Abstract
OBJECTIVE:
To review the concordance between pathologists in the diagnosis of atypical ductal hyperplasia of the breast.
METHODS:
Systematic review of articles registered in the PubMed database with the terms “pathologists” and “atypical ductal hyperplasia”, “atypical ductal hyperplasia” and “interobserver variability”, in Embase with “interobserver agreement in atypical ductal hyperplasia” and Lilacs with the terms in Spanish “patólogos” and “hiperplasia ductal atípica”, without the use of filters, between 03/16/2022 and 03/26/2022 searching for articles that assess inter-observer agreement in the diagnosis of atypical ductal hyperplasia by using the kappa statistic. Review articles, conference proceedings, and commentaries were excluded.
RESULTS:
With the search terms, 507 publications were found. Of these, 491 were excluded from the screening by title and abstract. Of the full reading of the remaining articles, 6 were selected for qualitative analysis. The kappa values ranged from slight agreement, kappa = 0.17, to substantial agreement, 0.69 obtained from pathologists dedicated or experienced in breast pathology and who received a prior tutorial session to review the general criteria for atypia.
CONCLUSIONS:
This study reviews the reproducibility in the diagnosis of atypical ductal hyperplasia. A better concordance was obtained with a previous teaching process offered to pathologists dedicated or experienced in breast pathology.
Introduction
Atypical ductal hyperplasia (ADH) is the most common high-risk proliferative lesion in breast cancer [1]. It is defined as a proliferative epithelial lesion that has cytological and architectural characteristics similar to those of low-grade ductal carcinoma in situ (DCIS) but with less compromise in the architecture, degree of involvement of the terminal lobular duct unit, and extension [2]. Page et al. [3] proposed a cut-off of <2 involved spaces to classify a lesion as ADH, but Tavassoli and Norris proposed that lesions ≤2 mm in contiguous extent be classified as ADH considering that using the involvement of at least two spaces was prone to confusion [4].
It is considered a non-obligatory precursor and independent risk factor for developing invasive breast cancer [5–7]. Its diagnosis is associated with a 3 to 5 fold-increased risk of developing invasive breast carcinoma in either breast, although some studies have found a higher risk in the ipsilateral breast [2]. The cumulative incidence of breast cancer with a previous diagnosis of hyperplasia atypical ductal is estimated between 17 and 26% at 10 years and almost 30% at 25 years [8–10].
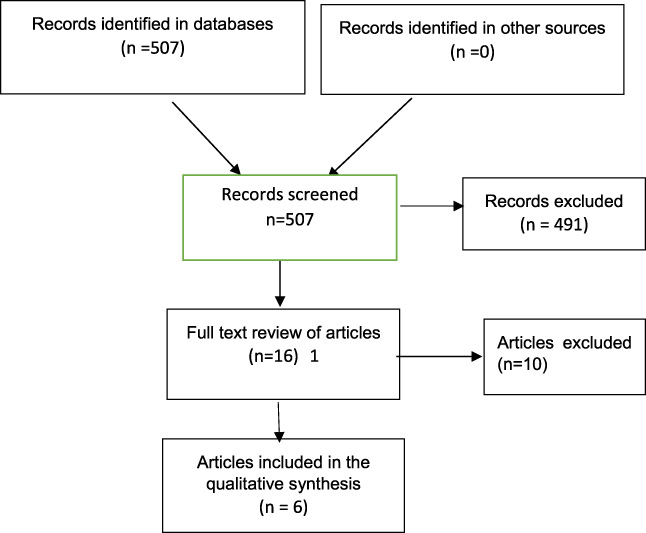
Information flow through the different phases of the systematic review.
Percutaneous core needle biopsy is a widely used procedure for diagnosing breast disease [11–14] due to its diagnostic performance, low rate of complications, minimal incision, acceptance [14], and ease of the technique in an outpatient setting. However, in atypical ductal hyperplasia, because a major difference from low-grade ductal carcinoma in situ is the extent of the lesion, the distinction between these two histologies is not always easy on breast core needle biopsies because the size of the biopsy creates the possibility of undersampling the lesion. To remedy this limitation, if what is identified in the biopsy is atypical ductal hyperplasia, it is recommended to perform an open biopsy [7] which is considered the standard management [15] and with which 10 to 20% of cases are reclassified in recent studies as ductal carcinoma in situ or invasive cases [2]. Even with open biopsy, it is unlikely that a perfect agreement will be achieved, even strictly complying with the current criteria, considering these are arbitrary and more quantitative than qualitative [2].
The pathological diagnosis of breast biopsy is often considered the gold standard for patient management, but it has significant diagnostic variation, being more susceptible to errors than in other areas of surgical pathology [16]. This variation frequently occurs among pathologists in the diagnosis of atypia so that many benign without atypia breast biopsies might be interpreted as having atypia [17], but also in the analysis of other parameters, so the diagnosis can result in a subjective assessment, prone to variability between observers and limited by the amount of tissue obtained [18]. In breast pathology, a particularly problematic area is the distinction between atypical ductal hyperplasia ADH and low-grade ductal carcinoma in situ of limited extention [2]; These are lesions that exist in a biological continuum but have been separated artifactually based on subtle histologic and extensional differences.
Learning the reproducibility of the diagnosis of atypical ductal hyperplasia becomes important to surgeons who rely on the pathologist’s interpretation to guide their management recommendations. This study reviews the studies that have assessed the consistency of the histological diagnosis of atypical ductal hyperplasia of the breast using the kappa statistic [19].
A systematic review of articles registered in the PubMed database with the terms “pathologists” and “Atypical ductal hyperplasia”, “Atypical ductal hyperplasia” and “interobserver variability”, in Embase with “interobserver agreement in Atypical ductal hyperplasia” and Lilacs with the Spanish terms “patólogos” and “hiperplasia ductal atípica”, without the use of filters, between 03/16/2022 and 03/26/2022 searching for articles with the evaluation of inter-observer agreement in the diagnosis of atypical ductal hyperplasia by using the kappa statistic. Review articles, conference proceedings, and commentaries were excluded.
Search terms were provided by the author; with another reviewer, the articles on the objective of the research were selected, according to the title, or by additional information in the abstract. Discrepancies were resolved by reviewing the full article and by a mutual agreement. In the articles that continued to be evaluated, the author reviewed the entire article to ensure the relevance of the articles with the objective of the research.
Results
With the search terms, 507 publications were found. Of these, 491 were excluded in the screening by title and abstract, 55 of them because they were repeated articles in the search. The rest were excluded because they did not correspond to the objective of the research, were part of the exclusion criteria and in one the reason was the language (Chinese). From the remaining review of 16 full articles, 10 were excluded mostly because they did not assess inter-observer agreement in the diagnosis of atypical ductal hyperplasia by using the kappa statistic, and 6 were finally selected for qualitative analysis (Fig. 1). These are summarized in Table 1. The kappa values were scored according to Table 2. The strength of agreement in the extracted studies ranged from slight agreement, kappa = 0.17, to substantial agreement, 0.69, obtained by pathologists dedicated or experienced in breast pathology and who received a prior tutorial.
Characteristics and values of the kappa statistic in the selected studies
Characteristics and values of the kappa statistic in the selected studies
∗Number of pathologists. ∗∗Not defined. ∗∗∗The U.K. National Cancer Screening Evaluation Unit. Regional Coordinators (one for each of the 14 English health regions and Scotland, Wales, and Northern Ireland).
Interpretation of the kappa statistic [19]
The evaluations of concordance were made among dedicated pathologists, with experience or interest in breast pathology in two publications [20,21], one publication describes many pathologists with interest in breast pathology [22], a pathologist with experience and training compared to general pathologists [23] and in two publications, the affinity of pathologists with breast pathology was not established [24,25].
Atypical ductal hyperplasia represents a diagnostic dilemma, with wide variation in reproducibility among pathologists, mainly due to problems in differentiating atypical ductal hyperplasia from low-grade ductal carcinoma in situ. The low consistency observed in the diagnosis raises serious concerns regarding the robustness of the diagnostic criteria. Elston et al. [21] raises two possible reasons for this inconsistency, the differences in interpretation of the specific histological characteristics, the most important, and the second in the selection of a field where the morphology is heterogeneous. These authors emphasize that fair consistency in their study is due to differences in morphological interpretation.
When evaluating the concordance for atypical ductal hyperplasia in the selected studies, it was found that the interobserver reproducibility ranges from slight to a substantial agreement, with reproducibility indices (kappa) ranging between 0.17 [22] and 0.69 [20]. A substantial agreement was found among pathologists with dedication or experience in breast pathology when a previous tutorial on the subject was included, as assessed in the study by Darvishian et al. [20], obtaining a concordance that is higher than that one of the other studies reviewed.
However, these strategies defined as adequate to improve concordance between pathologists are far from resolving these limitations in diagnosis that can lead to inadequate treatments [17]. Schnitt et al. [26]. In their study on proliferative lesions, observed a complete agreement of 58% among the participating pathologists. According to their study, interobserver variability can be reduced using standardized criteria for ductal lesions by using Page’s criteria [3] and providing training slides. In contrast, Palazzo and Hyslop [27] documented a fair kappa value (0.36) in the diagnosis of benign and malignant ductal lesions with the same criteria among the pathologists participating in the study (community and academic pathologists).
The inclusion of studies with immunohistochemistry in the diagnosis improved the concordance from fair to moderate in the study by MacGrogan [25], but continued to be moderate in the study by Jain [24], showing only a marginal advantage. The use of digitized images marking a specific field was not useful either (on the contrary, it slightly decreased the concordance) remaining in the range of a fair concordance [21] reflecting inconsistencies due to differences in the morphological interpretation [2].
The diagnosis of ADH is problematic as it represents a spectrum of atypical architectural patterns ranging from flat epithelial atypia to low-grade DCIS. Distinguishing between some atypical ductal hyperplasia lesions and small low-grade ductal cancers in situ remains a problem, which has implications for inter-observer variability, some less common patterns (solid and Pagetoid) can be difficult to distinguish from classic lobular carcinoma in situ [2] and additionally, there is a considerable variation in the diagnosis of atypia so that some benign without atypia breast biopsies can be considered as having atypia [17].
The correct identification of ADH is an essential step for the adequate clinical management of patients [28–31]. The current treatment for ADH is excision, while the options for DCIS are mastectomy, breast-conserving surgery followed by radiation treatment, and breast-conserving surgery alone. Any of these treatments can be followed by hormone therapy [31].
The included studies in this systematic review were defined by their evaluation with the kappa statistic, which is the most widely accepted concordance test when the data is categorical [19]. Its values are usually between 0 (no agreement) and 1 (absolute agreement). A negative value can be obtained in situations where the actual agreement is less than the value expected by chance [32] Table 2.
Strategies are required to reduce the risks of inter-observer variability in management, such as considering reviewing the diagnostic standards of this pathology as well as its differential diagnoses, referral of histological studies in the event of diagnostic suspicion of ADH to pathologists trained in breast pathology with continuous education processes in their practice and considering novel and creative approaches such as artificial intelligence and molecular markers [33].
Conclusion
Studies on concordance in the diagnosis of atypical ductal hyperplasia between pathologists show an important variation that is reduced when pathologists are dedicated or have experience in breast pathology. The histologic continuum of the atypical ductal hyperplasia and the low-grade ductal carcinoma in situ represents a major diagnostic problem that also has therapeutic implications. New developments are required to improve the performance in the diagnosis of this entity, such as artificial intelligence and molecular analysis.
Footnotes
Conflicts of interests
The author has no conflicts of interest.
Funding
The author has no funding to declare.
