Abstract
BACKGROUND:
Several investigations have demonstrated that follistatin-like 1 (FSTL1) is implicated in the initiation and progression of diverse cancers. It remains unclear whether FSTL1 acted as a cancer-promoting gene through its overexpression in HCC.
PATIENTS AND METHODS:
We detected FSTL1 protein expression in 210 consecutive HCC cases curatively resected in our hospital between 2004 and 2007. The correlation between FSTL1 expression in HCC tissues and post-surgical prognosis of HCCs was analyzed. The in vitro experiments including apoptosis assessment, MTT, BrdU incorporation ELISA assay, Western immunoblotting, and qRT-PCR were performed to determine the impact of FSTL1 on apoptosis and proliferation abilities of HCC cells and the relevant mechanisms.
RESULTS:
FSTL1 protein was found aberrantly increased in 172 of 210 HCC tissues (81.9%) compared to adjacent liver tissues. FSTL1 overexpression was apparently associated with larger tumor size, advanced TNM staging, portal vein invasion, intra-hepatic metastases. Patients with higher FSTL1 expression in tumors suffered from the worse overall survival rate as assessed by comparison of Kaplan-Meier survival curves. Higher FSTL1 expression in HCC tissues was identified as a independent poor post-surgical prognostic predictor for HCC. Silencing FSTL1 by siRNA promoted cell apoptosis and leaded to suppression of cell viability and proliferation in MHCC97h cells. Furthermore, enforced expression of FSTL1 obtained the opposite results in Huh7 cells. Mechanistic investigation showed that FSTL1 repressed HCC cell apoptosis through AKT/GSK-3
CONCLUSION:
These data proved that FSTL1 contributed to unfavorable post-surgical outcome of HCC patients via inhibiting cell apoptosis.
Abbreviations
Introduction
Hepatocellular carcinoma (HCC), which is one of the most aggressive malignant cancers, has been the second most leading cause of cancer-related deaths in males and the sixth in females because of its high-risk post-surgical recurrence [1, 2]. Despite the tremendous advancement has been achieved by physicians and researchers on diagnostic and innovative therapy strategies for HCC during decades, the prognosis of most HCC cases remains very poor with a 5-year survival of 11% [3, 4]. Moreover, the incidence of HCC remains increasing in the last decade [5]. Herein, it is imperative to explore the underlying molecular mechanisms of HCC progression, establish the novel diagnostic markers, and develop the effective therapy regimen.
Follistatin-like protein 1 (FSTL1), also known as TSC-36 (TGF
To the best of our knowledge, no investigation was performed to detect the expression of FSTL1 in HCC tissues and identify its role on HCC progression. In the present study, we found that FSTL1 was over-expressed frequently in HCC tissues in contrast to adjacent liver tissues, which was correlated with the worse post-surgical prognosis of HCC positively. The further experiments revealed that FSTL1 functioned as a pro-oncogenic factor on HCC progression via inhibiting tumor cell apoptosis in a AKT/GSK-3
Demographic information and clinical characteristics of 210 HCC patients
Demographic information and clinical characteristics of 210 HCC patients
Patients and tissue samples
The present study was carried out with the approval of the Ethical and Scientific Committees of Xi’an Jiaotong University according to the Helsinki Declaration of 1975. HCC and adjacent liver tissues recruited in this investigation were from 210 patients diagnosed with HCC. All HCC patients received hepatectomy for HCC in Department of Hepatobiliary Surgery at the First Hospital of Xi’an Jiaotong University from January 2004 to June 2007. Written informed consent was obtained from all patients. Diagnosis of HCC was identified by a experienced pathologist according to the criteria of the Chinese Society of Liver Cancer and Chinese Anti-Cancer Association. All patients included in this study had not received preoperative radiotherapy, chemotherapy, immunotherapy, TACE or target therapy before liver resection. Specimens of both HCC and adjacent liver tissues were achieved during surgery and kept in paraformaldehyde for the further assessment. The study involved the following clinicopathological parameters: age, gender, serum AFP level, HBV infection, tumor diameter, liver cirrhosis, Edmondson-Steiner Classification, TNM stage, portal vein invasion, and intra-hepatic metastases, which were obtained from the medical records. The histopathologic information was confirmed by two experienced pathologists. The follow-up information was gained from 175 of 210 HCCs (83.3%) and the median follow-up time was 42.5 months (from 3 to 75.4 months). The demographic and clinicopathological characteristics are presented in Table 1.
Immunohistochemistry staining assay
Immunohistochemistry (IHC) staining was cond- ucted as described in our previous study [14]. Both HCC tissues and adjacent liver tissues were formalin-fixed, paraffin-embedded and cut into 4
Cell culture
The human hepatocellular carcinoma cell lines Hep3B, SK Hep1 and HepG2 were obtained from Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences (Shanghai, China) and MHCC97h were from Fudan University (Shanghai, China). Prof. Kefeng Dou (Department of Hepatobiliary Surgery, Xijing Hospital, Fourth Military Medical University) provided Huh7 cells as a kind gift. All HCC cells were grown as monolayer in DMEM or RPMI medium (Invitrogen, Carlsbad, CA) with 10% FBS, and maintained in a humidified 5% CO2 incubator at 37
Establishment of FSTL1 stable transfectant clones
FSTL1 expressing plasmid (Origene, Beijing, China) and pCMV6-Entry control plasmid were transfected into Hep3B cells using TurboFect transfection reagent, respectively. After 2-week selection with Geneticin (500 g/mL), we got FSTL1 stable transfectant clones (Hep3B FSTL1 cells) and control stable transfectant clones (Hep3B Vector cells).
Establishment of FSTL1 knockdown cells
siRNA sequences targeting FSTL1 and the scramble siRNAs were both from Sigma-Aldrich (St. Louis, MO,USA). MHCC97H cells were plated at the concentration of 0.2
Quantitative reverse-transcription-polymerase chain reaction (qRT-PCR)
Total RNA was extracted from HCC cells using Trizol reagent (Invitrogen, USA) according to the suggested manufacturer’s protocol. the PrimeScript RT Master Mix (TaKaRa, Osaka, Japan) were used for converting mRNA to cDNA. Expression levels of FSTL1 mRNA were detected by SYBR Green qRT-PCR with the following primers: FSTL1: Forward 5’-C CAGAACTATGATAATGGAGACGCT-3’, Reverse 5’-TAAGATGAACTATGAACCTCCTGCC-3’; GAPDH: Forward 5’-GAAGGTGAAGGTCGGAGTC-3’, Reverse 5’-GAGATGGTGATGGGATTTC-3’. For each sample, the relative FSTL1 mRNA level was normalized to GAPDH expression.
Western immunoblotting
Western immunoblotting was carried out as described previously [15]. Briefly, HCC cells were lysed using RIPA protein extraction reagent mixed with PMSF. 30
Cell viability and proliferation assessment
The MTT assay was carried out to determine cell viability at 24, 48, 72 and 96 h and BrdU incorporation was detected by ELISA assay to assess the impact of FSTL1 on HCC cell proliferation. HCC cells were inoculated into 96-well plate at the concentration of 1
Detection of HCC cell apoptosis
Cell apoptosis was examined by both Annexin V-FITC/PI double staining assay and Caspase 3/7 activity assessment. For Annexin V-FITC/PI double staining assay, approximately 1
Statistical analysis
All data were presented as the mean
Results
Aberrant FSTL1 over-expression in HCC tissues was found frequently and associated with the worse prognosis of HCC patients after liver resection
FSTL1 protein expression was detected by IHC staining in the HCC tissues and adjacent liver tissues from 210 HCC patients. As shown in Fig. 1(A), FSTL1 protein located predominantly in cytoplasm. And FSTL1 expression was increased in 172 of 210 HCC tumor tissues (81.9%) compared to adjacent liver tissues. Mann-Whitney U test verified that IHC score of FSTL1 protein in HCC tissues was significantly higher than that in adjacent liver tissues (
Univariate and multivariate analyses of predictive factors in HCC patients with liver resection
Univariate and multivariate analyses of predictive factors in HCC patients with liver resection

FSTL1 was found increased frequently in HCC tissues, which was associated with poor prognosis of HCC patients after liver resection. A. the FSTL1 IHC staining in adjacent liver tissues (a) was stronger apparently than that in HCC tissues (b); B. Mann-Whitney U test revealed that the score of FSTL1 IHC staining in HCC tissues was significantly higher that one in adjacent liver tissues; C. Comparison of the Kaplan-Meier survival curves demonstrated that patients with higher FSTL1 expression in HCC tissues (higher FSTL1 group) had the worse post-surgical prognosis compared to those with lower FSTL1 expression in HCC tissues (lower FSTL1 group).
As shown in Table 2, univariate analyses demonstrated that the unfavorable predictive factors consisted with higher FSTL1 expression in HCC tissues, advanced TNM staging, portal vein invasion, as well as intra-hepatic metastases. Furthermore, higher FSTL1 expression in HCC tissues, portal vein invasion, and intra-hepatic metastases were established as the independent post-surgical prognostic factors for HCC patients by multivariate analysis. These data implied strongly that FSTL1 promoted HCC progression potentially. To confirm the oncogenic function of FSTL1 on liver carcinogenesis and explore the underlying mechanism.
As shown in Fig. 2(A), we examined FSTL1 expression in 5 kinds of HCC cell lines by both qRT-PCR and Western immunoblotting assays and found that MHCC97h cells expressed the highest level of FSTL1 among all 5 kinds of HCC cell lines. Transfection with siRNA sequences against FSTL1 resulted in a sharp decrease of FSTL1 expression in MHCC97h cells (Fig. 2(B)). Annexin V-FITC/PI double staining assay showed that knockdown of FSTL1 (MHCC97h FSTL1 siRNA cells) reinforced tumor cell apoptosis apparently (Fig. 2(C)). And Caspase 3/7 activity measurement also found that FSTL1 deletion expedited cell apoptosis of MHCC97h cells (Fig. 2(D)). As assessed by MTT, cell viability of MHCC97h cells was attenuated by getting rid of FSTL1 expression (Fig. 2(E)). Consistently, cell proliferation of MHCC97h cells were found inhibited after silencing FSTL1 expression by BrdU incorporation examination (Fig. 2(F)). Therefore, it demonstrated that FSTL1 contributed to suppressing cell apoptosis and accelerating cell growth in HCC cells.
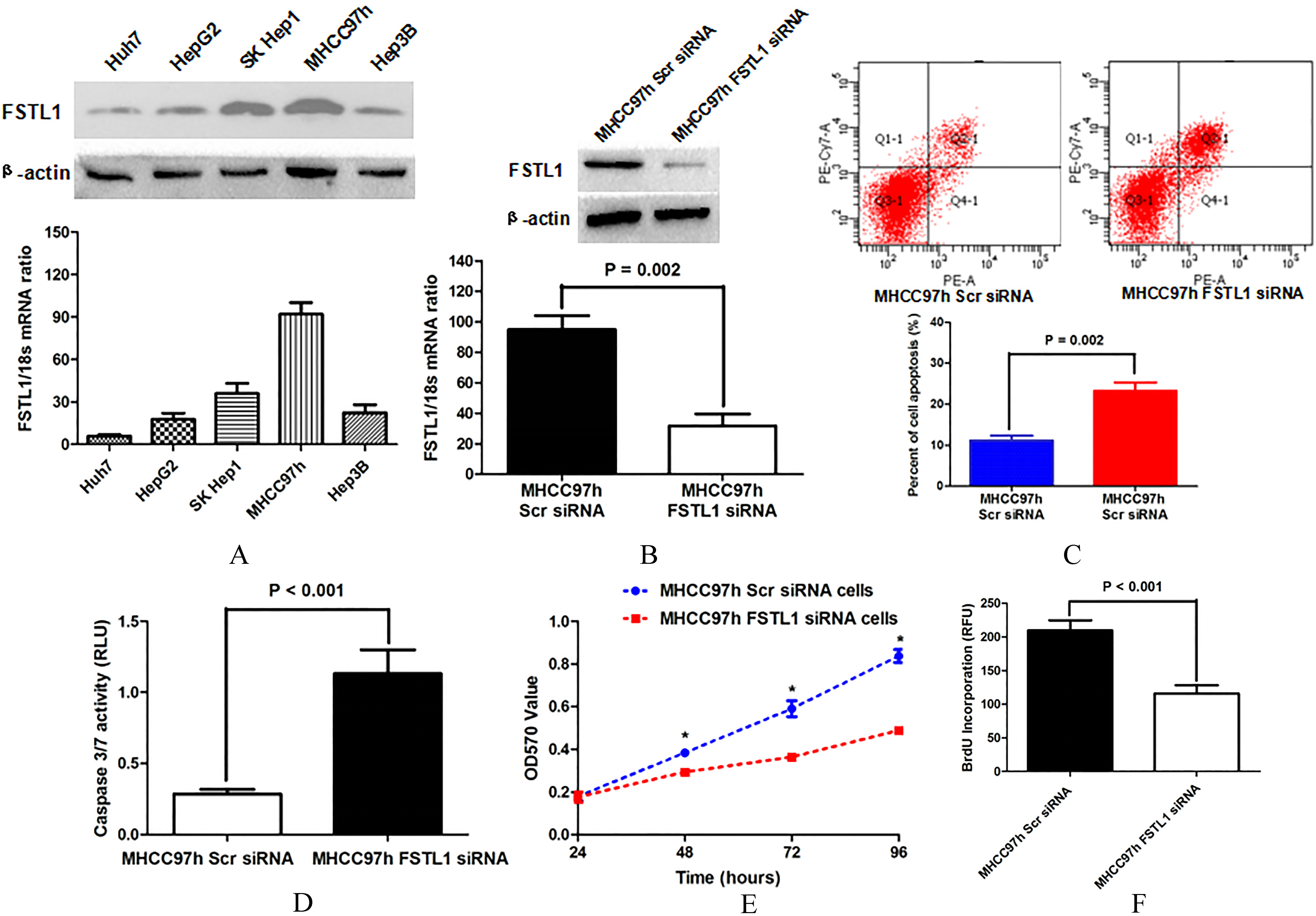
Knockdown of FSTL1 in MHCC97h cells accelerated cell apoptosis and promoted cell viability and proliferation. A. MHCC97h cells expressed the highest level of FSTL1 among 5 kinds of HCC cell lines, whereas Huh7 cells had the lowest level of FSTL1, as assessed by both qRT-PCR and Western immunoblotting; B. Both qRT-PCR and Western immunoblotting verified that siRNA sequences inhibited FSTL1 expression in MHCC97h cells notably; C. Annexin V-FITC/PI double staining assay displayed that there were more apoptotic tumor cells in MHCC97h FSTL1 siRNA cells than MHCC97h Scr siRNA cells; D. ELISA assessment showed that the MHCC97h FSTL1 siRNA cells had significantly higher Caspase3/7 activity than MHCC97h Scr siRNA cells; E. MTT assay displayed that the cell viability of MHCC97h cells was remarkably restrained by FSTL1 silencing at 48, 72 and 96 h; F. ELISA assay showed that MHCC97h FSTL1 siRNA cells possessed less BrdU incorporation than MHCC97h Scr siRNA cells notably.
As shown in Fig. 2(A), wild type Huh7 cells had the lowest FSTL1 expression among the all 5 HCC cells. Thus, we enhanced the expression of FSTL1 in Huh7 cells by transfecting with FSTL1 expressing plasmid, which was validated by both qRT-PCR and Western immunoblotting (Fig. 3(A)). Huh7 Vector cells had the higher Caspase 3/7 activity than Huh7 FSTL1 cells (Fig. 3(B)). And more apoptotic cells were found in Huh7 Vector cell group than Huh7 FSTL1 cell group during Annexin V-FITC/PI double staining assay (Fig. 3(C)), which illuminated that FSTL1 over-expression lead to suppression of Huh7 cell apoptosis. MTT assay revealed that cell viability of Huh7 FSTL1 cells was significantly higher than one of Huh7 Vector cells (Fig. 3(D)). BrdU incorporation examination showed that enhanced expression of FSTL1 promoted cell proliferation of Huh7 cells (Fig. 3(E)). Thus, the present results further illustrated that FSTL1 overexpression was involved closely in HCC progression.

Enforced expression of FSTL1 restrained cell apoptosis and increased cell viability and proliferation. A. Both qRT-PCR and Western immunoblotting assays confirmed that FSTL1 expressing plasmid transfection leaded to up-regulation of FSTL1 expression in Huh7 cells; B. As examined by ELISA assay, Huh7 Vector cells had significantly more Caspase3/7 activity than Huh7 FSTL1 cells; C. Annexin V-FITC/PI double staining assay also demonstrated that Huh7 FSTL1 cells had the lower percentage of apoptotic cells in contrast to Huh7 Vector cells; D. MTT assay displayed that cell viability of Huh7 FSTL1 cells were obviously stronger than one of Huh7 Vector cells at 48, 72 and 96h, respectively; E. BrdU incorporation of Huh7 cells was found up-regulated by FSTL1 overexpression, as measured by ELISA assay.
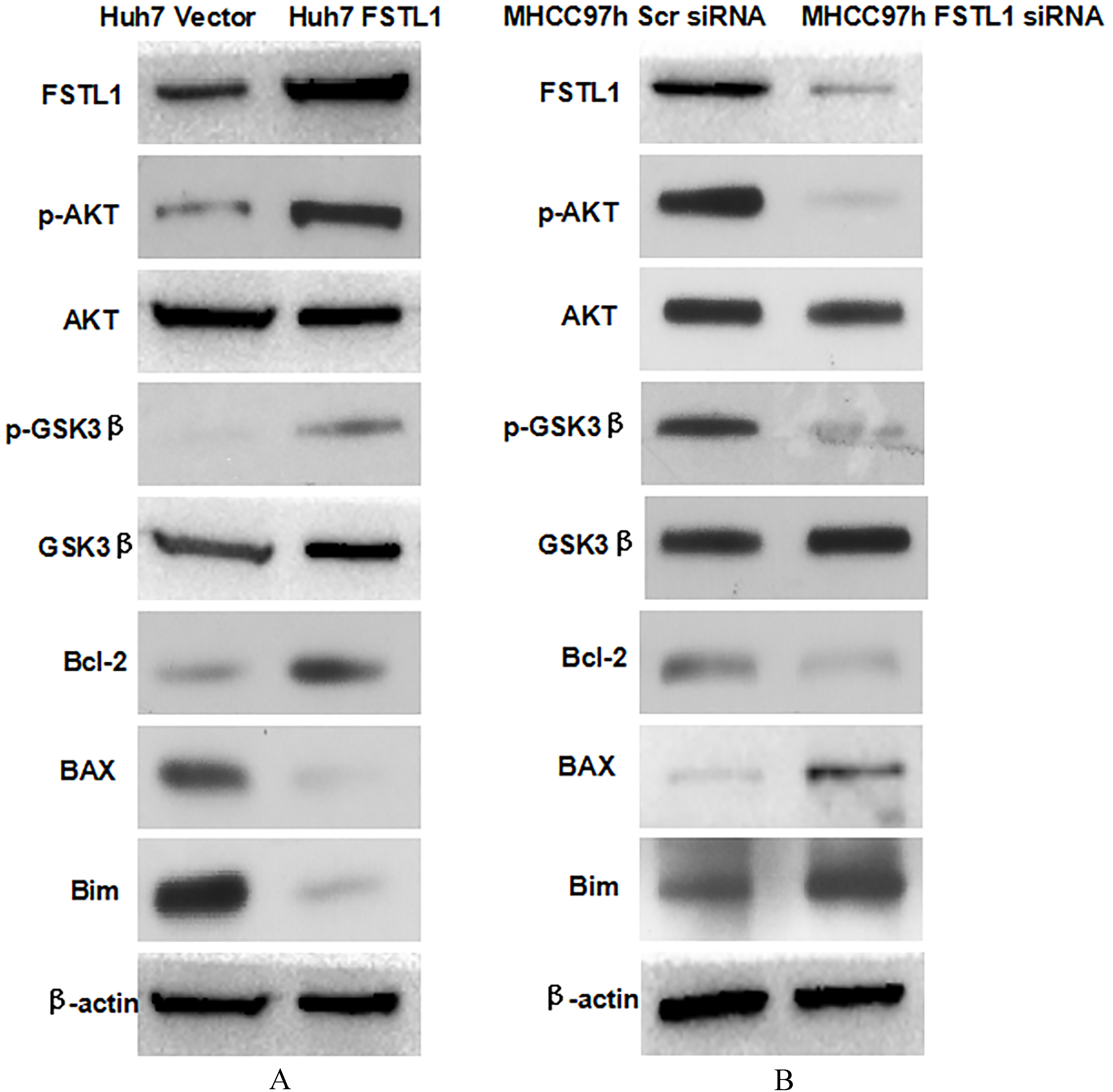
FSTL1 up-regulated Bcl-2 expression and inhibited the expression of BAX and Bim simultaneously via activating AKT/GSK3
To determine the mechanism that FSTL1 inhibited HCC cell apoptosis, we examined whether FSTL1 mediated AKT/GSK-3
Discussions
FSTL1 was found as a secreted glycoprotein which widely expressed in various human tissues [16, 17]. It was characterized by the presence of a follistatin-like domain with the similar sequence to the endometrial protein follistatin [18]. Previous study revealed that follistatin-like protein 1 promoted follistatin binding and inactivating activin which is a member of the TGF-b family. However, the role of FSTL1 on human pathology remain unclear. Several investigation displayed that FSTL1 was involved closely in human inflammatory diseases via regulating multiple signal pathway. Li group reported that serum FSTL1 were notably up-regulated in patients with different inflammation diseases, including rheumatoid arthritis, ulcerative colitis, systemic lupus erythematosus, Sjogren’s syndrome, systemic sclerosis, and polymyositis [19]. FSTL1 was found to mediate negatively proinflammatory cfos and IL-6, while repressing the expression of matrix metalloproteinases and prostaglandin E2 in synovial fluid of rheumatoid arthritis [20, 21]. FSTL1 was found by Gorelik group to be aberrantly over-expressed in acute macrophage activation syndrome (MAS) and treatment of MAS decrease its expression to the normal level [22].
There were different results about the role of FSTL1 on the progression of various cancer. Wilson group revealed that FSTL1 was up-regulated in prostate cancer and play a critical role during androgen receptor promoting prostate cancer progression [11]. Yoon reported that FSTL1 facilitated cellular proliferation and inhibited cell apoptosis in lung cancer cells via increasing phosphorylation of Erk1/2 [13]. Kawakami et al. reported that FSTL1 expedited bone metastasis of breast cancer by driving immune incapacity [12]. However, there were also some investigations finding out that FSTL1 exerted the anti-tumor function in diverse cancers. FSTL1 promoter was found frequently hypermethylated in nasopharyngeal carcinoma cells and clinical tumor biopsies, which leaded to down-regulation of FSTL1 in nasopharyngeal carcinoma by Zhang group. The further investigation revealed that FSTL1 down-regulation resulted in dysfunctional innate responses to surrounding macrophages which accelerating nasopharyngeal carcinoma immune evasion, though repression of FSTL1 inhibited the proliferation and migration of nasopharyngeal carcinoma cells [23]. It seemed that FSTL1 inhibited carcinogenesis of nasopharyngeal carcinoma via mediating tumor immune positively. Zhang et al. found by gene expression microarray analysis that FSTL1 up-regulation was closely associated with inhibition of the invasion and metastasis ability of pulmonary giant cell carcinoma cells [24]. The study about ovarian and endometrial cancer reported that FSTL1 functioned as a tumor suppressor through inducing cell apoptosis [25].
In this study, we found for the first time that FSTL1 was frequently up-regulation in HCC tissues in contrast to adjacent liver tissues. And overexpression of FSTL1 in HCC tissues was related significantly with unfavorable clinical features including larger tumor diameter, advanced TNM stage, portal vein invasion, and intrahepatic metastasis. Analysis of the follow-up information displayed that HCC patients with higher FSTL1 expression in HCC tissues had the worse post-surgical outcome. Multivariate analysis by Cox proportional hazards model identified higher FSTL1 expression in tumor tissues as the independent predictive biomarker for HCC patients. Next, we sought to address whether FSTL1 played a critical role on HCC progression and could be a candidate therapy target for HCC recurrence. Silencing FSTL1 expression by siRNA sequences induced cell apoptosis and inhibited cell viability and proliferation capacities of MHCC97h cells. On the other hand, enhanced expression of FSTL1 in Huh7 cells repressed cell apoptosis and strengthened cell viability, proliferation and colony-formation ability. These data supported strongly that FSTL1 promoted HCC cell growth via restraining cell apoptosis. The further mechanistic study showed that FSTL1 increased Bcl-2 expression and down-regulated BAX and Bim via inducing hyperactivation of AKT/GSK-3
Taken together, this was the first report to demonstrate that FSTL1 was frequently up-regulated in HCC tissues compared to the adjacent liver tissues and accelerated HCC progression via inhibiting cell apoptosis in the AKT/GSK-3
Footnotes
Acknowledgments
This study was supported by grants from National Natural Scientific Foundation of China (81301743 and 81572733 to Xin Zheng), Research Fund for the doctoral Program of High Education of China from Ministry of Education (No. 20120201120090 to Xin Zheng), Key Science and Technology Program of Shaanxi Province (No. 2014K11-01-01-21 to Xin Zheng) and the Fundamental Research Funds for the Basic Research Operating Expenses Program of Central College sponsored by Xi’an Jiaotong University to Xin Zheng.
Conflict of interest
No conflicts of interest exist.
