Abstract
BACKGROUND:
Around 5%–10% of breast cancers are due to hereditary breast and ovarian cancer syndrome. Genetic testing is important to identify these cases, enabling the adoption of specific risk-reducing treatment strategies.
OBJECTIVE:
To analyze the performance of genetic testing and its implications in patients with indication of genetic testing to identify hereditary predisposition to breast cancer.
METHODS:
This is a retrospective observational cross-sectional study, including 176 patients with clinical indication of genetic testing for pathogenic variants related to breast, ovarian and pancreatic cancers (among others), managed from 1999 to 2021 in an Oncology private clinic located in the city of Teresina (PI), Brazil.
RESULTS:
There was a predominance of female patients (98.9%) and those with a family (91.0%) and personal history (64.2%) of cancer. In the study, 102 patients (57.9%) received genetic testing. BRCA1 and BRCA2 pathogenic variants occurred in 26 cases (90%). Another three PALB2 and TP53 pathogenic variants were detected. Eleven pathogenic variant carriers (38%) underwent risk-reducing surgeries.
CONCLUSIONS:
BRCA1/BRCA2 pathogenic variants occurred in around 25% of tested patients. Approximately 42.0% of the patients did not undergo genetic testing, despite clinical indication.
Keywords
Introduction
Around 5%–10% of breast cancers (BC) are due to hereditary breast and ovarian cancer syndrome, mainly related to the BRCA1 and BRCA2 pathogenic variants, among others [1]. In Brazil, the National Institute of Cancer estimates that in the 2020–2022 triennial, there will be about 66,280 new BC cases per year. Therefore, it is estimated that around 3,314 to 6,628 of these new cases/year will be due to highly penetrant inherited cancer predisposition genes [2].
Inequitable access to genetic testing is still a reality, mainly due to the high costs involved. Nevertheless, the cost of multigene panel testing has been decreasing in the last years, and more patients can be tested [3].
The aim of this study is to analyze the performance of genetic testing after genetic counseling and its implications in patients diagnosed with BC or with a family history of BC in a real world scenario.
Methods
This is a retrospective observational cross-sectional study, including 176 patients with a clinical indication for genetic testing due to their increased likelihood of having pathogenic variants related mainly to breast, ovarian and pancreatic cancers according to the criteria for genetic testing to identify hereditary predisposition to BC established by the National Comprehensive Cancer Network (NCCN) [4,5]. These patients were managed from 1999 to 2021 in a private oncology clinic in the city of Teresina (PI), Brazil.
After the diagnosis of BC in patients with a positive family history, pre-test counseling was conducted and genetic testing by next-generation sequencing (NGS) was recommended, preferentially multigene panel testing that is covered by the Brazilian National Supplementary Health Agency (ANS, acronym in Portuguese): (1) ATM; (2) BARD1; (3) BRCA1; (4) BRCA2; (5) BRIP1; (6) CDH1; (7) CHEK2; (8) EPCAM; (9) MLH1; (10) MSH2; (11) MSH6; (12) NBN; (13) NF1; (14) PALB2; (15) PMS2 (PMS2CL); (16) PTEN; (17) RAD51C; (18) RAD51D; (19) STK11; and (20) TP53. In NGS, the target region includes the coding DNA and splicing sites (+25 bp intronic). Minimum expected vertical coverage is 250×, and horizontal coverage is 100% at 20× and >99% at 100×. Bioinformatics analysis is performed using a previously validated platform, being able to detect single-nucleotide variants (SNV), small insertions and deletions (indels) and copy number variations (CNV). Biological material (saliva or blood) was collected after the patient signed an informed consent term specifically for genetic testing to identify hereditary predisposition to cancer [6].
The following variables were retrospectively obtained: age, sex, diagnosis or not of breast carcinoma, family history of cancer, management/outcome, pathogenic variants and indication of bilateral mastectomy – BM (unilateral therapeutic mastectomy and contralateral risk-reducing mastectomy) and risk-reducing bilateral salpingo-oophorectomy (RRBSO).
The research protocol for this study was approved by the Research Ethics Committee of the State University of Piauí, Teresina (PI), Brazil, with report number 4.311.835. All ethical principles in current research in the National Health Council resolution number 466/12 and international documents were followed.
Results
The mean age of patients in the study was 48 years. Female patients predominated (174 [98.9%]) and the majority had a family (160 [91.0%]) and personal history (113 [64.2%]) of cancer (Table 1). As shown in Table 1, 113 patients (64.2%) had a previous diagnosis of cancer. Of these, 84 (74%) were treated with chemotherapy and 57 (50%) with radiotherapy, 7 (6%) had cancer recurrence and 5 (4%) died.
Clinical profile of study patients
Clinical profile of study patients
Among the patients with a family history of cancer (Fig. 1), there was a mean number of 3,20 cancer cases per family and mean age at diagnosis was 40 years.
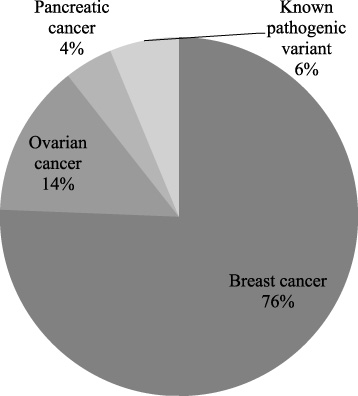
Distribution of study patients with family history of cancer according to tumor site or presence of known pathogenic variants in their families.
Of the 176 patients with a recommendation to research genetic mutation, 102 (57.9%) were assessed by genetic testing. Ninety-six of these probands (94.1%) had a diagnosis of BC and 6 (5.9%) were relatives of BC patients. Table 2 shows the frequency of pathogenic variants found in tested study patients (BRCA1/2, PALB2 and TP53), indicating the risk-reducing surgical treatment adopted in cases of BRCA1 and PALB2 pathogenic variants.
Results of genetic tests performed and prophylactic treatment adopted
MU: Unilateral mastectomy. BM: Unilateral therapeutic mastectomy and risk-reducing contralateral mastectomy. RRBSO: Risk-reducing bilateral salpingo-ophorectomy.
According to Table 3, of the 29 pathogenic variants in probands of the study, 5 concomitant variants were found: the c.3331_3334del (p.Gln1111fs) type, 2 of the c.6724A>G (p.Ser2242Gly) type and 2 of the c.791_794del (p.Ser264fs) type–all occurring in the BRCA1 gene. The remaining BRCA1, BRCA2, PALB2 and TP53 pathogenic variants were the only ones that occurred.
Pathogenic variants in study probands and management/outcomes
*Access available at: https://ncbi.nlm.nih.gov/clinvar/. **Performed with chemotherapy and radiotherapy. UM: Unilateral therapeutic mastectomy. BM: Unilateral therapeutic mastectomy and contralateral risk-reducing mastectomy. RRBSO: Risk-reducing bilateral salpingo-ophorectomy. MRI: Magnetic resonance imaging.
Patients with BRCA1 and BRCA2 pathogenic variants have an increased risk of developing breast, ovarian and pancreatic cancer at an earlier age, in comparison to the general population. Therefore, it is important to identify these patients, to enable the modification of the natural history of cancer in these individuals and family members. Risk-reducing surgical strategies can be adopted (BM and RRBSO). Periodical follow-ups can offer magnetic resonance imaging and chemoprophylaxis, using targeted drugs for breast carcinoma, such as PARP inhibitors, and avoiding radiotherapy in case of TP53 pathogenic variant [7,8]. These gene mutations are also associated with increased risk of developing other cancers, including prostate, pancreatic and ovarian cancers and melanoma, among others [4].
Patients with clinical criteria to undergo genetic testing for cancer risk should receive pre-test counseling. It should be highlighted that although genetic testing can guide subsequent management, it does not fully eliminate the patient’s risk of developing some type of cancer. Whenever possible, multigene panel testing including high-penetrance genes or even moderate-penetrance genes associated with cancer should be prioritized. In this study, a multigene panel revealed BRCA1, BRCA2, PALB2 and TP53 pathogenic variant carriers. The risk of developing breast and ovarian cancers could be decreased in these patients, especially after discussing the possibility of BM and RRBSO. Furthermore, these patients were counseled about the importance of lifestyle changes in the prevention of cancers in general (healthy nutrition, regular physical activity, avoid smoking or drinking alcohol) [9].
The majority of pathogenic variants detected in the study patients investigated were BRCA1 and 2 (90% of the pathogenic variants detected). Other pathogenic variants related to an increased risk of developing BC and ovarian cancer at a lower rate (PALB2 and TP53) were also detected. BRCA1 and BRCA2 are autosomal dominant high-penetrance genes that have the ability to form molecular complexes with proteins of a number of other genes, participating in homologous recombination. Failures in homologous recombination lead to a large accumulation of errors in DNA, accelerating the process of carcinogenesis, which makes these individuals more susceptible to developing malignancies at an earlier age [10,11].
TP53 (a high-penetrance gene responsible for the Li-Fraumeni syndrome [LFS]) gene is known for its relation to several cancers in humans. The five most common cancers associated with LFS are: adrenocortical carcinoma, BC, central nervous system tumors, osteosarcomas and soft tissue sarcomas. In addition, LFS is related to the occurrence of leukemia, lymphoma and gastrointestinal, head and neck, renal, laryngeal, lung, skin (including melanoma), ovarian, pancreatic, prostate, testicles and thyroid cancers. The lifetime risk of developing cancer in patients with LFS is ≥70% in males and ≥90% in females, and the risk of developing BC is around 85% until age 60 in female LFS carriers. In breast cancer it is very important to identify SLF carriers, since BC patients with TP53 pathogenic variants should undergo a contralateral risk-reducing mastectomy, owing to the high risk of developing cancer in the contralateral breast. Furthermore, in these patients radiotherapy is contra-indicated due to increased risk of carcinogenesis from radiotherapy [12,13]. In this study, the TP53 pathogenic variant occurred recently. Genetic counseling was offered and periodical clinical follow-up with whole-body MRI and mammogram was proposed, since the patient refused to undergo risk-reducing surgery.
PALB2 (moderate-penetrance gene) codes a protein that interacts with the BRCA2 gene during homologous recombination and repair of double-strand breaks [14]. Both PALB2 pathogenic variant carriers in this study had BC. One patient was treated with surgery, chemotherapy and radiotherapy and the other underwent BM and RRBSO.
Owing to the predisposition to BC in BRCA1/BRCA2 pathogenic variant carriers, a risk-reducing mastectomy is essential to prevent the development of breast malignancies in these patients and also decrease mortality. A study conducted by Heemskerk-Gerritsen et al. indicated a reduced mortality in BRCA1/BRCA2 pathogenic mutation carriers undergoing BM (0.5% [BM] × 1.7% [control group]) [15].
In BRCA1/BRCA2 pathogenic variant carriers, it is also important to discuss the possibility of RRBSO due to their increased lifetime risk of developing ovarian cancer at an earlier age. In younger patients, the procedure may be delayed until after age 35–40, in order to decrease the deleterious effects of early menopause and preserve fertility. After RRBSO, histopathology exam of the surgical specimen using a specific protocol is carried out to detect in situ or early invasive cancer. The surgical technique recommended for the performance of RRBSO is laparoscopy with complete removal of the fallopian tubes and ovaries. An endobag should be used to collect surgical specimens [16–18].
In this study, eleven patients with pathogenic variants underwent risk reducing surgeries: eight had a BM and seven had an RRBSO. Of the total, four underwent BM and RRBSO. Two cases with BRCA1 pathogenic mutations are scheduled to undergo prophylactic surgeries: one patient will receive a BM and the other will undergo RRBSO. All patients undergoing prophylactic surgeries did not have tumors on histopathology exam of the surgical specimens and have no evidence of malignancy (median follow-up of 30.7 months). Among the remaining pathogenic variant carriers included, four received the result recently and are still under follow-up care, four with magnetic resonance imaging of the breast, five with chemotherapy and radiotherapy. One patient had a unilateral therapeutic mastectomy and two died from disease progression–one case developed metastatic pancreatic cancer and another had clinical stage IV BC with Central Nervous System metastases.
Conclusions
Of the 102 cases selected for genetic testing, only 29 were found to have either BRCA1/BRCA2, PALB2 or TP53 mutations. Pathogenic variants in BRCA1/BRCA2 genes were detected in around 25% of patients undergoing genetic testing. Genetic risk for hereditary breast, ovarian and pancreatic cancer was not assessed in 42% of the patients, despite clinical indication. The main limitation of genetic testing for cancer risk was financial.
Footnotes
Acknowledgements
None.
Conflicts of interest
None.
Funding
None.
Author contributions
DRSF: study concept, data curation, formal analysis, methodology, project management, writing (original draft, review and editing). REARC: study concept, data curation, formal analysis, methodology, project management, writing (original draft, review and editing). MKSM: study concept, data curation, formal analysis, methodology, project management, writing (original draft). SCV: study concept, data curation, formal analysis, methodology, project management, writing (review and editing). All authors have read and approved the final draft.
