Abstract
Tuberculosis (TB) remains in 2022 a significant public health issue as it remains endemic in some areas of the globe, with a high prevalence in underdeveloped countries (Pujani, Khan, Hassan, Jetley, Raina,
Keywords
Introduction
Tuberculosis (TB) remains a significant public health issue that is endemic in some areas of the globe, with high prevalence in underdeveloped countries [1]. Pulmonary TB is the most common form, but it can also have extrapulmonary manifestations like tubercular lymphadenopathy. Tuberculous lymphadenitis is the most extrapulmonary form of tuberculosis. It used to be called scrofula, which is from the Latin meaning “breeding sow [2,3]”. It is a common cause of peripheral lymphadenitis and is seen mostly in developing countries, but it is also re-emerging among intravenous drug users and immunocompromised populations.
Cervical nodes are the most commonly detected nodes in tuberculous lymphadenitis, accounting for 63% of the cases, followed by mediastinal (27%) and axillary nodes (8%) [4]. Tuberculous lymphadenitis predominantly affects young populations and children, and there is also a slight female predilection. To our knowledge, there have not been any reported cases of post-menopausal axillary tuberculous lymphadenitis thus far, which is the focus of this report.
A 51-year-old female patient was referred to the breast cancer center with tenderness in the right axilla followed by a right palpable axillary mass. This was preceded by flu-like symptoms one month prior to her referral. Except for prior pregnancies, she reported no relevant medical history and no family history of breast cancer. The patient emigrated to Canada in 1994 from Ecuador and had gone there for a one-month visit before the onset of her symptoms.
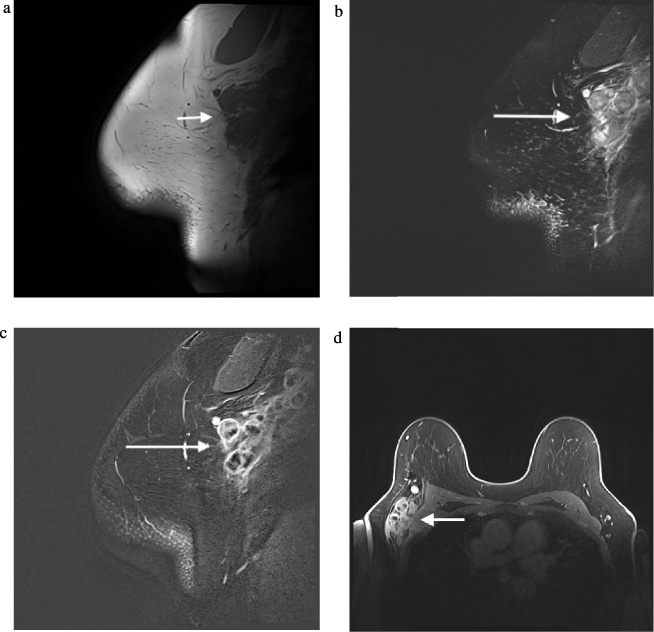
MRI images showing right axillary abnormal lymph nodes with enhancement of the capsule, hypointense centres and enhancement of the surrounding axillary fat. Figure 3a: sagittal T1 sequence. Figure 3b: sagittal T2 sequence. Figure 3b: sagittal T1 with fat sat post contrast. Figure 3d: axial t1 with fat sat post contrast.
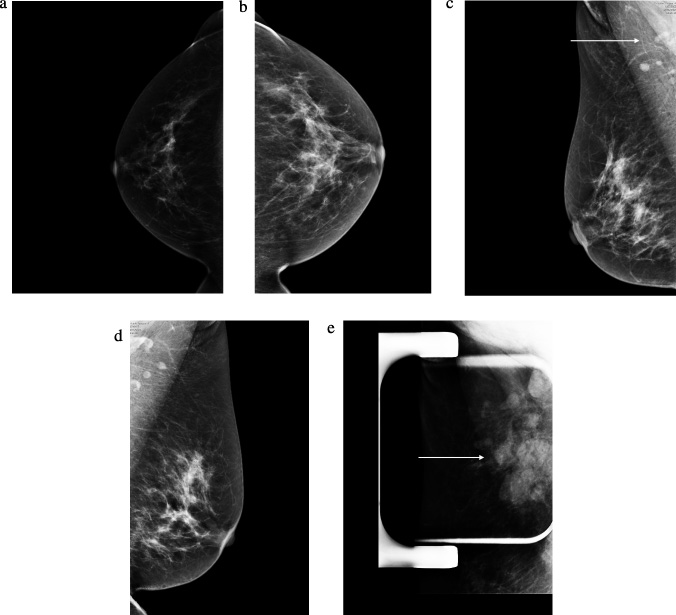
Initial mammograms showed multiple right axillary masses (Fig. 1). One of the largest deeper masses appeared ill defined and was only partially included in the field of view. No microcalcifications were seen within the right axilla. These findings were suggestive of matted lymph nodes. No abnormal left axillary lymph nodes were identified, and there were no other suspicious masses, microcalcifications, or distortion in the right breast. There was no suspicious mass, suspicious calcifications, or architectural distortion to suggest malignancy in the left breast.
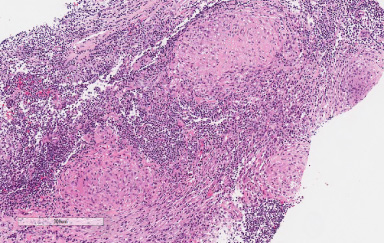
Core biopsy of one of the abnormal node shows inflammatory reaction with changes suggestive of granulomatous inflammation but no clearly formed granulomata readily appreciated.
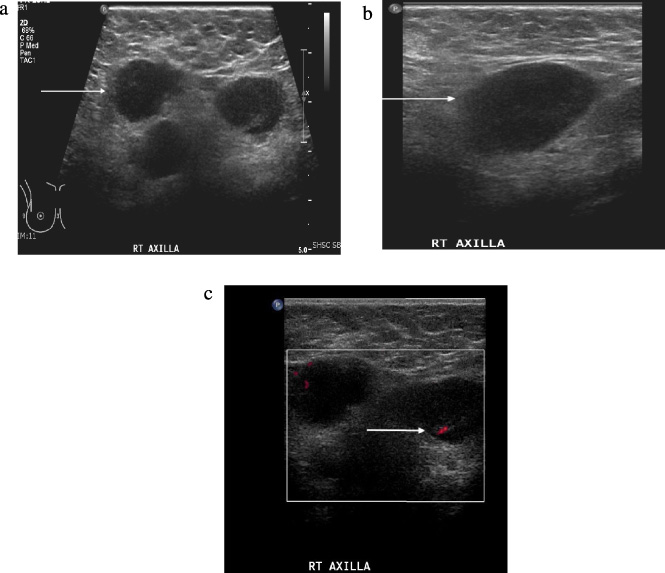
Additional whole right-breast and axilla ultrasound was subsequently performed (Fig. 2). More than 10 hypoechoic conglomerated masses without fatty hilum were demonstrated in the right axilla with ill-defined margins and hyperechoic infiltrated surrounding fat, suggesting matted inflammatory lymphadenopathy. There was no sonographic evidence of level 2 or interpectoral lymphadenopathy. Since the initial workup showed no suspicious mass in the right breast, subsequent breast MRI was recommended.
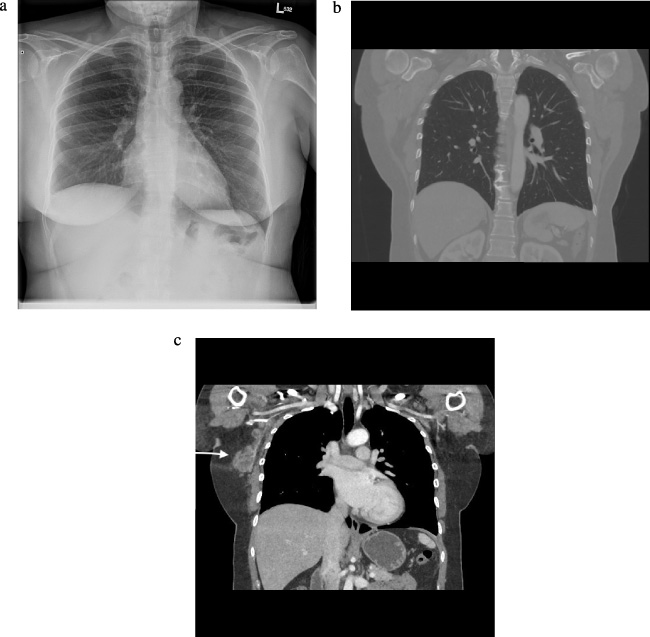
MRI showedextensive right axillary lymphadenopathy involving levels 1–3. Multiple enlarged lymph nodes had indistinct margins with enhancement of the capsule of the lymph nodes and the surrounding axillary fat. There was no abnormal finding in the right breast (Fig. 3). Additional chest X-ray and a thoracic computed tomography scan showed no additional abnormalities (Fig. 4).
One of the largest lymphadenopathies was biopsied with a 14-gauge Tru-Cut Marquee disposable biopsy device needle under ultrasound guidance. Pathology showed no evidence of malignancy by morphology, immunohistochemistry, or flow cytometry (Fig. 5). The appearance was suggestive of necrotizing granulomatous inflammation, but there was no clearly formed granulomata. Although a definite lymph node structure was not identified, the findings could be consistent with necrotizing lymphadenitis given the site of the biopsy.
The differential diagnosis of necrotizing lymphadenitis suggested by the pathologist was necrotizing granuloma, which includes infections such as cat scratch disease, TB, and fungal organisms, as well as autoimmune conditions such as Kikuchi-Fujimoto, systemic lupus lymphadenitis, and drug reaction. Special stains (GMS, ZN) were negative for fungal elements and mycobacterial organisms, but this did not rule out the possibility of fungal or mycobacterial infection.
A second biopsy was performed six weeks after, and multiple granulomas with necrosis were found. Special stains for acid fast bacilli and fungi were again negative. Therefore, samples were sent for microbiological studies as the stains have a low sensitivity.
The patient was referred to the infectious diseases clinic, and quadruple therapy for extrapulmonary tuberculosis was started, which she continued for 6 months. No follow up imaging of the axilla was done, but a physical examination revealed that the lymph nodes were no longer palpable.
The axillary space is a triangular space located between the proximal aspect of the arm, the side of the chest, and the base of the neck [5]. The base of the triangle is located at the inferior aspect of the neck, and the apex is located toward side of the chest and proximal aspect of the arm. The boundaries include several chest muscles, fascia, and bones.
The axilla contains mesenchymal tissues with mostly fat content. They also include vessels, nerves, and lymph nodes. The most common finding of axillary mass is lymph node metastases from breast cancer. However, there are some other findings, which are possibly benign or malignant. Some of these findings have specific characteristics in imaging findings that may help radiologists in the differential diagnosis.
The most common axillary mass is axillary node metastasis from breast carcinoma. Most often, it spreads from level I to II and then III. A prognostic factor for the breast is the presence or absence of metastatic axillary lymph nodes [6]. Metastatic lymphadenopathy usually appears with either displaced or absent fatty hilum or increased cortical thickening.
Breast metastatic lymphadenopathy is not distinguishable from another metastatic lymphadenopathy. However, there are some differences compared to a reactive lymph node. There are usually multiple reactive lymph nodes, and they tend to be elliptical, while metastatic nodes are round and often adjacent to a normal lymph node. Metastatic nodes tend to have poorly defined margins, while benign reactive lymph nodes usually have sharp margins. Only 0.3–1% of breast carcinomas are occult (i.e., onset of breast carcinomas with axillary lymphadenopathy as initial presentation without any concurrent lesion seen in mammograms or clinical examinations) [6].
Radiologists must be aware of the differentiation between tuberculous and metastatic cervical nodes. Yang et al. showed that tuberculous lymph nodes show some heterogeneity (64%), intracystic necrosis (60%), and abnormal surrounding tissues (49%) [7]. There is also a specific pattern with a hypoechoic center and posterior enhancement, which is not seen with nodal metastasis because compacted tumor cells are not associated with posterior enhancement. However, some authors report that there are no definite distinguishable imaging features between metastatic lymph nodes versus granulomatous disease of the lymph nodes.
Interestingly, reports have indicated the coexistence of necrotizing granulomatous inflammation in an ipsilateral axillary lymph node with invasive ductal breast carcinoma [1]. Some authors report that this could be explained by an incidental finding of retrograde spread from the mediastinal nodes that is not clinically palpable or detected with regular investigations [8,9]. This could also be explained by reactivation of a silent
Conclusion
Axillary lymphadenopathy is not always related to metastatic breast carcinoma. Although it is the most common etiology, radiologists should be aware of other possibilities such as systemic disease (such as sarcoidosis or rheumatoid arthritis) or infectious processes like tuberculosis. The patient’s history is very significant in the management, and if needed, a procedure like fine needle aspiration or biopsy should be considered. When there is stranding/hyperechoic surrounding fat, infectious processes should be excluded. Furthermore, microbiological studies should also be considered in addition to pathology and flow cytometry.
Footnotes
Competing interest
All authors declare that there are no financial disclosures and personal relationships with other people or organizations that could inappropriately influence (bias) our work.
