Abstract
BACKGROUND:
Block of programmed cell death protein 1 (PD-1) interaction with its ligand, PD-L1, enhances anti-tumor activity.
OBJECTIVES:
We aimed to assess the association between PD-L1 expression in tumor cells and CD8+ tumor infiltrating T cells (TILs) as well as soluble (s)PD-L1 serum levels in patients with triple negative breast cancer (TNBC) compared to triple positive (TPBC).
METHODS:
A total of 113 tumor sections and 133 serum samples were available from 144 patients with breast cancer (72 TNBC and 72 TPBC). Dual immunohistochemistry staining was applied to determine differential PD-L1 expression in tumor cells and CD8+ TILs. Soluble PD-L1 serum levels were also evaluated in patients compared to 40 healthy women by ELISA method.
RESULTS:
Despite TPBC patients which were mostly grades 1/2, TNBC patients were grade 3 (72% versus 66.7%, P < 0.001). Most of the TNBC patients were stages I/II, whereas most of the TPBC patients were stages III/IV (57.3% versus 68.3%,P = 0.005). There was no difference in tumor size and metastasis between TNBC and TPBC patients, although the number of involved lymph nodes was significantly more in TPBC patients (P = 0.0012). PD-L1 expression was detected in 11.5% of samples mostly in TNBC subtype and was associated with advanced grades (P = 0.039). There was no relationship between PD-L1 expression and tumor stage. PD-L1 expression in CD8+ TILs was nonsignificantly higher than tumor cells. Serum levels of sPD-L1 showed no difference between patients and healthy women. We found no correlation between PD-L1 expression in tumor lesions and serum levels of sPD-L1 in patients.
CONCLUSION:
PD-L1 expression was more detected in our patients with TNBC. It seems that, these patients who are resistant to standard chemotherapy regimens may get benefit from PD-L1 inhibition therapy and because of its low serum levels, sPD-L1 cannot interfere with this therapy.
Introduction
Cancer is one of the main causes of death with an estimated 12.7 million involved patients around the world and expected to rise to 21 million by 2030 [1]. Breast cancer is one of the most common diagnosed cancer and the leading cause of mortality in females worldwide [2]. As reported by Iranian cancer registry, the incidence of breast cancer shows an increasing trend among Iranians since 1999 and currently it is the fifth most common cause of death among women [3]. Because of great improvements in therapeutic strategies, mortality rate of breast cancer has been declined in past decades [4,5]. However, due to the lack of competent markers for predicting disease courses and detecting chemo-resistance patients, the efficacy of existing treatments remain unsatisfactory [6]. To improve patients’ survival, it is crucial to find specific biomarkers in breast cancer as potential therapeutic targets [7]. Although immune system is continually attempting to destroy tumor cells, these cells also escape the antitumor efforts by various mechanisms [6,7]. Checkpoint inhibitors which are crucial for the maintaining of self-tolerance, are also used by cancer cells for immune evasion. Interaction between checkpoint inhibitors on tumor infiltrating T cells and their corresponding ligands on tumor cells can be readily blocked by antibodies or modulated by recombinant forms of ligands or receptors. Antibody therapy against checkpoint inhibitors is likely to be a major treatment for patients with a variety of malignancies [8]. Antibodies produced against cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) and blocking of programmed cell death protein 1 (PD-1) interaction with its ligands have been effective in antitumor immunity. Phenotype comparison of PD-1 knockout mice that develop delayed-onset organ-specific inflammation and CTLA-4 knockouts with uncontrolled global T cell proliferation reveals that although CTLA-4 regulates de novo immune responses, PD-1 exerts its major effect on ongoing immune responses. As it seems, PD-1 pathway is more selective and controllable with less autoimmune-related toxicity. PD-1 is expressed by CD4+ and CD8+ T cells and following interaction with its ligands, PD-L1 and PD-L2 belong to the B7 family, leads to classical programmed cell death. Despite PD-L2 expression which is limited to antigen presenting cells (APCs), PD-L1 is expressed on tumor cells, APCs and activated T cells. Furthermore, PD-1 interaction with PD-L1 suppresses both T cell proliferation and cytokine secretion [9,10]. Blocking antibodies against PD-1 or PD-L1 have been used promisingly for the treatment of various solid tumors by restoring T-cell activity [11].
Based on molecular typing, triple negative breast cancer (TNBC) is characterized by lack of the expression of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 (HER2). This subtype is an aggressive disease with a high rate of distant metastasis and poor response to standard chemotherapy regimens and represents about 20% of all breast cancers [12]. Tumor infiltrating lymphocytes (TILs) especially CD8+ T cells are massively recruited into TNBC due to high rate of genetic mutations and production of neoantigens, but probably become inefficient in tumor cell killing because of high expression of PD-L1 by these cells [13,14]. Accordingly, anti-PD-L1/anti-PD-1 therapy might be effective in the treatment of PD-L1+ TNBC.
Powles et al. found a stronger association between PD-L1 expression by TILs and immune evasion than PD-L1 expression by tumor cells [15]. Therefore, determination of PD-L1 expression either by tumor cells or by CD8+ TILs would be important before therapeutic approaches using anti-PD-L1/anti-PD-1. Moreover, therapeutic anti-PD-L1 might be absorbed by high rate of soluble (s)PD-L1 before reaching the tumor. So, evaluation of sPD-L1 serum level would be helpful to ensure the effectiveness of the medication. This study was designed to evaluate the expression of PD-L1 on tumor cells and CD8+ TILs as well as its association with sPD-L1 serum levels in patients with TNBC compared to those with triple positive breast cancer (TPBC).
Materials and methods
Patients and clinical samples
In this retrospective study, a total of 144 patients with breast cancer (72 TNBC and 72 TPBC) diagnosed between 2009 and 2019 at Faghihi Hospital affiliated with Shiraz University of Medical Sciences, Iran, were included. All of the patients had provided written informed consent before their operation to permit the use of their samples in future research works. The protocol of this study was approved by the ethics committee of our university (Decision Number: IR.SUMS.REC.1397-45) in accordance with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Demographic information for each patient and their pathology reports were collected from their medical files. None of these patients had received any preoperative chemotherapy or radiotherapy. All tumors were graded based on current pathology standards [16]. Tumor stages were determined according to the American Joint Committee on Cancer (AJCC) staging system [17]. There were 133 serum samples from these patients in the biobank of Institute for Cancer Research and 113 formalin-fixed paraffin-embedded tumor sections in pathology archive as both samples of 102 patients were available.
Detection of PD-L1 in tumor cells and CD8+ TILs
PD-L1 expression was first screened by typical immunohistochemistry (IHC) test using 3,3 ′ -Diaminobenzidine (DAB) staining. To evaluate PD-L1 expression on CD8+ TILs, IHC double staining was done using fast red dye for the detection of PD-L1 and green chromogen for CD8 (Zytomed, Berlin, Germany). Briefly, tissue sections were deparaffinized and endogenous peroxidase was inactivated. Then antigen retrieval was done in a pressure cooker for 9 min in Tris-EDTA buffer, pH 9. After blocking with 1% goat serum, anti-PD-L1 rabbit monoclonal antibody (clone E1L3N, Cell Signaling, Danvers, MA, USA) 1:100 (in single and double staining) and anti-CD8 mouse monoclonal antibody (clone C8/144B, Invitrogen, Carlsbad, California, USA) 1:75 (only in double staining) in phosphate-buffered saline were added to each section and incubated for 1 h at room temperature. Immunostained sites were visualized with the related conjugates, horse radish peroxidase-polymer anti-mouse and alkaline phosphatase-polymer anti-rabbit secondary antibodies (Zytomed) and their appropriate substrates. Then tissue sections were counterstained with hematoxylin and mounted. PD-L1 and CD8 stained sections were scored in a blind manner by three different pathologists. Scoring was done based on membranous or cytoplasmic expression of PD-L1 in tumor cells and CD8+ cells in tumor stromal compartments. Samples were categorized as PD-L1 positive if this molecule was expressed by more than 1% of the cells [18–20]. Adjacent normal tissue in each section was considered as negative control and human tonsil tissue was used as a positive control. Background compensation was done by the omission of primary antibodies in positive control.
Evaluation of sPD-L1 serum levels
Soluble PD-L1 levels were determined in serum samples of 133 patients with breast cancer and 40 age-matched healthy women using a commercially available ELISA Kit (R&D, France & Munich, Germany) with a sensitivity of 4.52 pg/mL. The concentration of sPD-L1 in each sample was obtained from a linear standard curve of known sPD-L1 concentrations ranging from 25 to 1600 pg/mL, based on the manufacturer’s protocol.
Statistical analysis
Normal distribution of the data was analyzed by the Shapiro–Wilk test. Parametric statistical tests were used for data with normal distribution, otherwise nonparametric analyses were applied. The relationship between PD-L1 expression in tumor tissues and each of the clinicopathological parameters were evaluated using chi-square and the nonparametric Fischer’s exact test. Soluble PD-L1 levels were compared between patients and controls by t-test. The association between sPD-L1 serum levels and each of the clinicopathological parameters were evaluated by Mann–Whitney U test. The correlation between PD-L1 expression in tumor tissues and sPD-L1 serum levels was assessed with the Spearman’s rho test. All statistical analyses were done with SPSS v. 23, and two-sided P-values less than 0.05 were considered statistically significant for 2 × 2 tables. When there was more than two groups, corrected P-value (Pc) was calculated by 𝛼∕n, where 𝛼 was original P-value and n was the number of groups.
Results
To study the association between PD-L1 expression in tumor tissues and sPD-L1 serum levels in patients with breast cancer, a total of 144 patients including 72 patients with TNBC (mean age of 46.6 ± 12.6 years and age range from 26 to 84 years at the time of diagnosis) and 72 patients with TPBC (mean age of 46.9 ± 10.4 and age range from 28 to 77 years) were examined. Pathological characteristics of the patients are summarized in Table 1. As shown, tumor grades and stages were significantly different between TNBC and TPBC patients. A total of 52/72 (72%) of TPBC patients were grades 1 and 2, whereas 48/72 (66.7%) of TNBC patients were grade 3 (P < 0.001). A total of 103/144 (71.5%) of the patients were stages I/II whereas 41/144 (28.5%) of the patients were stages III/IV (P = 0.005), as most of the TNBC patients were stages I/II [59/103 (57.3%)] and most of the TPBC patients were stages III/IV [28/41 (68.3%)].
Comparison of pathological characteristics in patients with triple negative and triple positive breast cancer
Comparison of pathological characteristics in patients with triple negative and triple positive breast cancer
∗ Pc < 0.017, ∗∗ Pc < 0.0125.
There was no difference in tumor size and tumor metastasis between TNBC and TPBC patients, although the number of involved lymph nodes (N2∕N3) in TPBC patients was significantly higher than TNBC patients (P = 0.0012).
Immunohistochemical staining of PD-L1 was done for 113 of the patients whose tissue blocks were available. The results of IHC test is summarized in Table 2. As shown, 12 out of 13 PD-L1+ samples were TNBC and just 1 sample was TPBC (P = 0.005). There was an association between PD-L1 expression and tumor grade (P = 0.039), as 11/13 (84.6%) of PD-L1+ samples belonged to the patients with grade 3 (P = 0.017). There was no relationship between PD-L1 expression and tumor stage, tumor size, metastasis and lymph node involvement.
PD-L1 expression in patients with breast cancer
∗ Pc < 0.017, ∗∗ Pc < 0.0125.
To evaluate differential expression of PD-L1 on tumor cells and CD8+ TILs, dual IHC test was performed on breast cancer tissues (Fig. 1). However, in most of the samples, the percentage of PD-L1 expression was higher in CD8+ TILs than tumor cells (Table 3), this difference was not statistically significant (P = 0.105).
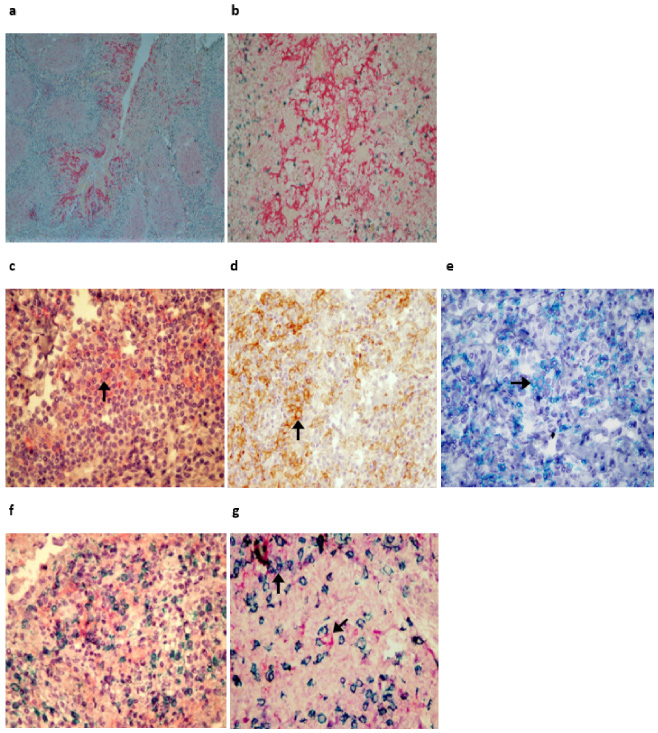
Immunohistochemical staining of PD-L1 in breast cancer lesions. a and b: Tonsil as positive control for PD-L1 and CD8; c and d tumor lesions, arrows show PD-L1+ tumor cells stained with Fast red and DAB, respectively; e CD8+ TILs in a breast cancer lesion, arrow indicates membrane expression of CD8 stained with Green chromogen; f and g breast cancer tumor lesions, PD-L1+ tumor cell is marked in the centre and PD-L1+ CD8+ TIL is displayed in the upper left corner (original magnification: a. 40×, b–f. 100×, and g. 400×).
PD-L1 expression in tumor cells compared to CD8+ tumor infiltrating T cells
∗ Pc = 0.0038.
Serum levels of sPD-L1 showed no difference between patients and controls. The level of sPD-L1 also revealed no difference between TNBC and TPBC and between each of the TNBC and TPBC patients compared to the controls (Table 4). There was no association between PD-L1 expression in tumor lesions and sPD-L1 serum levels in 102 studied patients (P = 0.069, r = −0.039).
Serum levels of sPD-L1 in patients with breast cancer compared to the healthy controls
SEM: Standard Error of Mean.
In this study, the expression of PD-L1 in tumor cells and CD8+ TILs was investigated in TNBC and TPBC patients and correlation between PD-L1 expression in tumor lesions and sPD-L1 serum levels was determined. While, TNBC is usually occurs in younger ages [21], we did not find any difference between the mean age of TNBC and TPBC patient and more than 60% of the patients in both group were under 50 years old (Table 1). As it seems, the incidence of breast cancer is increasing in young women [22].
Consistent with the results of previous studies, most of our TNBC patients were grade 3 [23,24]. Although, TNBC is considered as an aggressive disease with a high rate of distant metastasis [25,26], we did not find any difference in tumor size and metastasis between two studied subgroups. Regardless of small size, TNBC tumors can be invasive [23]. Based on our results, metastasis was detected in 2 TNBC and 3 TPBC patients (Table 1). Moreover, higher rate of N2∕N3 was identified among TPBC patients than in TNBC. This observation might result from the metastasis pathway of TNBC which occurs hematogenously rather than via the lymphatics, [27]. Thus, the lower percentage of clinical stages III/IV in TNBC (28.5%) versus TPBC (68.3%) can be interpreted in our study.
In this study, PD-L1 expression was detected in 13/113 (11.5%) of the tested samples, as 12/63 (19%) of TNBC samples were PD-L1+ with a range of 10% to 80% which 92% of those were grade 3 (Tables 2 and 3). Previous studies showed higher expression of PD-L1 in TNBC than in hormone receptor-positive breast cancers [28,29]. More frequency of PD-L1 positive samples among patients with higher histological grades of breast cancer has also previously been reported [30]. Higher PD-L1 expression by TNBC than TPBC might be explained by higher infiltration of TILs into TNBC microenvironment due to high rate of genetic mutations and production of neoantigens by this type of tumor [14]. There is some evidence for the involvement of TILs in a phenomenon called “adaptive resistance” which occurs by production of cytokines such as IFN-𝛾 that led to the induction of PD-L1 expression in tumor cells [28].
Circulating sPD-L1 which is generally produced following proteolytic cleavage of membrane-bound PD-L1 molecules by matrix metalloproteinase or translation of alternatively spliced mRNA, can also bind to PD-1 receptor [31]. Following administration of therapeutic anti-PD-L1, these antibodies might be absorbed by sPD-L1 before reaching into tumor site. On the other hand, lately, sPD-L1 serum level has been reported to be considered as a noninvasive prognostic factors in different solid tumors [32]. Thus, we measured serum levels of sPD-L1 in patients with breast cancer. Although we found no difference between patients and controls (Table 4), our results were in agreement with the results of Li et al. who also did not detected any difference in serum concentration of sPD-L1 between TNBC patients and healthy women [33]. It seems that, evaluation of sPD-L1 serum level cannot be considered as a predicting biomarker for the prescription of anti-PD-L1 therapy in TNBC patients. Moreover, due to higher frequency of PD-L1+ tumor lesions among TNBC patients than TPBC, anti-PD-L1 can be considered a promising therapy even for a small group of TNBC patients. Nevertheless, targeting the PD-1/PD-L1 pathway alone does not result in complete restoration of T cell function [34]. Thus, it is required to identify other molecules and inhibitory pathways that are involved in T cell dysfunction. Moreover, the predictive value of high PD-L1 expression in malignancies is not properly clear, correlation with worse [35] and favorable outcome [36] has been reported while some showed no association [37]. These contradictions might be due to the interpretation of IHC results to evaluate PD-L1 expression in tumor lesions that strongly affected by the clone of applied monoclonal antibodies which directly determine their sensitivity. There is no precise cut-off to mark a sample as positive, moreover, PD-L1 is not uniformly expressed in different parts of a tumor mass [14,38]. Therefore, further investigations are required to find more biomarkers. A combination index is essential for better identification of patients benefit from PD-1/PD-L1 checkpoint blockade treatment. Hence, using PD-L1 as an exclusive predictive biomarker for cancer immunotherapy is still open to discuss.
Compliance with ethical standards
Funding
This work was supported by a grant from Shiraz University of Medical Sciences (No. 14586).
Conflict of interest
Ethical approval
All procedures involving human participants were approved by the Ethics Committee of Shiraz University of Medical Sciences (Decision no. IR.SUMS.REC.1397-45) in accordance with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
This retrospective study was approved by the research board of Shiraz University of Medical Sciences with a waiver for the need to obtain informed consent, because all of the patients had provided written informed consent before their operation to permit the use of their samples in future research work.
Footnotes
Acknowledgments
This work was performed in partial fulfillment of the requirements for the MSc degree in Immunology (Parvaneh Yazdanpanah). We thank K. Shashok (AuthorAID in the Eastern Mediterranean) for improving the use of English in the manuscript.
