Abstract
BACKGROUND:
The Notch signaling pathway is an evolutionary conserved cell signaling pathway that plays an indispensable role in essential developmental processes. Aberrant activation of Notch pathway is known to initiate wide array of diseases and cancers.
OBJECTIVE:
To evaluate the clinical significance of Notch receptors in Triple Negative Breast Cancer.
METHODS:
We evaluated the association between Notch receptors and clinicopathological parameters including disease-free survival and overall survival of one hundred TNBC patients by immunohistochemistry.
RESULTS:
Positive expression of nuclear Notch1 receptor (18%) was found be significantly correlated with positive lymph node (p = 0.009), high BR score (p = 0.02) and necrosis (p = 0.004) while cytoplasmic expression of Notch2 receptor (26%) was significantly correlated with metastasis (p = 0.05), worse DFS (p = 0.05) and poor OS (p = 0.02) in TNBC patients. Membrane (18%) and cytonuclear (3%) Notch3 expression were significantly associated with poorly differentiated tumors (p = 0.007), high BR score (p = 0.002) and necrosis (p = 0.03) respectively. However, cytoplasmic Notch3 and Notch4 expression were negatively correlated with poor prognostic factors.
CONCLUSIONS:
Our data indicated that Notch receptors play a key role in promoting TNBC and mainly, Notch2 may contribute to poor prognosis of the disease. Hence, it is implicated that Notch2 may serve as a potential biomarker and therapeutic target for TNBC.
Introduction
Breast cancer is the most commonly diagnosed type of cancer, accounting for 24.5% newly registered cases in women and 11.7% newly registered cases in both sexes in a year (https://gco.iarc.fr/today/data/factsheets/populations/900-world-fact-sheets.pdf). Globally, it is second leading cause of death in women after lung cancer (https://gco.iarc.fr/today/data/factsheets/populations/900-world-fact-sheets.pdf). Breast cancer is a phenotypically and biologically heterogeneous disease with diverse response to the treatments offered [35]. Immunohistochemical profiling categorizes breast cancer into four molecular subtypes – luminal A, luminal B, Her2 enriched and Triple negative breast cancer [3]. Luminal A, luminal B and Her2 enriched subtypes are characterized by hormone receptor positivity and better prognosis than TNBC. Triple negative breast cancer is defined by the lack of expression of hormone receptors (ER-, PR-, HER2-), which accounts for 15–20% of breast cancer cases [15,28]. TNBC subtype has unfavorable prognosis and patients with TNBC are at greater risk of disease relapse or distant metastasis [17]. TNBC is characterized as an aggressive form of cancer which includes features such as higher tumor size, high histological grade, advanced stage and lymph node involvement [6]. Currently treatment available for TNBC comprises of surgery, traditional chemotherapy with anthracyclines, taxanes, platinum agents and radiotherapy [30]. Recently, Poly (ADP-Ribose) Polymerase (PARP) inhibitors are used as targeted therapy for TNBC patients with BRCA1/2 gene mutations and programmed death-ligand 1 (PD-L1) positive TNBC patients with locally advanced or metastatic disease are benefited from PD-L1 inhibitors [24,27]. Nonetheless, these therapies are beneficial to a small number of TNBC population.
The Notch signaling pathway is a highly conserved pathway which regulates fundamental developmental processes such as cellular differentiation, proliferation, organ formation, morphogenesis and apoptosis [34]. The Notch signaling pathway is constitutively activated by the interaction of transmembrane Notch receptors and cell-surface ligands on adjacent cells [33]. Upon ligand activation, cascade of events is triggered which includes sequential cleavage by tumor necrosis factor alpha (TNF) converting enzyme (TACE) and gamma-secretase complex, releasing the Notch intracellular domain (NICD). The cleaved NICD translocates to the nucleus and binds with transcriptional regulators such as CBF-1/RBPjk, Suppressor of Hairless, and Lag-1 (CSL), Mastermind-like (MAML), p300 in order to initiate the expression Notch target genes [10,14]. Abnormal expression of Notch receptors is known to play a crucial role in malignant transformation in haemopoietic malignancies as well as solid tumors [31]. Notch signaling pathway is responsible for normal development of mammary glands and the deregulation of Notch signaling pathway is involved in breast carcinogenesis [18]. Studies have frequently shown that Notch1, Notch3 and Notch4 are responsible for aggressiveness of TNBC by contributing to increased cell proliferation, anti-apoptotic activity and stem cell maintenance while few studies have reported tumor suppressive role of Notch2 receptors [1]. However, the role of Notch receptors in TNBC is quite debatable. The present study evaluated Notch1, Notch2, Notch3 and Notch4 expression by immunohistochemistry and their clinical significance in TNBC.
Materials and methods
Patients
One hundred female patients diagnosed with TNBC who had undergone surgery and treatment at a research institute, in Ahmedabad during 2014 to 2019 were enrolled in this retrospective study. Detailed clinical history of the patients including age, menopausal status, tumor size, nodal involvement, American Joint Committee on Cancer (AJCC) TNM stage, histological type, histological grade and treatment offered was noted from the institutional records. Table 2 depicts the clinicopathological characteristics of the patients. Patients who had received chemotherapy or radiotherapy prior to the surgery were excluded from the study. Formalin fixed paraffin embedded (FFPE) blocks were retrieved from archives of Histopathology Department of the institute where the blocks were stored and preserved under optimal conditions. This study was approved by Scientific Review and Ethics committees of the institute.
Immunohistochemistry
Immunohistochemical analysis was used to detect the presence of Notch1, Notch2, Notch3 and Notch4 receptors in formalin fixed paraffin embedded (FFPE) blocks of TNBC patients. 4 μm sections from FFPE tumor tissue blocks were taken using a microtome (Leica Biosystems, Germany) and the sections were mounted on 3-Aminopropyl triethoxysilane (APES) coated slides. Immunohistochemistry was performed using autoimmunostainer instrument, Ventana Benchmark XT (Ventana Medical Biosystems, USA) and Ventana reagents according to the manufacturer’s protocol. Briefly, the protocol includes deparaffinization with EZ prep solution, antigen retrieval using cell conditioning solution (CC1) at 95 °C, followed by incubation with Ultra view DAB inhibitor for 4 min, 100 μl of respective primary antibody for Notch1 (Monoclonal, 1:50, Invitrogen) for 32 min, Notch2 (Polyclonal, 1:25, Invitrogen) for 60 min, Notch3 (Polyclonal, 1:50, Invitrogen) for 60 min and Notch4 (Polyclonal, 1:150, Invitrogen) for 32 min, incubation with Ultra view Horseradish peroxidase (HRP) multimer for 8 min. The detection of antigen-antibody complex was carried out by 3,3’-diaminobenzidine (DAB), counterstaining using haematoxylin for 8 min and post counterstaining with bluing reagent for 4 min. Ultra view DAB detection kit was used for staining of Notch2, Notch3, Notch4 receptors and Opti view DAB detection kit was used for staining of Notch1. The protocol of Opti view DAB detection kit includes additional incubation with HQ linker for 8 min, counterstaining and post counterstaining for 16 min and 8 min respectively. The slides were later mounted using Dibutylphthalate Polystyrene Xylene (DPX) and xylene. Table 1 represents the details of antigen retrieval and antibodies used.
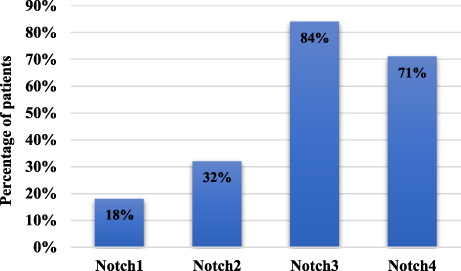
Total cases expressing Notch receptors.
Expression of Notch receptors (Notch1, Notch2, Notch3 and Notch4) was assessed by semiquantitative scoring method by employing Immunoreactive scoring (IRS) system. The percentage positive cells were graded as 0(negative staining), 1(<10% positive cells), 2(10%–50% positive cells), 3(51%–80% positive cells) and 4(>80% positive cells). Further, staining intensity of the cells was ranked as 0(negative), 1+(weak), 2+(moderate) and 3+(strong). The immunoreactive score (IRS) for each case was obtained by multiplying the percentage of positive cells and the staining intensity which gives the range of 0–12. Median score was calculated and cases below the median score were considered as negative or low expression and cases above the median score were considered as over expression.
Statistical analysis
Statistical analysis was performed using SPSS 26.0 (SPSS, Inc., Chicago, USA). The Chi-square test was to used determine the association between the expression of Notch receptors and clinicopathological parameters and the association between the Notch receptors. The correlation between the two parameters was determined by Pearson’s correlation coefficient (r). Yate’s continuity correction value and Fisher’s exact test were used where values were less than 5 in cells of 2 × 2 tables and 2 × 3 tables respectively. Disease-free survival (DFS) time was calculated from date of diagnosis of the patient to date of disease relapse or date of last follow-up. Overall survival (OS) time was calculated from date of diagnosis of the patient to last follow-up or date of death due to cancer. Univariate survival analysis was performed by Kaplan-Meier method and significance of survival differences was calculated using log-rank test. Multivariate survival analysis was performed by Cox regression model with forward stepwise selection, Wald’s statistics and hazard ratio (HR) with 95% confidence interval (CI) were calculated to estimate the prognostic significance. P value ≤0.05 was interpreted as statistically significant.
Results
Correlation of Notch receptors with clinical and pathological parameters
Notch receptors were found to be localized in membrane, cytoplasm and nucleus of the tumor cells. As shown in Fig. 1, positive expression of Notch1 was observed in 18 patients (18%), Notch2 in 31 patients (31%), Notch3 in 84 patients (84%) and Notch4 in 71 patients of TNBC (71%).
Notch1 receptor expression
Notch1 receptor was found to be predominantly localized only in the nucleus of the tumor cells of 18% patients as represented in Fig. 2 and Fig. 3a. As shown in Table 3, Notch1 nuclear positivity was significantly associated with lymph node positive status (60%, 03/05, p = 0.009), high BR score (27%, 17/62,p = 0.02) and necrosis (31%, 13/42, p = 0.004).
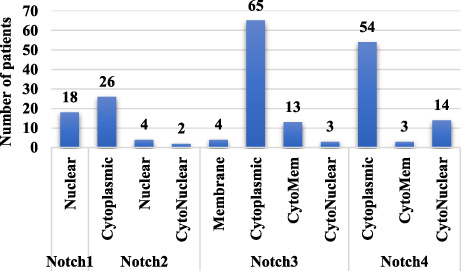
Localization of Notch receptors.
Representative image of Notch1 nuclear staining.
Antigens and antibodies details used in IHC
Clinical and pathological data of TNBC patients (N = 100)
T1 (≤2 cm), T2 (2–5 cm), T3 (>5 cm), T4 (Tumor of any size growing into chest wall or skin). N0 (Negative), N1 (1–3 axillary lymph nodes and/or cancer is found in internal mammary lymph nodes), N2 (4–9 axillary lymph nodes or cancer has enlarged the internal mammary lymph nodes), N3 (Cancer has spread to 10 or more axillary lymph nodes, with at least one area of cancer spread greater than 2 mm).
Correlation of Notch1 and clinicopathological characteristics
∗Fisher’s exact test, ∗a Yate’s continuity correction value. T1 (≤2 cm), T2 (2–5 cm), T3 (>5 cm), T4 (Tumor of any size growing into chest wall or skin). N0 (Negative), N1 (1–3 axillary lymph nodes and/or cancer is found in internal mammary lymph nodes), N2 (4–9 axillary lymph nodes or cancer has enlarged the internal mammary lymph nodes), N3 (Cancer has spread to 10 or more axillary lymph nodes, with at least one area of cancer spread greater than 2 mm).
Notch2 was localized in cytoplasm of tumor tissue of 26% patients (Fig. 3b) and in nucleus of 4% patients. In 2% patients, coexistent cytoplasmic and nuclear expression of Notch2 was detected. As shown in Table 4, the incidence of cytoplasmic Notch2 expression was significantly higher in patients with local or distant metastasis (47%, 7/15, p = 0.05), while there was no significant association observed between nuclear and cytonuclear expression of Notch2 and clinicopathological parameters.
Representative image of Notch2 cytoplasmic staining.
Correlation of cytoplasmic Notch2 and clinicopathological characteristics
∗Fisher’s exact test, ∗a Yate’s continuity correction value. T1 (≤2 cm), T2 (2–5 cm), T3 (>5 cm), T4 (Tumor of any size growing into chest wall or skin). N0 (Negative), N1 (1–3 axillary lymph nodes and/or cancer is found in internal mammary lymph nodes), N2 (4–9 axillary lymph nodes or cancer has enlarged the internal mammary lymph nodes), N3 (Cancer has spread to 10 or more axillary lymph nodes, with at least one area of cancer spread greater than 2 mm).
It was observed that Notch3 receptor was found to be most frequently localized in the cytoplasm (65%) (Fig. 3d), while 4% patients exhibited membrane expression (Fig. 3c), 14% patients and 3% patients exhibited cytomembrane and cytonuclear expression (Fig. 3e) respectively. Although, membrane Notch3 expression was found in few patients, it was observed that tumors which were poorly differentiated exhibited high membrane Notch3 levels (23%, 15/62, p = 0.008). Also, a significant association of Notch3 expression was observed in patients with high BR score (24%, 15/62, p = 0.002) (Table 5). Moreover, cytoplasmic Notch3 expression was more frequently detected in patients with well differentiated to moderately differentiated tumors (43%, 16/37, p = 0.02), low BR score tumors (52%, 14/27, p = 0.04), lymph node negative status (51%, 29/57, p = 0.006) and perinodal extension negative status (44%, 35/79, p = 0.03). Further, a trend of significant association was observed between high cytoplasmic expression and early TNM stage (100%, 02/02, p = 0.06) (Table 6). It was observed that cytonuclear Notch3 positivity was strongly correlated with necrosis (07%, 03/43, p = 0.03) (Table 7).
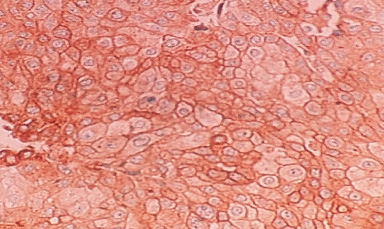
Representative image of Notch3 membrane staining.
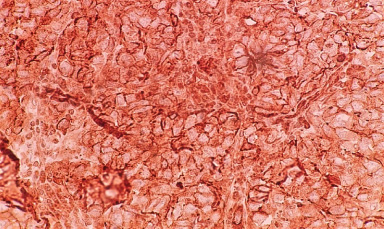
Representative image of Notch3 cytoplasmic staining.
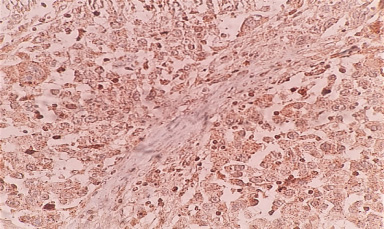
Representative image of Notch3 nuclear staining.
Correlation of membrane Notch3 and clinicopathological characteristics
∗Fisher’s exact test, ∗a Yate’s continuity correction value. T1 (≤2 cm), T2 (2–5cm), T3 (>5 cm), T4 (Tumor of any size growing into chest wall or skin). N0 (Negative), N1 (1–3 axillary lymph nodes and/or cancer is found in internal mammary lymph nodes), N2 (4–9 axillary lymph nodes or cancer has enlarged the internal mammary lymph nodes), N3 (Cancer has spread to 10 or more axillary lymph nodes, with at least one area of cancer spread greater than 2 mm).
Correlation of cytoplasmic Notch3 and clinicopathological characteristics
∗Fisher’s exact test, ∗a Yate’s continuity correction value. T1 (≤2 cm), T2 (2–5 cm), T3 (>5 cm), T4 (Tumor of any size growing into chest wall or skin). N0 (Negative), N1 (1–3 axillary lymph nodes and/or cancer is found in internal mammary lymph nodes), N2 (4–9 axillary lymph nodes or cancer has enlarged the internal mammary lymph nodes), N3 (Cancer has spread to 10 or more axillary lymph nodes, with at least one area of cancer spread greater than 2 mm).
Correlation of nuclear Notch3 and clinicopathological characteristics
∗Fisher’s exact test, ∗a Yate’s continuity correction value. T1 (≤2 cm), T2 (2–5 cm), T3 (>5 cm), T4 (Tumor of any size growing into chest wall or skin). N0 (Negative), N1 (1–3 axillary lymph nodes and/or cancer is found in internal mammary lymph nodes), N2 (4–9 axillary lymph nodes or cancer has enlarged the internal mammary lymph nodes), N3 (Cancer has spread to 10 or more axillary lymph nodes, with at least one area of cancer spread greater than 2 mm).
Localization of Notch4 receptor was observed in cytoplasm of 54% cases (Fig. 3f), cytonuclear expression was observed in 13% cases and cytomembrane expression was observed in 4% cases. As represented in Table 8, low incidence of cytoplasmic Notch4 expression was detected in younger patients (72%, 36/50,p = 0.02) and necrotic tumors (72%, 31/43, p = 0.04). However, cytomembrane and cytonuclear expression of Notch4 did not show any significant correlation with clinicopathological parameters. The association between Notch4 and clinicopathological parameters is listed in Table 8.
Intercorrelation of Notch receptors
As depicted in Table 9, it was observed that expression of Notch2 and Notch3 receptors was significantly correlated with expression of Notch4 receptor. Patients with increased levels of Notch2 expression exhibited increased levels of Notch4 receptor (84%, r = 0.19,p = 0.05) and similarly, higher levels of Notch4 receptor were detected in patients with positive Notch3 expression (76%, r = 0.26, p = 0.008). In addition, Notch1 expression was strongly correlated with nuclear expression of Notch3 (99%, r = 0.22, p = 0.02). Further, it was seen that patients with increased levels of Notch1 receptor had decreased levels of cytoplasmic Notch3 receptor (95%, r = −0.26, p = 0.007).
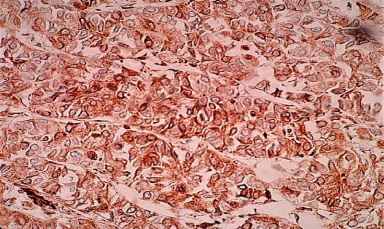
Representative image of Notch4 cytoplasmic staining.
Correlation of cytoplasmic Notch4 and clinicopathological characteristics
∗Fisher’s exact test, ∗a Yate’s continuity correction value. T1 (≤2 cm), T2 (2–5 cm), T3 (>5 cm), T4 (Tumor of any size growing into chest wall or skin). N0 (Negative), N1 (1–3 axillary lymph nodes and/or cancer is found in internal mammary lymph nodes), N2 (4–9 axillary lymph nodes or cancer has enlarged the internal mammary lymph nodes), N3 (Cancer has spread to 10 or more axillary lymph nodes, with at least one area of cancer spread greater than 2 mm).
Intercorrelation between Notch receptors
The association between expression of Notch receptors and disease-free survival time and overall survival time of TNBC patients was investigated using Kaplan-Meier survival curves. The mean follow-up time was 37.05 months and maximum follow-up time was 75 months. During this period, 15 (15%) patients had local or distant metastasis and 15 (15%) patients had died due to the disease. As depicted in Fig. 4, with regard to disease-free survival, patients expressing higher cytoplasmic Notch2 (27%, 7/26, 50.71 ± 4.50 months) had worse disease-free survival than patients expressing lower cytoplasmic Notch2 (11%, 8/74, 66.83 ± 2.71 months, 𝜒2 = 3.77, df = 1, p = 0.05). As depicted in Fig. 5, with regard to overall survival, it was observed that patients with higher incidence of cytoplasmic expression of Notch2 (31%, 8/26, 50.60 ± 3.87 months) had poor overall survival than patients with low expression of cytoplasmic Notch2 (10%, 7/74, 67.93 ± 2.48 months, 𝜒2 = 5.44, df = 1, p = 0.02). Expression of other Notch receptors did not show any statistically significant relationship with disease-free survival and overall survival.
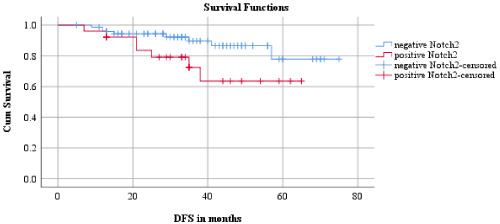
Relationship between disease-free survival and Notch2.
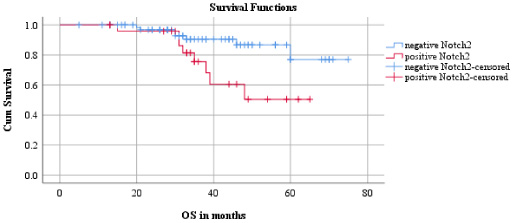
Relationship between overall survival and Notch2.
Multivariate survival analysis using Cox regression model with forward selection method (likelihood ratio) revealed that, with regard to disease-free survival advanced TNM stage (Wald = 9.36, HR = 0.19, 95% CI = 0.066–0.551, p = 0.002) entered at step 1 and high BR score (Wald = 7.94, HR = 4.93, 95% CI = 1.626–14.977, p = 0.005) entered at step 2, indicating their role as poor prognostic factors. With respect to overall survival, greater tumor size (Wald = 10.69, HR = 0.57, 95% CI = 0.10–0.321, p = 0.001) entered at step 1, positive lymph node status (Wald = 5.482, HR = 7.209, 95% CI = 1.380–37.676, p = 0.019) entered at step 2, high BR score (Wald = 8.355, HR = 7.787, 95% CI = 1.936–31.317, p = 0.004) entered at step 3 and higher grade (Wald = 8.762, HR = 0.012, 95% CI = 0.001–0.223,p = 0.003) entered at step 4. Multivariate analysis carried out by Cox regression model to evaluate the prognostic role of Notch receptors expression, independent of clinicopathological parameters revealed that with respect to overall survival, cytoplasmic expression of Notch2 (Wald = 4.87, HR = 3.152, 95% CI = 1.138–8.734, p = 0.027) entered at step 1.
Discussion
Notch signaling pathway is known to play a vital role in normal development and growth. Notch signaling pathway is involved in embryonic development, cellular differentiation, proliferation, homeostasis and apoptosis. Numerous studies have reported that aberrant Notch expression is involved in neoplastic transformation and tumorigenesis. There are four known Notch paralogs which display different patterns in their expression and function across different malignancies. Oncogenic role of Notch signaling was first reported in T-ALL [7] and later reported in lung cancer, ovarian cancer, breast cancer, colorectal cancer [2,21,25,37,39] while tumor suppressive role of Notch signaling was reported in pancreatic cancer and hepatocellular carcinoma. [8,32]. However, research about relationship between Notch receptors and TNBC, and the association among the Notch receptors is limited and unclear.
In this study, we have examined the role of Notch receptors in TNBC cohort by analyzing the cellular localization of all four known Notch receptors by immunohistochemistry. We observed that Notch1 was expressed in the nucleus of tumor cells suggesting the translocation and accumulation of Notch intracellular domain (NICD1) in the nucleus of tumor cells. The accumulation of NICD1 in the nucleus was strongly correlated with positive lymph node status, high BR score and necrosis, indicating the tumor aggravating role of Notch1. Our data was in concordance with studies reporting the association of high-grade tumors and poor prognosis of TNBC patients with up-regulated Notch1. [13,26,38].
Furthermore, upon examination of Notch2 protein it was revealed that Notch2 was localized in the cytoplasm and nucleus of tumor cells. Despite, various studies showing evidence of tumor suppressive role of Notch2, [9,12,36] our data suggests that Notch2 receptor plays a tumor promoting role in TNBC. It was observed that high cytoplasmic Notch2 expression was significantly correlated with local or distant metastasis, however nuclear and cytonuclear expression of Notch2 did not show any statistical significance with clinicopathological parameters. Nonetheless, our study was in line with evidence presented by Lee et al. and Sehrawat et al., stating that inhibition of Notch2 was responsible for anti-apoptotic activity and inhibition of migration in TNBC cells [5,20]. Also, our study demonstrated that high levels of cytoplasmic Notch2 was positively correlated with worse disease-free survival and poor overall survival proving its tumor promoting role. Earlier studies regarding Notch pathway emphasized that nuclear translocation of NICD was required in order to trigger the downstream target genes. Although, recently it has been demonstrated that in non-canonical Notch pathway, nuclear translocation is not necessary for the pathway to be activated and it may interact with other molecules in the cytoplasm to activate the pathway [29]. Therefore, it may be suggested that the association between cytoplasmic Notch2 expression and poor prognosis of the patients might be due to non-canonical Notch pathway.
Moreover, our data represents that majority of TNBC samples exhibited high Notch3 protein levels in the cytoplasm. Along with cytoplasmic expression, membrane, cytomembrane and cytonuclear protein expression were also detected. Notably, it was revealed that up-regulated cytoplasmic Notch3 expression in tumors was associated with well differentiated tumors, low BR score tumors, lymph node negative status and perinodal extension negative status indicating that patients with up-regulated cytoplasmic Notch3 might have favorable prognosis. Our data was in line with studies demonstrating tumor suppressive role of Notch3. Zhang and colleagues observed that high expression of Notch3 inhibited cellular proliferation and tumorigenesis by activating PTEN resulting in better prognosis of breast cancer patients [40]. Another study by Chen and colleagues provided evidence that overexpression of Notch3 inhibited cell cycle progression by upregulating levels of Cdh1 in MDA-MB-231 cell lines [11]. However, contrary to above findings, it was observed that membrane and cytonuclear expression of Notch3 were strongly correlated with high BR score, poorly differentiated tumors and necrosis respectively. Although, cytonuclear expression of Notch3 was not detected frequently it was strongly associated with necrotic tumors suggesting the role of nuclear translocation in tumor promoting activity of Notch3. These results were parallel to the findings of Choy and colleagues that revealed the involvement of nuclear Notch3 expression in tumor promotion [23]. The association of increased levels of Notch3 with poorly differentiated tumors might be due to the non-canonical pathway, where membrane-bound Notch3 may have interacted with cell regulators present in the cytoplasm. These results of Notch3 suggests that nuclear translocation of Notch3 is potentially involved in promoting tumor aggravating activity, whereas membrane-bound Notch3 may act as an oncogene via activation of non-canonical pathway which does not essentially require nuclear translocation. However, the strong association of high cytoplasmic Notch3 expression with well differentiated tumors, low BR score tumors, lymph node negative status and perinodal extension negative status might be due the immature state of the receptor (as it was not translocated to the nucleus) or interaction of cytoplasmic Notch3 with tumor suppressing molecules which might be responsible for favorable prognosis of TNBC patients.
In addition, in the majority of the patients expressing Notch4 receptor, it was frequently detected in the cytoplasm. This study revealed that high levels of cytoplasmic Notch4 were negatively associated with younger age and necrotic tumors. Contrary, to studies which stated that Notch4 was associated with poor prognosis for TNBC patients [20,22], our data represents that Notch4 is inversely correlated to poor prognostic factors of the disease and may function as a good prognosticator. Concurrently, few studies have presented over expression of Notch4 receptor as tumor suppressive in melanoma and endometrial cancer [4,16]. Nonetheless, a significant association was not seen between Notch4 and survival time and therefore, the role of Notch4 in TNBC needs to be further evaluated in a larger cohort.
Further, we evaluated the co-expression patterns of Notch receptors in TNBC patients. It was observed that Notch2 and Notch3 receptors showed positive correlation with Notch4 receptor. These results were in line with the findings of Song et al. which revealed that Notch2 and Notch4 were positively correlated in pancreatic adenocarcinoma [19]. This may be interpreted as Notch receptors are significantly associated with each other and even though Notch4 might function as a good prognostic factor, together with Notch2 they may contribute to tumor promotion and poor survival of TNBC patients since, multivariate survival analysis revealed that Notch2 appeared to be a dominant receptor in relation to Notch4 and patients with Notch2 expression had worse disease-free survival and poor survival. Moreover, it was observed that nuclear expression of Notch3 was positively associated with nuclear expression of Notch1 suggesting the oncogenic role of nuclear localization of both Notch1 and Notch3 receptors. Additionally, it was noted that nuclear Notch1 expression was negatively correlated with the cytoplasmic Notch3 expression indicating opposing role of both the receptors based on their localization, as cytoplasmic Notch3 is associated with better prognosis and nuclear Notch1 is linked to poor outcome of the disease. Therefore, our results imply that interplay of Notch receptors is rather complex mechanism.
In conclusion, our study demonstrated that Notch receptors exhibit a pleiotropic role in TNBC. Notch1 and Notch2 receptors play a tumor promoting role and contribute to poor outcome in TNBC patients. It was also revealed that although membrane and cytonuclear Notch3 had tumor aggravating effects, cytoplasmic Notch3 had tumor suppressive effect. It is implicated that the tumor promoting activity of Notch receptors might be due to the canonical Notch signaling pathway which essentially requires the nuclear translocation of NICD to trigger the transcriptional regulation of target genes or it might be due to the non-canonical pathway which is activated through the interaction of Notch receptors with transcriptional regulators present in the cytoplasm to exert its effects. The tumor suppressive effect of cytoplasmic Notch3 expression may be due to the inactive state of the receptor or its interaction with tumor suppressive molecules in the cytoplasm. Therefore, it is important to assess the subcellular localization of the Notch receptors as they are ubiquitously distributed in the cells in order to evaluate their individual effects in TNBC. Hence, our results strongly indicate that Notch receptors are involved in promoting TNBC and chiefly, Notch2 receptor may serve as a potential prognostic marker and therapeutic target for patients with TNBC.
Footnotes
Acknowledgements
This research work was supported by The Gujarat Cancer Society and The Gujarat Cancer & Research Institute. The authors have no conflict of interest.
Conflicts of interest
The authors declare no conflicts of interest.
