Abstract
Lymphedema (LE) in a breast cancer patient usually results from axillary lymph node dissection, sentinel lymph node biopsy, and/or axillary radiotherapy. Although lymphatic obstruction and consequent LE can occur due to axillary lymph node involvement, such presentation in breast cancer patients has rarely been reported. This report describes the case of a 50-year-old lady with right breast carcinoma who presented with right upper limb lymphedema. Surprisingly, she did not have any bulky axillary lymphadenopathy that could have accounted for lymphedema of the upper limb, but a suspicious breast mass was present on clinical examination. Diagnosis of breast cancer on clinical examination can be challenging in such patients due to breast edema and careful imaging of the breast and axilla with appropriate imaging tools (mammogram, ultrasound, MRI, PET mammography) should be resorted to before excluding malignancy. Lymphoscintigraphy confirmed the findings of lymphedema in the right upper limb and revealed uptake in the epitrochlear lymph nodes of the contralateral limb suggesting that she might had pre-existing widespread “subclinical lymphedema” which presented as “clinical lymphedema” in right upper limb after axillary lymph node/lymphatic involvement by right breast carcinoma.
Introduction
Lymphedema is characterized by chronic and often debilitating swelling of the limbs due to lymphatic stasis-induced inflammation, fibrosis, and adipose tissue deposition. Based on the etiology, it can be classified into two groups- primary and secondary. Primary lymphedema is mainly due to embryological maldevelopment whereas secondary lymphedema is due to some acquired lymphatic obstruction [1]. Secondary lymphedema is more common and typical examples include upper limb LE following axillary dissection with or without radiotherapy for the treatment of breast cancer [2]. Lymphedema can affect up to 20–30% of breast cancer survivors [3,4].
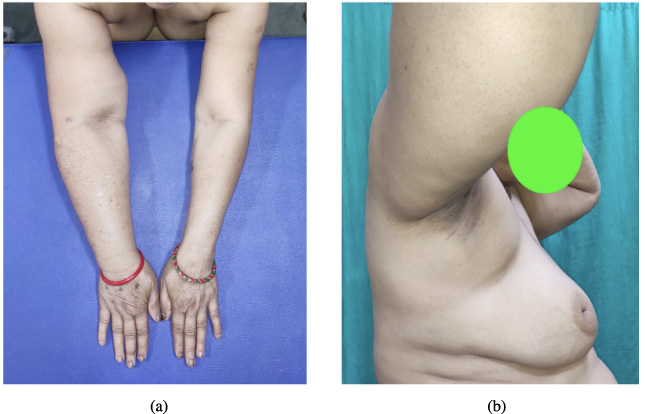
(a) Edema of the right arm and breast with retraction of the nipple and retracted skin in UOQ. (b) Lymphedema of the right upper limb.
LE in a patient of breast cancer is usually consequent to axillary lymph node dissection, sentinel lymph node biopsy, and/or axillary radiotherapy. Taxane-based chemotherapy has also been suggested as a risk factor for lymphedema, at least temporarily [5]. Although lymphatic obstruction and consequent LE can occur due to axillary lymph node involvement, such presentation in breast cancer patients has rarely been reported. We found only one case report in the English language where a patient with breast carcinoma had presented with lymphedema of the upper limb [6]. Two more such cases have been reported in French language by Vignes et al. In both cases, breast cancer was not evident on clinical examination but revealed itself on a mammogram in one patient and a breast MRI in another patient [7]. This report describes the case of a 50-year-old lady with right breast carcinoma who presented with right upper limb LE.
A 50-year-old postmenopausal lady presented with complaints of swelling in the right upper limb for five months. On clinical examination, there was edema of the right upper limb and right breast. The right mid-arm circumference was 7.5 cm higher than the left (Fig. 1a,b). There was puckering of the skin in the upper outer quadrant of the breast. On palpation, there was a hard, irregular lump of 4 × 4 cm in the upper outer quadrant (UOQ) of the right breast which was free from skin and underlying structures. There were no palpable axillary or supraclavicular lymph nodes. There were no signs of deep vein thrombosis in the upper limb and arterial pulses were palpable normally.
On evaluation by mammography, an irregular, high-density mass with spiculated margins, and retracted overlying skin was seen in the upper outer quadrant. It was associated with diffuse skin thickening, a retracted nipple, and an enlarged lymph node in the axilla (Fig. 2a). A core needle biopsy was done from the breast mass and histopathology revealed it to be infiltrating ductal carcinoma- not otherwise specified. Immunohistochemistry suggested it to be positive for hormone receptors (estrogen and progesterone receptor positive) and negative for human epidermal growth factor receptor (HER- 2) with a ki-67 index of 50–60%. There was no evidence of visceral or skeletal metastasis on the CECT of the chest, abdomen, pelvis, and bone scan. A color doppler examination of the upper limb ruled out any venous or arterial pathology and skeletal muscles were unaffected. Since the axillary lymphadenopathy was not sufficient to explain the LE of the upper limb, we subjected the patient to lymphoscintigraphy (LSG). On LSG, there were collaterals observed in the right cubital fossa and lower third of the arm. A dermal flow pattern was seen in the arm and forearm and no axillary nodes were visualized. It was suggestive of obstruction at the level of axillary lymph nodes (Fig. 3). Contralateral epitrochlear lymph nodes were also visualized on LSG. We arrived at a diagnosis of carcinoma of the right breast stage (cT4bN1M0) with LE- stage 2 of the right upper limb.
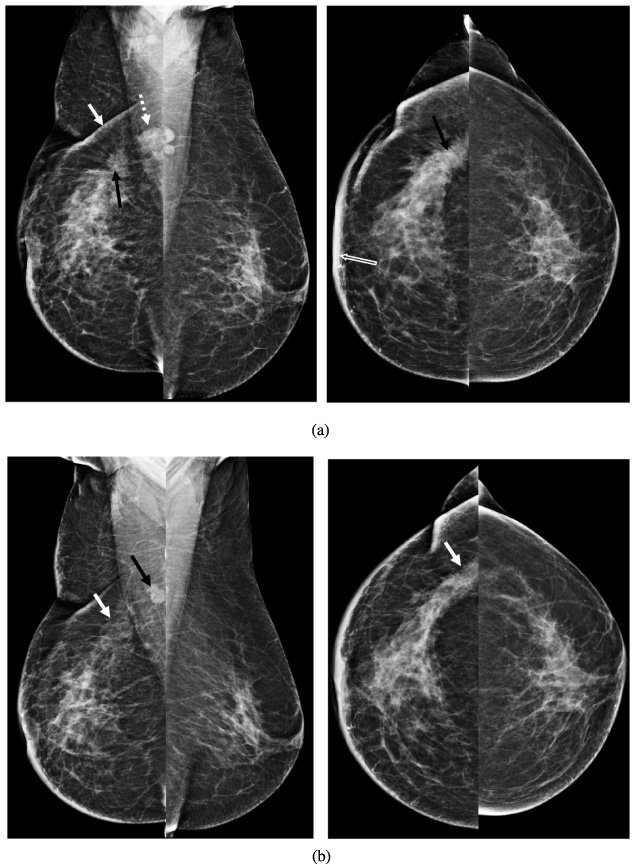
(a) Pre-NACT bilateral mammogram: irregular, high-density mass with spiculated margin (black arrow) in UOQ, with retracted overlying skin (white arrow), associated diffuse skin thickening (open white arrow) with retracted nipple. An enlarged lymph node is seen in the axilla (dashed white arrow). (b) Post NACT bilateral mammogram: reduction in the size, as well as density of the mass (white arrow) in UOQ, the size of the lymph node, has also reduced (black arrow).
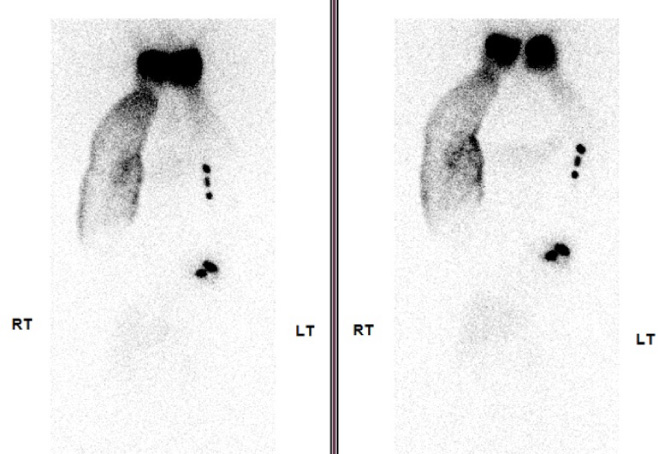
Lymphoscintigraphy images (2 hr and 4 hr after injection) showing collaterals in the right cubital fossa and lower third of the arm, dermal flow pattern in the right arm and forearm, and non-visualization of axillary lymph nodes. Left epitrochlear LNs can also be visualized.
After a multidisciplinary tumor board (MDT) discussion, the patient was planned for neoadjuvant chemotherapy (NACT). She responded to NACT, and no lesion was clinically palpable after NACT but the edema of the breast skin persisted. Post-chemotherapy mammogram (Fig. 2b) revealed an asymmetric density seen in the UOQ of the right breast- suggestive of residual mass. Nipple retraction with diffuse skin thickening persisted. An enlarged axillary lymph node (reduced in size) was also seen in the axilla.
After discussion in the MDT, the patient was planned for a modified radical mastectomy. Intraoperatively during the axillary dissection, although no enlarged/palpable LNs were visualized, multiple extranodal, sub-centimetric deposits were identified in the axillary fat both medial and lateral to the thoracodorsal pedicle. Histopathological examination of the MRM specimen revealed a residual tumor of 5 cm in the UOQ. All the resected margins were free. Fifteen out of seventeen resected LNs were found to have metastasis and multiple extranodal deposits were found in the axillary fat.
The patient received adjuvant radiotherapy to the chest wall and is currently doing well on hormone therapy (tablet letrozole 2.5 mg per day) after 17 months of follow-up following diagnosis. Her limb volume has been stable with decongestive physiotherapy and elastic compression garments (Fig. 4).
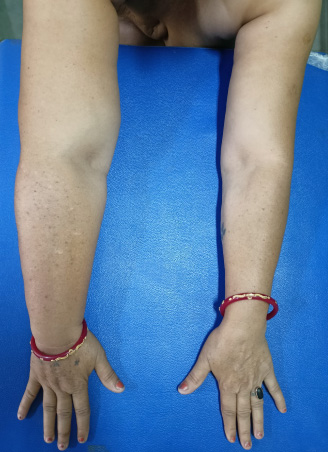
Status at seventeen months of follow-up: Stable swelling of right arm.
Our patient presented with a complaint of upper limb swelling. Although she was aware of the lump and swelling in her right breast, it was revealed only on clinical examination. Delayed presentation of breast cancer is common in developing countries like ours. Occasionally, patients present with LE of the upper limb and breast due to advanced axillary nodal disease. However, this case is unique in the sense that the patient had presented to us with a complaint of swelling in the upper limb, and although a breast lump was discovered on clinical evaluation, there was no advanced axillary lymphadenopathy, neither on clinical examination nor on radiological assessment. On final histological examination of the axillary lymph node dissection specimen, 15 out of 17 lymph nodes were found to have metastasis along with extranodal spread.
It must be emphasized that the presence of lymphedema in the upper limb due to the involvement of axillary nodes/lymphatics may be associated with edema /LE of the breast as well which may reduce the sensitivity of clinical examination. This can potentially lead to misdiagnosis unless the breast and axilla are imaged for the presence of malignant masses and lymph nodes respectively. Mammography and ultrasonography can be diagnostic in most patients, but for equivocal cases, the use of breast MRI or even a 18F-FDG-PET mammography is appropriate [7]. The volume of axillary lymph node burden can also be well appreciated by contrast-enhanced CT scans of the axilla.
Lymphedema is a condition characterized by the regional accumulation of an excessive amount of interstitial protein-rich fluid because of an imbalance between the demand for lymphatic flow and the capacity of lymphatic circulation. The major causes of secondary lymphedema are lymph node dissection, malignant obstruction, radiation, and infection. Be it primary or secondary, the pathological progression of the disease is the same. There are four stages of lymphedema- (i) stage 0- normal extremity clinically with abnormal lymph transport (diagnosed by lymphoscintigraphy), (ii) stage 1- early edema improving with limb elevation, (iii) stage 2- pitting edema not resolving with elevation, (iv) stage 3- deposition of fibroadipose tissue and skin changes (7). The severity of lymphedema is categorized as mild (<20
For the visualization of lymphatic vasculature, lymphoscintigraphy is the first-line imaging modality. The findings in lymphedema patients vary considerably but features seen in primary lymphedema are distinct from the lymphoscintigraphic finding of secondary lymphedema. The prominent findings in primary lymphedema include the complete absence of radiotracer transport or early migration of radiotracer into the interstitium (dispersion or dermal backflow) without evidence of lymphatic truncal flow (consistent with truncal aplasia or hypoplasia). Lymphoscintigraphy in secondary lymphedema demonstrates prominent lymphatic trunks in contrast to attenuation or absence in congenital or hypoplastic syndromes and other findings, such as delayed radiotracer transport and faintly visualized regional lymph nodes, may also be seen in secondary LE [9]. In our patient, there were collaterals in the right cubital fossa and lower 1/3rd of the arm, the dermal flow was seen in the right arm and forearm and axillary nodes were not visualized suggesting secondary lymphedema.
An interesting finding in the lymphoscintigraphy was the visualization of contralateral epitrochlear lymph nodes. The presence of epitrochlear lymph nodes may indicate the rerouting of lymph to the deep system and has been hypothesized to indicate “subclinical lymphatic insufficiency” [10]. Thus, it might be the case that our patient had widespread “subclinical lymphatic insufficiency” and the axillary lymphatic involvement by breast cancer on the right side may have resulted in the development of “clinical lymphedema”.
Footnotes
Acknowledgement
We are thankful to our patient who allowed us to share this case report.
