Abstract
Objective
To explore prognostic factors and develop an accurate prognostic prediction model for angioimmunoblastic T-cell lymphoma (AITL).
Methods
Clinical data from Chinese patients with newly diagnosed AITL were retrospectively analysed. Overall survival (OS) and progression-free survival (PFS) were estimated using Kaplan-Meier method survival curves; prognostic factors were determined using a Cox proportional hazards model. The sensitivity and specificity of the predicted survival rates were compared using area under the curve (AUC) of receiver operating characteristic (ROC) curves.
Results
The estimated 5-year OS and PFS of 55 eligible patients with AITL were 22% and 3%, respectively. Multivariate analysis showed that the presence of pneumonia, and serous cavity effusions at initial diagnosis were significant prognostic factors for OS. Based on AUC ROC values, our novel prognostic model was superior to IPI and PIT based models and suggested better diagnostic accuracy.
Conclusions
Our prognostic model based on pneumonia, and serous cavity effusions at initial diagnosis enabled a balanced classification of AITL patients into different risk groups.
Introduction
Angioimmunoblastic T-cell lymphoma (AITL) is an uncommon subtype of mature peripheral T-cell lymphoma (PTCL). 1 Possessing unique clinical and pathological features, AITL typically affects elderly men and accounts for approximately 1–2% of non-Hodgkin lymphomas (NHLs) and 15–20% of PTCLs. 1 At the time of diagnosis, most AITL patients have advanced-stage disease (Stage III–IV) which is often accompanied by lymph node disease, hepatosplenomegaly, extra nodal involvement and systemic symptoms.1–4 Currently, there is no standard treatment regimen for AITL and the combination therapy of cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) is often adopted as first-line chemotherapy. 5 The disease generally has a poor prognosis and studies have estimated the 5-year overall survival (OS) and 5-year progression-free survival (PFS) rates to be 33–48% and 18–25%, respectively.2,3,6,7
Given the diversity of the clinical manifestations of AITL and poor response to current treatment strategies, the identification of prognostic indicators would be of great clinical significance. The International Prognostic Index (IPI) and the Prognostic Index for PTCL (PIT) have been widely used in the prognostic assessment of NHLs.8,9 The IPI is based on age, performance status, lactate dehydrogenase (LDH), disease stage, and extra nodal involvement, 8 whereas PIT is based on age, performance status, LDH, and bone marrow involvement. 9 However, the use of IPI and PIT as prognostic models for AITL remains controversial.1,3,10 Interestingly, several studies have found that other clinical characteristics, such as age, elevated plasma β2 microglobulin (β2-MG), anaemia, oedema, and thrombocytopenia were key prognostic factors for AITL.11–16 The purpose of this present study was to explore prognostic factors for AITL in a group of Chinese patients and establish a new prognostic model that had better predictive performance than IPI or PIT-based models.
Methods
Data from newly diagnosed patients with AITL who were treated at the Guangxi Medical University Affiliated Cancer Hospital, between January 2007 and June 2019 were retrospectively analysed. Eligible patients were ≥18 years and had a diagnosis of AITL made by at least two experienced haemato-pathologists according to the 2008 WHO lymphoma classification criteria. Data on past medical history and results of physical, laboratory and imaging examinations were extracted from patient clinical records. Laboratory examinations had included: complete blood count; liver and kidney function tests; BM biopsy results; serum levels of albumin (ALB), LDH, β2-MG and C-reactive protein (CRP). Imaging examinations had involved computed tomography (CT) or positron emission tomography/computed tomography (PET/CT) supplemented by ultrasound. Biopsy specimens had been examined using histological and immunohistochemical techniques. Disease staging was performed using the Ann Arbor staging system. Risk stratification was performed using IPI 8 and PIT scores. 9 Treatment responses were assessed using the Lugano 2014 response criteria based on PET-CT or CT treatment response, and were defined as complete remission (CR), partial remission (PR), stable disease (SD), or progressive disease (PD). 17 The overall response rate (ORR) was defined as the proportion of patients who achieved CR or PR after treatment.
Written informed consent was obtained from all patients before publishing their anonymised data. Ethical Committee approval was obtained from the Ethics Committee at the Guangxi Medical University Affiliated Cancer Hospital and the study was reported in accordance with Strobe Guidelines. 18
Statistical analyses
Data were analysed using the Statistical Package for Social Sciences (SPSS®) for Windows® version 22 (IBM Corp., Armonk, NY, USA). A P-value <0.05 was considered to indicate statistical significance. For this study, OS was defined as the time from initial diagnosis of AITL to death or the last follow-up and PFS was defined as the time from initial diagnosis of AITL to disease progression, relapse, death from any cause, or the last follow-up. The OS and PFS were estimated using Kaplan-Meier plots, and survival curves were compared using the log-rank test. Cox proportional hazards model was used for univariate and multivariate analyses to evaluate the prognostic value of clinical factors associated with survival. Differences between categorical variables were analysed using a χ2 test. The sensitivity and specificity of the predicted survival rates were compared using area under the curve (AUC) of receiver operating characteristic (ROC) curves.
Results
In total, 55 newly diagnosed patients with AITL were included in this retrospective analysis. The clinical characteristics of the patients are shown in Table 1. Median age at diagnosis was 61 years (27–85 years), 55% (30/55) of patients were >60 years of age, and 78% (43/55) were male. Clinically, 93% (51/55) of patients had Ann Arbor stage III/IV disease, 36% (20/55) had an Eastern Cooperative Oncology Group Performance Status (ECOG PS) score >1, and 29% (16/55) had concomitant B-symptoms. Clinical presentations during disease onset included concomitant extranodal involvement in 49% (27/55) of patients, with BM involvement being the most common (20%; 11/55), while involvement of >1 extranodal site occurred in 16% (9/55) of patients. Of the 55 patients, 26 (47%) had invasion of the spleen and 22 (40%) had oedema or serous cavity effusions (i.e., pleural or pericardial effusion or ascites,). Other concomitant conditions included ‘rash’ (7 patients), ‘invasion of the liver’ (6 patients), and ‘invasion of the lungs’ (4 patients). Overall, 27% (15/55) of patients had concomitant pneumonia at initial diagnosis and 38% (21/55) exhibited pyrexia (body temperature ≥37.4°C). The proportions of patients with a platelet count <150 × 109/l and anaemia (haemoglobin level <120 g/l) were 29% (16/55) and 60% (33/55), respectively. Furthermore, 86% (47/55) of patients had an elevated serum LDH (normal range 180–285 U/l), 49% (27/55) had hypoalbuminemia (albumin level <35 g/l), 61% (25/41) had elevated CRP (normal range 0–10 mg/l), and 75% (36/48) had elevated β2-MG (normal range 1–3 mg/l). Based on IPI scores, patients were classified into four risk groups, with 16%, 31%, 36% and 16% at low, low-intermediate, high-intermediate, and high-risk, respectively. Based on PIT scores, 7%, 33%, 35% and 26% of patients were in groups 1 to 4, respectively.
Clinical characteristics of patients with angioimmunoblastic T-cell lymphoma (AITL).

Values are shown as median (range), or n (%).
Abbreviations: ECOG PS, Eastern Cooperative Oncology Group performance status; LDH, lactic dehydrogenase; β2-MG, β2-microglobulin; CRP, C-reactive protein; IPI, International Prognostic Index; PIT, Prognostic Index for Peripheral T-Cell Lymphoma.
*Some patients had more than one extranodal organ involvement; αLDH normal range 180–285 U/l; ββ2-MG normal range 1–3 mg/l; γCRP normal range 0–10 mg/l.
Of the 55 patients included in the study, 43 (78%) underwent chemotherapy, four (7%) received chidamide monotherapy (30 mg twice weekly) and eight (15%) declined treatment. Of the 43 patients that underwent chemotherapy, six (14%) received <4 cycles of treatment. Thirty-seven (86%) patients received a CHOP regimen; 29 (67%) received this regimen alone and eight (19%) received CHOP plus etoposide (CHOEP) 75 mg/m2 for three consecutive days. The remaining six (14%) patients received an anthracycline-free chemotherapy regimen. Five (12%) patients who received chemotherapy also received concurrent treatment with chidamide at a dose of 20 mg twice weekly.
The response rates for the various treatment regimens are shown in Table 2. The overall response rate (ORR) did not differ significantly between patient treatment groups. However, patient numbers were small in some groups. Median follow-up for all patients was 62 months; 36 patients (66%) died during this period. The 2-year and 5-year OS rates were 51% and 22%, respectively. The median OS time was 25 months (range 0–120 months; Figure 1a). The 2-year and 5-year PFS rates were 20% and 3%, respectively. The median PFS time was 7 months (range 0–73 months; Figure 1b).
Tumour response rates for the 55 patients with angioimmunoblastic T-cell lymphoma (AITL) according to treatment.
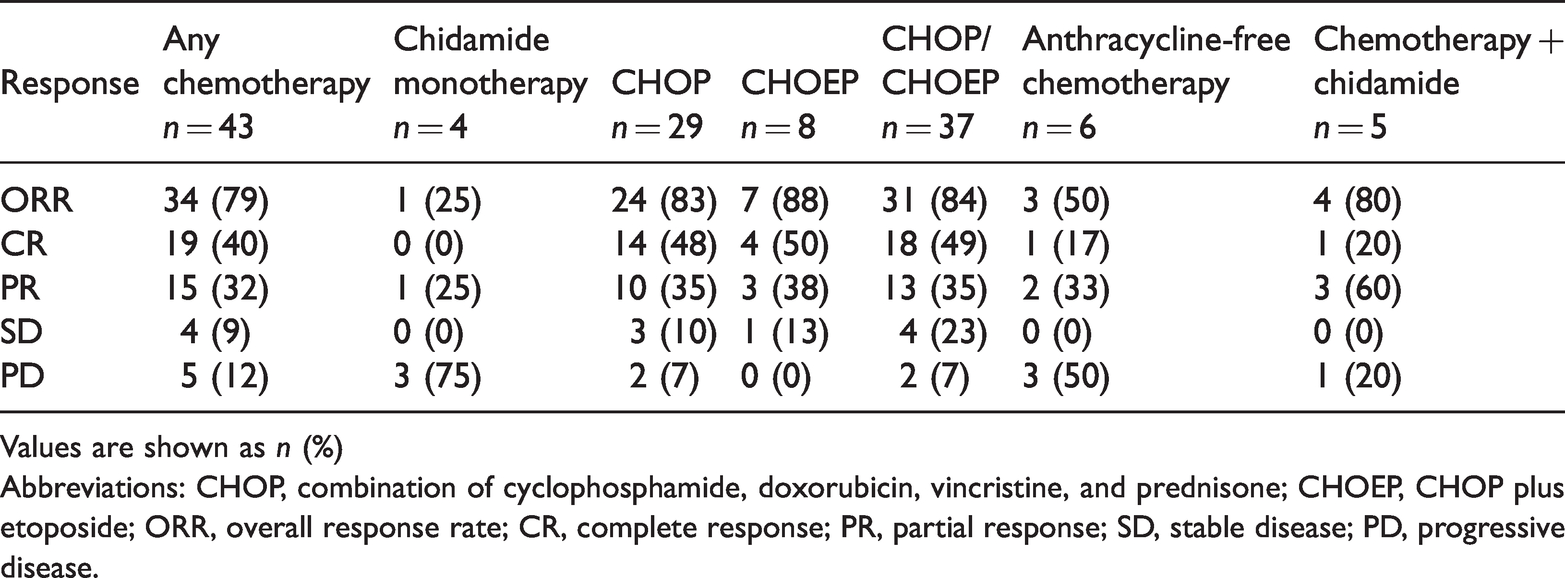
Values are shown as n (%)
Abbreviations: CHOP, combination of cyclophosphamide, doxorubicin, vincristine, and prednisone; CHOEP, CHOP plus etoposide; ORR, overall response rate; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease.

(a) Overall survival (OS) curve and (b) progression free survival (PFS) curve for 55 patients with angioimmunoblastic T-cell lymphoma (AITL).
Based on the IPI score, the 47 patients who received treatment were classified into different risk groups. The 3-year OS for the low, low-intermediate, high-intermediate, and high-risk groups were, 80%, 49%, 29%, and 17%, respectively (P = 0.018, log rank test) (Figure 2a). Based on the PIT score, patients who received treatment were classified into four risk groups. The 3-year OS for groups 1, 2, 3, and 4 were, 0%, 66%, 40%, and 13%, respectively. (P = 0.016, log rank test) (Figure 2b).

(a) Overall survival for patients with angioimmunoblastic T-cell lymphoma (AITL) using the International Prognostic Index (IPI) classified into four risk groups (low, low-intermediate, high-intermediate, and high-risk) and (b) Prognostic Index for Peripheral T-Cell Lymphoma (PIT) classified into four risk groups (Groups 1–4).
Results of univariate analysis suggested that ECOG PS score >1 (P = 0.001), B-symptoms present (P = 0.013), pyrexia (P < 0.001), pneumonia (P < 0.001), serous cavity effusions (P < 0.001), anaemia (P = 0.021) may be prognostic factors that influenced OS. A subsequent multivariate analysis confirmed that serous cavity effusions (P = 0.022), and pneumonia (P = 0.020) were independent prognostic factors of OS (Table 3). Therefore, we attempted to construct a novel prognostic model using these two risk factors. Accordingly, based on the presence of serous cavity effusions and/or pneumonia, patients were separated into three AITL groups: low-risk (no risk factors, n = 26); intermediate-risk (one risk factor, n = 12); high-risk (two risk factors, n = 9). The 3-year OS rates for the three groups were 66%, 19%, and 11%, respectively (P < 0.001) (Figure 3).
Univariate and multivariate analysis to evaluate the prognostic value of baseline characteristics in patients with angioimmunoblastic T-cell lymphoma (AITL) (n = 55).
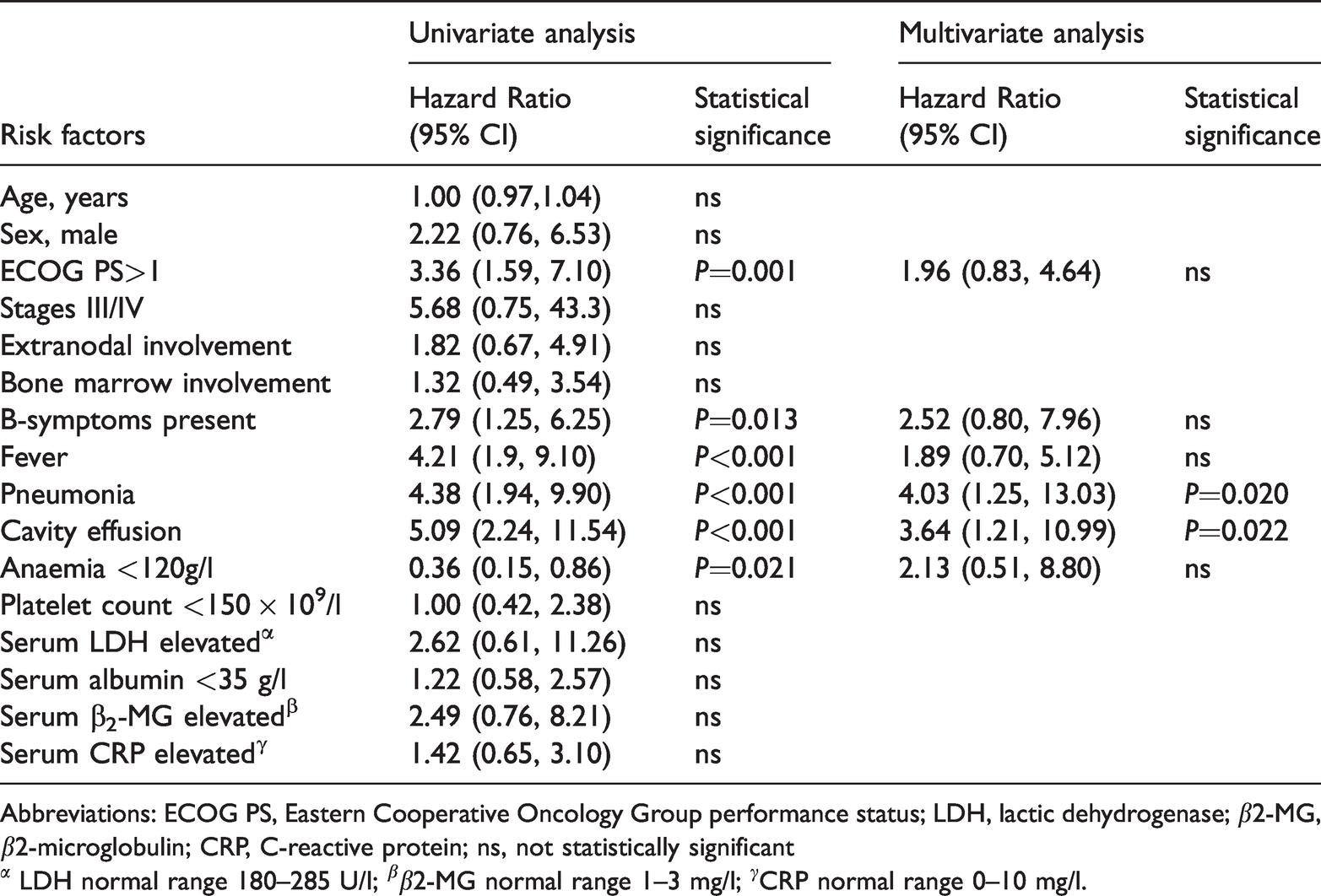
Abbreviations: ECOG PS, Eastern Cooperative Oncology Group performance status; LDH, lactic dehydrogenase; β2-MG, β2-microglobulin; CRP, C-reactive protein; ns, not statistically significant
α LDH normal range 180–285 U/l; ββ2-MG normal range 1–3 mg/l; γCRP normal range 0–10 mg/l.
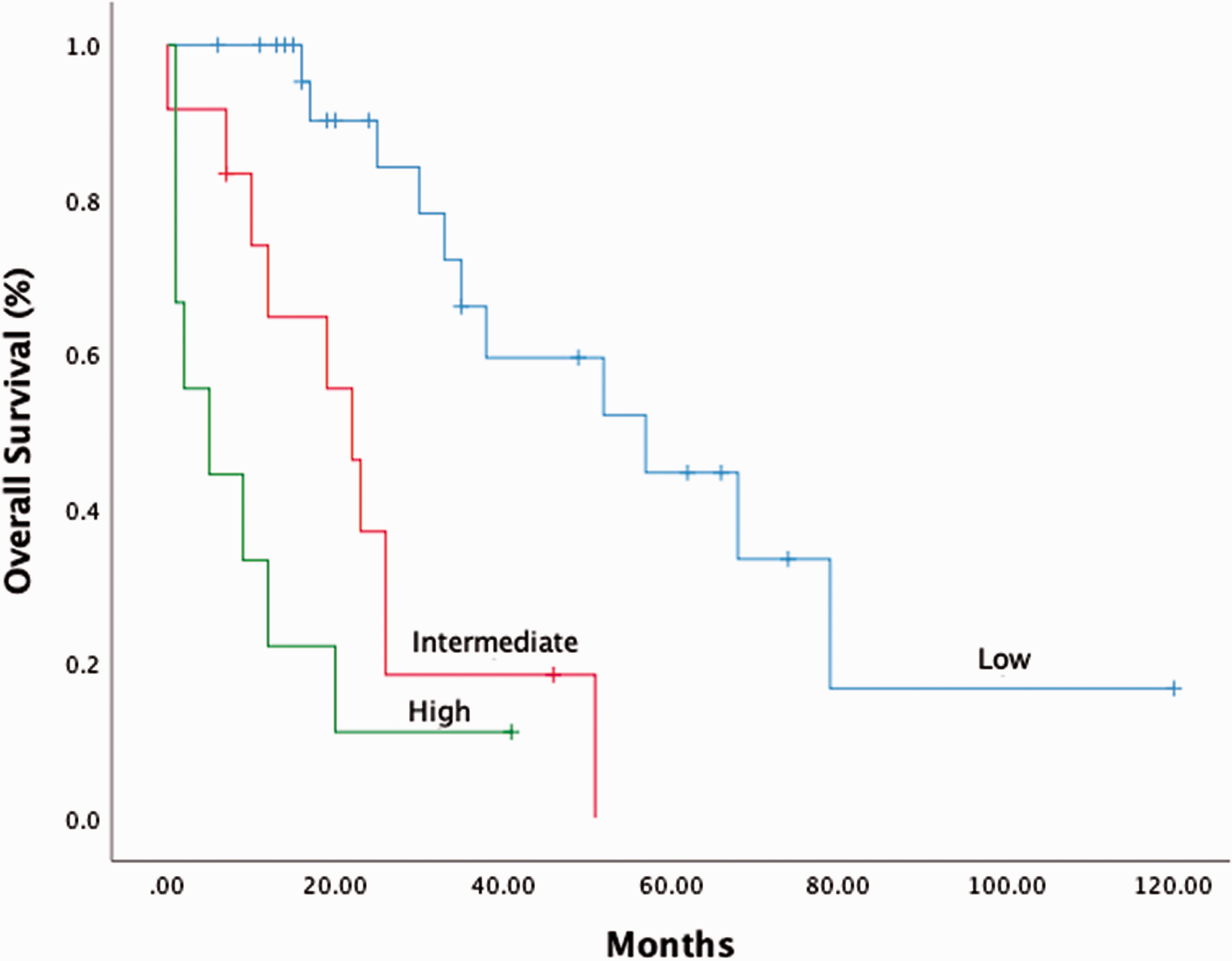
Overall survival (OS) for patients with angioimmunoblastic T-cell lymphoma (AITL) according to a new prognostic model based on presence of ‘serous cavity effusions’ and/or ‘pneumonia’. Patients were separated into three groups: low-risk (no risk factors, n = 26); intermediate-risk (one risk factor, n = 12); high-risk (two risk factors, n = 9). The 3-year OS rates for the three groups were 66%, 19%, and 11%, respectively (P < 0.001).
According to AUC ROC values, our novel prognostic model based the presence of serous cavity effusions and pneumonia at initial diagnosis, was superior to the IPI and PIT based models (i.e., novel model, 0.66; 95% CI: 0.49-0.82; IPI model, 0.63; 95% CI: 0.45-0.80; PIT model, 0.57, 95% CI: 0.40-0.74) and suggested a better diagnostic accuracy (Figure 4).

Receiver operating curves (ROC)s comparing the sensitivity and specificity of survival predictions for the new diagnostic model with a reference line and International Prognostic Index (IPI) and (B) Prognostic Index for Peripheral T-Cell Lymphoma (PIT) based models. The area under the ROC curves were 0.66 (95% CI: 0.49–0.82), 0.63 (95% CI: 0.45–0.80), 0.57 (95% CI: 0.40–0.74) for the novel, IPI- and PIT-based models, respectively.
Discussion
Due to its rarity, the precise clinical features and accurate prognostic factors for AITL have not been established. In our study, we found that serous cavity effusions and pneumonia were significant prognostic factors for OS in patients with AITL. Based on the area under ROC curves, our new model, which was based on these two prognostic factors, provided better diagnostic accuracy than our IPI or PIT based models. Although our analysis is based on retrospective data, we believe that our findings may be useful in the development of optimal treatment strategies for these patients.
Our analysis was based on clinical data from 55 Chinese patients newly diagnosed with AITL at our hospital. The clinical characteristics of our patients were similar to those from other studies in both Asia and Europe.2,3,8 Consistent with previous studies,1–4 a large proportion of our population was male, patients tended to be elderly and most were at Stage III/IV disease. However, the proportion of patients with BM involvement (20%) was lower than that recorded in a previous retrospective study in 33 Chinese patients (70%) 19 The bone marrow examinations of the patients in this present study included bone marrow cytology and bone marrow pathology and although most patients had both types of bone marrow assessment, a large proportion only had cytology which may have led to some underreporting.
While out remission rates following first-line treatment were similar to previously reported data, 3 our 5-year OS and PFS rates (22% and 3%) were lower than those from previously reported studies.2,3,6,7 Although our patient demographics and distribution of IPI and PIT scores were similar to those of several large cohort studies,2,3,8 differences in survival rates between our study and previous studies may have been due to the high proportion of patients (15%) at our centre who declined treatment. Furthermore, although 86% of our patients received a CHOP based regimen as first-line treatment, their treatment may have not been optimal. Interestingly, in another study involving 157 patients with AITL that found a 7-year survival rate of 30%, many of the patients had received intensive courses of CHOP and some had even received autologous hematopoietic stem cell transplantation. 3 Currently, there is no standard regimen for the first-line treatment of AITL, and the CHOP regimen remains a commonly used treatment. 20 However, as this study demonstrates, there is a need to develop targeted treatments and alternative therapies for AITL. Therefore, the development of a novel prognostic model specially designed for AITL may facilitate risk-based stratification of patients and therapeutic targets.
Despite the widespread application of IPI and PIT scores in prognostic risk stratification for NHLs, the use of these indices for AITL remains a subject of debate.1,3,10 For example, in one study IPI was found to be negatively correlated with survival, 21 whereas in another study it was found to be a prognostic factor for survival. 8 Similarly, PIT scores have been shown to be positively correlated with survival in one large-scale study of patients with AITL, 2 whereas in another study there was no correlation. 3 Our results showed that although IPI and PIT scores were factors that affected the survival of patients with AITL, our various patient risk groups could not be effectively delineated using these classifications. This may be related to the fact that IPI and PIT only include a limited number prognostic factors in their assessment and patients with AITL may have other clinical factors that affect outcome.15,16
We identified serous cavity effusion as a novel prognostic factor in AITL. This factor was found in 40% of our patients which is more than observed in a previous study in Chinese patients (26%). The mechanisms of fluid build-up in patients with AITL are uncertain. Vascular endothelial growth factor (VEGF) was previously known as vascular permeability factor because of its ability to induce vascular leakage. 22 We hypothesise that, in patients with AITL, increased vascular permeability mediated by VEGF may be the cause of the systemic oedema.22,23 Further biological investigations on the role of cavity effusion and VEGF should be encouraged for a better understanding of the aetiology of AITL. We also identified pneumonia as a significant prognostic factor in our patients with AITL. Concomitant active pneumonia was found in 27% of our patients at initial diagnosis. To our knowledge, the presence or absence of pneumonia in patients with AITL at initial diagnosis has not been reported previously. We have observed that patients with pneumonia often undergo dose reductions in chemotherapy and additional therapy for the bacterial infection. Therefore, the presence of concomitant pneumonia, will undoubtedly have a significant effect on survival.
Our study had some limitations. For example, it was a retrospective study involving a small sample of patients. Therefore, unrecognized biases cannot be excluded, and careful interpretation of the results is required. In addition, the treatment strategies were based on institutional protocols and physicians' decisions and so treatment outcomes may have varied. Nevertheless, we suggest that our novel prognostic model based on serous cavity effusion and pneumonia may provide useful information for future studies.
In conclusion, the prognostic model constructed in this study, based on the presence of serous cavity effusion and pneumonia at initial diagnosis, enabled a balanced classification of our patients with AITL into different risk groups. This information may be beneficial in establishing optimal treatment regimens for patients with AITL. Further studies are required to validate our findings.
Footnotes
Acknowledgements
We are grateful to the staff of the diseases follow-up centre of the Guangxi Medical University Affiliated Cancer Hospital for their valuable contribution.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This work was supported by grant from Guangxi Provincial Natural Science Foundation (2019GXNSFAA245086)
