Abstract
Structural properties and chemical bonding of new composites (ZnMnO2 and activated carbon) with honeycomb structure have been studied by using X-ray diffraction and Fourier transform infrared. The composition of the powder of ZnMnO2 has been studied by X-ray fluorescence in waste derived from a conventional battery. X-ray diffraction data show that the average crystallite size and the dislocation density for the composite are 90.17 nm and 12.30 × 1013 m−2, respectively. The C–O and C=O bonding for composites seen shifted to the lower wave number of pure ZnMnO2 and activated carbon indicated size reduction as an effect of Zn or Mn in the site of activated carbon. In addition, the reflection loss was analyzed by vector network analyzer and shows reflection loss of about −21.72 dB, the frequency range being 4.40–4.59 GHz for a thickness of 6 nm. The honeycomb structure of composite ZnMnO2–AC in this study promises new types of composite materials for various applications.
Keywords
Introduction
Composites of carbon-based material have increasing usage in automobiles, aircraft, and spacecraft due to being light, environmentally stabile, and stronger than some other metals.1–3 However, composite-based carbon demonstrates some weakness during performance in device applications; decreased compressive strength and decreased thermal conductivity may be due to unstable bonding between the base materials.3–13 Employing materials with high volume pore offers advantages such as increased stability performance.4–7 The function of the pore as a matrix and using the semiconductor magnetic materials can be optimized and may address stabilizing bonding during the operation of the device applications. This approach may provide best performance materials in device applications to achieve new generation composite materials.
Recently, materials from waste are gaining attention due to environmental problems. Some waste materials can be recycled to produce new materials which can be used in many other applications. Waste from agriculture products is usually used as a source of carbon material in the form of carbon black, carbon dot, carbon active, and also as a source of new types of energy.4–7 Other types of waste, household or industrial waste, can cause environmental problems due to their containing toxic material, 4 for example, conventional batteries. Conventional batteries contain semiconductor magnetic materials and other materials hazardous to the environment. The reuse of semiconductor magnetic materials from conventional batteries can provide other functional materials which can help to reduce problematic side effects in the environment.
Semiconductor magnetic materials are widely used for various applications in the form of compounds: MnFe2O4, SrCO3, Fe2O3, TiO2, MnCO3, and ZnFe2O4.13,14 These materials also are usually used as a filler in polymeric materials or carbon type materials as a matrix. To the best of our knowledge, there is no report on the synthesis, bonding characteristics, and structural properties of magnetic materials derived from waste as a filler for honeycomb strucure. In this study, magnetic materials of ZnMnO2 we investigate the structural properties, bonding characteristics, and reflection loss properties of ZnMnO2 derived from the waste of conventional batteries as a filler for activated carbon (AC) in paper honeycomb structure by using X-ray diffraction (XRD), Fourier transform infrared (FTIR), and vector network analyzer (VNA), respectively.
Experiments
Materials
Materials used in this study include powder ZnMnO2 obtained from the waste of conventional batteries produced by PT. ABC Indonesia. The powder was immersed several times in 150 ml of aquabidest for removal of the Cl element, which was controlled by X-ray fluorescence (XRF) to form ZnMnO2. The ZnMnO2 was dried by using a furnace at 80°C for 3 h. Polyvinyl alcohol (PVA), purity 99.5%, was purchased from Merck, AC was supplied by local company PT. Cahaya Indo Abadi Indonesia, with an average diameter <10 μm, purity >95%, surface area >240 m/g, and honeycomb paper was supplied by World Expert Indonesia.
Composite Preparation
AC, about 9.42 g, was mixed with 24 ml PVA using a magnetic stirrer for 60 min to obtain a solution. Honeycomb paper with thicknesses of 2 mm, 4 mm, and 6 mm was dipped into the solution. Subsequently, AC attached to the paper honeycomb and was dried at 80°C for 2 h. MnZnO2, about 4.71 g, was added into 12 ml aquabidests and stirred using a magnetic stirrer for 3 min to obtain a slurry form and then poured onto the AC honeycomb structure, then dried using a furnace with a temperature of 60°C for 5 h. The procedure for fabrication of the composite’s honeycomb structure materials can be seen clearly in Figure 1.
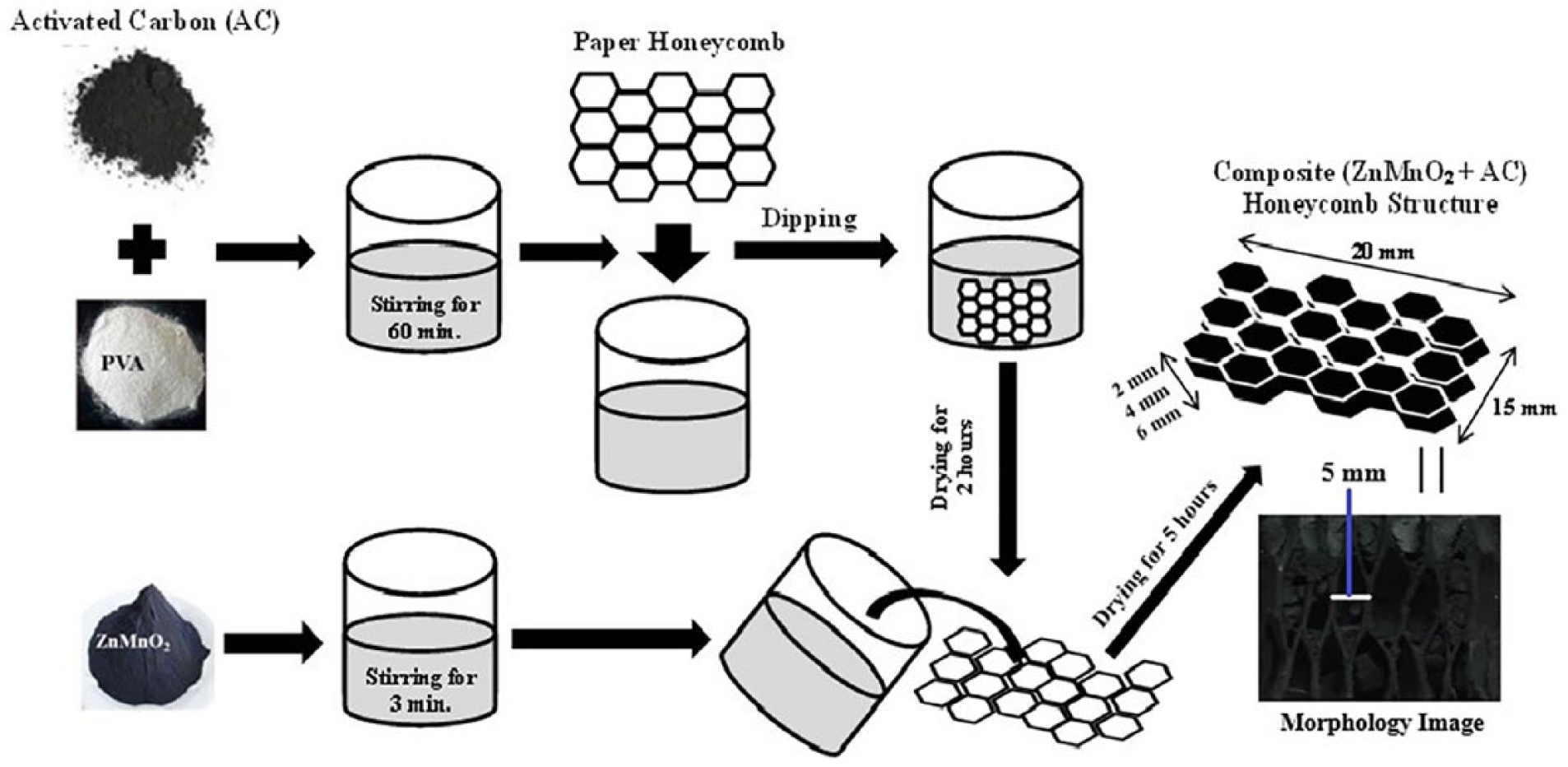
Procedure for fabrication of the composite (ZnMnO2+AC) honeycomb structure materials in this study.
Characterization
XRD data were used to determine the crystallite size of composites which were collected on XRD spectroscopy (Shimadzu 7000) with Cu Kα radiation (λ =1.5405 Å) over the angular range 10o ⩽ 2θ ⩽ 80o, operating at 30 kV and 10 mA. XRF spectroscopy, Model Thermo ARL Quant, was used to determine the elemental composition of the samples. FTIR spectroscopy was carried out on IRPrestige-21 FT-IR spectrometer (Shimadzu Corp.) equipped with a bright ceramic light source, a KBr beamsplitter, and a deuterated triglycine sulfate doped with L-alanine (DLATGS) detector. The measurements of the sample were collected over the range of 4000–600 cm−1 and 16 co-added scans. All spectra of FTIR were in transmittance units. VNA (Rohde & Schwarz. ZVHB) with the frequency range 2.5–8 GHz was used to determined electromagnetic wave absorber performance (reflection loss).
Results and Discussion
Chemical composition analysis performed on the powder from the waste of conventional batteries by XRF found 60.47% MnO, 27.2% ZnO, and 12.33% Cl. The final chemical composition of ZnMnO2, AC, honeycomb paper, and composite’s (ZnMnO2+AC) honeycomb structure is shown in Table 1. ZnMnO2 from the waste of conventional batteries shows the chemical compositions of MnO 51.07% and ZnO 48.93%. Based on the oxide composition of ZnO and MnO as shown in Table 1, we concluded that the material from the waste of conventional batteries is ZnMnO2. The main chemical oxide existing in AC is 38.41% SiO2, 36.47% Fe2O3, and K2O 13.18%, while for honeycomb papers it is 61.22% CaO, 18.59% Fe2O3, and 15.18% K2O. As reported,2,3,8–10,14 the combination oxide of Fe, Mn and Zn to form composites has been analyzed and showed good performance for electromagnetic wave absorber materials. Composites in this study show potential for application as electromagnetic wave absorber materials due to the main contribution from magnetic oxide materials of 31.41%, 30.03%, and 15.99% of MnO, ZnO, and Fe2O3, respectively.
Chemical composition determined by X-ray fluorescence spectroscopy for ZnMnO2, activated carbon, honeycomb paper, and composite (ZnMnO2+AC) honeycomb structure.
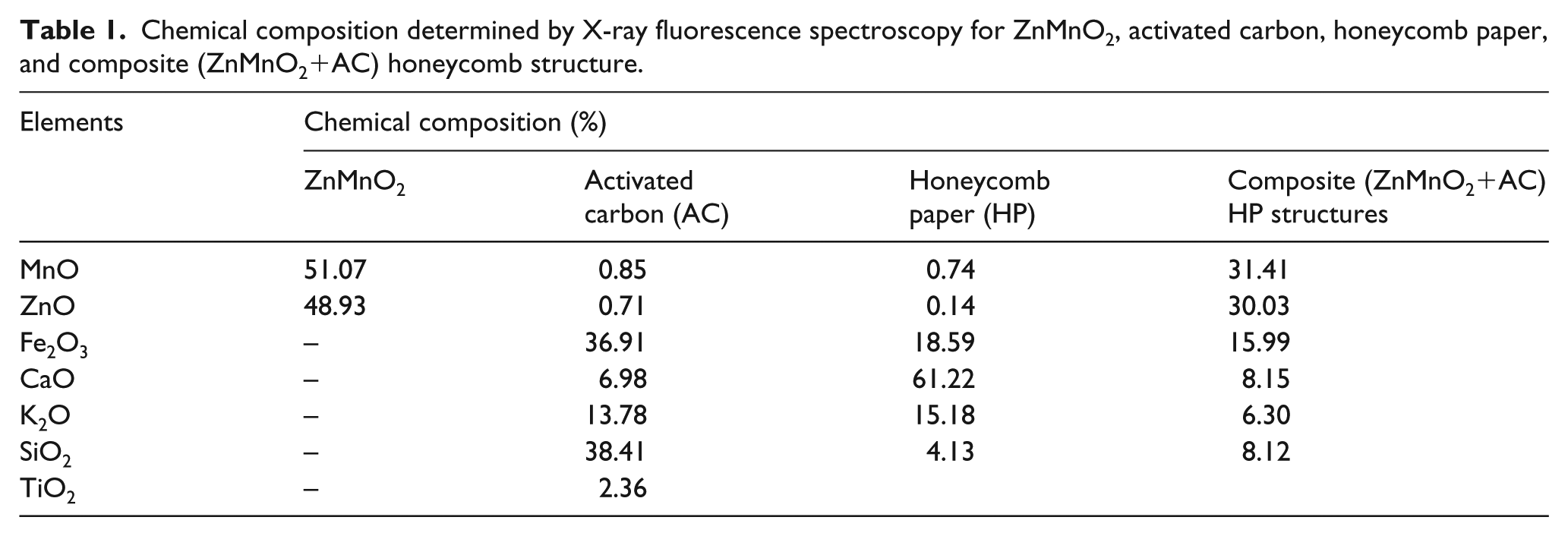
Figure 2 shows the characterization of ZnMnO2, AC, honeycomb paper, and composite using FTIR. ZnMnO2 showed the O-H vibration band of water molecules located at the wave number 3400 cm−1, indicating the existence of water absorbed on the surface of nano powder. Asymmetric and symmetric stretching of C=O is observed of the band at 1403 to 1617 cm−1 and stretching vibration of C-O band at wave number 1014 cm−1 due to the ambient environment.15–18 At wave numbers 526 and 712 cm−1 vibration of Mn-O is shown, and the wave number 470 cm−1 and 901 indicates the vibration band of Zn-O. The vibration mode for all samples by FTIR in this study is shown in Table 2.
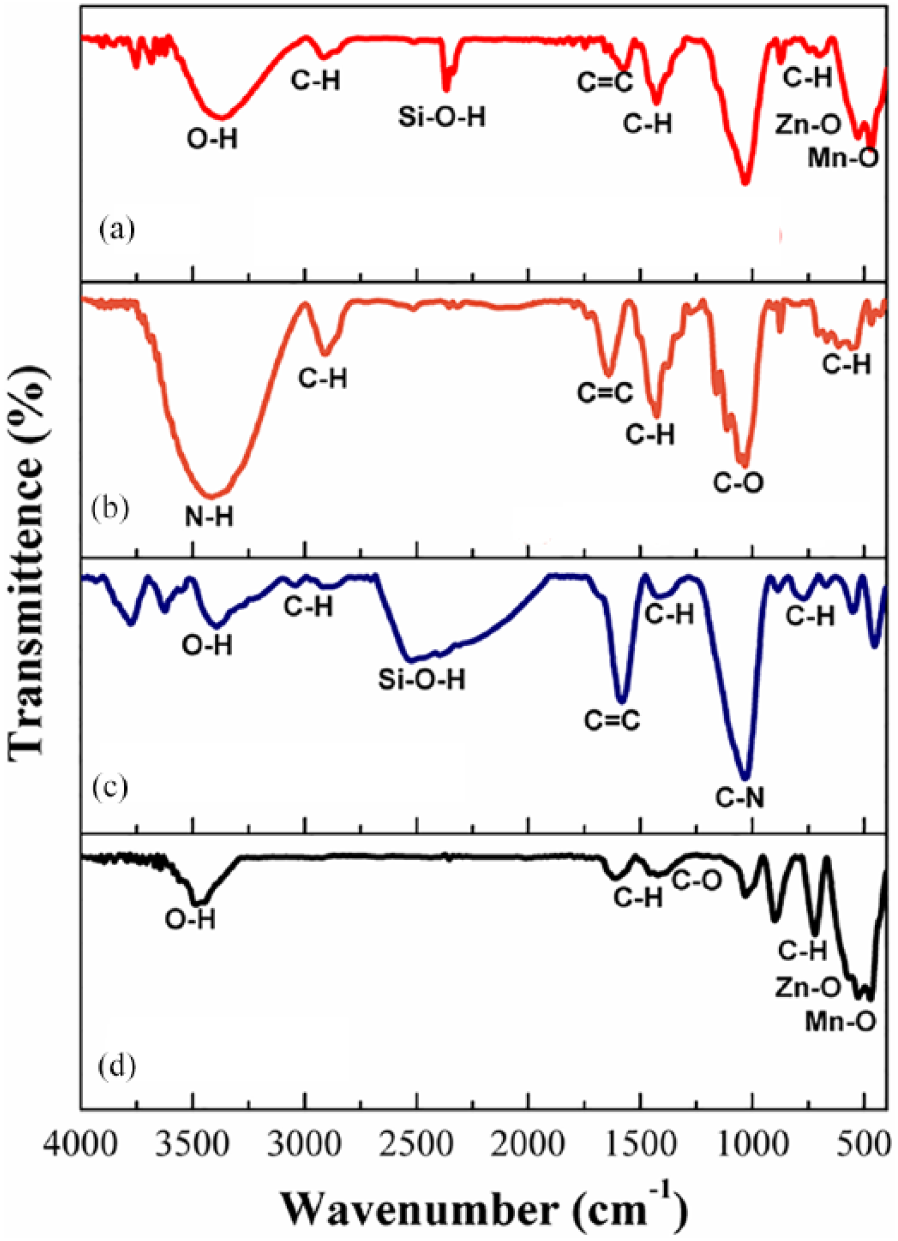
Vibration mode spectra from Fourier transform infra-red spectroscopy for (a) composite (ZnMnO2+AC) honeycomb structure, (b) honeycomb paper, (c) activated carbon, and (d) ZnMnO2.
Vibration mode determined by Fourier transform infra-red spectroscopy for ZnMnO2, activated carbon, honeycomb paper, and composite (ZnMnO2+AC) honeycomb structure.
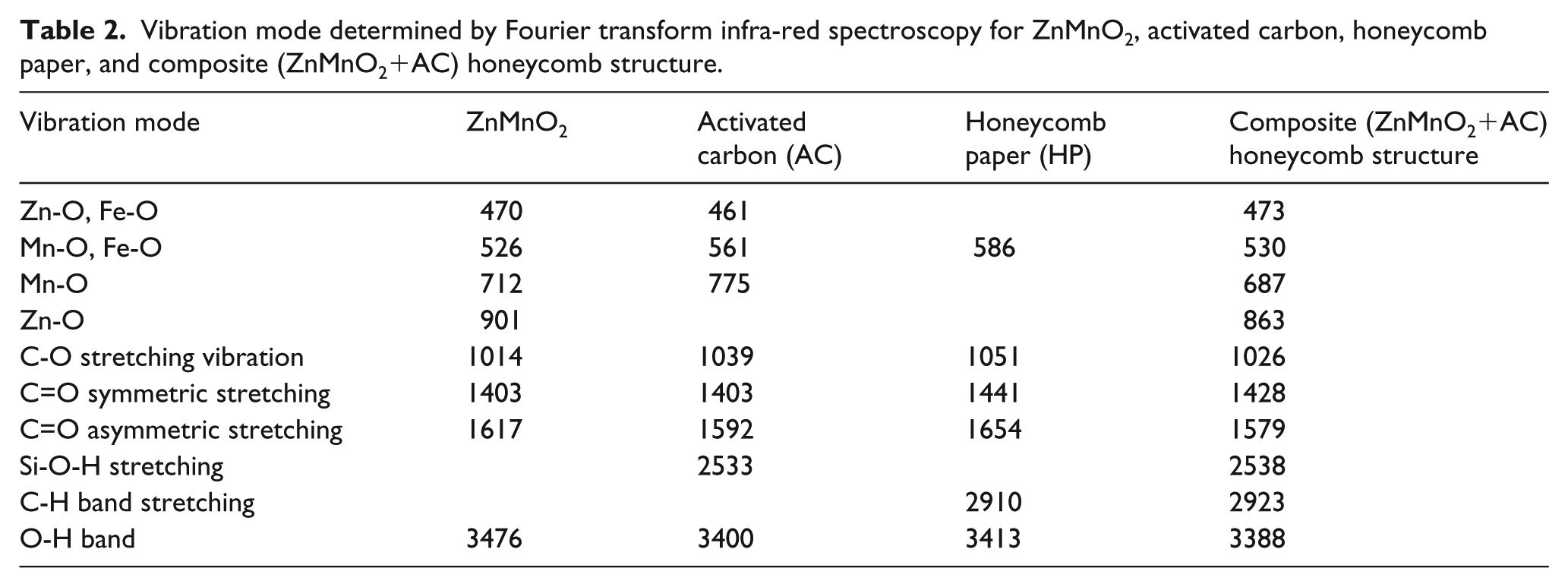
AC shows a band similar to that observed for ZnMnO2, but with a peak at 2500–2600 cm−1 due to O-H stretching mode vibration, which is identified by hydrous silica.19–22 For honeycomb paper there is another band vibration at 2910 cm−1, which is identified as C-H stretching vibration bonding with Ca due to the existence of CaO, as can be seen in Table 1. All vibration modes in the composite show incorporation from the individual materials. It shows that Zn, Mn, Ca, and Fe are successfully incorporated in the composite honeycomb structure. Here we notice that the wave number for the composite shifted to lower wave numbers for contribution from AC indicated size reduction as an effect of Zn or Mn bonding in the site of AC. This finding endorses the stable bonding by involving the atom Zn or Mn in the site of the AC group which was attached in the honeycomb pore of honeycomb paper. The C=C bonding responsible for stabilizing the Zn and Mn atom sits on the pore of AC sites, which has happened inside the pore of honeycomb paper, as can be seen in Figure 3. This figure clearly shows the mechanism to localize the Zn and Mn atoms inside the honeycomb paper and position them on the pore of AC, resulting in stable bonding for a new generation of composite. The absorption processes take place inside the honeycomb pore and result in the vibration between the atoms as a product of interaction between the electromagnetic waves and the atoms. XRD spectra used to determine the crystallite size of samples at the angle 2

where D is crystallite size (Å), λ is X-ray wavelength of source (Å), B is full width at half maximum (FWHM), θ is angle of diffraction peak. The crystallite size for each material in this study is shown in Table 3. Crystallite size for ZnMnO2 is 103.27 nm, activated carbon 86.43 nm, honeycomb paper 140.72 nm, and for the composite it is 90.17 nm. It shows that the FWHM is the main parameter with high contribution to the crystallite size of materials. Dislocation density (δ) was calculated from Williamson and Smallman’s relation 19 as follows:

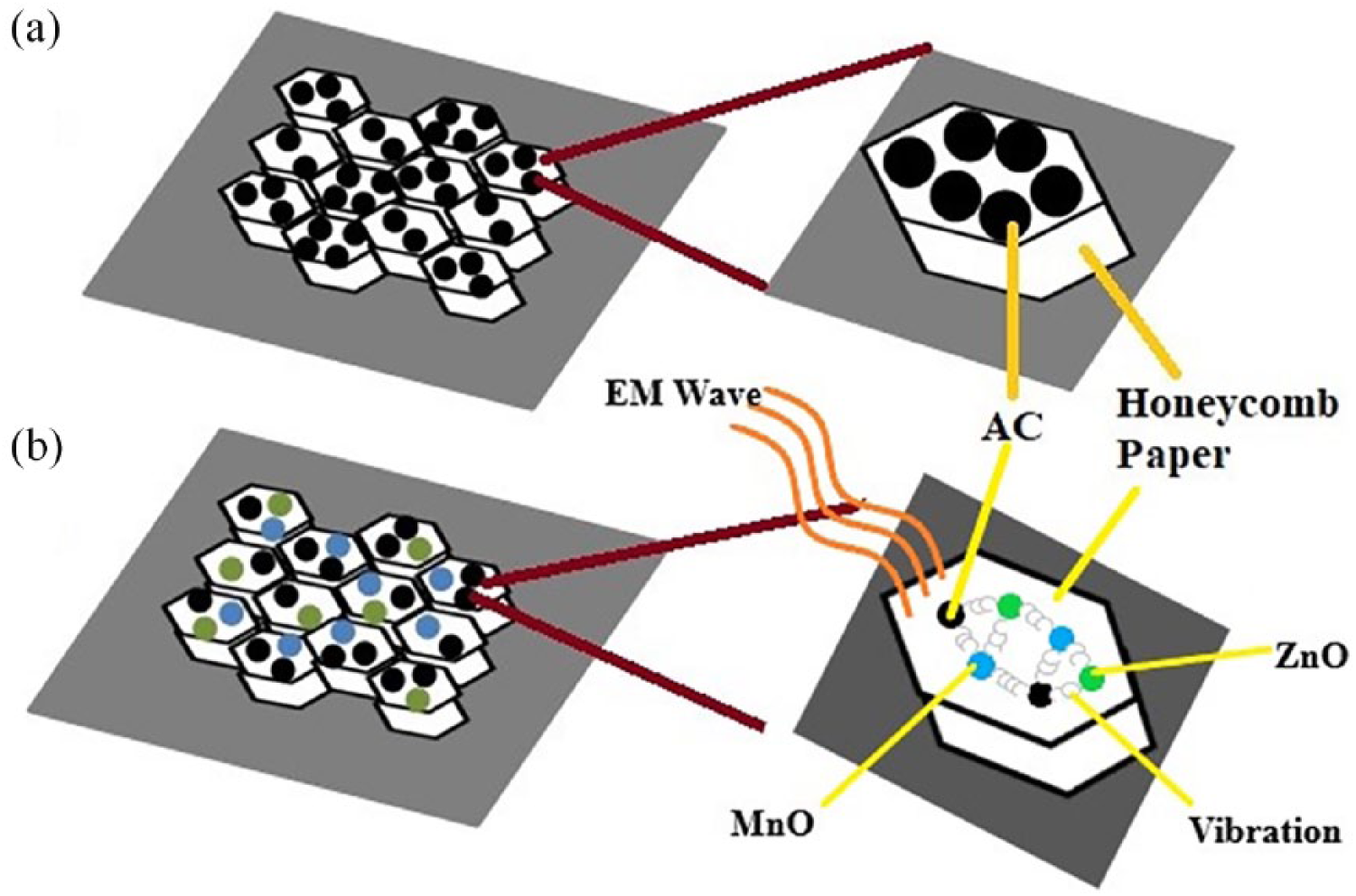
Illustration of paper honeycomb structure (a) filled with activated carbon (AC) and (b) filled with composite (ZnMnO2+AC), and the electromagnetic (EM) interaction.
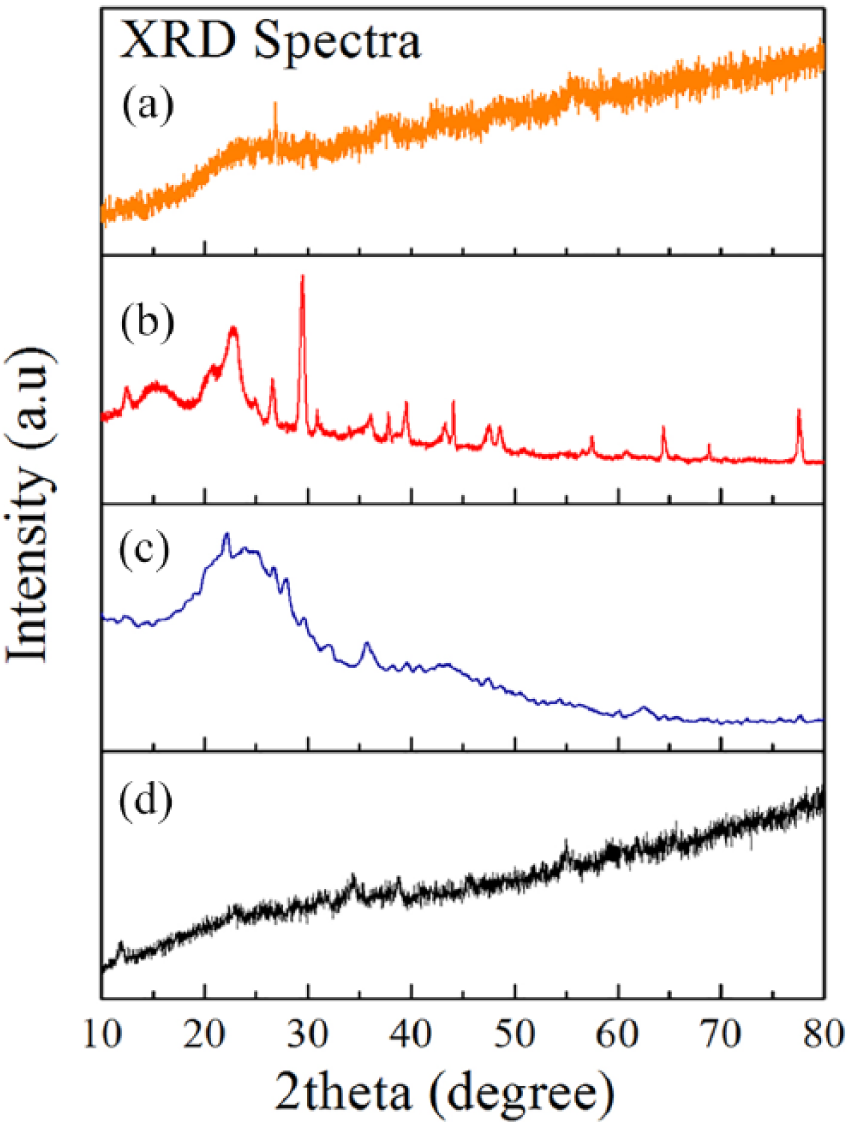
X-ray diffraction (XRD) spectra for (a) composite (ZnMnO2+AC) honeycomb structure, (b) honeycomb paper, (c) activated carbon, and (d) ZnMnO2.
Crystallite size determined from the X-ray diffraction spectroscopy spectra for ZnMnO2, activated carbon, honeycomb paper, and composite (ZnMnO2+AC) honeycomb structure.
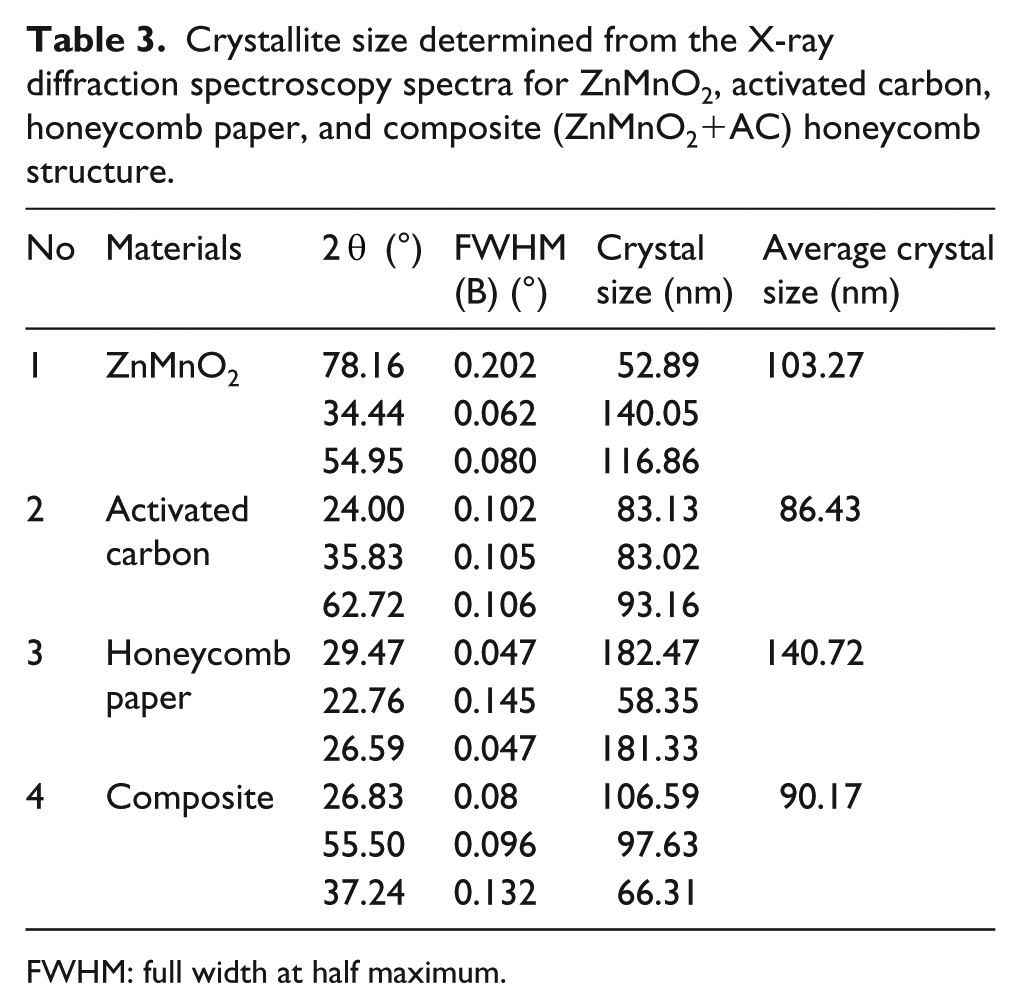
FWHM: full width at half maximum.
The dislocation density of the composite is 12.30 × 1013 m−2. The average of crystallite size and the dislocation density indicated good interaction between ZnMnO2 and AC in the pore of honeycomb paper. VNA analysis for a frequency range of 2.5–8 GHz is used for determining the reflection loss of the composite, which is related to the ability of the composite to absorb the electromagnetic waves. Figure 5 shows that for the thickness 2 mm, the reflection loss is −17.19935 dB at the frequency 4.34 GHz; for thickness 4 mm, the reflection loss is −18.41658 dB at the frequency 4.56 GHz; and for thickness 6 mm, the reflection loss is −21.72315 dB at the frequency 4.65 GHz. It is indicated that the thickness increases the reflection loss and the level of absorption of electromagnetic waves. Dai et al. 23 and Abdullah et al. 24 reported that reflection loss (R) values less than −10 dB can be designed to attenuate electromagnetic waves. In this study composite honeycomb structure based on ZnMnO2 derived from the waste of the conventional battery and AC for the thickness 6 mm shows the best performance of absorption of electromagnetic waves for frequency 4–5 GHz and shows promise as a new type of electromagnetic wave absorber material.
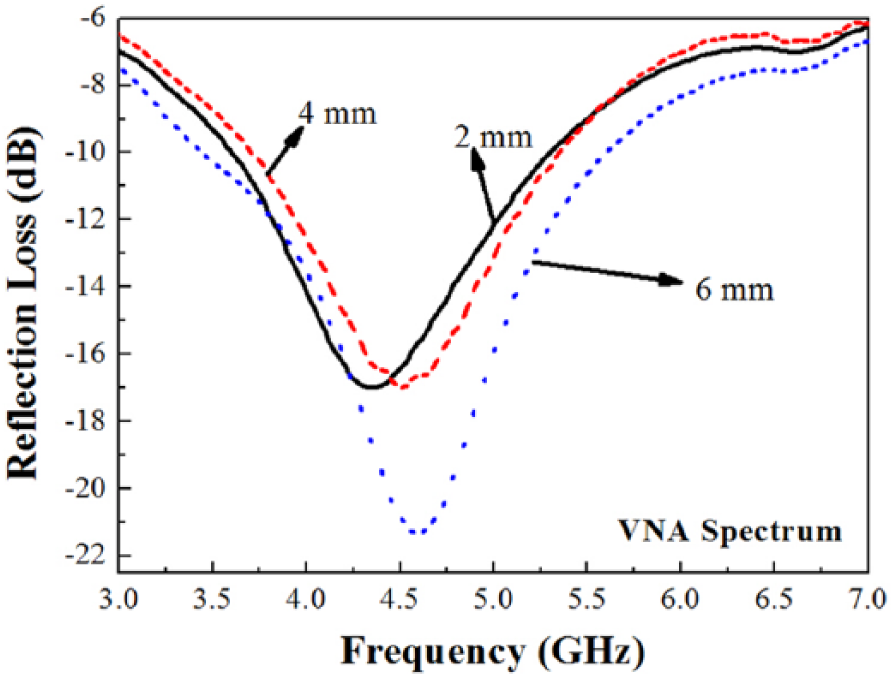
Reflection loss spectra from vector network analysis at frequencies between 2.5 GHz and 8 GHz for composite (ZnMnO2+AC) honeycomb structure with varied thickness.
Conclusions
Composite (ZnMnO2+ AC) honeycomb structure was successfully fabricated in this study, using ZnMnO2 synthesized from the waste of conventional batteries. Chemical composition and vibration mode shows existence of ZnMnO2 and AC in composites. The crystallite size and dislocation density shows C=C bonding responsible for good interaction between ZnMnO2 and AC in the pore of honeycomb paper. Composite honeycomb structure based on ZnMnO2 derived from the waste from conventional batteries and AC for the thickness 6 mm shows the best performance as an absorber in the absorption of electromagnetic waves at frequency 4–5 GHz.
Footnotes
Acknowledgements
We thank Hasanuddin University, Indonesia by Internal Research Program 2018 for facilities in support of this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
