Abstract
OBJECTIVE:
The study is to explore the role of tunicamycin-induced endoplasmic reticulum stress (ERS) in human ovarian cancer (OC) SKOV3 cells proliferation, migration and invasion by modulating the activity of PI3K/AKT/mTOR pathway.
METHODS:
The collected human OC SKOV3 cells were randomly separated into three groups: The control group, the Tun group (treated with tunicamycin to induce ERS) and the CHOP-si group (transfected with CHOP-siRNA before tunicamycin treatment). CCK-8 method was applied for testing cell proliferation, while flow cytometry was conducted to detect cell apoptosis. Scratch test and Transwell test were used to determine the level of cell migration and invasion, respectively. Western blotting was performed to determine the related proteins expressions in ERS and PI3K/AKT/mTOR pathway.
RESULTS:
The cell survival rate in the Tun group was enhanced than that in the CHOP-si group, both of which were declined with the passage of time. The cell apoptosis rate in the Tun group was increased compared to the CHOP-si group, both of which were significantly elevated. The horizontal migration distance and the number of invasive cells in the Tun and CHOP-si groups were inhibited; however, the horizontal migration distance and the number of invasive cells in the CHOP-si group were enhanced than that in the Tun group. In comparison with the control group, the expressions of CHOP and TRB3 were increased in the Tun group but decreased in the CHOP-si group. The PI3K, p-AKT and p-mTOR expressions were remarkably declined in the Tun group than those in the control group (
CONCLUSION:
The study provides strong evidence that tunicamycin-induced ERS induces the apoptosis of human OC SKOV3 cells through inhibiting PI3K/AKT/mTOR signaling pathway.
Introduction
Ovarian cancer (OC) is the sixth commonest malignancy cancer worldwide and one of the three main types of gynecological tumors, which ranks in the third position after cervical carcinoma and uterine corpus cancer [1]. In 2008, it was estimated that 225,500 women were diagnosed with OC and 140,200 women died from this malignant disease in the world [2]. Even though OC accounts for only 3% of all cancers, this malignant tumor is disproportionally deadly owing to the lack of either obvious symptoms or effective strategies of screening and early observation, resulting in more than 70% of patients being diagnosed with advanced OC, which generally confers a poor prognosis and a low survival rate even treated aggressively and immediately [3]. To suppress the elevating morbidity and mortality of OC at advanced stage attributed to the absence of early diagnostic techniques and adequate chemotherapy, previous evidence devoted to investigating the etiology of the disease from the aspect of molecular mechanism [4]. The present study thus aims to find out the association between endoplasmic reticulum stress (ERS), a complex membranous network that participates in multiple cellular processes, and OC SKOV3 cells proliferation, migration and invasion, in the hope of developing a new alternative treatment for OC.
The endoplasmic reticulum (ER) is responsible for a large part of the total protein output in a mammalian cell, however, even though the protein flux through the ER is carefully observed, it is still hard to avoid the presence of misfolded proteins within the ER [5]. Accordingly, an unfolded protein response (UPR) is built in a mammalian cell to induce apoptosis in the ERS environment, thus ERS and UPR activation are identified to play crucial roles in the occurrence and progression of many solid tumors [6]. Phosphatidylinositol 3-kinase (PI3K) alteration in cancer is well-documented as a leading cause of malignancy, and PI3K inhibitors have been widely studied for control of OS [7]. The Akt kinases are homologous kinases and function as a major downstream effector of PI3K, and it has been pointed out that signals generated by activated Akt can affect the entire signaling networks [8]. PI3K/AKT/mammalian target of rapamycin (mTOR) network plays a vital regulatory role in cell proliferation, and has been identified as a therapeutic target in breast cancer treatment [9]. However, the reports about relationship among ERS, PI3K/AKT/mTOR pathway and OC are still limited, thus this study is designed to explore whether ERS can increase the apoptosis rate of OC SKOV3 cells through suppressing the activity of PI3K/AKT/mTOR pathway, in the hope of developing new treatment with great clinical efficacy for OC patients.
Materials and methods
Cell culture and grouping
The human OC SKOV3 cell lines were bought from the Institute of Biochemistry and Cell Biology, SIBS, CAS and grown in RPMI-1640 medium with fetal bovine serum (FBS) (10%) in an incubator (Sanyo, Japan) with 90% relative humidity in 37
Cell proliferation by cell counting kit-8 (CCK-8) assay
SKOV3 cells in each group were inoculated in the 96-well plate. The well with medium alone was assigned as the blank control group. After cells attached to the wall, cell number was counted at a specific time and the steps were as follows: Discarding the medium, adding fresh medium (100
Cell apoptosis by flow cytometry
Cells were collected and adjusted to 1
Cell migration ability by scratch assay
Cells were inoculated in the 6-well plate with 5
Cell invasion ability by Transwell
Forty
Western blotting
Protein lysis solution (Beyotime Biotechnology Co. Ltd., Shanghai, China) was applied to extract the total protein of cells in each group. Protein quantification was performed using Bradford (Thermo Fisher Scientific Inc.Waltham, MA, USA). After 50
Statistical analysis
The statistical analyses were conducted with SPSS21.0 (SPSS Inc., Chicago, IL, USA). All measurement data were expressed with the means and standard deviations (
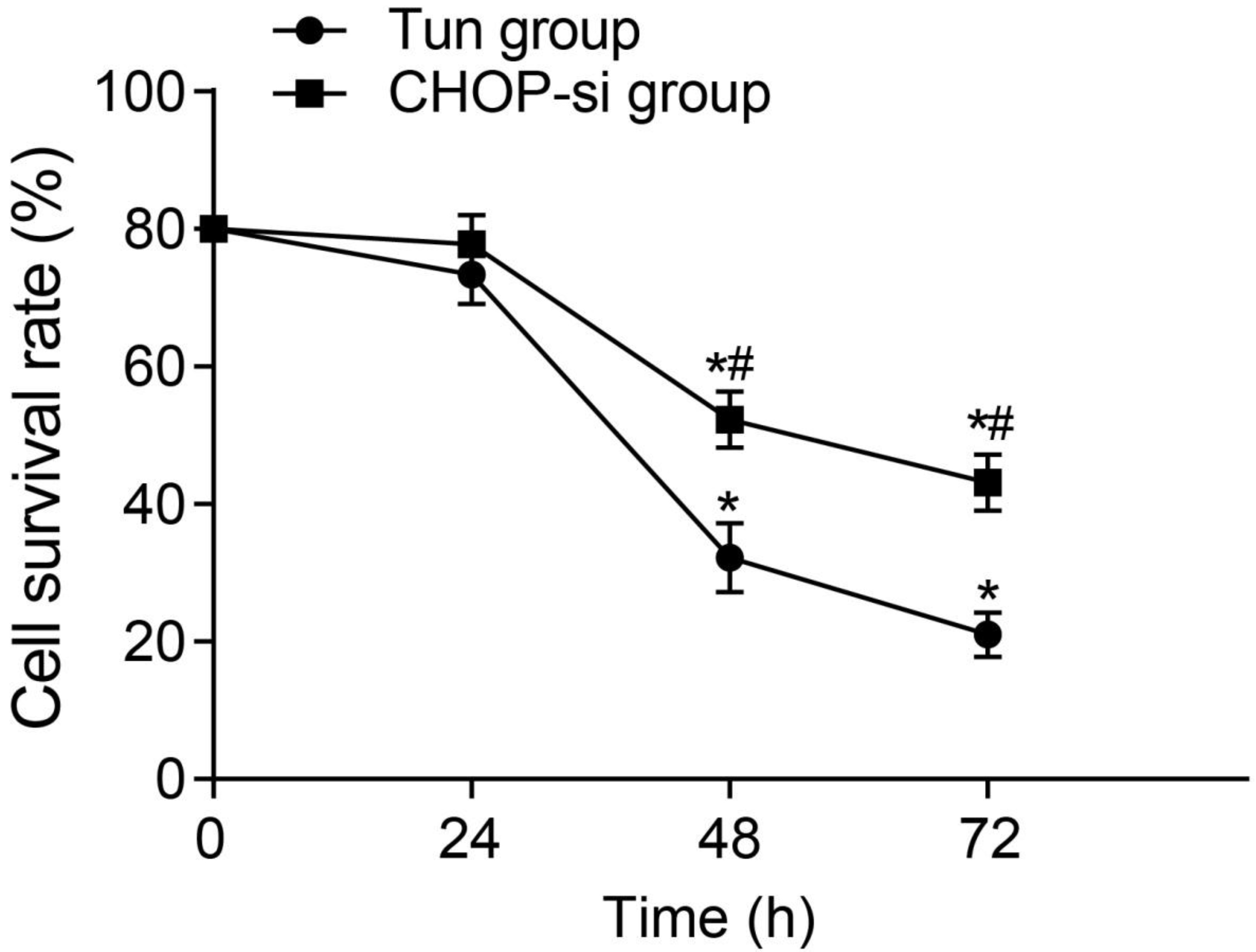
The effect of CHOP-siRNA transfection on the cell survival rate of tunicamycin-induced ERS in OC SKOV3 cell model. Note: ERS, endoplasmic reticulum stress; OC, ovarian cancer.
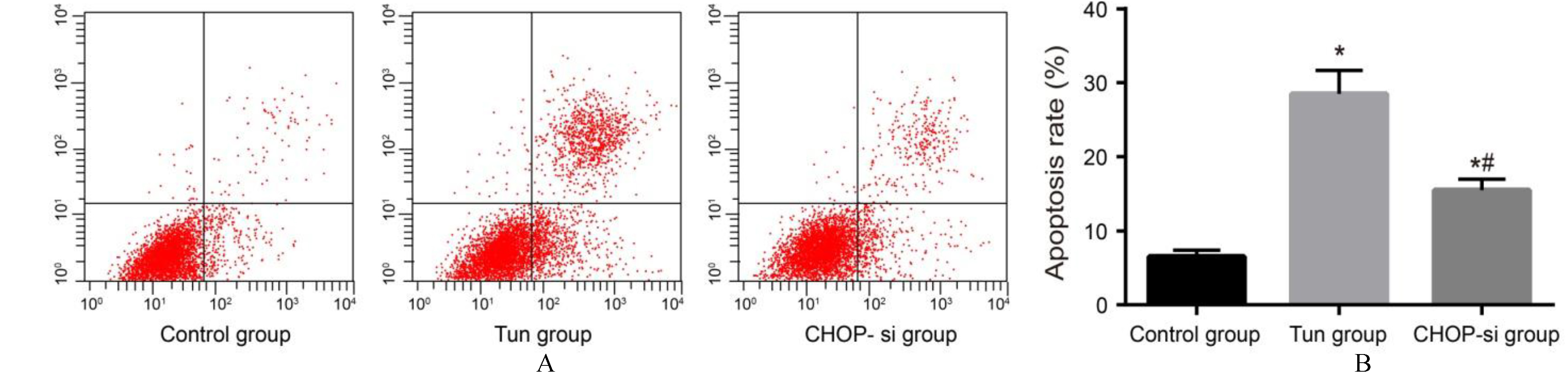
Comparisons of the cell apoptosis rates (B) of OC SKOV3 cells among the control, Tun and CHOP-si groups by Flow cytometry (A) (%). Note: OC, ovarian cancer.
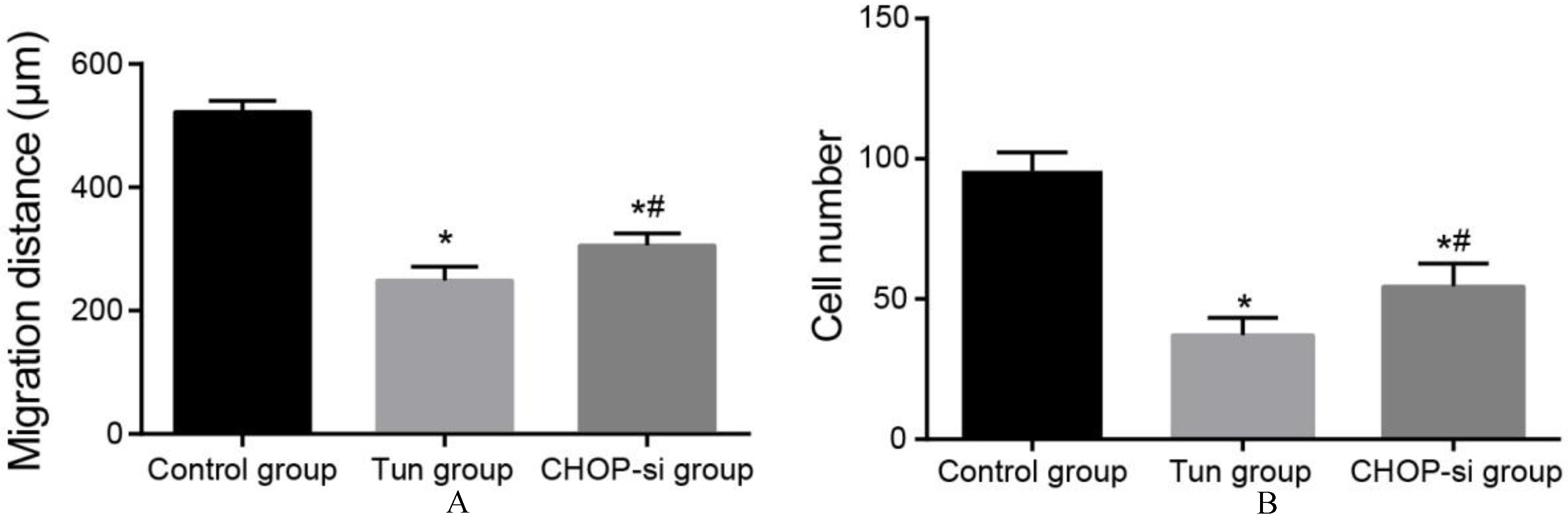
The migration distance (A) and the number of invasive cells (B) in three groups. Note:
Effect of silencing CHOP on cell viability of tunicamycin-induced ERS in OC SKOV3 cell model
The results of CCK-8 assay (Fig. 1) showed that, the cell survival rates gradually declined over time in the Tun and CHOP-si groups, which were also significantly different at the time points of 48 h and 72 h compared with at 0 h, respectively (both
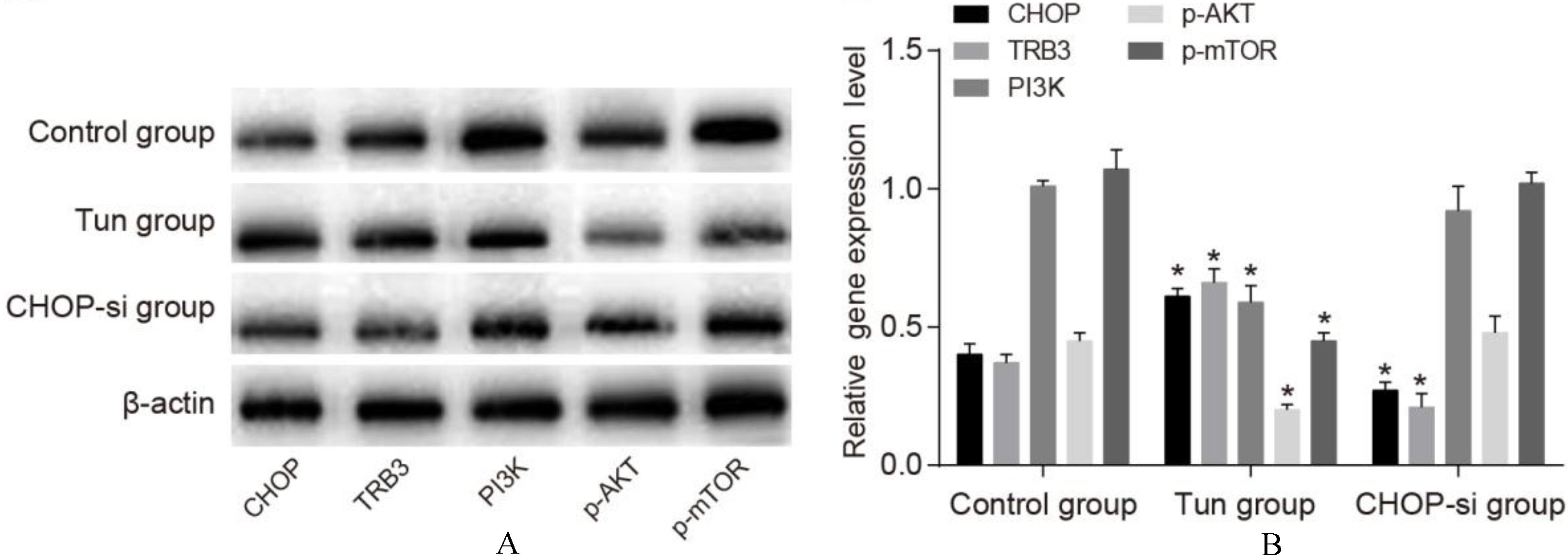
The expressions of ERS and PI3K/AKT/mTOR signaling pathway related proteins in three groups by western blotting (A) and histogram (B). Note:
Flow cytometry (Fig. 2) showed that in comparison with the control group (6.52
Comparisons of cell migration and invasion abilities of OC SKOV3 cells among three groups
The scratch assay and Transwell assay results (Fig. 3) revealed that compared with the control group, horizontal migration distance and the number of invasive cells in the Tun group and the CHOP-si group were reduced (both
Comparisons of expressions of ERS and PI3K/AKT/mTOR signaling pathway related proteins in OC SKOV3 cells among three groups
Western-blotting results (Fig. 4) revealed that the expressions of CHOP and TRB3 were upregulated in the Tun group than those in the control group (both
Discussion
The high variability among subtypes of OC has made the treatment for individual tumor subtypes a huge clinical challenge [10]. ER dysfunction is reported to be linked with tumor progression [11]. Therefore, our study aims to explore the role of ERS in the proliferation, migration and apoptosis of OC SKOV3 cells and to further investigate the regulatory mechanisms. The results of our study revealed that tunicamycin can increase the ERS level and ERS can induce cell apoptosis through inhibiting PI3K/AKT/mTOR signaling pathway, indicating that tunicamycin treatment may be a new regimen for OC patients.
Firstly, the findings in the study revealed that tunicamycin could increase ERS level in OC SKOV3 cells, and tunicamycin was also able to suppress proliferation, promote apoptosis and inhibit the migration and invasion levels of SKOV3 cells, while silencing CHOP may attenuate the protective effects of Tun. CHOP and TRB3 is significantly increased in the Tun group and decreased in the CHOP-si group. Tunicamycin is known as a typical ERS inducer applied in clinical studies, and it enhances ERS mainly through interfering with the first step of N-linked protein glycosylation which cause extensive UPR activation, thereby promoting apoptosis [12]. TRB3 is related to apoptosis and increases during ERS [13]. CHOP, also known as gadd153, is a primary cellular response for the control of ERS-induced apoptosis, and is also able up-regulate the transcription of pro-apoptotic factors and down-regulate anti-apoptotic genes [14]. B cell lymphoma-2 (Bcl-2) is a human proto-oncogene, and its product is called Bcl-2 which can suppress tumor cell apoptosis by preventing the activation of the caspases in the process; however, Bcl-2 associated X protein (BAX) is a pro-apoptotic member of Bcl-2 protein family, and increased Bax expression combined with reduced Bcl-2 expression have been identified to cause tumor cell apoptosis [15]. The decreased level of Bcl-2 is known as a transcriptional suppressing target of CHOP [14]. Furthermore, Bcl-2 is reported to be a downstream target gene inhibited by CHOP and suppressed by tunicamycin at the same time [12]. Therefore, upon CHOP-silence, we assume that Bcl-2 may continue to decrease the apoptosis rate of OC SKOV3 cells, thus diminish the effects of Tun. The research of Li et al., report that ERS activation is associated with the promoted breast cancer cell invasion and migration [16]. The mechanism of tunicamycin suppressing OC SKOV3 cells migration and invasion remains unclear, it is previously reported that strong activation of pancreatic ER kinase (PERK) in response to severe ERS inhibited medulloblastoma cells migration and invasion level, which is consistent with our study [17].
In addition, further investigation improved our understanding of the mechanism of ERS in inducing SKOV3 cell apoptosis, indicating that ERS may promote the apoptosis of SKOV3 cells through suppressing the activity of PI3K/AKT/mTOR pathway. The PI3K/AKT signaling pathway is a pivotal medium in the signal transmitting and the regulation of critical cellular responses, including cell proliferation, apoptosis, differentiation and senescence, and PI3K/AKT signaling pathway has been reported recently to play a crucial role in the control of cell survival through suppressing ERSinduced cell apoptosis [18]. The mTOR kinases are known as critical downstream kinases of the AKT pathway which play a dual role in malignant tumors, and the activation of the AKT pathway usually phosphorylates mTOR to have a positive effect [19]. And the inhibited PI3K/AKT/mTOR pathway activity is reported to contribute to cell apoptosis in colorectal cancer [20], and promote osteosarcoma cells proliferation and invasion [21]. As we have discussed above, the balance between Bcl-2 and BAX is greatly important in the progression of malignancy, and PI3K/AKT/mTOR pathway has been demonstrated to up-regulate Bcl-2 expression while downregulate Bax expression with co-function with ERS [22], which resembles our findings.
All in all, the study provides evidence that tunicamycin is capable of increasing ERS level in OC SKOV3 cells and suppressing the proliferation, promoting the apoptosis and inhibiting the migration and invasion ability of SKOV3 cells, while silencing CHOP may downregulate the protective effects of Tun. We have also observed that ERS may promote the apoptosis of SKOV3 cells via suppressing the activity of PI3K/AKT/mTOR signaling pathway. Therefore, tunicamycin therapy may be a promising regimen with great efficacy in the treatment for OC. However, given that we have only discussed about the role of tunicamycin in the growth of OC SKOV3 cell lines, studies about the effects of tunicamycin in other OC cell lines are still needed to determine the benefits of tunicamycin treatment for OC patients.
Footnotes
Acknowledgments
This work was supported by the Department of Hea- lth Project of Heilongjiang Province (No. 2013013). The authors acknowledge all the reviewers who had given supports for our article.
Conflict of interest
We declare that we have no conflict of interest.
