Abstract
Background
Sarcopenia has been associated with poor outcomes in patients with cirrhosis and solid tumours.
Objective
Analyse the influence of sarcopenia on survival and treatment duration in patients with advanced hepatocellular carcinoma (HCC) treated with sorafenib.
Methods
We conducted a multicentre, retrospective study on 96 patients with advanced HCC treated with sorafenib, all with available abdominal computed tomography (CT) scan within 30 days from treatment start. Anthropometric, laboratory, treatment and follow-up data were collected. Sarcopenia was defined by reduced skeletal muscle index calculated from an L3 section CT image.
Results
Sarcopenia was present in 49% of patients. Patients were divided into two groups according to sarcopenia: age was significantly higher in the sarcopenic group (SG) (66 years (31–87) versus 72 years (30–84), p = 0.04], with no difference in other baseline characteristics. The SG showed shorter overall survival (OS) (39 (95% confidence interval (CI) 26–50) versus 61 (95% CI 47–77) weeks (p = 0,01)) and shorter time on treatment (12.3 (95% CI 8–19) versus 25.9 (95% CI 15–33) weeks (p = 0.0044)). At multivariate analysis, sarcopenia was independently associated to reduced OS (p = 0.03) and reduced time on treatment (p = 0.001).
Conclusion
Sarcopenia is present in almost half of patients with advanced HCC, and is associated with reduced survival and reduced duration of oral chemotherapy.
Key summary
Sarcopenia has been associated with poor outcomes in patients with cirrhosis and hepatocellular carcinoma (HCC), and with reduced tolerance to chemotherapy. Our study indicates that sarcopenia is present in almost half of cirrhotic patients with advanced HCC selected to undergo sorafenib treatment. Sarcopenia was found to be independently associated with a twofold risk of mortality and reduced sorafenib treatment duration. Baseline measurement of sarcopenia could improve prediction of mortality in patients with advanced HCC.
Introduction
Hepatocellular carcinoma (HCC) is the third cause of cancer related deaths, with rising incidence worldwide, with nearly 750,000 new cases per year. 1 Epidemiological data suggest that the incidence of HCC is still similar to the death rate, and thus almost all patients who are diagnosed with HCC will die because of it. 1 In developed countries, HCC predominantly affects the elderly, and is diagnosed at an advanced stage (Barcelona Clinic Liver Cancer (BCLC-C)) in up to 40% of all patients. 2 At this stage, the only approved treatment in Italy is sorafenib (Nexavar®, Bayer), an orally administered multikinase inhibitor shown to improve survival, but burdened by relatively high costs and toxicity rates, limiting patients’ chances of receiving full dose treatment and hindering their quality of life. 3
Therefore, the search for prognostic tools able to stratify risk, predict toxicity and allow for therapy tailoring for each patient is of major clinical interest. 4
The condition of reduced muscle mass, defined as sarcopenia,5,6 has been identified as an independent predictor of reduced survival, tolerance to chemotherapy and higher rates of toxicity in patients with different neoplasias.7–10 In the research setting, imaging techniques such as computed tomography (CT) scanning are considered the gold standard for estimating muscle mass. 6 In the field of hepatology, sarcopenia has been linked to reduced survival rates in different therapeutic settings such as liver transplantation, hepatic resection and intra-arterial treatments for HCC.11–15 Its presence has also been associated with increased toxicity in patients with HCC treated with sorafenib 16 and reduced survival, but the few data available come mainly from oriental series, where ethnic and anthropometric differences limit reproducibility in a Western setting.17–19 Only one Western study is available, with a limited number of patients treated with various tyrosine kinase inhibitors. 20
The aim of the present study was to identify the prevalence and influence of sarcopenia in a homogeneous population of European Caucasian patients with advanced HCC undergoing sorafenib therapy. We aimed to evaluate the influence of sarcopenia on patients’ survival, as well as on the duration and tolerance of sorafenib treatment.
Materials and methods
Study design
This is a multicentre, retrospective study to assess the added value of sarcopenia evaluation in patients with advanced HCC and patients with an intermediate HCC who were not eligible for or failed ablative therapies, treated with sorafenib according to international guidelines. 2 Patients were followed and included in four different units in three hospital centres from 2008 to 2016.
Patients and data collection
All patients with advanced HCC, i.e. classified as BCLC stage C and those with a BCLC-B stage who were unfit to any or failed to respond to locoregional therapies, 2 were enrolled. HCC was diagnosed by the criteria of the American Association for the Study of Liver Disease 21 and staged by abdominal dynamic contrast-enhanced CT scan or gadolinium-enhanced magnetic resonance imaging (MRI). Each patient underwent also chest X-ray/CT scan and bone scanning as requested by the attending physicians.
The main inclusion criteria were those specified by the Italian Medicines Agency (AIFA) and the availability of an abdominal CT scan within 1 month before the start of sorafenib treatment. We analysed gender, age, pre-treatment anthropometric features, laboratory findings (including serum albumin, creatinine, international normality ratio (INR) and alpha-fetoprotein (AFP)), dry weight body mass index (BMI), macrovascular invasion and extra-hepatic spread. Cirrhosis, diagnosed either by histology or clinically, was graded according to the Child–Pugh and Model for End-stage Liver Disease (MELD) scores as a measures of liver impairment.22,23 Performance status (PS) was scored according to the Eastern Cooperative Oncology Group (ECOG). 24 All data were obtained within 1 month before the start of sorafenib treatment. Follow-up was ended at patients’ death, last visit or on 10 December 2016.
After initial data collection, all patients were given an identification number to guarantee anonymity and information was entered in a dedicated database for statistical analysis. All patients gave informed consent for the use of clinical, laboratory and radiological data at the time of the first examination. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by local the institutional review board (Comitato Etico Milano Area 2, Università degli Studi di Milano, 3 December 2017).
Image analysis and treatment modality
CT scans were obtained routinely at baseline for tumour staging purposes within 1 month prior to initiation of sorafenib treatment and used to determine the skeletal muscle index (SMI). A transverse image at the level of the third lumbar vertebrae (L3) was selected from each scan. Muscles at this level include psoas, erector spinae, quadratus lumborum, transversus abdominis, external and internal obliques, and the rectus abdominis.10,13,25 Images were analysed with SliceOmatic V 5.0 software (Tomovision, Montreal, Quebec, Canada), which enables specific tissue selection by using previously determined Hounsfield units (HU). Skeletal muscle is selected and quantified by thresholds between –29 to +150 HU. 26 These specific thresholds permit evaluation of skeletal muscle regardless of ascites in patients with cirrhosis, excluding muscular fat infiltration in the selected section. All CT images were analysed by two trained observers (GA and PB). Cross-sectional areas were calculated and normalized for height (cm2/m2) as reported elsewhere. 27 The MRI imaging data were excluded to avoid methodological heterogeneity. L3 SMI was expressed in cm2/m2. L3 SMI was considered to be indicative of sarcopenia if ≤53 cm2/m2 for male patients with a BMI ≥25 and ≤43 cm2/m2 in male patients with a BMI <25 and ≤41 cm2/m2 for women, independently of BMI. 9
Sorafenib treatment was administered according to guidelines. Therapy was discontinued whenever the patient developed unacceptable toxicity, when radiological or symptomatic progression of HCC occurred, or by investigator’s judgment of the patient being unlikely to benefit from further treatment with sorafenib. Tumour progression was defined either radiologically by modified response evaluation criteria in solid tumours (mRECIST) 28 or clinically in terms of worsening of PS or onset of symptoms unrelated to liver failure. 29 Another reason for treatment discontinuation was unacceptable treatment toxicity, i.e. grade 2–4 adverse events (AEs) not responding to dose reductions and/or temporary interruption of treatment as suggested by the manufacturer.24,30 Toxicity was assessed in all patients who received at least one dose of sorafenib; AEs were graded according to the National Cancer Institute’s Common Terminology Criteria (version 3.0). 31
Statistical analysis
Patients were divided in two groups according to the presence of sarcopenia and characteristics of the two groups were then compared. Continuous data were reported as mean ± SD or as median (range) and compared using a Student’s t-test or Mann–Whitney test as appropriate. Categorical variables were expressed as number (percentage) and compared using the χ2 test with Fisher’s exact test, with Yates’ correction as appropriate. Survival curves were estimated using the Kaplan–Meier method and compared using the log-rank test. Overall survival (OS) time was defined as the period between treatment start and patient death, and the time on treatment was intended as time from treatment beginning to its end. Univariate analyses were performed using Cox’s proposal hazards model and hazard ratios (HRs) of independent predictive factors were obtained. The variables significantly associated with endpoints in univariate analysis were entered in a sex- and age-adjusted multivariate model. All p-values were two sided and the level of significance was set at p < 0.05. Results are reported as HR and 95% confidence interval (CI). All statistical analyses were carried out on MedCalc® 12.5 statistical software. Data are expressed as percentage (number/total) median (range) for demographic and laboratory data in tables and text unless otherwise indicated.
Results
Clinical and biochemical characteristics of advanced HCC patients treated with sorafenib
Baseline clinical and biochemical characteristics of 96 patients with advanced hepatocellular carcinoma treated with sorafenib.

HCV: hepatitis C virus; HBV: hepatitis B virus; NASH: non-alcoholic steatohepatitis; BMI: body mass index; INR: international normalized ratio; MELD: Model for End-stage Liver Disease; ECOG PS: Eastern Cooperative Oncology Group performance status; BCLC: Barcelona Clinic Liver Cancer; L3 SMI: Muscle Skeletal Index at the third lumbar vertebra.
Median (range).
Lumbar SMI and sarcopenia distribution
At baseline, the median value of the L3 SMI was 47.2 cm2/m2 (range 23.4–70). The median SMI L3 value was 49.7 cm2/m2 (range 33.3–70) in males and 36 cm2/m2 (range 23.4–49.8) in females: 47 (49%) patients were defined sarcopenic. Inter-observer concordance on L3 SMI values was >95%.
Comparison of clinical and biochemical characteristics among sarcopenic and non-sarcopenic patients.
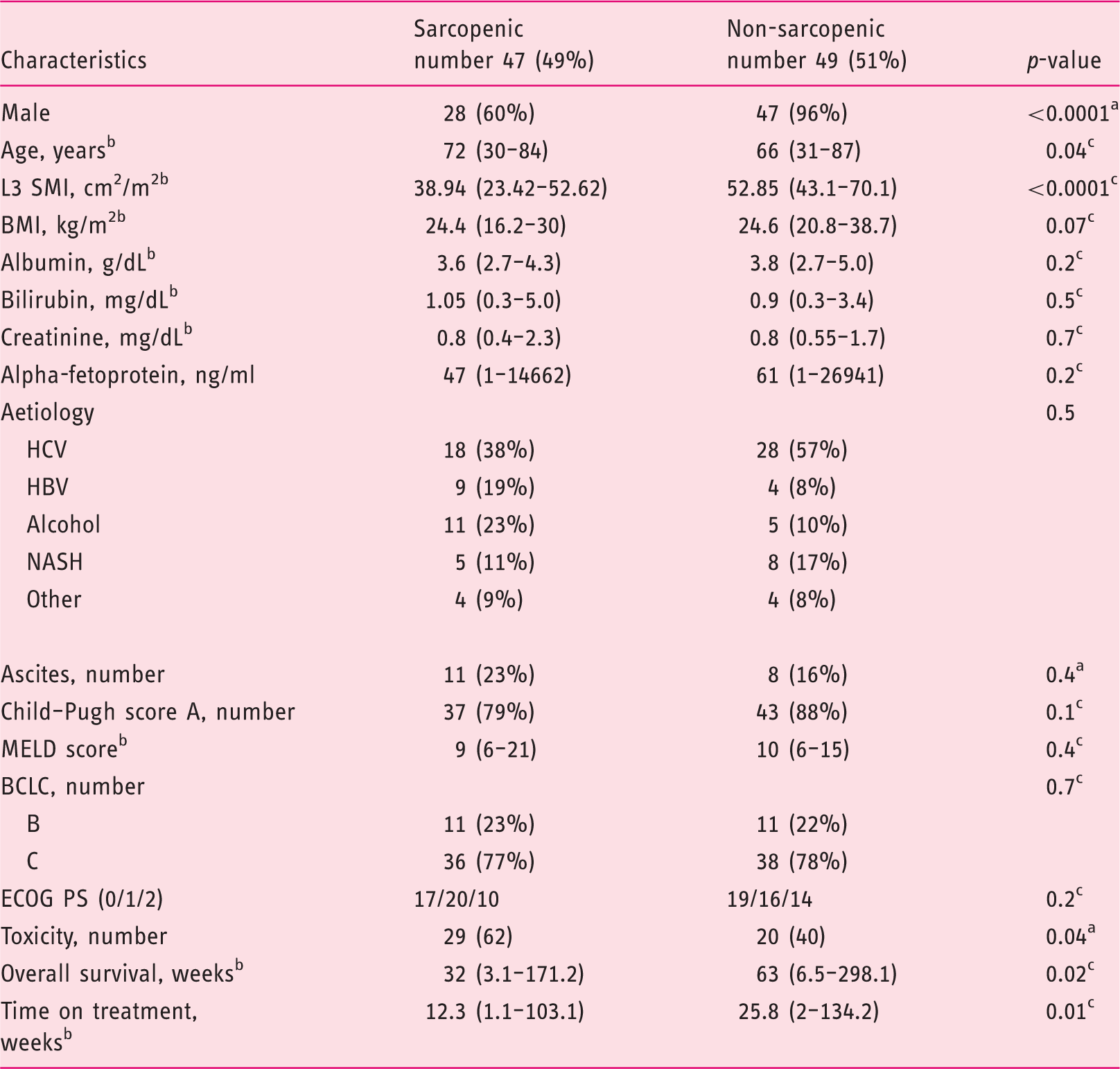
L3 SMI: Muscle Skeletal Index at 3rd lumbar vertebra; BMI: body mass index; INR: international normalized ratio; HCV: hepatitis C virus; HBV: Hepatitis B virus; NASH: non-alcoholic liver disease; MELD: Model for End-stage Liver Disease; BCLC: Barcelona Clinic Liver Cancer; ECOG PS: Eastern Cooperative Oncology Group performance status.
Fisher’s exact test.
Median (range).
Mann–Whitney test.
Factors influencing survival in advanced HCC patients treated with sorafenib
Overall survival in our cohort was 50 (3.1–298.1) weeks. According to the Kaplan–Meier method, median OS was significantly shorter among sarcopenic patients (63 versus 32 weeks, HR 1.69, p = 0.02) (Figure 1).
Kaplan Meier Curve for Overall Survival comparing sarcopenic and non sarcopenic patients.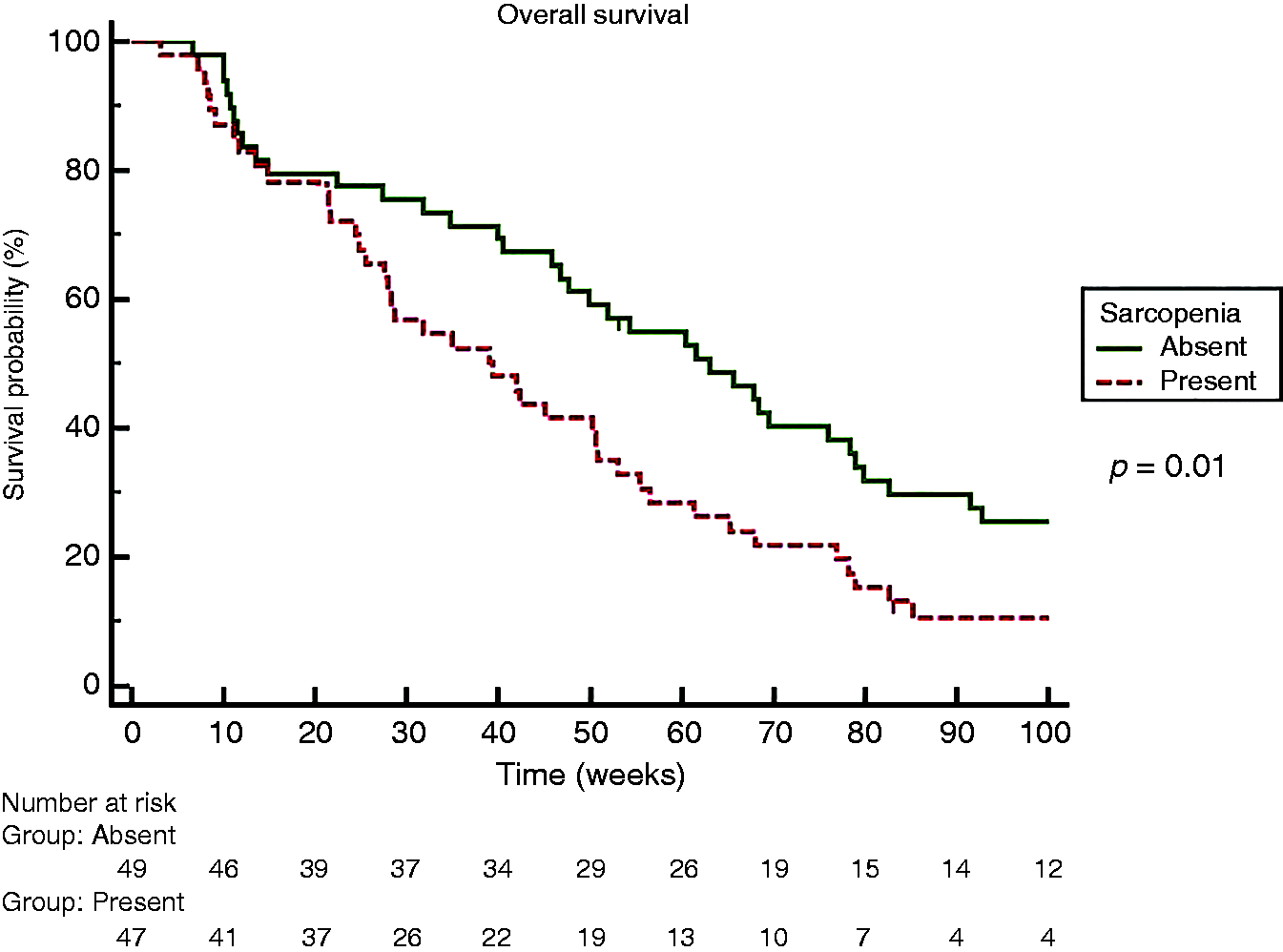
Univariate and multivariate analysis of risk factors for overall survival of hepatocellular carcinoma patients treated with sorafenib by the Cox proportional hazards model.
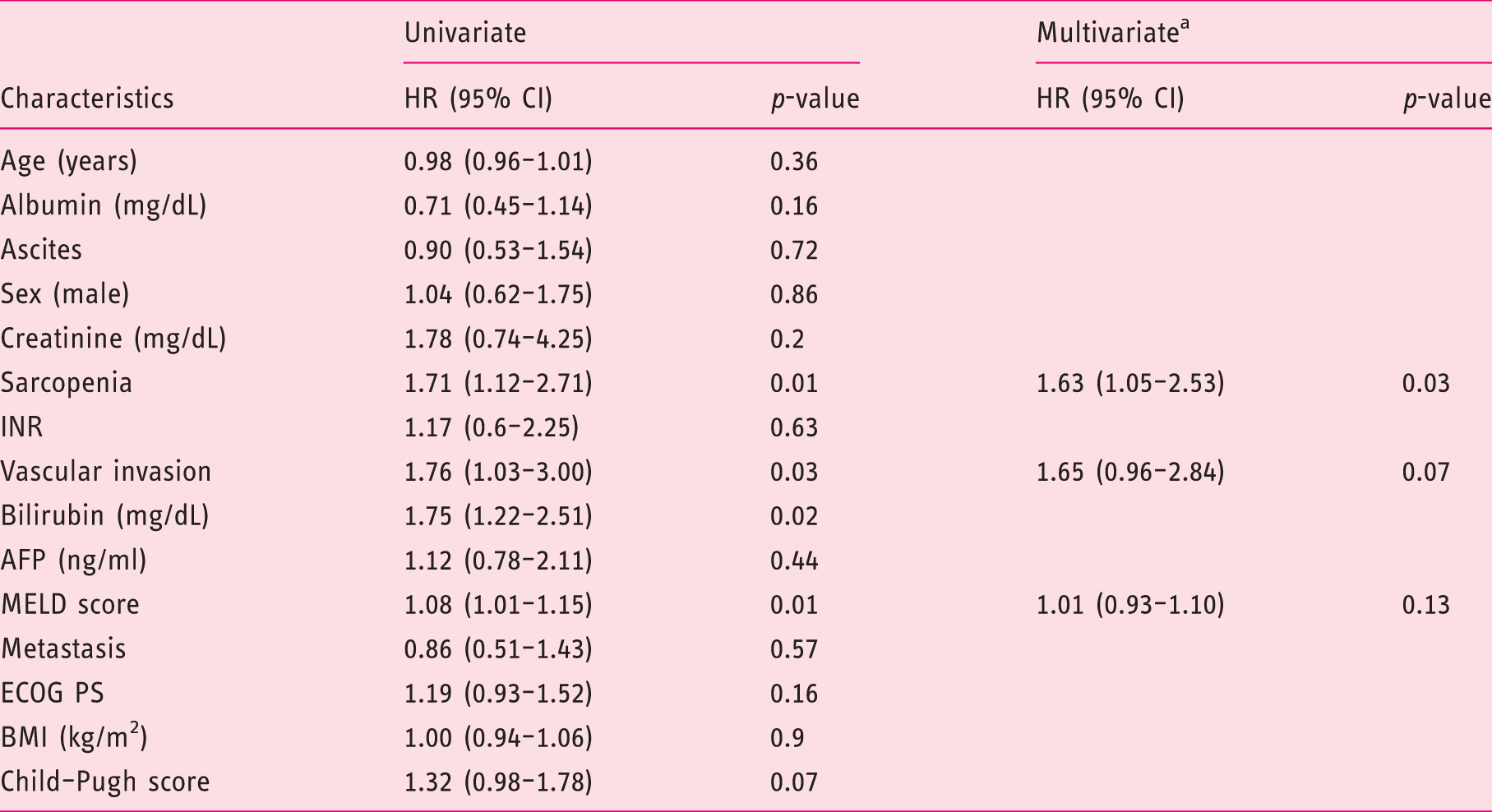
HR: hazard ratio; CI: confidence interval; INR: international normalized ratio; AFP: alpha-fetoprotein; MELD: Model for End-stage Liver Disease; ECOG PS: Eastern Cooperative Oncology Group performance status; BMI: body mass index.
Bilirubin was not included in the multivariate analysis to avoid colinearity, since it was included in the MELD score.
Factors influencing time on treatment and sorafenib tolerance
Overall, in our cohort, time on treatment was 19.2 (1.1–134.2) weeks. By Kaplan–Meier analysis, time on treatment was significantly shorter among sarcopenic patients (25.8 versus 12.3 weeks, HR 1.75, p = 0.0044) (Figure 2). All patients at the end of follow-up had discontinued sorafenib: 47 (49%) for disease progression, 42 (44%) for toxicity and 7 (7%) for liver decompensation. Sarcopenic patients significantly suffered sorafenib-related toxicity (AEs grade 3 and 4) as compared to non-sarcopenic patients (62% versus 40%, p = 0.04). Information concerning drug dose reduction was available for 55 patients. When available, there was no difference in dose reduction between sarcopenic and non-sarcopenic patients (43% versus 37%, p=not significant).
Kaplan Meier Curve for Sorafenib treatment duration comparing sarcopenic and non sarcopenic patients.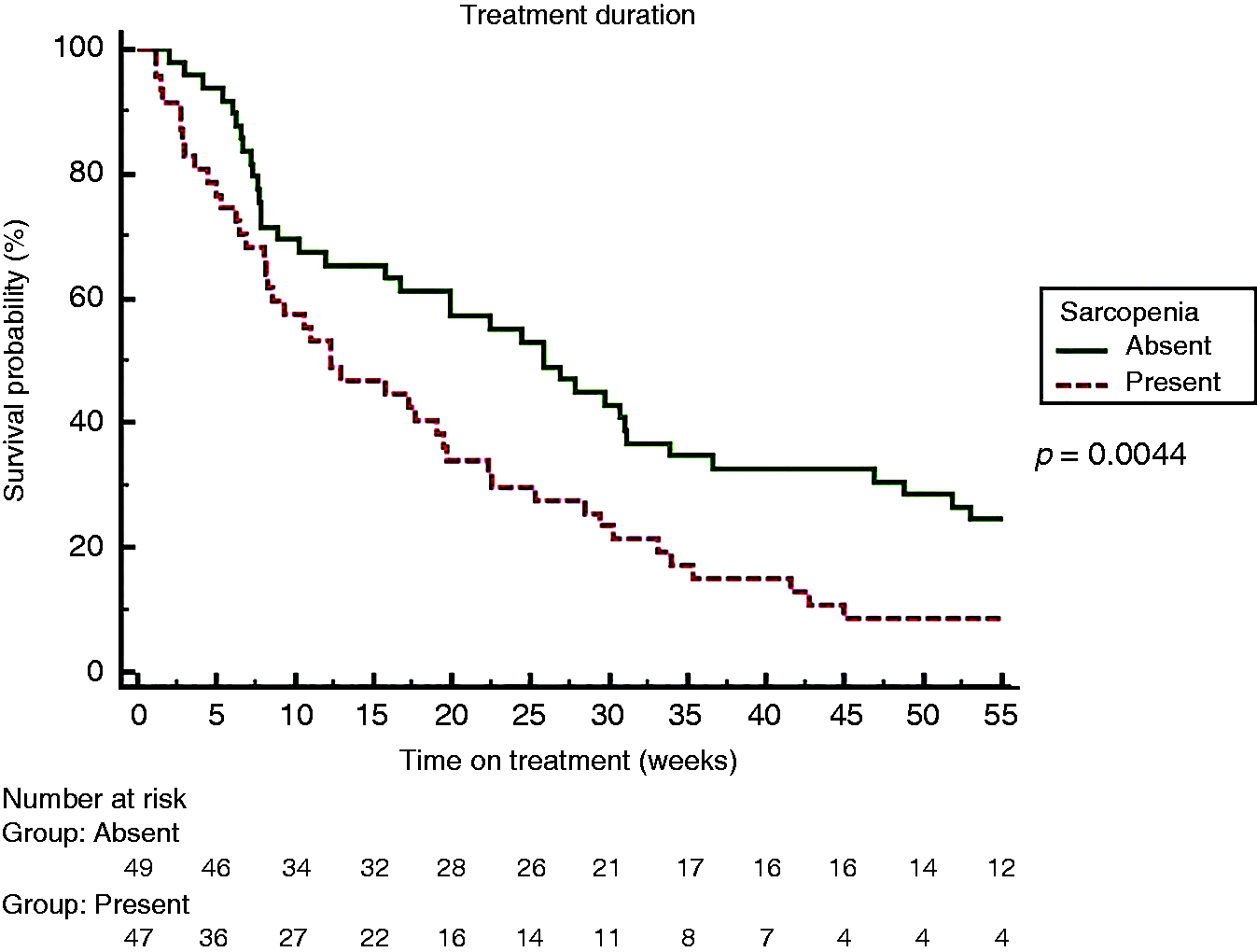
Univariate and multivariate analysis of risk factors for time on treatment of hepatocellular carcinoma patients treated with sorafenib by the Cox proportional hazards model.
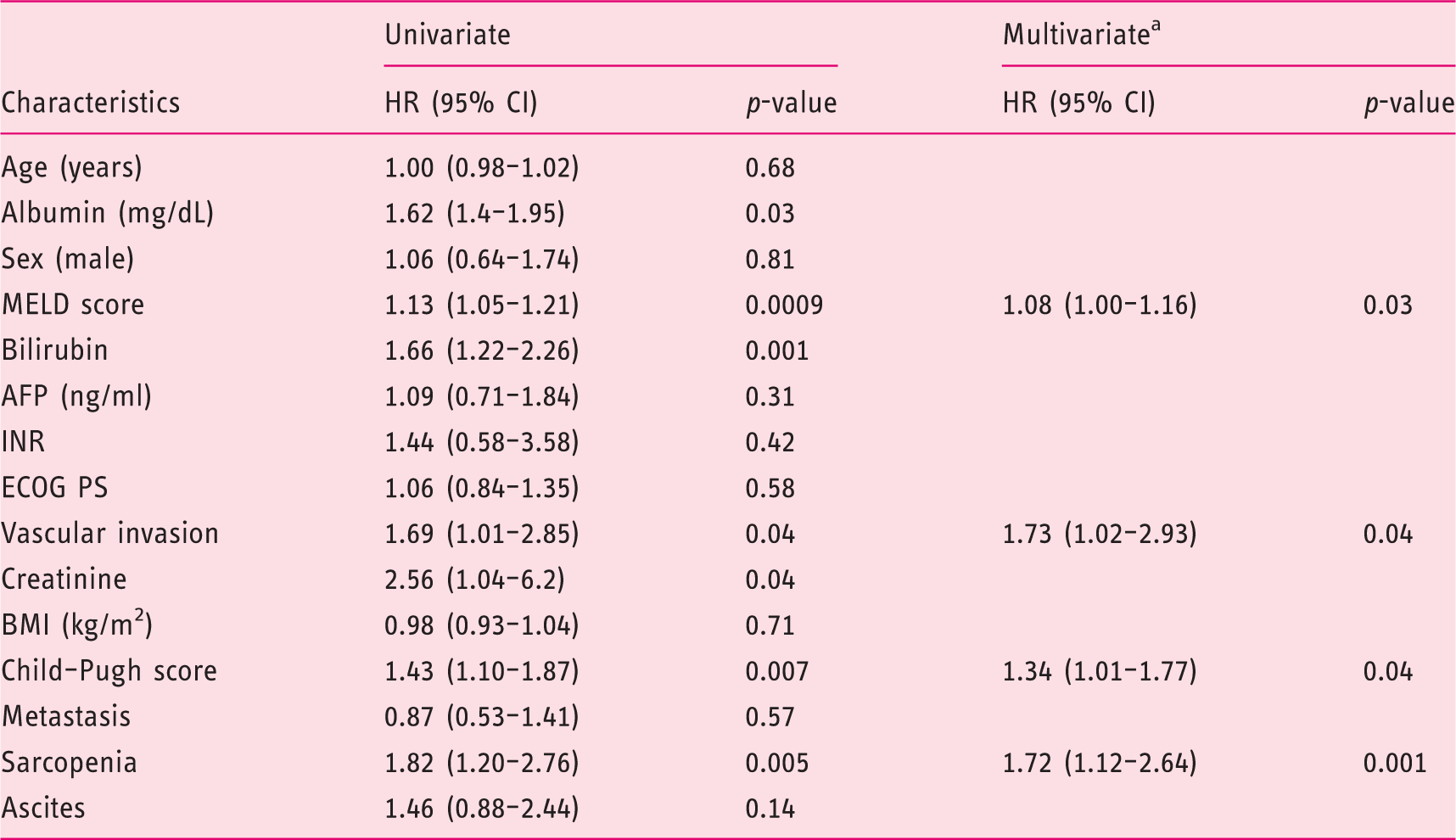
HR: hazard ratio; CI: confidence interval; INR: international normalized ratio; AFP: alpha-fetoprotein; MELD: Model for End-stage Liver Disease; ECOG PS: Eastern Cooperative Oncology Group performance status; BMI: body mass index.
Bilirubin, creatinine and albumin were not included in the multivariate analysis to avoid colinearity, since they were included in either the Child–Pugh or MELD scores.
Discussion
To the best of our knowledge, this is the largest multicentre study addressing the relationship between sarcopenia and survival in a homogeneous cohort of European Caucasian cirrhotic patients with advanced HCC treated with sorafenib. Our study indicates that sarcopenia is present in almost half of cirrhotic patients with advanced HCC selected to undergo sorafenib treatment. In our cohort, sarcopenia was independently associated with a twofold risk of mortality and reduced treatment duration, and inclusion of its measurement at baseline could be associated with an improvement in the prediction of mortality in patients with advanced HCC.
Available data demonstrate that sarcopenia is commonly observed among patients with cirrhosis and associated with significant morbidity and mortality, contributing to reduced quality of life. Among cancer patients, prevalence of pre-therapeutic sarcopenia was found to be 38.6% in a recent comprehensive review of 32 studies. 32
In our population of cirrhotic patients with advanced HCC, the prevalence of sarcopenia, as derived by a measurable and reproducible technique, was even higher, probably due to the association of advanced stage cancer and underlying liver disease. This percentage is in line with those observed among HCC cirrhotic patients and in our previous paper in Caucasian patients with HCC using similar L3 SMI thresholds.11,15 As expected, sarcopenia was linked to gender and age. A paper from Japan previously reported a higher prevalence of sarcopenia in patients treated with sorafenib (close to 65%), probably linked to different thresholds chosen from Asian series published in the literature, and as suggested by the authors, could be unfit for Caucasian patients. 17 At odds with oriental data, sarcopenia was less represented in our patients, further suggesting a role of ethnicity in influencing results in this setting.
Unexpectedly, sarcopenia was more prevalent in female patients, a discordant result when compared to most of the available literature. Our finding may be due to the homogeneous composition of our cohort, which was comprised exclusively of patients of Caucasian-Mediterranean origin, again suggesting a possible role of ethnicity in determining the development of sarcopenia, and to the generalized post-menopausal state of our female patients, as it is well known that the protective effect of oestrogens in counteracting sarcopenia development is lost after menopause. 33
Sarcopenia measurement can be obtained at marginally increased costs from CT scans already available at baseline for staging purposes, providing a further independent prognostic index to plan appropriate patient management. Furthermore, the dedicated software used overcomes the intrinsic limits of bioimpedance and anthropometric measurement, hampered by elevated BMI and ascites.6,34,35 In addition, since the measurements we performed were not limited to the psoas only, but comprised all the muscles represented in the transverse image selected, it maximized the opportunity to obtain a representative sample of muscle mass.
Sarcopenic patients experienced more serious AEs and remained on treatment for a shorter period of time, which suggests that sarcopenia could assist in performance status evaluation, normally applied to HCC patients for staging and treatment allocation. 2 The role of reduced muscle mass in exposing patients to an increased risk of developing sorafenib-related side effects has been reported in a previous study. 16 Our study, conducted in a larger and homogeneous sample representative of advanced HCC, including different degrees of liver functional reserve and performance status, confirms and completes these findings.
An additional relevant finding of our study is that sarcopenia was prevalent in 49% of overweight/obese patients (BMI ≥ 25). This observation confirms the warning that sarcopenia is not exclusively to be sought in patients with low BMI, strengthening the importance and prevalence of sarcopenic obesity.10,11,15
Our data further contributes to the definition of the role of sarcopenia as a possible predictor of reduced survival and cancer treatment toxicity, as has been previously shown in other oncological settings. 10 Accurate measurement tools are also needed in HCC, and CT-based technologies provide measurable and repeatable muscle mass measurement. We believe that sarcopenia assessment can certainly be an additional component in the complex decision-making process surrounding therapeutic choices in advance HCC.
The main limitation of our study is the retrospective design, which cannot exclude selection bias, that was nonetheless minimized by cross-referencing clinical and radiological databases, and the unavailability of full data regarding sorafenib initial dose and dose reduction.
In conclusion, our data show that European Caucasian cirrhotic patients with advanced HCC have a high prevalence of sarcopenia, and that its measurement by CT scan could be a readily available tool to identify them, improving prognostic evaluation at minimally increased costs.
Footnotes
Declaration of conflicting interests
Massimo Iavarone is on the speaker bureau for Bayer. Angelo Sangiovanni is on the speaker bureau for Bayer. All other authors declare no conflict of interest.
Funding
No funding was obtained for this study.
Authors’ contributions
GA, EG and MM conceived the study, enrolled patients, performed statistical analysis, and wrote and revised the manuscript. MI, AS and PL enrolled patients and wrote and revised the manuscript. GA, PB, EI and PB retrieved and elaborated CT scan data. AMP, LM and PM enrolled patients. All authors read and approved the final version of the manuscript.
Informed consent
Written informed consent was obtained from each patient included in this study.
Ethics approval
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by local institutional review board (Comitato Etico Milano Area 2, Universita` degli Studi di Milano, 3 December 2017).
