Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040. 1
Keywords
Primary Objective
Objective BR2.7: Factors Affecting Response and Prognosis of Breast Cancer. Explain the prognosis and likelihood of recurrence and response to therapy for breast cancer patients based on knowledge of molecular classification and/or gene expression profiling, morphologic classification, grade, prognostic marker studies, and other predictive factors.
Competency 2: Organ System Pathology, Topic: Breast (BR), Learning Goal 2: Molecular Basis of Breast Neoplasms
Patient Presentation
A 65-year-old woman with no relevant past medical history is found to have a spiculated right breast mass measuring approximately 1.6 cm on routine mammogram. She has had unremarkable annual screening mammograms since her first mammogram at age 50. Her family history is significant for breast cancer in her mother and maternal grandmother, both older than 75 years at diagnosis.
Diagnostic Findings, Part 1
The patient undergoes an ultrasound-guided core needle biopsy, shown in Figure 1. The biopsy demonstrates small clusters and single cells with enlarged and pleomorphic, hyperchromatic nuclei and moderately increased mitoses infiltrating through stroma. A diagnosis of poorly differentiated invasive ductal carcinoma is rendered. There is no evidence of lymphovascular invasion or in situ carcinoma.
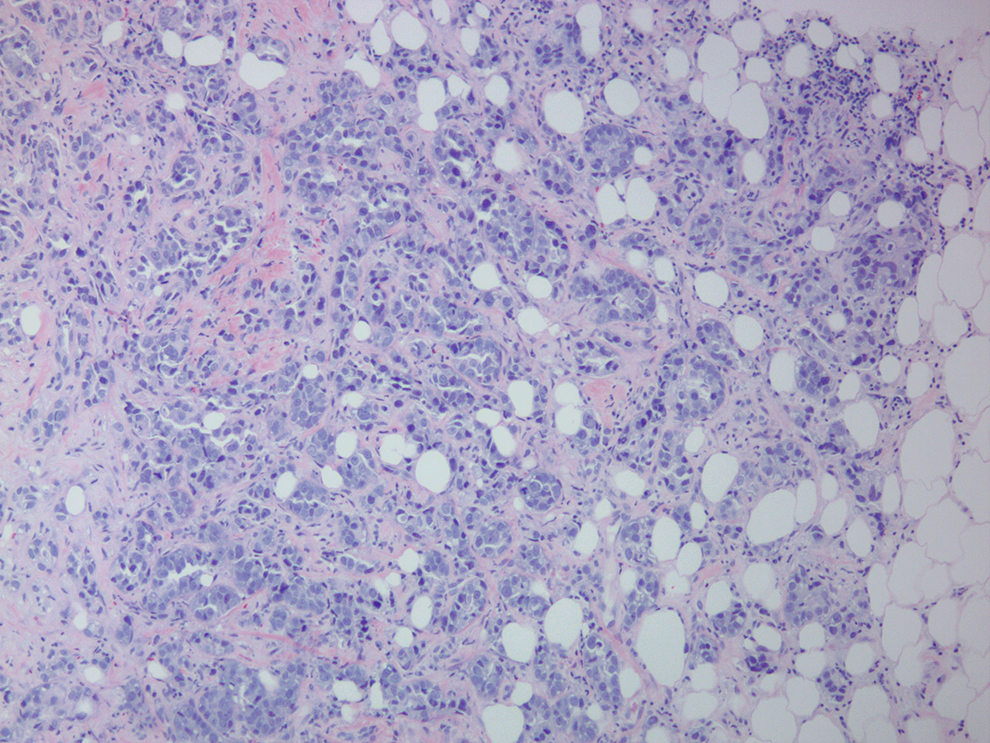
Results of breast core needle biopsy. A hematoxylin & eosin–stained tissue section from the breast core needle biopsy demonstrates poorly differentiated invasive ductal carcinoma (10× objective).
Questions/Discussion Points, Part 1
What Biomarkers Are Routinely Performed on New Diagnoses of Breast Cancer?
Newly diagnosed cases of breast carcinoma undergo testing for estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor (HER-2) receptor.
How Are These Biomarkers Evaluated, and What Is Their Clinical Significance?
Both ER and PR status are evaluated by immunohistochemistry alone, while HER-2 status can be determined by immunohistochemistry and/or fluorescence in situ hybridization (FISH). The expression of these markers contributes to prognosis, as well as targeted therapy. 2
Estrogen receptor and PR are both nuclear hormone receptors that operate as ligand-dependent transcription factors. Approximately 50% to 60% of breast cancers are ER positive. Hormone receptor positivity predicts tumor progression and mortality benefit from endocrine therapies such as tamoxifen. 2
HER-2 is a transmembrane receptor with tyrosine kinase activity that mediates growth, differentiation, and survival of cells via multiple signal transduction pathways. Approximately 15% to 25% of breast cancers overexpress this gene which is typically associated with more aggressive tumor behavior. 3 In fact, the quantitative amount of HER-2 overamplification has been found to be in direct association with worse clinical outcomes. 4 HER-2 overexpression and amplification have also been found in cancers of other sites, such as the stomach, ovary, colon, bladder, and esophagus. 2
The first step in routine evaluation of HER-2 is immunohistochemistry, following a scoring system illustrated in Figure 2A to D. A tumor demonstrating circumferential membrane staining that is complete, intense, and in >10% of the tumor cells is scored immunohistochemistry (IHC) 3+ and is considered HER-2 positive. Conversely, a tumor with incomplete membrane staining that is faint and in >10% of tumor cells is classified as IHC 1+ and is considered HER-2 negative. Weak to moderate complete membrane staining observed in >10% of tumor cells is classified as IHC 2+ and is considered equivocal. In equivocal cases, the sample should be evaluated with FISH.
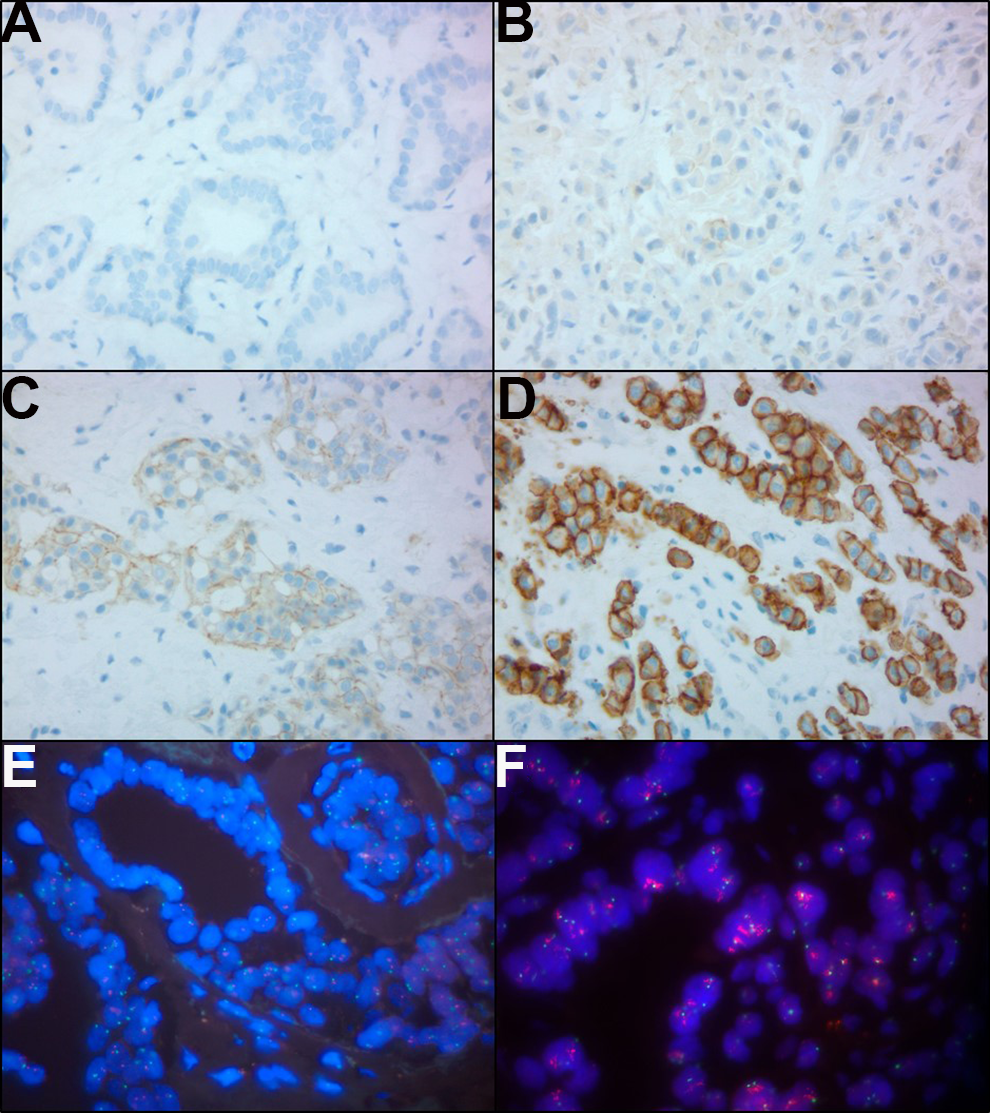
Human epidermal growth factor (HER-2) immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH) guideline summary. (A) No perceptible staining or barely perceptible incomplete membrane staining in <10% of tumor cells (IHC 0; 40× objective), (B) faint, barely perceptible incomplete membrane staining in >10% of tumor cells (IHC 1+; 40× objective), (C) weak to moderate complete membrane staining in >10% of tumor cells (IHC 2+; 40× objective), (D) intense, complete circumferential membrane staining in >10% of tumor cells (IHC 3+; 40× objective), (E) negative HER-2 FISH demonstrating HER-2 copy number <4.0 and HER-2/CEP17 ratio <2.0 (HER-2: red signal, CEP17: green signal; 100× objective), (F) positive HER-2 FISH demonstrating HER-2 copy number >6.0 and HER-2/CEP17 ratio >2.0 (HER-2: red signal, CEP17: green signal; 100× objective).
Fluorescence in situ hybridization is used to identify and quantify amplification of the HER-2-neu gene and is viewed as the “gold standard” for determining which patients would benefit from HER-2-targeted therapy. Fluorescence in situ hybridization is a molecular technique that utilizes fluorescent probes complementary to the nucleic acid sequence of interest, in this case, the HER-2-neu gene. The HER-2-neu copy number can either be evaluated alone or in comparison to the copy number of internal centromeric control (typically the region termed CEP17), as summarized in Table 1. Under normal circumstances, each cell will demonstrate 2 copies of both the HER-2-neu and CEP17 regions (1:1 ratio), but in the presence of an amplified HER-2-neu gene, there will be an excess of HER-2-neu signals in relation to CEP17 signals. Examples of both negative and positive FISH results are included in Figure 2E and F. A downside of FISH testing is that it is often more expensive than immunohistochemistry. 2,5
Summary of HER-2 FISH Reporting Guidelines.

Abbreviations: FISH, fluorescence in situ hybridization; HER-2, human epidermal growth factor; IHC, immunohistochemistry.
* Additional workup varies depending on the HER-2/CEP17 ratio and average HER-2 copy number in a particular case but may include review of HER-2 IHC, repeat FISH scoring by a second observer, and/or use of alternate probe sets.
What Other Factors Affect the Prognosis of Breast Cancer?
Tumor grade (well, moderately, or poorly differentiated), determined through scoring of tubule formation, nuclear atypia, and mitotic count, is a major factor affecting the prognosis of breast cancer. In addition, molecular subtypes, including luminal, normal breast-like, HER-2-enriched, and basal-like types, identified through extensive molecular profiling of a large number of breast cancer samples, play a major role in prognosis and treatment response. These topics are further discussed in a previously published educational case. 6
Diagnostic Findings, Part 2
This patient’s breast cancer is negative for ER and PR. Immunohistochemistry staining results for HER-2 are shown in Figure 3. HER-2 IHC is scored as 2+ (equivocal) for HER-2, demonstrating weak to moderate complete membrane staining in >10% of tumor cells. Due to this result, the sample is tested reflexively by FISH.
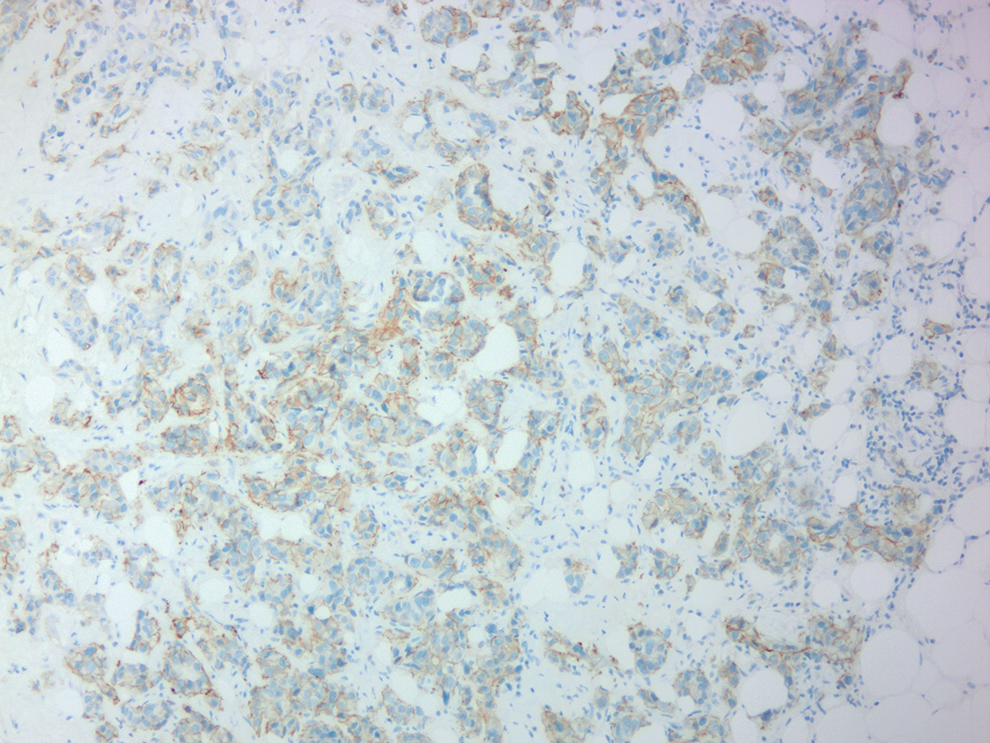
Human epidermal growth factor (HER-2) immunohistochemistry (IHC) on breast core needle biopsy. The HER-2 IHC demonstrates moderate complete membrane staining in >10% of tumor cells (IHC 2+; 10× objective).
Fluorescence in situ hybridization results demonstrate amplification of the HER-2/neu gene. Using the CEP17 gene as an internal centromeric control, the HER-2/CEP17 ratio is 3.7, while the average nucleus contains 9.4 copies of HER-2. This result indicates that this patient’s breast cancer is HER-2 positive.
Questions/Discussion Points, Part 2
Discuss the Biology of the HER-2 Protein and Its Role in Breast Cancer
The HER-2/neu belongs to a family of 4 human epidermal growth factor receptors: HER-1, HER-2, HER-3, and HER-4. Each is characterized by a cysteine-rich extracellular ligand-binding site, a transmembrane lipophilic segment, and an intracellular domain with tyrosine kinase activity. More specifically, the HER-2 receptor is a 1255 amino acid, 185 kD transmembrane glycoprotein located on the long arm of human chromosome 17 (17q12). It has no direct activating ligand and thus requires heterodimerization with other epidermal growth factor receptor (EGFR) family receptors such as HER-1 and HER-3. Dimerization and autophosphorylation of the HER-2 receptor tyrosine residues initiates various signal transduction pathways including mitogen-activated protein kinase, phosphatidylinositol-4,5-bisphophate 3-kinase, and protein kinase C. 2 When amplified or overexpressed, uncontrolled overactivation of these pathways leads to tumorigenesis and increased tumor aggressiveness. The HER-2 overexpression in breast cancer is related to the amplification of the otherwise normal gene rather than mutation. For this reason, HER-2 targeted therapy does inherently carry the risk of damaging normal tissues which express the protein.
Discuss the Epidemiologic Features of HER-2 Positive Breast Cancer
It has been found that ER/PR+HER-2− breast cancers are associated with better survival rates, followed by ER/PR+HER-2+ breast cancers, while triple-negative cancers (ER/PR-HER-2−) are associated with the shortest survival rates of these categories. Although HER-2 positive breast cancers were historically among the most aggressive cancers with the worst prognosis, the advent of HER-2-targeted therapy has improved survival rates significantly, such that HER-2 positive breast cancers now demonstrate similar or better outcomes than triple-negative breast cancers when matched for stage. The distribution of breast cancer subtypes varies by age, race, ethnicity, and other factors. Compared with women who have ER/PR+/HER-2− breast cancer, those diagnosed with other subtypes (ER/PR+/HER-2+, ER/PR-/HER-2+, or ER/PR-/HER-2−) are more likely to be younger, belong to minority groups, and be diagnosed with cancer at a later stage. 7 BRCA1/BRCA2 mutations are associated with lower rates of HER-2 positive breast cancer. 8
What Is the Prognosis of HER-2 Positive Breast Cancer?
HER-2 positive breast cancers have been found to have a worse overall prognosis than HER-2 negative breast cancers. Population-based studies have shown that overexpression of HER-2 is associated with poorly differentiated, high-grade tumors, high proliferative rates, lymph node involvement, and relative resistance to certain types of chemotherapy. 9
Studies have shown that HER-2 positive breast cancers carry an increased risk of cerebral metastasis compared with ER/PR+ breast cancers and that HER-2-positivity is associated with earlier cerebral metastasis in the disease course. 10 It is believed that once HER-2 amplification occurs, the HER-2 phenotype is fixed throughout the tumor’s lifetime. As a result, testing for HER-2 may be performed on either a primary tumor or a metastatic deposit. 2
Discuss the Management of HER-2 Positive Breast Cancer
The advent of HER-2-targeted therapies has significantly improved the outlook for patients with HER-2 positive breast cancer. Trastuzumab (Herceptin), a monoclonal antibody targeting the HER-2 receptor, has been found to cause internalization and downregulation of the receptor. 11 It has also been found to reduce mortality and recurrence rates in HER-2 positive breast cancers. Studies have shown that the addition of trastuzumab to adjuvant chemotherapeutics such as paclitaxel, doxorubicin, and cyclophosphamide can reduce the rates of recurrence by half. 3,11 Due to the persistence of the HER-2 phenotype, trastuzumab has also been found to improve outcomes for both local and distant disease. 2 Additional therapy options include pertuzumab, a monoclonal antibody thought to interrupt HER-2/HER-3 dimerization, and neratinib, a tyrosine kinase inhibitor believed to interact with HER-2 and other EGFR kinases. 12
Approximately half of HER-2 positive breast cancers are also ER positive, PR positive, or both. However, the levels of these hormone receptors are typically lower than in HER-2 negative, hormone receptor positive tumors. For this reason, most HER-2 positive breast cancers are relatively resistant to tamoxifen and other endocrine therapies. 9
Current guidelines do not recommend the use of trastuzumab or other anti-HER-2 therapies in HER-2 negative breast cancers. Treatment for these tumors typically includes endocrine therapy (if ER/PR+) or single-agent chemotherapy.
Diagnostic Findings, Part 3
The patient’s stage is determined to be a clinical stage cT1cN0 based on physical examination and imaging findings.
Questions/Discussion Points, Part 3
How Is Breast Cancer Staged?
Breast cancer is staged using the TNM (tumor, node, metastasis) system which assesses the size and local extent of the primary tumor (T), presence or absence and number of axillary lymph node (N) metastases, and presence or absence of metastasis (M) at distant sites. Although clinical staging, designated by the prefix “c” is performed prior to surgery using data gathered from physical examination and imaging studies, including breast, axillary, and whole body imaging as indicated; pathologic staging, designated by the prefix “p,” is performed after surgery using data gathered from the resulting excision specimen. 13 In this patient case, stage cT1cN0 refers to a localized breast cancer measuring 1 to 2 cm on imaging without evidence of nodal metastasis.
How Does the Clinical Stage Affect Initial Management of Breast Cancer?
Treatment of breast cancer is interdisciplinary, utilizing expertise from medical oncology, radiation oncology, and surgery. Although low stage, localized cancer is often treated initially by surgical excision, higher stage cancers may require up-front neoadjuvant chemotherapy to reduce the tumor burden prior to surgery. Patients with known or suspected lymph node metastasis may undergo complete removal of the axillary lymph nodes at the time of surgery, as well. However, patients such as the one in this case without suspected lymph node metastasis may undergo sentinel lymph node biopsy only (the primary node draining from the breast lymphatics, as identified by a blue dye and/or radiotracer injected into the breast at the time of surgery). If the sentinel node is positive for metastatic tumor, further surgery or radiation may be pursued to treat the remaining axillary lymph nodes. The presence of distant metastasis may preclude surgery altogether, with a focus on palliative chemotherapy and radiation.
Teaching Points
Pathologic factors affecting the prognosis of breast cancer include tumor grade, molecular subtype, and expression of ER, PR, and HER-2.
HER-2 is part of a family of receptors that work together within various signal transduction pathways to promote growth, differentiation, and survival of cells. Amplification and overexpression of HER-2 may be seen in a subset of breast cancers.
Primary evaluation for HER-2 status should be performed by immunohistochemical staining, with equivocal results evaluated by FISH. A positive FISH result is the gold standard for determining whether a patient would benefit from HER-2-targeted therapy.
HER-2 positive breast cancers are often more aggressive, less responsive to treatment, and associated with decreased survival rates, though the advent of HER-2-targeted therapy has significantly improved prognosis.
Current management recommendations for HER-2 positive breast cancer include HER-2-targeted therapy (such as Trastuzumab) with adjuvant chemoradiation. Endocrine therapies (such as tamoxifen) have not been found to be effective for tumors with HER-2 amplification.
Footnotes
Acknowledgments
The authors would like to acknowledge Dr Dina Kandil for her assistance in reviewing the content of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
