Abstract
Breast cancer (BC) is the most frequent type of malignancy affecting females worldwide. Molecular–based studies resulted in an identification of at least four subtypes of breast carcinoma, including luminal A and luminal B, Human growth factor receptor (HER-2)-enriched and triple-negative tumors (basal-like and normal breast-like). A proportion of BC cases are of the triple-negative breast cancer (TNBC) type. TNBC lacks the expression of estrogen receptor (ER), progesterone receptor (PR), and HER-2, and is known to express androgen receptor (AR) at considerable levels. AR has been shown to promote the progression of TNBC. However, the exact mechanisms have yet to be unraveled. One of these mechanisms could be through regulating the expression of microRNA (miRNA) molecules, which play an important regulatory role in BC through post-transcriptional gene silencing. Activation of AR controls the expression of miRNA molecules, which target selective mRNAs, consequently, affecting protein expression. In this review we attempt to elucidate the relations between AR and miRNA in TNBC.
Introduction
Breast cancer (BC) is the leading cancer and the second deadly cancer affecting women in the USA [1]. It is a heterogeneous disease that has been classified into several subtypes based on two main approaches: histopathological and molecular [2]. Histopathological classification depends on the tumor grade, stage, and immunohistochemical assessment of expression of ER, PR, and HER-2. Such protein markers are of great importance in guiding the treatment strategy and predicting patient’s prognosis [3,4]. Histopathologic examination is currently the standard method for diagnosing and classifying BC; however, it is time-consuming and highly dependent on personal error [5]. Histopathology reports alone do not reflect the true biology of the tumor, which is needed for determining best prognosis and treatment options [6].
Microarray-based gene expression profiling of BC is the basis for their molecular classification. Sørlie et al. selected 427 unique genes (from a total of 8,102 genes) whose expression significantly varied among different tumor samples [7]. These genes were used to reflect the unique intrinsic properties of five different BC categories, each with distinct clinical outcome and histopathological features. These subtypes are: luminal A, luminal B, HER-2 enriched, basal-like tumors, and normal-like tumors [8,9]. Table 1 lists the five intrinsic subtypes with their key histopathological properties. New molecular subtypes have emerged later such as the claudin-low, molecular apocrine, and interferon-related subtypes [2].
The molecular classification of BC depends on scrutinizing the variations of gene expression that drive cancer cells [9]. Furthermore, it has predictive significance beyond the traditional predictive indexes and can support determining the appropriate cure for patients [9,10].
Luminal subtypes
About 70% of BC cases are hormonal receptor (HR)-positive including ER and PR, which display more affirmative prognosis than HR-negative BCs. Around 90–95% of HR-positive/HER-2-negative BC cases are either luminal A or luminal B subtypes [8,11]. The luminal B subtype pinpointed as ER-positive/HER-2-negative exhibits greater expression of proliferation genes including the proliferation marker Ki67 compared to luminal A subtype [12,13] and poorer baseline distant recurrence-free survival at five to ten years, in spite of adjuvant systemic therapy [11,13]. Whereas Luminal A subtype has low Ki67expression [14], luminal B subtype is positive for the proliferation markers and is more aggressive with higher histological grade and poorer diagnosis. The treatment of this subtype of BC chiefly depends on endocrine therapy [15].
HER-2 Enriched subtype
HER-2, a tyrosine kinase receptor found in the cell membrane, contributes to the signal transduction pathways that result in cell differentiation and proliferation [16]. HER-2-enriched tumors are typically proliferative and aggressive [17]. About 15–20% of all BC cases coincide with HER-2 overexpression [18]. HER-2 is a target for BC therapy as a result of its distinct function in the pathogenesis of BC [17]. Tumors that overexpress HER-2 showed an elevation in histological grade, bad prognosis, but higher susceptibility to chemotherapy [17,19]. Meanwhile, the HER-2 subtype that is hormone-receptor (HR)-positive is associated with improved disease free-survival (DFS) and overall survival in comparison with HR-negative tumors [20].
Basal-like subtype
These tumors have the poorest prognosis. Potential illustration for the discrimination of this type include distinct cell of origin, cancer subtype-specific inherited and epigenetic events to every tumor subtype [21]. The term basal-like subtype has been given because tumors of this type express genes that are typically presented in normal breast myoepithelial cells [18]. Scrutiny depends on the Cancer Genome Atlas Network data, which has proven that the basal-like subtype comprises a distinct subtype among BCs, the plurality (86%) of TNBCs cases that display as ER-negative, PR-negative, and HER-2-negative related to the basal-like subtype [11,12]. The terms of TNBC and basal-like are used reciprocally to denote to the same tumor subtype, nevertheless, within the set of TNBCs [22]. Basal-like subtypes have diverse clinical presentations, different response to chemotherapy, unique histological structures, and several complications [23,24].
TNBC
TNBCs denote 60–90% of all basal-like cases and about 10–20% of all BC cases [8,25]. Moreover, TNBCs deemed to be extremely aggressive with a greater proliferative index compared to other BC types [26]. In addition, TNBC has a poor prognosis with rapid relapses in relation to other BC subtypes [27]. TNBC is divided into six molecular BC subtypes, including two basal-like (1 and 2), immunomodulatory, mesenchymal, mesenchymal stem-like, and luminal AR [28]. The mesenchymal subtype displays the mischievous consequences and the immunomodulatory subtype shows the best prognosis [11]. Additionally, TNBC has a unique molecular profile and its track of metastasis usually leads to the brain, lungs, and bones [29]. It is vital to differentiate among both basal-like and non-basal-like subtypes of TNBC in order to select the most suitable chemotherapy; for example, carboplatin and docetaxel are interchangeable in basal-like subtypes in terms of efficacy, but carboplatin is less active in other intrinsic subtypes in the metastatic condition [30]. A proportion of TNBCs possess
The morphological appearance of TNBC has heterogeneous features and is classified into different types including infiltrating secretory carcinoma, ductal carcinoma or not otherwise specified, metaplastic carcinoma, medullary carcinoma, squamous carcinoma, adenoid cystic carcinoma, myoepithelial carcinoma, apocrine carcinoma, or carcinoma arising in the background of micro-glandular adenosis [32]. TNBC has variations in aggressiveness; for instance, adenoid cystic carcinomas are deemed slow growing and have a good prognosis status post-surgical excision [33].
Basal-like 1 and basal-like 2 subtypes
This subtype includes basal-like 1 and basal-like 2. The basal-like 1 subtype forms rapidly from dividing cells related to the elevation in cell proliferation and damaged cell cycle checkpoints harmonious with increment expression of DNA damage response genes such as ataxia telangiectasia; Rad3-Related Protein (ATR) or BRCA [28]. As a result of elevated proliferation rate, raised Ki67 mRNA expression is more restraint to anti-mitotic agents’ diathesis cell cycle [30]. On the other hand, the basal-like 2 subtype shows unique gene ontology including epidermal growth factor signaling, glycolysis, and gluconeogenesis pathway [34]. There is an elevation in the expression of specific genes including epidermal growth factor receptor (EGFR), TP63, and MET [35,36]. Therefore, as a treatment strategy, DNA damage-activated pathways are potential targets in this type of BC [37]. The platinum salt and poly ADP-ribose polymerase 1 (PARP) inhibitors blemish the homologous recombination DNA repair system that results in DNA cross-linking strand break inhibitors [37,38]. PARP inhibitors likewise are promising drugs for basal-like tumors [39]. Prognostic biomarkers are needed to grasp the rate of medical response of such agents that target the DNA repair system [37].
Mesenchymal and mesenchymal stem-like subtypes
Various biological events are involved in tumors with the mesenchymal subtype [40]. Genomic records proposed that gene collections based on gene expression profiling of processes like cell motility, extracellular matrix interaction, epithelial-mesenchymal transition (EMT), and growth factor signaling pathways participate in the distinctive qualities of mesenchymal cancers [37]. The mesenchymal stem-like subtype may be sentient to mammalian target of rapamycin (mTOR) inhibitors since these tumor cells have stimulated phosphoinositide 3-kinase (PI3K) or protein kinase B (PKB) signaling [28]. An additional treatment choice for mesenchymal stem-like subtype is eribulin mesylate, which considerably suppresses the EMT pathway in BC cells [41]. It is clear that the fibroblast growth factor receptor pathway has to be suppressed in order to effectively target the EMT pathway and treat mesenchymal stem-like TNBCs [40,42]. In addition, the mesenchymal stem-like subtype is characterized by genes involved in angiogenesis such as vascular endothelial growth factor receptor 2, which was found to has vast responsiveness to the mammalian target of rapamycin inhibitors and dasatinib, a tyrosine kinase (TK) inhibitor [34].
Immune-enriched subtype
The immune-enriched subtype is histologically considered an uncommon discrete form of TNBC that is characterized by promising prognosis even when high grade [34]. The improved gene collections of the immunomodulatory subtype are involved in immune cell signaling, antigen presentation signaling, cytokine signaling, and immune signal transduction. The latter includes different pathways such as the nuclear factor
Luminal AR subtype
The luminal AR subtype is identified by active AR signaling and ER-negative expression with activated genes responsible hormonally controlled metabolic pathways such as steroid synthesis, porphyrin metabolism, and androgen-estrogen metabolism [34]. AR mRNA is expressed more than nine times compared to other subtypes [37]. Thus, this subtype appears to be susceptible to AR antagonists like bicalutamide. Patients with this subtype have decreased DFS and overall survival [43,46]. Numerous studies have provided medical evidence that AR-positive TNBC patients can gain a clinical value from AR blocking [37]. PI-3K inhibitors in increment to AR antagonist are expected to be high effective in treating AR-positive TNBC as a result of frequent mutations detected within the catalytic subunit alpha of phosphatidylinositol-4,5-bisphosphate 3-kinase, which trigger these tumors [47].
Human AR
AR has a few signal sequences, two transcriptional activation functions termed the ligand-independent activation function 1 (AF-1) found within the N-terminal domain, which is vital for the highest function of the AR [58]. Furthermore, the ligand-dependent activation function (AF-2), positioned in the LBD, is vital for creating the co-regulator binding site besides mediating direct interactions among the N-terminal and LBDs (N/C interactions) [59,60]. The nuclear localizing signal is responsible for importing of AR into the nucleus and a nuclear export signal is responsible for exporting the AR to the cytoplasm upon ligand withdrawal. Both are found between the DBD and hinge region and in the LBD, respectively [45] .
AR is a nuclear receptor that belongs to the nuclear receptors super family, which includes receptors for other steroid hormones [53]. There are two different modes of actions of ligand-dependent AR signaling, either by controlling the transcription of target genes through DBD signaling [54] or independent of DNA binding (i.e. non-genomic) [61] Fig. 1. It has been reported that androgen/AR axis exerts non-genomic action by triggering the AR/Src complex, which leads to alterations of membrane elasticity and increase of the activity of second messenger pathways. The latter events result in the activation of different responses including cell differentiation, proliferation, survival, invasion, motility, and cell cycle progression [62]. Furthermore, the expression of the insulin growth factor receptor type I (IGF-R I) and the epidermal growth factor receptor (EGFR) are considered part of the non-genomic response of AR [63–65]. Several findings support the role of AR/Src complex assembly in enhancing the oncogenic properties of the TNBC cell lines in association with cell motility and invasion through activation of PI3K/Akt pathway [66–73]. These pathways are critical in the rapid action of second messenger pathways taking place within seconds to minutes of androgen entry into cells [74,75].
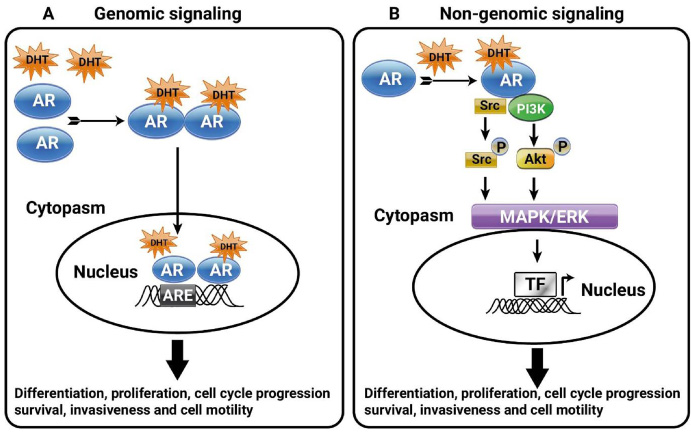
Genomic and non-genomic AR signaling in cancer cells. (A) Genomic AR signaling. the activated form of androgen, 5α-dihydrotestosterone (DHT), bind to AR receptor then AR undergoes translocated to the nucleus. In the nucleus, AR binds to the enhancer regions on the promoter called androgen response elements (AREs), in order to activate the transcription of different responses. (B) Non-genomic signaling. After DHT binding to AR in the cytoplasm, the ligand-bound receptor can recruit and activate phosphatidyl-inositol 3-kinase (PI3K) and Src to trigger several signaling pathways leading to modulation of transcription factors involved in different cell responses.
Indirect gene trans-repression can also arise from DBD-independent signaling by AR binding and sequestering transcription factors such as AP-1 [76], which are ordinarily needed to up-regulate expression of target genes in the absence of the AR binding, e.g. nerve growth factor receptor and matrix metalloproteinase-13 [77].
Androgens are usually considered as male hormones, but they are identified at physiologically significant levels in the circulation of females, and play important biological roles in female development and physiology [78,79]. There are numerous kinds of androgenic hormones in the blood stream including dehydroepiandrosterone sulphate (DHEAS), dehydroepiandrosterone (DHEA), androstenedione (A4), testosterone, and DHT [80]. The potent androgen is DHT and to a lesser extent the precursor androgens DHEA and A4, which are formed by the ovaries, whereas DHEA, DHEAS, A4, and testosterone are formed by the adrenal gland [80]. In addition to endocrine provenance testosterone, DHT, and their metabolites are similarly manufactured in peripheral tissues including the breast, bone, and brain [81]. Testosterone levels fluctuated through the menstrual cycle and are progressively reduced, even though not entirely following menopause [81,82]. By disparity, synthesis of adrenal androgens continues after menopause [81]. Androgens and estrogens bind to steroid hormone-binding globulin (SHBG) in the circulation that dominates the bioavailability of hormones to breast, ovary, bone, and other tissues. In some studies, raised serum testosterone level is related to an elevation of BC risk in postmenopausal females [80]. Nevertheless, inconsistent results among studies are often reported, possibly due to limitations in the methods used to measure testosterone levels and the complexity in explaining these results in relation to intracrine [80].
The AR is found to be expressed in 12–55% of cases of TNBC [46,83]. While the exact percentages vary across studies, it is obvious that a considerable proportion of TNBCs expresses AR. Preclinical findings suggest that AR-positive TNBCs may depend on AR signaling for growth [84]. AR is also expressed in cell lines representing the basal-like 1 and 2, mesenchymal, and mesenchymal stem–like TNBC molecular subtypes [28]. If AR-targeted therapy is valuable in AR-positive TNBC, it would appear to be the first successful targeted therapy and would be significantly benefit this population of females [85]. Contradictory, findings about AR role in BC cell lines are present. Whereas many published data support the idea that AR can be a tumor suppressor gene, others studies have found that AR is an oncogenic protein [86,87]. It has been proposed that the role of AR depends on the ratio of AR to ER [88]. AR is a tumor suppressor gene in the presence of comparable levels of AR and ER-α since AR antagonizes the proliferative effect of ER-α. On the other hand, AR is an oncogene in the absence of ER-α where AR functions as an ER-α mimic [89]. The specific molecular mechanisms underlying this event have not been expansively demonstrated.
The function of AR in BC has yet to be elucidated. Numerous preclinical studies have reported AR as a potential tumor suppressor in ER-positive BC, with anti-proliferative findings resulting from the cross talking among the steroid receptor signaling pathways [89]. The expression manners of ER, PR, HER-2, CK5/6, EGFR, Ki-67 and AR were studied by immunohistochemistry analysis in BC cell lines [90]. Androgens can have both a suppression or an activation role on different BC cell lines relying on the co-expression of other SHRs and availability of breast adipose tissue fibroblasts (BAFs) [91]. In the presence of BAFs, testosterone promotes cell divisions in ER-positive MCF7 and T47D cell lines, but not in MDA-MB-231 TNBC tumor cells. This could be due to the elevated level of expression of aromatase enzyme that facilitates the conversion of androgens to estrogens, followed by ER-mediated cell proliferation [91,92]. DHT treatment results in AR conformational change and its translocation from the cytosol to the nucleus; this facilitates the transcriptional regulation of target genes that can be modulated by various AR co-regulators [56,93,94]. MDA-MB-231 cell line has been previously reported as either AR-negative [90,95] or AR-positive [87,96].
AR in BC
Androgens and estrogens have contradictory leverage on BC cell growth and development, for instance, ER-α positive BC is more expected to be AR-positive compared to ER-α negative tumors where AR has an inhibitory effect on proliferation of ER-α positive BC cells [97]. Therefore, the ratio of AR to ER-α has been considered an important in the parameter of BC cell propagation and clinical consequences [80]. ER-α negative BCs are more aggressive than ER-α positive tumors. It has been proposed that in ER-α -negative tumors, AR acts more as ER-α functioning as an oncogene [98]. The exact mechanism of androgen action in breast mammary progression and initiation of BC has still need to be elucidated [80].
The role of AR in breast tumorigenesis is contradictory. It has been stated that women with increased levels of androgens have an increased risk of BC [99]. Yet, AR expression in BC is considered as a favorable prognostic indicator [100,101]. Although accumulating evidence indicates that androgens have an inhibitory function in the growth of MCF7 cells [97,102,103], other studies illustrated a stimulatory role of androgens in MCF7 cells as well as MDA-MB-231 cells [104–106]. The expression of AR has also been reported to be associated with increased invasiveness [87,107] and a predictor and a facilitator of metastasis [108]. Androgen treatment of the BC T47D and MCF7 cells induced cell motility and metastasis
miRNA molecules
The miRNAs are short non-coding RNAs that facilitate post-transcriptional gene silencing. They form single-stranded RNA molecules of 19–24 nucleotides in length after cleavage by an endonuclease called DICER. These molecules never translated. miRNAs form RNA-RNA duplex with matching coding mRNA molecules at the 3’ UTR region either halting translation or accelerating mRNA degradation [110]. Although The first miRNA discovered was reported in
miRNA and BC
Several molecular changes appear to take place during the progression of BC. Among these changes is the differential expression of miRNA molecules. Previous studies have explained the capability to discriminate human cancers types, diagnosis, and prognosis in relation to the expression manner of mRNA [118–120]. Similarly, the variable expression of miRNA molecules has been found to differentiate not only human cancers; but also their histological criteria [121,122]. The same is true for BC whereby the expression pattern of miRNA molecules is associated with BC classification [123]. The expression patterns of miRNA in BC tissues has designated a new class with a distinctive expression of miRNA molecules between primary and metastatic breast tumors [124,125]. Interestingly, differential expression of miRNAs was enough to classify TNBC into an independent cluster of other types of BCs [126]. Further analysis revealed the existence of some miRNAs that are specifically up- and down-regulated [127]. For example, luminal-specific miR-200 family is down-regulated in combination with reduced epithelial phenotype.
AR and miRNA in BC
The potential relationship between AR expression and miRNA signature was well investigated in prostate cancer (PC). A miRNA profiling study of six PC cell lines, nine PC xenograft models, and 9 prostate carcinoma samples recognized a significant link between miRNAs and AR expression [128]. On the other hand, studies on AR-associated miRNAs in BC are limited, in order to reveal the association between miRNAs and AR in BC; miRNA expression profiling was performed in BC cell lines with variable expression levels of AR [129]. Some miRNAs were inconsistently associated with androgen expression. For example, androgen activation resulted in over-expression of miR-125b [129]. Moreover, this miRNA is considered a tumor suppressor gene in BC, which is capable to negatively regulate the expression of HER-2 [130]. Another miRNA, miR-34, has been described to be absent in androgen-refractory cell lines [131]. The expression of the latter molecule has been shown to be triggered by DNA injury and oncogenic stress in a p53-dependent pattern resulting in cell cycle arrest and eventually apoptosis by regulation of some regulatory proteins [132–134]. Androgen-independent cell lines exhibit over-expression of miR-21 including those of the breast. Interestingly, miR-21 is linked to increasing cell migrations as well as invasion via regulation of myristoylated alanine-rich protein kinase C substrate (MARCKS) among other proteins [135]. MiR-146 is similarly up-regulated in androgen-dependent cells and appears to block cell migration by repressing the Rho-associated coiled-coil-containing protein kinase 1 (ROCK1) expression, an activator of cell invasion and metastasis [136,137]. MiR-221/miR-222 are upregulated in androgen-independent cells [138]. This miRNA seems to participate in androgen-independent growth as well as maintain the androgen-independent phenotype by repression of the cell cycle suppressor cyclin dependent kinase inhibitor 1B [139,140]. Research studies on AR-associated miRNAs in BC are not as common as in PC. A few studies have shown the regulation of miRNA expression by AR (Table 2). A study was performed by Al-Othman et al. (2018) revealed that treatment of MDA-MB-231 cells with DHT resulted in up regulation of 35 miRNAs and down-regulation of three other ones [141,142]. Earlier, a study conducted by Nakano et al. (2013) on AR-positive MCF-7 cells treated with DHT revealed the up-regulation of 13 miRNAs and down-regulation of 28 others [143]. Lyu et al. (2014) investigated the overall miRNA expression in MDA-MB-453 cell line treated with DHT; four miRNAs were up-regulated whereas six miRNAs were down-regulated [144]. The latter cell line was also included with two other ones, T47D, and MCF-7, to investigate the effect of AR activation by its agonist CI-4AS-1 on expression of 84 miRNAs. Although a number of miRNAs were differentially expressed upon treatment, there were very few common miRNAs among the three cell lines. In addition, among the miRNAs with altered expression in MDA-MB-453 cells were miR-100 and miR-125, which were considerably down-regulated concurrently with the increase in expression and extracellular release of their target metalloprotease-13 [145].
DHT induced alteration in miRNA Expression in BC cell lines
Alterations in miRNA expression level after treatment with DHT in MDA-MB-231, MCF-7 and MDA-MB-453 BC cell lines.
DHT induced alteration in miRNA Expression in BC cell lines
Alterations in miRNA expression level after treatment with DHT in MDA-MB-231, MCF-7 and MDA-MB-453 BC cell lines.
A number of studies have suggested an association between miRNAs and AR in PC [129,146–149], but little evidences indicate the possible role of AR in regulating miRNA expression in BC. let-7a has a tumor suppressive role which was approved in AR-positive/ER-positive BC cells where DHT activation resulted in its transcriptional upregulation which led to reduction in cell proliferation, invasion and migration [150]. The expressions of AR, let-7a and CD44C/CD24-/low were studied in patients with invasive BC; patients who express AR and let-7a might have a good prognosis, suggestion that AR/let-7a pathway could be used as a new optional therapeutic target [150]. Different studies demonstrated the interaction between AR and miRNAs in regulating BC cells behavior. Three BC cell lines (MA-MDA-453, T47D, and MCF-7) were screened for 84 miRNAs after treatment with AR agonist CI-4AS-1 resulted in alterations in the expression profile of varied miRNAs; such as miR-100 and miR-125 which were found considerably downregulated concurrently with the increase and extracellular release of metalloprotease-13 [145].
In AR-positive tumors; AR negatively induced long non-coding RNA ARNILA that associated with poor progression-free survival, firmly related to AR and abled to sequester miR-204 which led to enhance the expression of SRY-box 4 (Sox4) gene which promotes the EMT, migration, and invasion in TNBC, as result ARNILA is repressed by the action of DHT and AR which leads to reduction in miR-204 adsorption and finally suppression of Sox4 expression, meanwhile AR-negative tumors Sox4 gene is activated [151]. DHT treatment resulted in variation of miRNAs expression, for example miR-328-3p was the most upregulated resulting in downregulation of CD44 a target of miR-328-3p that leads to reduction in cell adhesion and migration Fig. 2 and Table 2. The result was confirmed following miR-328-3p mimic transfection [141].
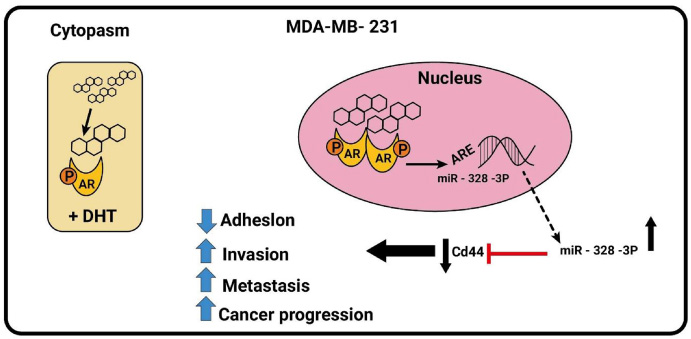
Androgenic-inducible miRNAs involved in the process of BC progression. In MDA-MB-231 cells, the DHT administration results in an androgen-induced AR activating signal pathway which upregulates miR-328-3P expression and negatively regulates CD44.
It was found that 153 miRNAs to be differentially expressed in AR-positive vs. AR-negative in BC cell lines. These findings revealed a role for these miRNAs in the regulation of AR in BC [152]. In MDA-MB-453 BC cells, miRNAs profiling was investigated after DHT treatment showed that miR-30a, b, and c expression were the most downregulated. AR did not bind to the promoter region of miR-30a a cancer suppressor gene, so downregulation could be due to AR-induced cell signaling pathways, stimulation of AR expression and AR-induction signal inhibits mir-30a expression which in turn activates AR availability and suppresses cell growth [153]. Treatment of BC cells with miboleron a synthetic androgen resulted in inhibition of miR-21 that led to inhibition of cell proliferation by negative modulation of the onco-miR-21. This provided an evidence that triggered AR works as a transcriptional inhibitor of miR-21 expression [154].
MiRNAs play a significant role in cancer progression, their levels are dysregulated in different kinds of cancers. There is an increasing interest in their potential application as therapeutic agents in chemotherapy or combined with anti-cancer therapies [155]. For example, the positive feedback mechanism between AR and miR-30a might be an initial point for additional research work regarding the role of miRNAs as a therapeutic predictive marker. Furthermore, perception the stability between AR and ER concerning the different BC subtypes, this will yeild different response to endocrine therapies in BC [155]. A recent interesting branch of miRceptor, which is involved in hormone-dependent tumor, needs to be evaluated regarding their tumor microenvironment. Therefore, it is realistic to predict how the relation between miRNAs and AR in BC can aid in the rising of the treatment chances and getting closer to BC personalized therapies [155].
Androgens trigger the AR/Src/PI3-K complex assembly and invasiveness in both luminal and mesenchyme subtype of TNBC cells. Thus, the AR/Src/PI3-K complex appears as a proper target for BC therapy[156]. Because of the role of AR in BC, understanding the molecular function of AR in BC might improve the therapeutic approach of these cancers. The regulation of miRNA by AR may facilitate this understanding and may lead to novel treatments. Additionally, androgen/AR signaling might also function at non genomic level to regulate miRNA. Until now, indeed, there is no evidence that non-genomic signaling causes alterations in miRNA expression.
Footnotes
Conflict of interest
The authors claim no competing financial or person interests with other people or organizations.
