Abstract
MR Spectroscopy (MRS) has been used for diagnosis of many neurological disorders such as central nervous system tumors and neuro-inflammatory diseases. Despite many studies on MRS in neurodegenerative disorders and particularly Alzheimer’s disease, its utility has been hampered by lack of standard techniques across studies and limited data on the association between the level of metabolites in MRS and pathological changes of AD. In this review we have summarized the findings of MRS in Alzheimer’s disease, discussed the limitation of the current data and the pitfalls in interpretation of the MRS results and also we presented the latest developments in this field and potential future steps in order to utilize MRS more effectively in clinical practice.
Introduction
A number of studies in the past two decades have investigated the role of MR Spectroscopy (MRS) in neurodegenerative disorders. Brain metabolites could act as surrogate markers for different pathological processes such as neuronal damage, glial proliferation, loss of membrane integrity, and even inflammatory changes. Therefore, MRS has the potential of detecting the early metabolic changes in Alzheimer’s disease (AD) while the structural MRI remains normal [14,49,55]. In addition, MRS is a readily available technique and relatively inexpensive with no risk of radiation. These characteristics make MRS a valuable potential biomarker to bridge the gap between the pathological changes [5,45] and clinical presentation of AD particularly in the clinical setting where access to PET is limited.
Changes in the level of MRS metabolites in Alzheimer’s disease
N-acetylaspartate
Decrease in N-acetylaspartate (NAA) is one of the main reported findings in AD [15,37,56] (Fig. 1). NAA is an amino acid that is predominantly synthesized intraneuronally in the mitochondria of the neuronal cell body, axons, and dendrites but not the glial cells. Its normal concentration is around 10–12 mmol/L [61] and it is a marker of neuroaxonal density and viability. It has been shown in different neurological disorders that the level of NAA indicates the neuronal vitality and function [34,46,57,58,60].
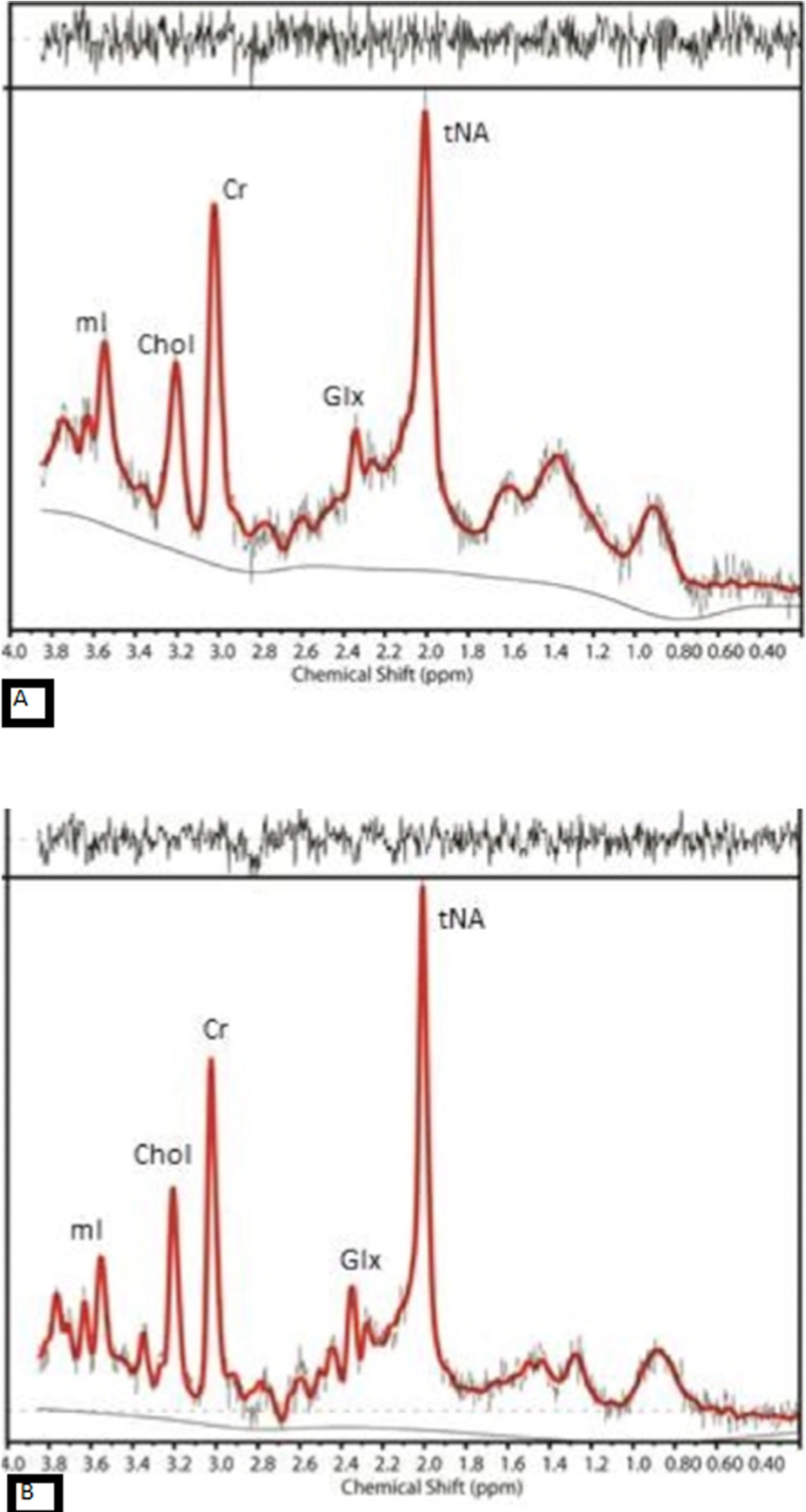
The MR spectra from posterior cingulate gyrus of (A) an AD case with amyloid deposition and (B) an aged-match control with no evidence of amyloid in the brain, show reduction in NAA and increase in mI in AD compared to control.
Both NAA absolute value and the ratio of NAA over Creatine (NAA/Cr) have been assessed in separate brain regions as well as the whole brain [12]. Reduction of NAA has been reported in the medial temporal lobe [1,8], hippocampi [1,4,15,52], and posterior cingulate gyrus [7,10,24]. Hippocampal NAA/Cr value has been reported to have the highest sensitivity and specificity to differentiate AD from HC [10]. A study using Serial MRS confirmed longitudinal reduction of NAA in the posterior cingulate gyrus of AD cases [37]. It has been claimed that the combination of NAA and [Cr + PCr] has a 74% accuracy in distinguishing AD from healthy control (HC) [15].
Although reduction of NAA is a known feature in AD, the results are not very consistent in those with mild cognitive impairment (MCI). Some studies claimed significant difference in the level of NAA in MCI cases compared to HC [16,28,37], while others found a difference between MCI and AD but not between MCI and HC [15]. Moreover, some studies found an intermediate reduction of NAA in MCI which was not significantly different from either AD or HC [56]. Some studies have shown that reduction of NAA in MCI cases may predict future conversion to AD [25,38]. Compared with non-converters, converter MCI cases appear to have lower levels of NAA at baseline and further significant reductions in follow up studies in posteromedial parietal cortex [37].
In addition to NAA reduction, increase in myoinositol (mI) in several brain regions is considered another feature of AD [2,23,55] (Fig. 1). mI is a glial marker and increase in the level of mI can be a sign of increased glial content, size, and activation [11,58]. Increase in mI or mI/Cr is assumed to be an early event in the course of AD pathology and can precede the NAA reduction [7,23,35]. This means glial proliferation can potentially happen prior to neural dysfunction in the course of AD [20,23]. Moreover, there is no evidence of longitudinal increase in the level of mI corroborating the assumption that elevation of mI is an early change in the course of the disease and reaches the plateau relatively early on [22]. Compared to HC population, higher levels of mI/Cr have also been reported in MCI cases. Detection of NAA reduction and mI increase in AD compared to MCI and HC improves the specificity and accuracy of the clinical diagnosis significantly (100% for distinguishing AD from HC) [61]. These two metabolites provide complementary information about AD pathology and the combined ratio of these two metabolites (NAA/mI) has the strongest association with the pathological severity of the disease [26].
Choline
Elevation of Choline (Chol) in AD is another finding reported in some of the studies [23,61] with no significant longitudinal change in serial MRS scans during the course of the disease [2,10]. Chol in MR spectrum is the product of membrane phosphotidylcholine breakdown and its elevation in AD cases might be a consequence of increased membrane turnover secondary to neural damage and degeneration. Alternatively, raised Chol peak could be due to a compensatory mechanism to overcome the deficit in the acetylcholine production in AD by increasing in the catabolism of the phosphotidylcholine membrane to provide free Chol [32,65]. There are, however, some studies which didn’t find any significant difference in the level of Chol between AD and HC [29,36,39,43,52]. Most of the studies that didn’t find any change in Chol, measured metabolites in temporal lobes or hippocampi while region of interest in studies with increased Chol were mainly in posterior cingulate gyrus (PCG). This may explain the contradictory results between different studies. Raised Chol/Cr has also been reported in MCI cases compared to HC with no difference between stable and converter MCI at the baseline. Interestingly, the stable MCI cases had more reduction in the level of Chol/Cr over time compared with converter MCI and control. The authors hypothesized that these findings were suggestive of compensatory mechanism by upregulation of the choline transferase in stable MCI cases [37].
Glutamate + glutamine
The level of Glutamate + Glutamine (GLx) is also reported in some studies to be lower in those with AD and MCI [3,13,17]. Glutamate (Glu) is the most prevalent and the main excitatory neurotransmitter in the central nervous system [67]. Glutamine (Gln) is the product of Glu conversion in presynaptic neurons. The continuous cycle of Glu–Gln mediates glutamatergic neurotransmission [64]. The reduction in the level of Glx in AD is seen in the anterior cingulate cortex, hippocampi [19], medial temporal lobe [63], and PCG [17]. Its level has also been reported to be reduced in MCI cases[48]. There are, however, other studies which didn’t identify any significant difference in the level of Glx between AD and healthy controls [24]. Limitation of the current MRS techniques is a possible explanation for these conflicting results. With the current MRS technique, it is challenging to separate Glu from Gln, hence the combined peak is measured as Glx, even separating Glx peak from other metabolites and quantification of Glx in both normal and diseased individuals is proven to be difficult with low field MRS.
MRS metabolites, amyloid and tau
There are limited data on the spatial and temporal associations between changes in the level of MRS metabolites and presence or load of misfolded proteins as the hallmarks of AD. In one study, the correlation between the level of NAA/Cr in different brain regions and CSF biomarkers revealed an association between hippocampal NAA/Cr and CSF AB and parietal NAA/Cr and CSF-tau [4]. In another study, however, no significant correlation was identified between global cortical AB load as measured by 11C-Pittsburgh Compound B (11C-PiB) PET scan and the level of NAA/Cr in cognitively normal older adults [27]. In autopsy samples, the NAA/Cr was associated with neurotic AB plaques [26] and also with reduction of the synaptic vesicle immunoreactivity and accumulation of early neuritic and pre-tangle tau in the posterior cingulate gyrus but not with the extracellular Neurofibrillary tangle of tau. Based on these findings, the authors postulated that NAA/Cr is a marker of early tau-mediated dynamic processes in AD [39].
The mI/Cr ratio is also associated with both neuritic and diffuse AB plaques in autopsy samples [26,39]. Histopathology studies have also confirmed that there are foci of proliferated glial cells, source of increased mI, in the proximity of AB plaques [42]. Furthermore, global cortical and regional PiB uptake in the posterior cingulate gyrus (PCG) showed a correlation between amyloid burden and elevation of mI/Cr in cognitively normal elderly [27]. More recently, Voevodskaya et al showed similar associations between mI/Cr and 18F-flutemetamol uptake in the PCG in cognitively healthy adults [62].
Despite extensive literatures on the relationship between AB and Cholinergic system in animal models and in-vitro [6,31,40], the same results cannot be replicated in human. In human studies, elevation of Chol/Cr ratio was reported to be associated with higher amyloid uptake in PiB-PET in posterior cingulate gyrus in cognitively normal elderly [27]. The level of Chol/Cr was also higher in PCG/ precuneus of AB + MCI individuals compared to AB – controls [62]. Histopathology studies, however, have shown conflicting results; some studies did not find any association between level of Chol and neuritic or diffuse AB plaques [26,39], while in another study, Choline acetyltransferase (ChAT) activity was shown to be inversely associated with PiB binding in precuneus area in autopsy samples of subjects with AD, but not in MCI [21]. Moreover MCI subjects have shown up-regulation of ChAT in the frontal lobes despite high AB load [9]. The underlying mechanism for the region-specific differences in the relationship between AB and cholinergic activity at early stages of disease is unclear [21].
The relationship between Glx and AB is also not completely understood. Many in-vitro and animal studies have shown that AB affects the glutamatergic synapses and causes the dysregulation and excitotoxic neuronal damage [18,44]. However, the results of human studies are contradictory. In some studies, the reduction in the level of Glx in posterior cingulate gyrus was associated with higher total cortical PiB uptake [66] while others didn’t find any association between reduced Glx in PCG and AB load [48]. Tau is also shown to affect glutamate receptors in preclinical research [59], but there is lack of data on human to confirm this association. On the other hand, the association between Glx and FDG has been reported in many conditions such as epilepsy [47], tumor [50] and behavior variant of frontotemporal dementia (bvFTD) [30].
Controversial results and limitations of the previous studies
Although the results of the initial MRS studies in AD were promising, there is lack of transition to the clinical setting. The main reasons for the limited use of MRS in diagnosis of AD are lack of specificity and also the controversial results at early stage of the disease, particularly for metabolites such as Chol and Glx.
The discrepancy seen in the literature might be due to several factors: Heterogeneity and different selection criteria for the MCI/preclinical AD in different studies. Technical differences in acquiring MRS data across studies. Voxel position variations between different studies as the level of some of the metabolites are region dependent. Different composition of voxels (the percentage of gray matter, white matter and CSF) since metabolites also have different ratios in white and gray matter. Lack of knowledge about presence of underlying pathological changes such as presence of amyloid B (AB), tau, Lewy body Inherent limitations of lower field MRS to detect some of the metabolites (such as Glx, or GABA) reliably. Limited data on validation of the MRS changes against other more established imaging biomarkers and pathology results. The association between changes in the level of metabolites and other disease biomarkers have not been thoroughly investigated in human.
New developments in the field of MRS can potentially address some of the above-mentioned limitations. In the first PET-guided MRS study, level of metabolites was measured in the regions of AB deposition and hypometabolism identified by PET in order to assess the regional and temporal relationships between PET markers of AD and the level of metabolites. Despite its modest sample size, the study showed that the level of NAA had regional associations with both AB and hypometabolism identified by PiB and FDG-PET respectively. However, when the regression model was corrected for clinical diagnosis NAA was only associated with hypometabolism as a marker of neurodegeneration. There was also a stepwise reduction in the level of NAA from healthy controls to MCI to AD, while neither of PET SUVRs could differentiate MCI from early AD in the same cohort. In the same study, the level of mI was found to have strong regional and temporal associations with AB, presenting the same pattern of changes across the disease spectrum [53].
It has also been shown that the association between Chol and AB in PET-guided MRS becomes significant only after correcting for the voxel position, which confirms the regional variations in the level of Chol in the brain. It may also explain the discrepancy between the results of previous studies. The same study showed the level of Glx was associated with FDG uptake again only when the regression model was adjusted for voxel position [54]. These results however, need to be confirmed in larger studies and in different cohorts.
Utilizing ultra-high field MRI can also overcome many of the current limitations of the MRS studies. The main advantages of ultra-high field MRS are:
Significantly higher signal to noise ratio and capability of reducing the voxel size if required.
The ability to detect a higher number (14–15) metabolite peaks in the spectrum compared with lower field MRS. High field MRI is particularly beneficial for metabolites such as GABA or Glx as main inhibitory or excitatory neurotransmitters respectively as well as Ascorbate or Glutathione as main antioxidants markers in the brain. Many preclinical studies have confirmed the relationship between the level of neurotransmitters, connectivity and misfolded proteins in animal models and in vitro. However, they haven’t been translated into human study because of the limitation of current in vivo techniques. The initial data on 7T MRS are very promising [33,41] and indicating the potential role of this technique for better measurement of neurotransmitters.
There are also many on-going developments to improve the acquisition techniques, separating the overlapping peaks and measuring the level of metabolites more accurately and only in gray matter [51]. These advances are particularly very crucial for Alzheimer’s study as the PET markers of AB, tau and FDG are based on gray matter uptake only.
MRS can potentially fill the gap between the low sensitivity and specificity structural images currently used in the clinical setting and more advanced, very expensive and not widely available techniques, such as amyloid and tau PET, that are currently only used in research.
Changes in the level of certain metabolites in MRS can be surrogate markers of underlying pathological alterations in AD and potentially MRS can improve the diagnosis, predict the prognosis or assess the response to treatment.
The limitations of the previous studies have hampered the utility of MRS, however, recent advances can overcome some of those shortcomings.
Standardization of MRS techniques and validation of the regional and temporal changes of the metabolites against the known pathological hallmarks of AD with longitudinal follow up across the spectrum of the disease are required to prove the utility of MRS as a potential imaging biomarker for AD.
Conflict of interest
The author has no conflict of interest to report.
