Abstract
BACKGROUND:
Recent study has revealed that large intergenic non-coding RNA-ROR (linc-ROR) is aberrantly expressed in a number of cancers including breast cancer. However, whether circulating linc-ROR in plasma could be used for breast cancer diagnosis and dynamic monitoring is not clear.
OBJECTIVE:
The objective of this study is to determine if plasma linc-ROR could be applied as a biomarker for the diagnosis and dynamic monitoring of breast cancer.
METHODS:
We tested the expression levels of linc-ROR in 24 pairs of tissue samples and 96 plasma samples from breast cancer patients by quantitative real time-polymerase chain reaction (qRT-PCR), and analyzed the correlation between plasma linc-ROR levels and clinico-pathological characteristics. Receiver operating characteristic (ROC) curve was used to assess the diagnostic power of plasma linc-ROR, carcinoembryonic antigen (CEA) and carbohydrate antigen (CA)153 for breast cancer. Furthermore, we explored the monitoring values of plasma linc-ROR for breast cancer by analyzing the preoperative and postoperative plasma linc-ROR levels of the same patients.
RESULTS:
The expression levels of linc-ROR were significantly higher in breast cancer tissues and plasma than the levels in the control (
CONCLUSION:
Overexpressed linc-ROR may be a potential biomarker for the diagnosis and dynamic monitoring of breast cancer.
Introduction
Breast cancer is the most common malignancy diagnosed among women worldwide and is the leading cause of female cancer death [1]. It is globally estimated that over 1,000,000 people are diagnosed with breast cancer annually and at least 400,000 females will die from the disease every year, accounting for 14% of total cancer deaths [2]. The prognosis of breast cancer mainly depends on its early detection, and the early diagnosis greatly relies on some economical diagnostic biomarkers in circulation system. The current blood-based biomarkers for the diagnosis and monitoring of breast cancer, such as cancer antigen (CA) 153 and carcinoembryonic antigen (CEA), are criticized for both low sensitivity and low specificity [3]. Meanwhile, the levels of these biomarkers can also be elevated in the patients with only digestive tract inflammation [4]. Additionally, the present screening methods, such as breast ultrasound and mammography, have some disadvantages due to over-diagnosis [5]. Thus, more ideal biomarkers for early diagnosis and treatment of breast cancer are urgently to be explored.
As a new class of RNA molecules, long non-coding RNAs (lncRNAs) have become a hot topic in epigenetics. Recently, lncRNAs have been reported to be involved in some physiological processes and pathological conditions, and play an important role in many human diseases [6, 7]. Importantly, lncRNAs are protected by extracellular vesicles in the circulation and thus can be present stably in the serum for an extended period [8], not only in room temperature incubation, but also in the conditions when exposed to multiple freeze-thaw cycles, low or high pH, or RNase A [9]. Accumulating evidence reveals that lncRNAs detectable in serum have been identified as potential biomarkers of various types of cancers including breast cancer [10, 11]. Among lncRNAs, large intergenic non-coding RNA regulator of reprogramming (lincRNA-ROR or linc-ROR) is a member of subvariety of lncRNAs, was first discovered in induced pluripotent stem cells (iPSCs), and plays a central role in promoting survival in iPSCs and embryonic stem cells (ESCs) through preventing the activation of cellular stress pathways [12]. More and more studies have showed that linc-ROR is aberrantly expressed in a number of solid cancers and closely associated with the development of several malignancies [13]. Moreover, linc-ROR was found to be upregulated in breast tumor samples; overexpressed linc-ROR in immortalized human mammary epithelial cells subsequently could induce an epithelial-to-mesenchymal transition (EMT) program, and enhance the migration and invasion of breast cancer cells [14]. Mechanistically, linc-ROR regulates the invasion in triple-negative (ER-, HER2-, PR-) breast cancer (TNBC) by linc-ROR/miR-145/ARF6 pathway [15]. Up-regulation of linc-ROR was also observed in the multidrug resistance and invasion of breast cancer [16]. In addition, linc-ROR was reported to suppress gemcitabine-induced autophagy and apoptosis in breast cancer cells by silencing miR-34a expression [17]. All these reports demonstrated that linc-ROR enhances breast cancer cell proliferation, malignant transformation and metastasis, and that lncRNA regulatory processes are critical players in breast cancer tumorigenesis and multidrug resistance. However, whether circulating linc-ROR in plasma is a potential biomarker for breast cancer is not clear.
In the current study, for the first time, we detected linc-ROR expression levels in the plasma of breast cancer patients and compared the levels between plasma and tissue samples. Then, the correlations between plasma linc-ROR levels and clinico-pathological characteristics in breast cancer patients were analyzed. Lastly, the diagnostic and monitoring values of the plasma linc-ROR for breast cancer patients were evaluated.
Primers sequences, annealing temperature and products length
Primers sequences, annealing temperature and products length
Patients and samples
In this study, 96 breast cancer patients (including 85 infiltrating ductal carcinoma, 5 ductal carcinoma, 4 mucinous carcinoma and 2 no special types of invasive breast cancer) and 90 healthy women were enrolled at the Sichuan Cancer Hospital from January 2015 to December 2015. Blood samples were collected from the 96 breast cancer patients and 90 healthy women. Twenty-four paired tissue samples (breast cancer tissues and matched adjacent noncancerous tissues) from the same 96 breast cancer patients were obtained from surgeries. And 24 paired blood samples (preoperative and postoperative blood in 7 days after surgery) were collected from the same tissue samples patients. None of the patients had received any clinical treatment before sample collection and each specimen was confirmed as breast cancer by histopathological examination. Besides, according to tumor node metastasis (TNM) staging criteria for breast cancer of American Joint Committee on Cancer (AJCC) [18], 22 breast cancer patients were found with stage I; 36 patients were with stage II; 21 patients were with stage III and 17 patients were with stage IV. No diseases or injuries had been found in the healthy volunteers. The average age of breast cancer patients is 49.24
Ribonucleic acid (RNA) extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA from tissues (50 mg) was isolated by Trizol Reagent (TaKaRa, Tokyo, Japan) and RNA from plasma samples (200
Detection of serological tumor marker and immunohistochemical evaluation
To evaluate the diagnostic applicability of the plasma linc-ROR, CEA and CA153 for breast cancer, venous bloods (5 mL) of 45 early stage patients from the 96 breast cancer patients and 45 age and sex-matched healthy controls from the 90 healthy volunteers were collected. Serum samples were obtained by centrifugation, placed in freezing tubes. Serum CEA and CA153 levels were determined using an automatic electrochemistry luminescence immunoassay system (ROCHE E170; Roche, Germany). Estrogen receptor (ER)- and progesterone receptor (PR)-positive were defined as the presence of
Statistical analysis
Statistical analysis was performed by SPSS 22.0 software. Measurement data were expressed as means
Results
Expression of linc-ROR in breast cancer tissues and plasma
Previous studies have shown that linc-ROR is overexpressed in breast cancer cells and tissues [13, 14]. To validate the expression of linc-ROR in breast cancer tissues, quantitative RT-PCR was applied to test the levels of linc-ROR expression in 24 pairs of breast cancer tissues in this study. The average linc-ROR expression level of 24 breast cancer tissues was 2.17
elative expression of linc-ROR in breast cancer tissues and adjacent noncancerous tissues.
The linc-ROR expression in 24 pairs of breast cancer patients’ tissues was analysed by quantitative RT-PCR before surgery. Data were represented as the means
Relative expression of linc-ROR in plasma of breast cancer patients and normal controls.
The linc-ROR expression levels in plasma samples from 96 breast cancer patients and 90 healthy controls were tested by quantitative RT-PCR before surgery. Data were represented as the means
Associations between plasma linc-ROR expression levels and clinico-pathological characteristics were analyzed. As shown in Table 2, the levels of linc-ROR expression significantly correlated with lymph node metastasis (
Plasma linc-ROR and clinical characteristic correlations in breast cancer patients
Plasma linc-ROR and clinical characteristic correlations in breast cancer patients
TNM: tumor node metastasis; ER: estrogen receptor; PR: progesterone receptor; HER2: Human epidermal growth factor receptor-2; Ki-67: an indicator of cell proliferation. *
In order to evaluate the diagnostic applicability of the plasma linc-ROR, CEA and CA153 for breast cancer, we recruited 45 early stage patients from the 96 breast cancer patients and 45 age and sex-matched healthy controls from the 90 healthy volunteers to determine the plasma linc-ROR, CEA and CA153 levels and constructed the receiver operating characteristic (ROC) curves. The results demonstrated that the diagnostic power of linc-ROR (area under the ROC curve [AUC]
ROC curves compare the diagnostic power of linc-ROR, CA153, CEA and a combination of the three indexes to discriminate breast cancer patients from healthy controls.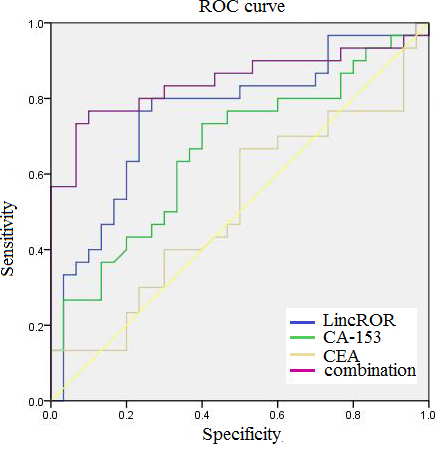
The plasma linc-ROR levels of 45 early stage patients from the 96 breast cancer patients and 45 ageand sex-matched healthy women from the 90 healthy women were examined by quantitative RT-PCR. The plasma cancer antigen (CA)153, carcinoembryonic antigen (CEA) levels of 45 early stage breast patients and 45 ageand sex-matched healthy women were tested by an automatic electrochemistry luminescence immunoassay system (ROCHE E170; Roche, Germany). ROC curves were used to compare the diagnostic power of linc-ROR, CA153, CEA and a combination of the three indexes to discriminate breast cancer patients from healthy controls.
Linc-ROR, CA153, CEA, and combined index sensitivity and specificity for breast cancer diagnosis
AUC
To investigate the value for dynamic monitoring of breast cancer, the expression levels of linc-ROR were analyzed in 24 paired preoperative and postoperative plasma samples from breast cancer patients. As shown in Fig. 4, linc-ROR expression levels were remarkably reduced in the postoperative samples (
Relative expression levels of plasma linc-ROR between postoperative and preoperative samples.
The linc-ROR expression in 24 breast cancer patients’ tissues was analyzed by quantitative RT-PCR before surgery and after surgery. “pre-operation” indicates the expression of linc-ROR in breast cancer tissues before surgery. “post-operation” indicates the expression level of linc-ROR in breast cancer tissues after surgery. “
RNAs work as bridges connecting the DNAs and proteins because coding RNAs can be messengers to translate genetic information from DNAs to proteins. Recent advances have prompted the realization that the human genome encodes an unappreciated large number of non-coding RNAs (ncRNAs), and many ncRNAs are expressed in temporally and spatially controlled manners and have potential transcriptional, post-transcriptional, and epigenetic regulatory functions [20]. Since non-coding RNAs greatly influence gene expression via affecting the stability of messenger RNA, once the stability is broken, serious consequences including tumor may be brought about [7]. LncRNAs are a class of ncRNAs, which lack the structure of functional open reading frames (ORFs), with transcripts longer than 200 nucleotides. Productions of lncRNAs are regulated by the mechanisms similar to those of protein-coding genes, such as histone modifications and RNA splicing [21]. An increasing number of studies have shown that lncRNAs are involved in the regulation of many biological functions and adverse events such as cancers and metastasis will happen when these functions are disrupted. Meanwhile, it is recognized that the endogenous RNA networks are composed of messenger RNA, lncRNA, and microRNAs, etc. These RNA molecules can compete against each other and greatly influence the post-transcriptional activity of tumor suppressors and oncogenes.
The large intergenic non-coding RNA regulator of reprogramming (lincRNA-ROR, linc-ROR) is a 2.6 kb lncRNA, firstly discovered in iPSCs and has a role in maintaining stem cell pluripotency, cellular reprogramming and cellular transformation [12]. Linc-ROR has been identified as an oncogenic lncRNA in multiple types of cancers because upregulations of linc-ROR levels were observed in various cancers, including breast cancer [17], pancreatic cancer [22, 23], hepatocellular cancer [24], bladder cancer [25], and nasopharyngeal carcinoma [26]. It has been reported that Linc-ROR can also promote cancer invasion, metastasis and tumor growth through activating ZEB1 pathway and subsequent epithelial-to-mesenchymal transition (EMT) [27]. And linc-ROR also acts as a ceRNA to increase stemness gene Nanog expression by sponging miR-145 in cancer cells [22]. Meanwhile, linc-ROR confers gemcitabine resistance to cancer cells via inducing autophagy and modulating the miR-124/ PTBP1/PKM2 axis [23]. These results suggested that linc-ROR enhances multiple cancer proliferation, malignant transformation, metastasis and multidrug resistance. However, whether plasma linc-ROR can be used as a biomarker for the diagnosis and dynamic monitoring of breast cancer is not clear as well as the significance of linc-ROR in breast cancer is still poorly understood. In this study, our results clearly demonstrated that the expression levels of linc-ROR, not only in tissues (Fig. 1,
Estrogen receptor (ER) and progesterone receptor (PR) are two members of hormone receptors, which play significant roles in the life processes of breast cell and the development of breast cancer. Most prediction and prognosis of breast cancer therapies are associated with these subtypes. A growing number of studies demonstrated that lncRNAs are emerging as critical determinants of hormone status in the complex network of hormone receptors. In epidemiology, the following have been proven: The levels of lncRNA-HOTAIR expression were related to ER-positive subtype breast cancer [28]; the levels of lncRNA-RP11-445H22.4 expression were associated with ER/PR-positive breast cancer patients [29]; the nine-lncRNA signature was also an independent prognostic variable in the subgroups grouped by ER status [30]. In basic medicine, the following have been proven: Several kinds of lncRNAs can be directly repressed by estrogen and induce tamoxifen resistance via regulating transcription and affecting ER signaling [31, 32]; ER regulates lncRNA transcription by decreasing binding of the two pioneer factors FoxA1 and AP-2
The human epidermal growth factor receptor 2 (HER2) is a 185-kd glycoprotein and approximately 20% to 30% of newly diagnosed invasive breast cancers showed the overexpression of HER2 (HER2-positive) [35]. Merry et al. [37] utilized several RNA-seq datasets that take advantage of both cell culture models of HER2-positive breast cancer and clinically relevant tumor data in vivo to identify the relationship between lincRNAs and HER2-positive breast cancer, but only three lncRNAs became deregulated in response to HER2 amplification both in vitro and in vivo. Ki67, a non-specific indicator of cell proliferation, is widely used in routine pathological examination, and no association between lncRNAs and Ki67 was observed in previous studies [28, 36]. In our current study, there was no marked difference in statistics between linc-ROR levels and HER2 or Ki67 (Table 2).
CEA is a member of cell surface glycoproteins, and plasma CEA levels are usually increased once cancer has metastasized [38]. CA153, a member of the mucin-1 (MUC-1) family of glycoproteins, is up-regulated in cancers [39]. Although CA153 and CEA are two of the frequently used serological biomarkers in cases of breast cancer, yet its application in screening and diagnosis is hindered by its limited sensitivity and specificity [36, 40]. Hence, it is essential to find more accurate and effective biomarkers with sufficient sensitivity and specificity in order to improve the clinical diagnosis for breast cancer. Here, we evaluated the diagnostic capabilities of the plasma linc-ROR, CEA and CA153 for breast cancer. Our results demonstrated that linc-ROR provided the highest diagnostic capability for detection of breast cancer (AUC
The released nucleic acids in the blood are thought to be associated with the apoptosis and necrosis of cancer cells in the tumor micro environment, which can stably exist in serum and other body fluids and thus may represent potential biomarkers [41]. As members of nucleic acids, some lncRNAs have been testified as biomarkers for monitoring tumor status due to overexpression in plasma. Zhang et al. reported that lncRNA H19 in plasma was overexpressed in preoperative compared with postoperative breast cancer patients and found a correlation between lncRNAH19 expression levels in tissue and plasma [40]. A recent study indicated that lncRNA HOTAIR expression was correlated with breast cancer tissue and plasma in the same way [36]. Similarly, we explored 24 paired preoperative and postoperative plasma samples and analyzed the correlation between linc-ROR expression in tumor tissue and in plasma for the first time. Our results determined that concentrations of plasma linc-ROR were significantly reduced in postoperative patients (Fig. 4). These findings indicate that linc-ROR expression in plasma may be a biomarker for monitoring breast tumor status.
In summary, we demonstrated for the first time that the levels of linc-ROR expression were significantly upregulated in both breast cancer tissues and plasma, and the increased levels of linc-ROR were associated with ER, PR, and lymph node metastasis. Among CEA, CA153 and linc-ROR, linc-ROR was the most powerful for diagnosis and monitoring of breast cancer. Although this study has limitation with the small sample size, our results indicated that up-regulation of linc-ROR played an important role in the development and progression of breast cancer. Thus, linc-ROR may be a novel marker for breast cancer diagnosis and monitoring. In the future, a large-scale prospective study should be carried out to validate the accuracy and effectiveness of linc-ROR as a representative biomarker for breast cancer.
Compliance with ethical standards
The study was approved by the Sichuan Cancer Hospital and meets the Ethics Committee of the Hospital. Informed consent was obtained from all the participating patients and healthy control.
Conflict of interest
The authors declare that they have no conflict of interests.
Footnotes
Acknowledgments
This work was supported by the grant from the National Science Foundation of China (No. 81372945) to Zunzhen Zhang.
