Abstract
BACKGROUND:
Cerium ions promote osteoclastogenesis and activate bone metabolism, while cerium oxide nanoparticles exhibit potent anti-inflammatory properties, making them promising for biomedical applications.
OBJECTIVE:
The purpose of this study was to develop and evaluate a synthesis method for sustained-release cerium-ion bioceramics containing apatite. Substituted apatite was found to be an effective biomaterial.
METHODS:
Cerium-containing chlorapatite was synthesized using a mechanochemical method employing dicalcium phosphate, cerium chloride heptahydrate, and calcium hydroxide as raw materials. The synthesized samples were characterized using X-ray diffraction (XRD), Fourier transform infrared (FTIR) spectroscopy, scanning electron microscopy, energy-dispersive X-ray spectroscopy, and Raman spectroscopy.
RESULTS:
Cerium chlorapatite was successfully synthesized in the 10.1% and 20.1% samples. However, at Ce concentrations higher than 30.2%, the samples consisted of three or more phases, indicating the instability of a single phase.
CONCLUSION:
The method used in this study was found to be more efficient and cost-effective than the precipitation method for producing substituted apatite and calcium phosphate-based biomaterials. This research contributes to the development of sustained-release cerium-ion bioceramics with potential applications in the field of biomedicine.
Introduction
Filling bone grafts and defects resulting from accidents or tumors have been long-standing issues in regenerative medicine. Bioceramics, specifically the functional and absorbable calcium phosphate, have been extensively studied as suitable filling materials [1]. Among various types of calcium phosphates, substituted apatite has attracted particular attention as a biomaterial [2]. Animal bones are composed primarily of carbonate apatite and collagen [3]. Hydroxyapatite has the chemical formula Ca10 (PO4)6 (OH)2. In vivo, the carbonate group in apatite is substituted with a phosphate group. Apatite can exchange cations and anions, making it suitable for drug delivery systems. Apatite nanocrystals can be synthesized using high-energy planetary ball milling, a process known as mechanochemical synthesis. This method has been widely adopted as a green synthesis approach for both organic and inorganic materials [4].
Zinc (Zn) and strontium (Sr) ions are expected to activate bone remodeling [5]. Furthermore, the incorporation of apatite promotes osteoblastic differentiation [6] due to the essential nature of these elements [7]. Sr ions, with a similar ionic radius to calcium ions, can easily replace them [8]. Therefore, Sr ions, such as those found in Sr ranelate, are used to treat osteoporosis [9]. However, they have also been reported to inhibit osteoblast differentiation [10]. Adding active ions to bioceramics can confer functionality.
Cerium (Ce) ions are rare earth elements with an atomic number of 58. They exist in trivalent and tetravalent states and are known for their catalytic properties [11]. Ce oxide nanoparticles have been extensively studied for their biomedical applications over the past few decades [12]. They exhibit potent anti-inflammatory properties and are of great research interest owing to their safe and effective use [13]. Ce ions also promote osteoclast differentiation [14], which may be beneficial for bone remodeling [15]. Ce apatite synthesized using a wet chemical precipitation method by Ciobanu et al. showed antibacterial activity [16].
In this study, we propose a method for introducing Ce ions into apatite via mechanochemical synthesis. Unlike the precipitation method, mechanochemically synthesized apatite can be synthesized in larger quantities (such as grams) depending on the size of the jar, allowing for scalability and environmental friendliness [4]. Apatite enriched with various ions is slowly released by the phagocytosis of osteoclasts. This release occurs through the formation of Howship’s lacunae which are created by the osteoclasts during the process of resorbing bone tissue and allows the use of apatite as a drug delivery system. Cerium-containing chlorapatite was synthesized via a mechanochemical method using dicalcium phosphate, cerium chloride heptahydrate, and calcium hydroxide as raw materials. This paper presents the mechanochemical synthesis and characterization of cerium chlorapatite resulting from the above-mentioned process.
Materials and methods
Materials
Calcium hydroxide, dicalcium phosphate dihydrate (DCPD), and cerium chloride heptahydrate were purchased from Fujifilm Wako Pure Chemical Corporation (Osaka, Japan). Calcium hydroxide and DCPD were Wako Special Grade and Guaranteed reagent grade.
Methods
Water, DCPD, cerium chloride heptahydrate, and calcium hydroxide were mixed and grounded in a planetary ball mill (P-6, Fritsch, Japan). The mixture was prepared with a (Ca + Ce)/P molar ratio of 1.67 and a Ce/Ca + Ce molar ratio ranging from 0 to 40.0%. The powder/water mixture was milled to a Powder/Liquid ratio of 0.5. The mixture was milled at 500 rpm for 1 hour using an 80 mL alumina jar containing 10 alumina balls, each with a diameter of 10. After milling, the samples were stored at 40 °C for more than 24 hours.
Characterization of prepared samples
The prepared samples were examined using X-ray diffraction (XRD), FT-IR, Raman spectroscopy, and scanning electron microscopy-energy dispersive X-ray spectroscopy (SEM-EDS). XRD was performed to evaluate crystallinity using an X-ray diffractometer (X’Pert PRO MPD, Malvern Panalytical Co., The Netherlands) with Cu-Kα radiation (45 kV, 40 mA) and a scanning step of 0.001°. Rietveld analysis based on the XRD patterns was performed using analysis software (HighScore Version 4.9; Malvern Panalytical B.V. 2020, The Netherlands).
For the FT-IR spectroscopy analysis, an FT/IR-4200 spectrometer (JASCO Co., Tokyo, Japan) equipped with an ATR accessory was used. The resolution was set to 1 cm−1, and the totalization was performed 64 times. The air spectrum was used as the background for the spectrum analysis.
Raman spectroscopy analysis was conducted using an excitation wavelength of 532 nm and an excitation power of 0.322122 mW. An ND filter with a transmittance of 99.6% was used, and the center wavenumber was set at 1700. The total exposure time for the analysis was 300 seconds. The prepared samples were also evaluated using SEM-EDS (SEM: FEI Quanta400; EDS: EDX Octane Elect Super). The accelerating voltage and magnification used were 15.0 kV and 1000 times, respectively.
Results
The evaluation of crystal phases is crucial in ceramic synthesis. XRD patterns are commonly used for this purpose. In our study, we evaluated the apatite synthesized by the mechanochemical method using XRD patterns obtained from previous studies [17,18]. Additionally, Rietveld analysis was applied to evaluate whether the crystal lattice was doped with the target ions. The effectiveness of the Rietveld method for doping zirconia into CuO was demonstrated by Hossein et al. [19].
The XRD patterns of the samples prepared via mechanochemical synthesis were analyzed (Fig. 1). The 0%, 10.1%, and 20.1% samples exhibited peaks at 26° and 32°, suggesting that they are apatite-type hexagonal crystals. The 30.2% and 40.1% samples displayed peaks at 11°, 14°, 20°, 25°, and 29°, suggesting that they did not exhibit apatite-like crystal structures. The 0% sample exhibited a peak pattern consistent with DCPD, suggesting a mixture of DCPD and nano-crystalline apatite.
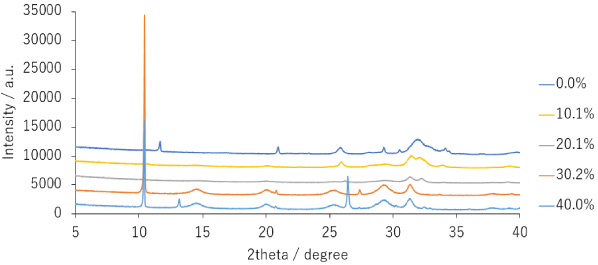
XRD patterns of prepared apatite sample.
The FTIR spectra of the prepared samples were obtained to characterize their properties (Fig. 2) [20]. The FTIR spectra are based on the vibrations of the ceramic, and changes in peak shapes can be attributed to phase transitions. The presence of carbonate in apatite is a particularly important factor for biocompatible materials. Featherstone et al. reported a method for presumptive quantification using 1415 cm−1 and 575 cm−1 bands in the FTIR spectrum of apatite with known carbonic acid content [21]. Studies on cerium apatite synthesis through precipitation methods have indicated that the carbonate content decreases with increasing cerium concentration [22].
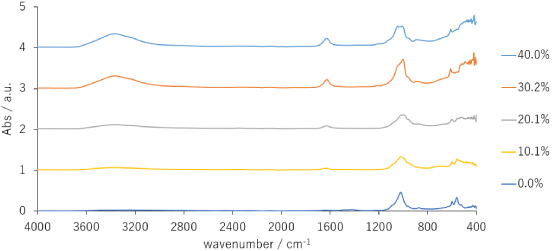
FT-IR spectra of prepared samples.
In this study, the 0% sample exhibited an absorption band around 1400 cm−1, whereas the other samples had no peak at these positions. These results suggest that the 10.1% and 20.1% samples did not incorporate carbonate ions into their crystals. The peak around 1000 cm−1 indicates the IR band of the phosphate group. Carbonate apatite synthesized from tetracalcium phosphate and DCPD has been reported to have a phosphate group band at 1024 cm−1 [23]. DCPD typically exhibits four characteristic phosphate group peaks from 1200 cm−1 to 900 cm−1, which were not observed in any of the samples.
Water molecules exhibit a relatively large peak from 2800 to 3600 cm−1 in the FTIR spectrum. Our results suggest that the 10.1%, 20.1%, 30.2%, and 40.1% samples contained bonded or free water, which aligns with the use of the wet mechanochemical synthesis method employed in this study.
The Raman spectra of the synthesized samples are shown in Fig. 3. Raman spectroscopy is an excellent non-destructive analytical tool that provides unique Raman bands dependent on the structural order. The magnified spectrum was acquired using a microscope, and a line measurement was performed on the sample surface. The phosphate group-based peak at 962 cm−1 is the most important peak for the characterization of apatite. The 0% sample, which is a mixture of DCPD and carbonate apatite, exhibited a peak at 963 cm−1. Similar peaks were observed for the 10.1% and 20.1% samples, suggesting that these three samples were transformed into apatite. The 10.1% and 20.1% samples displayed specific peaks at 960 cm−1 and 962 cm−1, respectively with the wavenumber shifting based on the Ce content. The shift in this peak is observed during apatite fluorination [24]. The XRD Rietveld analysis classified the 40.1% and 30.2% samples as non-apatite. Awonusi et al. reported that carbonate apatite synthesized using the precipitation method exhibited carbonate peaks at 1071 cm−1 and 1076 cm−1 in the Raman spectrum [25]. In the 0% sample, a peak appeared at 1072 cm−1, consistent with the presence of carbonate ions in the IR spectrum. Slight peaks at approximately 1070 cm−1 were also observed in the 10.1% and 20.1% samples.
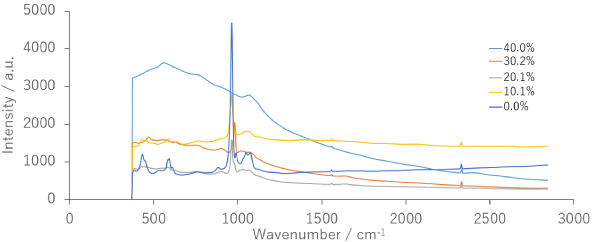
Raman spectra of prepared samples.
SEM-EDX analysis was performed on compressed tablets to evaluate their surface morphologies (Fig. 4A–E). Figure 4A suggests a smooth surface, which aligns with the XRD analysis indicating the presence of apatite and DCPD indicating the presence of apatite nanocrystals in the 0% sample. The 10.1% Ce and 20.1% Ce samples exhibited poor compression properties, with visible cracks on the surface. The 30.2% and 40.0% samples displayed sticky properties and agglomeration, resulting in periodic protrusions on the surface. The Ce content significantly affected the surface morphology owing to the differing phases.
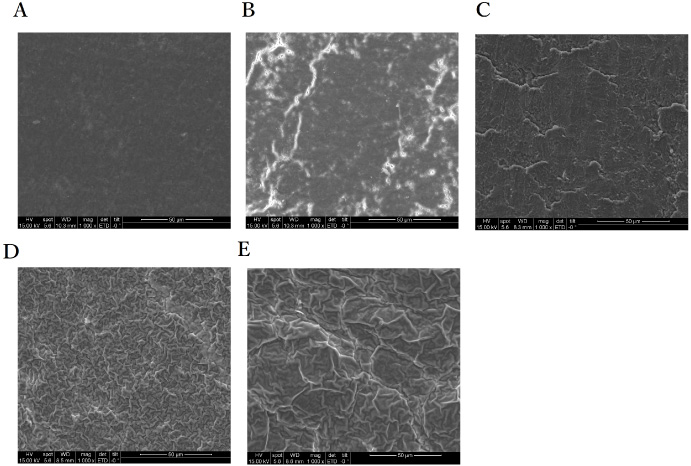
SEM of prepared samples (A) 0%, (B) 10.1%, (C) 20.1%, (D) 30.2% (E) Ce 40.0%.
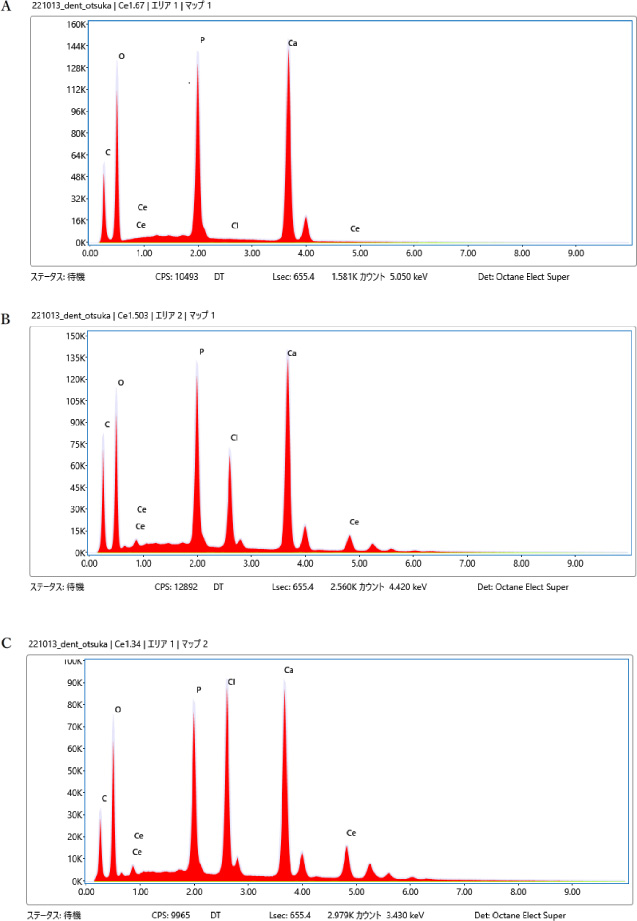
EDX spectra of prepared samples (A) 0%, (B) 10.1%, (C) 20.1%, (D) 30.2% (E) Ce 40.0%.
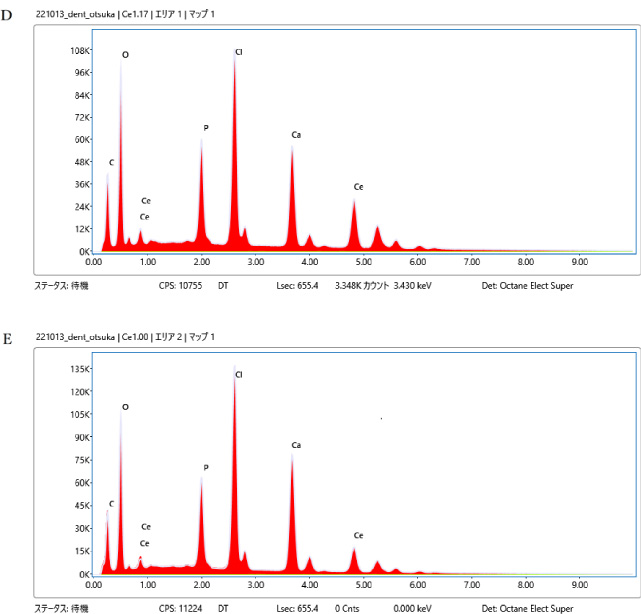
The EDX spectra of the prepared samples are shown in Fig. 5A–E. The combination of EDX and SEM is a powerful tool for analyzing the elemental content and supporting material chemistry. A qualitative assessment of the composition of the prepared samples from the EDX spectra was performed. The 0% sample exhibited no peaks corresponding to the presence of Ce and Cl (Fig. 5A). These peaks were observed in the 10.1%, 20.1%, 30.2%, and 40.0% systems containing cerium chloride. The results align with expectations and are considered reasonable.
The obtained XRD patterns were evaluated using the Rietveld method [26] and the whole powder pattern fitting method [27]. The 0% sample comprised a mixture of DCPD (98-001-6132) 24% and hydroxyapatite (98-003-4457) 76%. The Ce 10.1% sample consisted of cerium phosphate hydrate (01-074-0057) 4% and apatite (01-079-9908) 96%. The Ce 20.1% sample consisted of cerium phosphate (01-074-1889) 5%, and apatite (01-082-3638) 95%. The calculated lattice parameters and space groups are listed in Table 1.
Calculated lattice parameters and space groups
Calculated lattice parameters and space groups
The results revealed that pure apatite is not a stable phase in mechanochemical synthesis. Furthermore, cerium chlorapatite is not registered in the Rietveld database, indicating an unexplored area for cerium-doped apatite and potential for future research. Furthermore, the lattice constant changes with the Ce concentration, and the crystal lattice of apatite is affected by cerium ions.
Cerium apatite might possess antibacterial properties and be biocompatible [16]. While combining antibiotics and apatite can achieve antibacterial properties, there is a risk of organic matter combustion during the firing process. Thus, the antibacterial properties of cerium chlorapatite might provide an advantage. We hypothesize Ce ions are released during osteoclast fusion. According to Thill et al. [28], cerium compounds adsorb to the membranes of Gram-positive and Gram-negative bacteria through electrostatic interactions, impair transport exchange by disrupting ion pumps, and prevent bacterial growth. Additional possible effects include a direct attack on proteins, generation of reactive oxygen species, and hydrolysis of DNA oligomers [29]. Cerium apatite’s antibacterial properties make it a potential candidate for bone and tooth-filling materials.
For example, it could be used as lining material for cavities to reduce the risk of secondary caries. As the interior of a tooth is vulnerable to bacteria filling it with scraped cerium apatite could enhance its utility.
In this study, cerium-containing chlorapatite was successfully synthesized in the Ce 10.1% and 20.1% samples using a mechanochemical method. The synthesized samples were characterized using XRD, FTIR, Raman, and SEM-EDX analysis. At higher Ce concentrations, were synthesized. The mechanochemical method offers advantages over the precipitation method as it allows for the synthesis of larger quantities without requiring a large amount of solvent; therefore, it is an environmentally friendly synthesis method. Due to its potential antibacterial properties, cerium-containing chlorapatite is being considered for use as a bone replacement material and as a supplement for bone spaces that have been removed.
Footnotes
Acknowledgements
This study was partially supported by a Grant-in-Aid for Scientific Research No. 23K16044 from the Japan Society for Promotion of Sciences (JSPS).
