in SACC-LM and SACC-83 cell lines by immunofluorescence staining. High invasive SACC-LM cells were transfected with BTBD7 siRNA and the expression levels of BTBD7 and Slug were detected in both gene and protein levels by qRT-PCR and western blot analysis. Assays were performed to survey cell migration, invasion and proliferation capabilities with BTBD7 silencing.
RESULTS:
BTBD7 and Slug proteins were detected in SACC-LM and SACC-83 cell lines. BTBD7 silencing down-regulated the expression of Slug and MMP9 meanwhile up-regulated the expression of E-cadherin in SACC-LM cells, the migration and invasion abilities of cells were obviously suppressed but with no influence on cell proliferation.
CONCLUSIONS:
BTBD7 silencing inhibited EMT through regulation of Slug expression in SACC-LM cells and might act as a potential molecular target for gene therapy of SACC.
Introduction
Salivary adenoid cystic carcinoma (SACC) is a common subtype of malignant salivary gland tumors accounting for approximately 25% of malignant salivary gland tumors [1]. It is characterized by a high local invasion and distant metastasis to the lung, liver and bone [2]. Although substantial progress has been made in defining the genes that contribute to the initiation and progression of SACC, the biological mechanism for the increased metastatic capacity of SACC is still ambiguous and needs to be further investigated.
Epithelial-mesenchymal transition (EMT) recently attracted broad interest in the field of cancer research, tumor invasion and metastases which is a process that describes the development of motile, mesenchymal-like cells from non-motile parent epithelial cells [3]. During EMT, migratory and invasive capabilities of epithelial tumor cells are stimulated without a loss in viability, cells detach themselves from the basement membrane followed by metastasis [4]. Slug is one of the most effective transcription factors of EMT and is associated with invasion and metastasis in various tumors [5, 6, 7, 8]. Moreover, Slug silencing could inhibit the EMT process by downregulating EMMPRIN and upregulating E-cadherin in the PNI process of SACC [9].
Proteins that contain BTB domains are observed in combination with DNA-binding motifs, and many of these BTB family proteins function as transcriptional regulators [10, 11, 12]. BTB/POZ domain-containing protein 7 (BTBD7), one member of BTB family proteins, is initially identified as a promotor that promotes epithelial tissue remodeling and formation of branched organs [13]. In addition, downregulation of BTBD7 siRNA in lung cancer cells and hepatoma carcinoma cells significantly inhibits invasion of cancer cells and regulates the expression of identifiable EMT biomarkers [14, 15]. Based on our previous study, BTBD7 may contribute to patients’ poor clinical outcome of SACC and was significantly correlated with Slug expression [16]. In present study, the function of BTBD7 and the possible between BTBD7 and Slug are further investigated as possible target of gene therapy.
Material and methods
Cell culture
SACC-83 and SACC-LM, the two human SACC cell lines were kindly presented by Professor Wantao Chen (Department of Oral and Maxillofacial Surgery, Ninth People’s Hospital, College of Stomatology, Shanghai Jiao Tong University, Shanghai, China). The two cell lines were cultured in RPMI 1640 culture medium (Hyclone, Logan, UT, USA) with 10% heat-inactivated fetal bovine serum (FBS; Hyclone, Logan, UT, USA), 100 U/ml penicillin, and 100 mg/ml streptomycin. The cells were cultured in a 5% CO
Cell Immunofluorescence staining
Cells were grown on sterile slide in 24-cm cell culture plates and allowed to attach by overnight incubation, then washed with PBS, fixed with 4% paraformaldehyde for 15 min, and permeabilized with 0.5% Triton X-100 for 10–15 min. Subsequently, the cells were incubated with the rabbit anti-human polyclonal antibody BTBD7 (NBP19652, Novus, dilution1:50) and mouse anti-human monoclonal antibody Slug (ab128485, Abcam, dilution 1:50) for 4
Cell transient transfection
BTBD7 siRNAs (S1, S2, and S3) and control siRNA were obtained from Ribo (RiboBio Co., Ltd, Guangzhou, China). One day before transient transfection, 5*10E4 cells were seeded in 500
Real-time quantitative PCR
The total RNA of the cells that transfected with BTBD7 siRNA and control siRNA (Si-control) extracted using TRIZOL reagent (TaKaRa Co., Ltd, Japan). All samples were reverse transcribed into cDNA using a High Capacity cDNA Archive Kit (TaKaRa Co., Ltd, Japan). The primers (Sangon Biotech, Shanghai) for human were used as follows: BTBD7, forward, 5’-AGTCAAATGCCTGGTTACGG -3’, reverse, 5’-TGTCTGGCACATTGGACATT-3’; Slug, forward, 5’-CGAACTGGACACACATACAGTG -3’, reverse, 5’-CTGAGGATCTCTGGTTGTGGT-3’; MMP9, forward, 5’-GGGACGCAGACATCGTCATC-3’, reverse, 5’-TCGTCATCGTCGAAATGGGC-3’; E-cadherin, forward, 5’-ATTTTTCCCTCGACACCCGA T-3’, reverse, 5’-TCCCAGGCGTAGACCAAGA-3’; and GAPDH forward, 5’-TGTCTGGCACATTGGA CATT-3’, reverse, 5’-GCACCGTCAAGGCTGAGAA C-3’. GAPDH was chosen as the control.
Western blot analysis
Harvested transfected cells were lysed in ice-cold lysis buffer, and then the supernatant was collected after centrifugation at 4
Scratch-wound assay
Transfected Cells were seeded in six-well dishes at 5
Matrigel invasion assay
The assay were performed using a 24-well plate with 8-
MTT assay
The effect of BTBD7 silencing on cell proliferation was detected by3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyltetrazolium bromide (MTT) assay (Sangon Biotech Co., Ltd. Shanghai, China). The transfected cells in 96-well plates examined at 24, 48, 72 hour after adding MTT solution (5 mg/mL) at a final concentration of 0.5 mg/mL followed by incubation for 4 hour at 37
Statistical analysis
Statistical analysis was performed using the SPSS 17.0 software (IBM, Armonk, NY, USA). All experiments were performed in triplicate and the data were expressed as means
Results
Expression of BTBD7 and Slug in SACC-83 and SACC-LM cells
In vitro, BTBD7 and Slug Immunofluorescence staining were detected in two cell lines. Positive BTBD7 expression was mainly in the cytoplasm of two cell lines, positive Slug expression was both in the nuclear and cytoplasm of two cell lines (Fig. 1).
Positive BTBD7 expression was mainly in the cytoplasm of two cell lines; positive Slug expression was mainly in the nuclear and cytoplasm of two cell lines (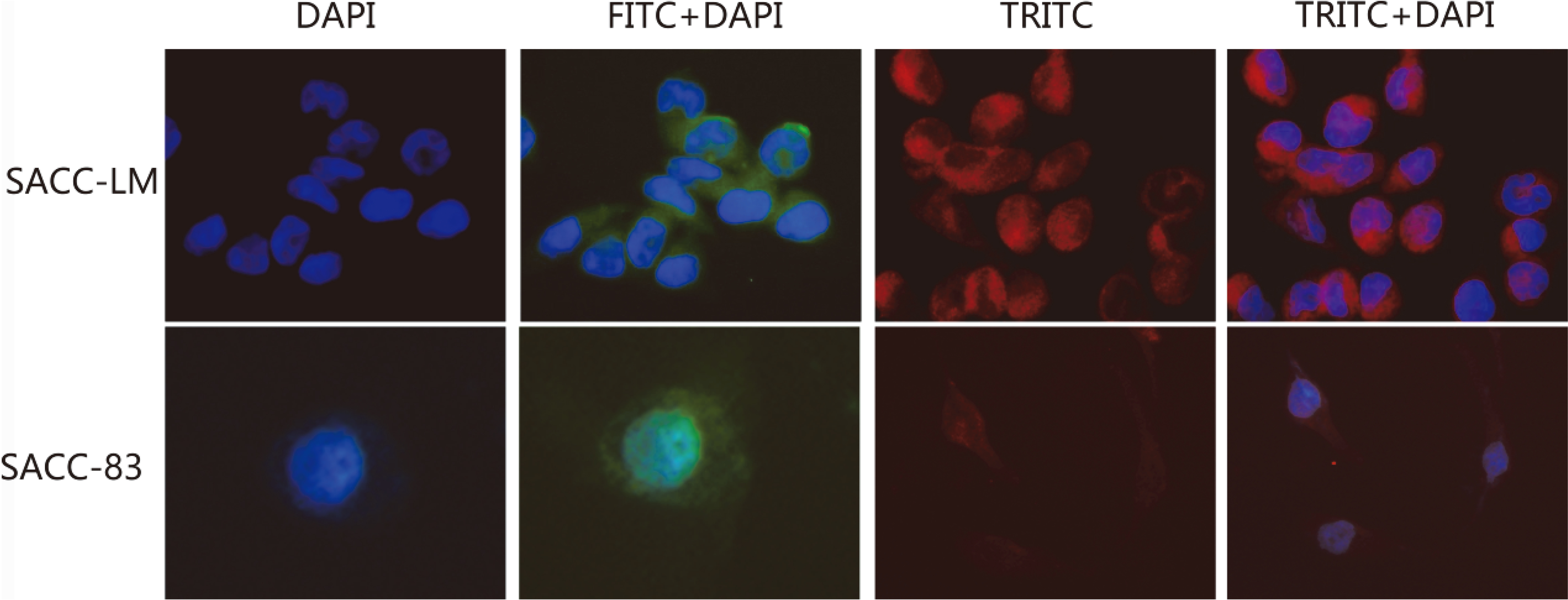
The expression of BTBD7 mRNA of transfected SACC-LM cells with S2 was significantly decreased compared to the SACC-LM cells transfected with S1 or S3 (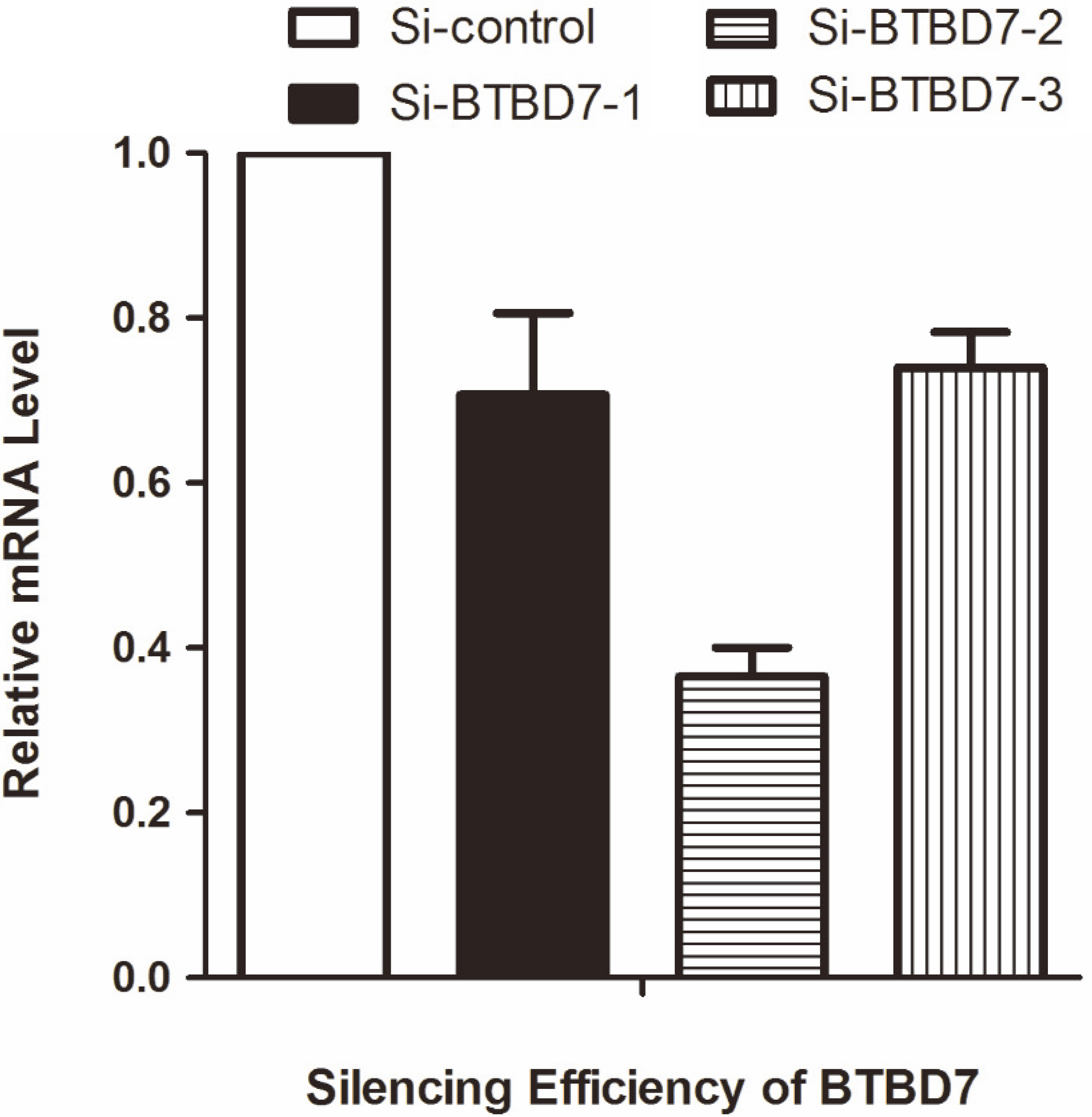
QRT-PCR showed that the expression of BTBD7, Slug and MMP9 mRNA of cells with S2 were significantly decreased meanwhile E-cadherin mRNA was increased compared with control group (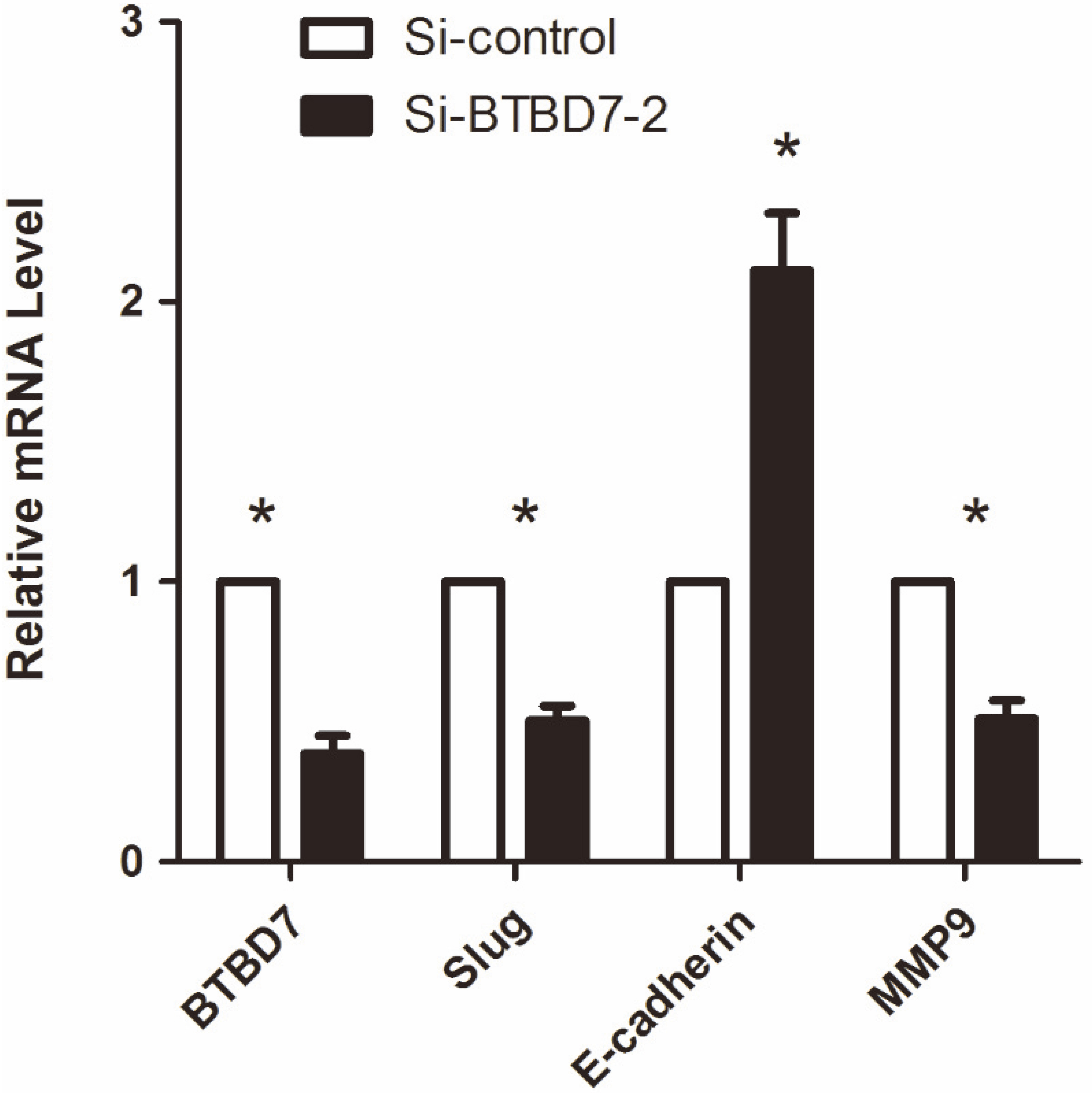
(A) Western blot analysis of BTBD7, Slug, E-cadherin and MMP9 protein levels in Si-control group and Si-BTBD7-2 group; (B) Western blot analysis showed that the expression of Slug and MMP9 protein level of cells with S2 were significantly decreased meanwhile E-cadherin protein level was increased compared with control group (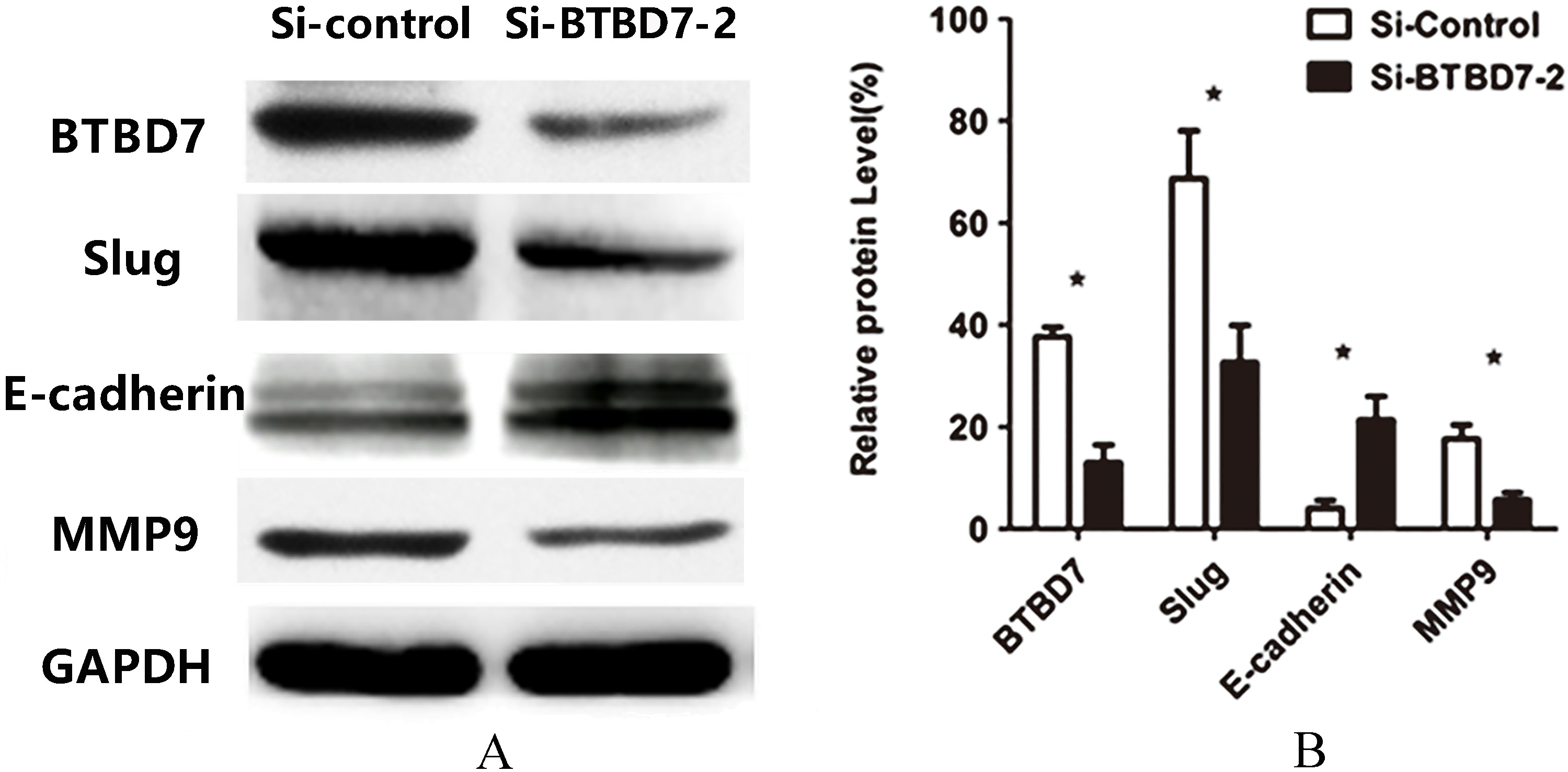
(A) Representative images of scratch test for Si-control group and Si-BTBD7-2 group (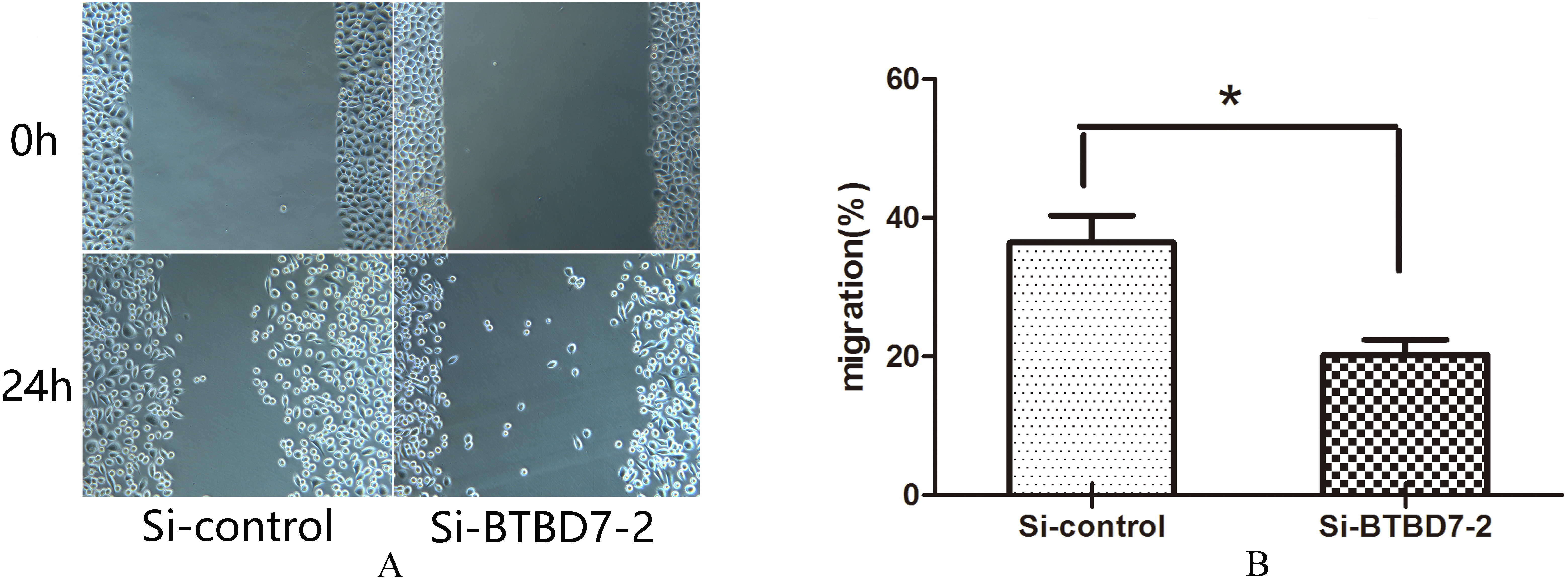
(A) Matrigel invasion assay established the invasion capability of Si-control and Si-BTBD7-2 with representative images (
BTBD7 silencing had no significant effect on proliferation of SACC-LM cells compared with the control group (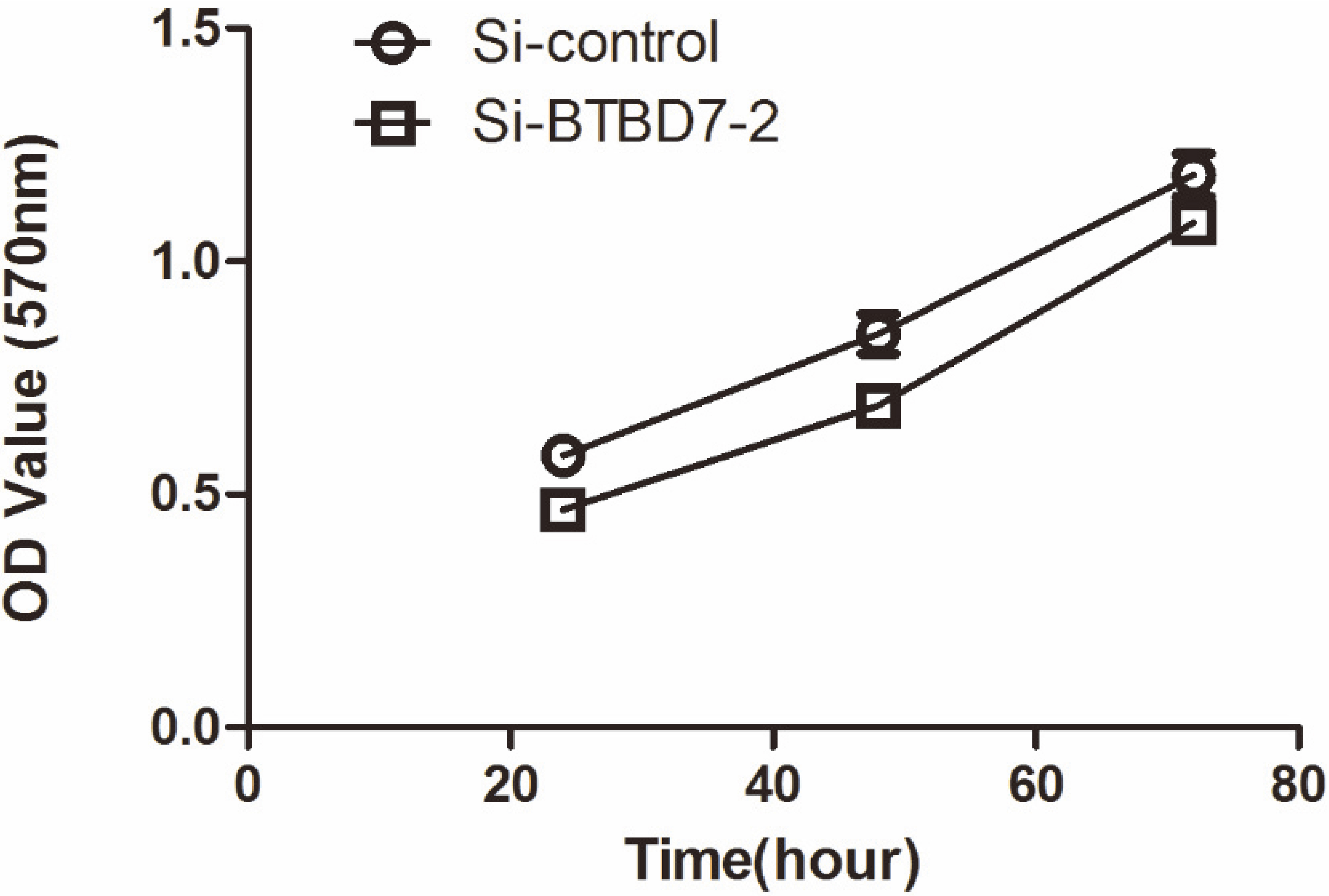
The expression of BTBD7 mRNA of transfected SACC-LM cells with S2 was significantly decreased compared to the SACC-LM cells transfected with S1 or S3 (
Effect of BTBD7 silencing on the mRNA expression of Slug and EMT markers in SACC-LM cells
Having the highest metastatic potential, SACC-LM cell is characterized by specific expression patterns of EMT markers, including E-cadherin, Slug, and MMP9. The expression of BTBD7, Slug and MMP9 mRNA of cells with S2 were significantly decreased meanwhile E-cadherin mRNA was increased compared with control group by qRT-PCR (
Effect of BTBD7 silencing on the protein expression of Slug and EMT markers in SACC-LM cells
Downregulate BTBD7 expression of SACC-LM cells by transfected with S2, Western blot analysis demonstrated significantly upregulated E-cadherin protein expression nevertheless downregulated Slug and MMP9 protein expression in SACC-LM cells (
BTBD7 silencing inhibited cell migration of SACC-LM cells
The results of the scratch test are shown in Fig. 5. After 24 hour of incubation, the width of the scratch area decreased with transfected cells moved to the scratch region. Compared with the control group, silencing of BTBD7 significantly inhibited the cell migration of SACC-LM cells (
BTBD7 silencing inhibited invasion ability of SACC-LM cells
The results of Matrigel invasion assay are shown in Fig. 6. After 24 hour of incubation, cells that invaded through the Matrigel and presented at the lower surface of the polycarbonate membrane were stained and counted. Compared with the control group, silencing of BTBD7 significantly inhibited the invasion ability of SACC-LM cells (
BTBD7 silencing had no significant effect on proliferation of SACC-LM cells
As shown in our data, BTBD7 silencing had no significant effect on proliferation of SACC-LM cells compared with the control group (Fig. 7) (
Discussion
BTB/POZ domain-containing protein 7 (BTBD7) as a critical regulator of cleft formation during in vitro branching morphogenesis, which functions via the down-regulation of E-cadherin and stimulation of epithelial cell motility, BTBD7 also promotes an epithelial-to-mesenchymal transition (EMT) in cultured tumor cells [17]. In our previous study, uncommon overexpression of BTBD7 in SACC was significantly related to malignant behavior of tumors and patients’ poor prognosis [16]. Therefore, in the present study, the molecular mechanisms of BTBD7 promoting invasion and metastasis in SACC were explored in vitro crucially.
Here, by downregulating of BTBD7 using BTBD7 siRNA, we found that downregulation of BTBD7 significantly inhibited the migration and invasion ability of the SACC-LM cells examined by Scratch-wounding assay and Matrigel invasion assay. These results indicated that BTBD7 may contribute to SACC-LM cell migration and invasion, which was consistent with the previous results [16] of tumor metastasis and patients’ poor clinical outcome. We also found that downregulation of BTBD7, could cause a rapid regression of EMT features. Downregulation of BTBD7 in SACC-LM cells significantly inhibited MMP9 expression and promoted E-cadherin expression that were associated with enhanced cell motility, invasive phenotypes, and consequently, metastasis in human malignancies [18]. The experimental results showed in molecular level that BTBD7 involved in EMT, contributed to malignant behavior of cancer cell, and was consistent with the results in vivo that distal metastasis and patients’ poor prognosis.
Slug which acts as a transcriptional repressor was over-expressed in various cancers [5, 6, 7, 8, 19] and has been shown to associate with a broad spectrum of biological functions in tumor EMT [20]. Our previous immunohistochemistry study [16] showed the overexpression of BTBD7 in SACC was significantly correlated with Slug expression which may imply a signaling pathway. Thus we used siRNA to downregulate BTBD7 expression in SACC-LM cell in vitro to investigate its possible relation. Our cytological experiments showed that downregulation of BTBD7 significantly inhibited the expression of Slug in SACC-LM cell. Other study has shown that Slug silencing upregulated the E-cadherin expression in the PNI process of SACC [9]. We found that BTBD7 depletion induced E-cadherin expression, but restrained Slug and MMP9 expression in SACC-LM cell, these results suggested that BTBD7 may be involved in SACC cancer cell through regulating Slug, which in turn induced overexpression of the MMP9 and suppresses E-cadherin levels, thereby altering cell morphology and reducing cancer cell invasion and metastasis. However, MTT assay revealed that down-regulation of BTBD7 had no significant effect on proliferation of SACC-LM cell although BTBD7 overexpression was previously shown to promote Bel7404 cell proliferation [21] but was ineffective in HCCLM3 cells proliferation [14]. Different results may due to different experimental conditions and tumor cells, this data also implied that BTBD7 may play complex functions in various tumors. Although we considered BTBD7 as an upstream activator of EMT, due to the limitations, further animal experiments and studies concerning deep and more regulatory mechanisms are still required to reveal the function of BTBD7 in SACC.
Conclusions
Following our previous study, the current research explored the function and molecular regulation mechanism of BTBD7 in SACC. Our findings suggested that BTBD7 was crucial for the migration and invasion of SACC-LM cells. We further demonstrated that downregulation of BTBD7 inhibited the EMT through downregulation of Slug in SACC. These results may provide experimental evidence in improving detection and treatment of SACC by targeting potential cancer biomarkers.
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Acknowledgments
This research is supported by Shandong Provincial Science and Technology Development Plan (No. 2014GGH218024), and Shandong Provincial Science and Technology Development Plan (No. 2010GSF 10239).
