Abstract
Amyloidosis rarely presents as localized lymphadenopathy. Various studies have elucidated the varied presentation and manifestations of this interesting disease. We reviewed the literature and found 36 cases of primary amyloidosis with lymph node enlargement as a presentation, and 17 of the 36 cases (47%) had systemic involvement on further work up. We describe a patient who presented with an isolated right axillary mass. Clinical examination and radiology were indicative of a lymph node enlargement with no evidence of malignancy in the breasts or lungs. Histopathological examination was indicative of amyloidosis. A further work up including serum, urine biochemistry, cardiac work up, bone marrow examination, and a kidney biopsy revealed systemic amyloidosis. Patient was treated with daratumumab and CyBorD (cyclophosphamide, bortezomib, and dexamethasone) followed by a stem cell transplantation. Patient is in remission for 1 year, at the time of submission of this report. Therefore, we conclude (1) systemic amyloidosis presenting as an isolated lymph node enlargement is rare, (2) a structured systemic work up is imperative for early diagnosis and proper management of amyloidosis, when there is an index of suspicion, and (3) use of novel therapeutic options such as CD38 + antibody (daratumumab) and stem cell transplant have positive impact on disease outcomes.
Introduction
Amyloidosis is a group of diseases resulting from the abnormal deposition of amyloid fibrils and may be considered under the broad term of protein misfolding disorders. These misfolded proteins deposit extracellularly causing organ damage. 1 The most commonly diagnosed type of systemic amyloidosis is light chain (AL) amyloidosis, which is characterized by a clonal population of bone marrow plasma cells that produce a monoclonal light chain of kappa or lambda type as either an intact molecule or a fragment. 1 AL amyloidosis, usually affects the kidneys and heart, but can also involve lungs, peripheral and autonomic nervous systems, gastrointestinal tract1,2 including liver, and soft tissues. 1 Systemic amyloidosis affects lymph nodes in a frequency ranging from 17% to 37%, but lymphadenopathy is rarely seen as an initial manifestation of this disease.2,3
A case of a patient with amyloidosis presenting with isolated axillary node enlargement and kidney involvement found on subsequent work up is described here.
We also reviewed the literature for cases of isolated lymph node enlargement as a presentation in AL amyloidosis, and found systemic involvement in 47% of cases. This case suggests that a search for systemic involvement is important in a setting of isolated lymph node presentation of AL amyloidosis.
Case Report
A 58-year-old black man with a past medical history of hypertension and diabetes presented with months-long history of a right axillary mass that had been growing in size, as well as fatigue, significant weight loss of 62 lbs (235 to 173 lbs). On physical examination, a right axillary mass was palpable, approximately 4 cm in diameter, rubbery, fixed, and was non-tender to palpitation. Ultrasound of the right axilla showed a 3.2 × 1.6 × 2.9 cm3 lobulated heterogeneous solid mass. Magnetic resonance imaging showed a 2.7 × 2.7 × 1.8 cm3 heterogeneous solid mass in the region of palpable lump in the right axilla indicating enlarged lymph nodes. An ultrasound-guided core biopsy of the right axilla suggested an amyloid tumor of the breast associated with moderate lymphocytic infiltrate (Figure 1A). Congo red stains supported the diagnosis of amyloidosis, and presence of AL amyloid was confirmed by immunochemistry (Figure 1B). There was no mammographic evidence of malignancy in the breast, but the mammogram was positive for mild bilateral gynecomastia. Computed tomography (CT) chest, abdomen, and pelvis showed right axillary lymph node with punctate calcification measuring 2.5 cm and a left axillary lymph node measuring 9 mm. Axillary lymph node dissection and excision of the lymph node was performed. Histopathological examination revealed lymph node replaced by hyaline-like amorphous eosinophilic depositions and foreign body type giant cell reaction consistent with amyloidosis. Patient had normocytic normochromic anemia with hemoglobin of 9 gm%. His creatinine was 1.88 and estimated glomerular filtration rate (eGFR) was 37. Bone marrow biopsy was hypocellular with 5% plasma cells with IgG/lambda excess and suspicious amyloid deposits. Fluorescence in situ hybridization (FISH) study of bone marrow cells was significant for IGH/CCND1 variant translocation and monosomy 13. Cardiac work up showed an elevated Pro-BNP 1580 pg/mL (normal: 11.1-125 pg/mL), Echocardiogram showed normal systolic function (ejection fraction: 70%), no septal wall thickening or global strain, and grade 1 diastolic dysfunction. Serum kappa chains were 2.12 mg/L (normal: 0.33-1.94 mg/L) and lambda chains were 6.08 mg/L (normal: 0.57-2.63 mg/L), and the ratio was 0.35 (normal: 0.26-1.65). IgG monoclonal protein was detected on serum immunofixation. The 24 hours urine protein was 9803 mg/24 hours (normal < 150 mg/24 hours). Albumin-creatinine ratio was markedly elevated to 10 606 mg/g of creatinine. To evaluate proteinuria, a renal biopsy was performed, which showed amyloidosis of light chain–lambda type, extensively involving glomeruli, blood vessels, and interstitial tissues (Figure 2). It also showed severe tubular atrophy and interstitial fibrosis. A fat pad biopsy was not done as a diagnosis of amyloidosis was made on kidney biopsy.
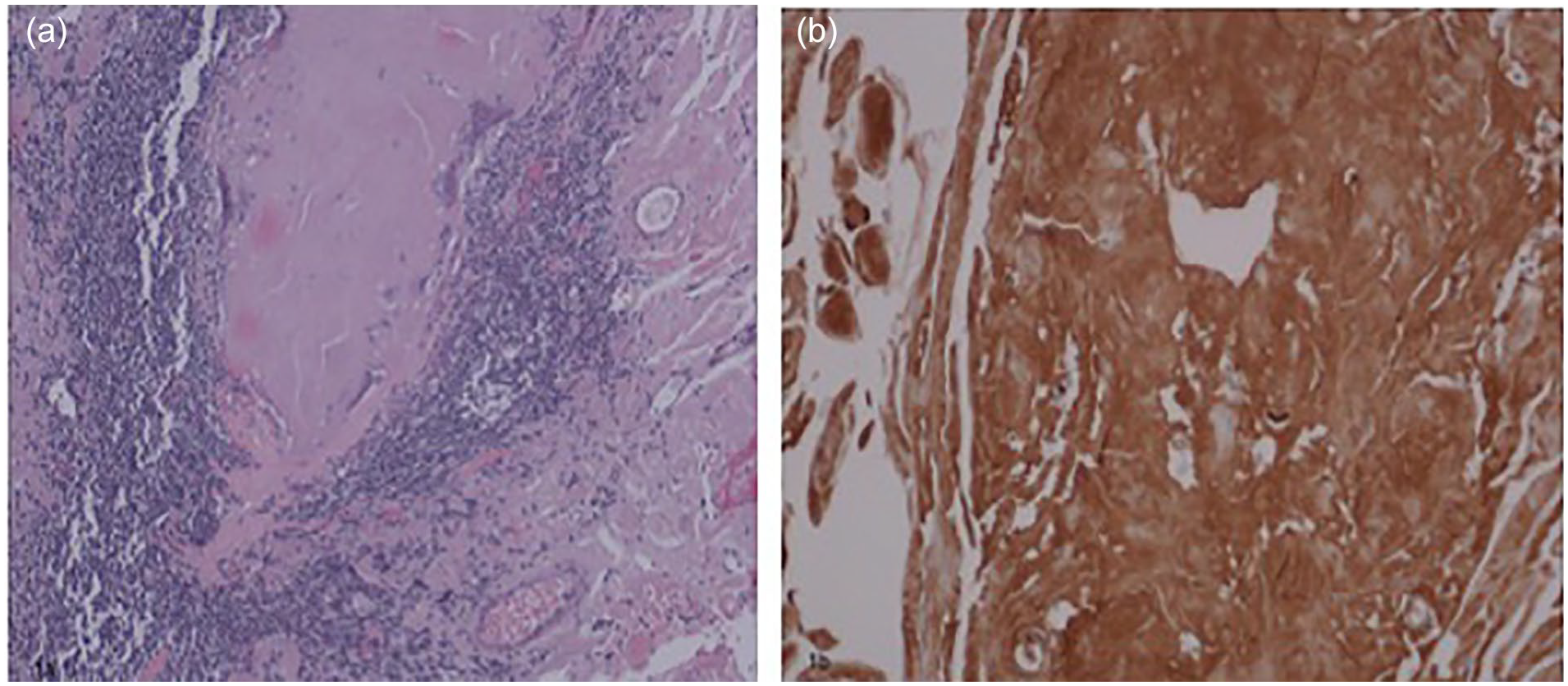
(A) Section of lymph node with effaced architecture and deposition of pink, amorphous material (H&E ×100). (B) Immunostaining of the lymph node highlighting AL amyloid (×400).

Renal biopsy showing amyloidosis, extensively involving glomeruli, blood vessels, and interstitium (H&E ×400).
Patient was started on daratumumab and CyBorD (cyclophosphamide, bortezomib, and dexamethasone). Patient received 6 cycles of chemotherapy. His creatinine was baseline (2.2) with an eGFR of 35, BNP was 74, troponin was negative (<0.01), kappa to lambda ratio was 0.9, and restriction in gamma region was seen but no M band was present in serum immunofixation after chemotherapy. Patient was subsequently referred to a New York hospital for a stem cell transplantation where he received Melphalan 200 mg/m2 prior to transplant as per European Hematology Association and International Society of Amyloidosis (EHA-ISA) guidelines. Patient developed renal failure after the stem cell transplant for which he temporarily required dialysis. His renal function recovered, and he is off dialysis. Patient is in remission for 1 year at the time of writing this report based on his normal serum and urine immunofixation.
Discussion
Lymphadenopathy as a presentation of systemic amyloidosis is rare. At a single center, 3008 new patients with amyloidosis (from 1994 to 2013) were evaluated, 4 47 (1.6%) presented with lymph node enlargement; leading to a biopsy and a diagnosis. Thirty-nine of those 47 patients were diagnosed with AL amyloidosis at initial presentation or on follow-up. In a study by Biewend et al, 5 an evaluation of 20 cases of localized amyloidosis was done and only one presented with isolated lymph node enlargement in axillary region, elucidating the rarity of such a presentation.
We reviewed the literature and found 36 cases of primary amyloidosis with lymph node enlargement as a presentation (Table 1). Seventeen of the 36 cases (47%) had systemic involvement. Twelve out of 36 (33.33%) manifested as single lymph node region involvement and out of those, 6 (50%) had systemic involvement. Whereas 24 cases (66.67%) had more than 1 lymph node region involved, and out of these 24, eleven (45.83%) had systemic disease. No follow-up data for the cases of isolated amyloidosis of lymph nodes are available. From the literature review on patients presenting as an isolated amyloidosis of lymph nodes, 50% were found to have systemic involvement. This observation suggests that a meticulous work up including abdominal fat pad and bone marrow biopsy and 24 hours urine protein should be screened for a possible systemic involvement in an isolated lymph node presentation of amyloidosis. As exemplified by our case, presence of elevated 24 hours urine protein led to performing a kidney biopsy on the patient. Such patients need systemic treatment including bone marrow transplantation.
Cases With Isolated Lymph Node Enlargement as Initial Presentation.

Abbreviation: CT, computed tomography ; B/L, bilateral; GI, Gastrointestinal; NHL, Non Hodgkin Lymphoma; HTLV, Human T Lymphotropic virus; MGUS, Monoclonal Gammopathy of Unknown Significance ; AA, Amyloid A.
High-dose melphalan supported by autologous stem cell transplantation (HDM-ASCT) remains the therapeutic standard of care in low-risk patients with AL amyloidosis. 34 CyBorD or CTD (cyclophosphamide, thalidomide, dexamethasone) are options for patients with intermediate risk. CyBorD and attenuated MDex (melphalan, dexamethasone) are used for high-risk cases, and lenalidomide or pomalidomide are available for relapsed/refractory cases. 34 A combination of melphalan–high-dose dexamethasone represents the current gold standard of treatment for patients ineligible for ASCT. 35 Bortezomib (a proteasome inhibitor) induces a rapid decrease in serum Free light chain (FLC) concentration and is currently the frontline therapy for AL amyloidosis.34,36,37 Daratumumab, a CD38-directed monoclonal antibody promises to be a well-tolerated option for patients who fail to achieve a complete response to standard light chain suppressive chemotherapy. In a study by Kaufman et al, 38 overall hematologic response rate to daratumumab was 76%, including clinical remission (CR) in 36% and very good partial response in 24% with a median time to response being 1 month. Jeryczynski et al describe daratumumab as an efficient first-line treatment strategy in AL amyloidosis. In their study, all 14 patients achieved hematological response, a substantial proportion also showed cardiac and renal response. The treatment was also well tolerated even in frail patients with cardiac involvement. 39 Palladini et al consider combination of bortezomib-based regimens with daratumumab as upcoming novel standards of care. Currently, daratumumab is not widely accessible, and CyBorD is preferred over BMDex (Bortezomib, melphalan, dexamethasone) in patients with renal failure and in those who may later become eligible for ASCT, whereas BMDex may overcome the negative effects of both t(11;14) and gain 1q21. 40 Sanchorawala share similar experiences with single-agent daratumumab which proved highly effective in inducing deep and lasting hematologic and organ responses in heavily pretreated patients. Deep hematologic responses were observed after 1 single infusion of daratumumab. 41 In other trials too, it was found that addition of daratumumab results in significantly higher hematologic (92% vs 77%), cardiac (42% vs 22%), and renal (54% vs 27%) response rates compared with CyBorD alone. A complete hematologic response was observed in 53% of patients treated with DaraCyBorD compared with 18% of patients receiving CyBorD at a median follow-up of 11 months.42,43
Autologous stem cell transplant has been used over 2 decades for amyloidosis now and remains highly effective in eligible patients. It is associated with improved survival and reduced treatment-related mortality. 44 Autologous stem cell transplant can achieve organ response rate up to 65% as it rapidly eradicates the amyloidogenic light chain produced by the clonal plasma cell populations. 36 According to Sidiqi et al, patients who have undergone ASCT have lesser plasma cell load in the marrow, less proliferative disease, and favorable genetics compared with patients with multiple myeloma. They found a response rate of 84% (CR, 39%) and 5-year OS (overall survival) of 84% in the most recent times highlighting the utility of ASCT in AL amyloidosis. Judicious patient selection for ASCT is critical and has shown promising results over time with reduced toxicity. 44 Our patient was started on daratumumab and CyBorD followed by stem cell transplant. A comparison in terms of long-term disease-free survival, treatment-related mortality between ASCT and novel agents will be required to choose between the two.
Number and type of organs involved affect the survival outcomes. Patients with isolated cardiac involvement have a similar outcome when compared with patients with cardiac plus multiorgan involvement. However, patients with isolated renal involvement have a better survival curve as compared with patients with renal plus multiorgan involvement. Hence presence of cardiac involvement is the most important determinant of prognosis. 45
Conclusions
Lymphadenopathy as a manifestation of amyloidosis is rare. Patients with amyloid lymphadenopathy should undergo an evaluation and/or follow-up to investigate systemic involvement. A detailed work up for systemic amyloidosis in the isolated lymph node presentation can lead to an early diagnosis of systemic amyloidosis and treatment. Chemotherapy with possible CD38+ positive antibody (such as daratumumab) and stem cell transplantation should be incorporated into the treatment regimen.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethics approval to report this case was obtained from Brookdale Hospital IRB Review Board.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report and on file.
