Abstract
BACKGROUND:
Breast-conserving surgery (BCS) as an alternative to total mastectomy (TM) in patients with early-stage triple-negative breast cancer (TNBC) is not widely spread.
OBJECTIVE:
We aimed to compare the overall survival (OS) and disease-free survival (DFS) between both surgical approaches in early-stage TNBC patients at 10 years.
METHODS:
We conducted a retrospective cohort study in TNBC female patients with stage I-IIa, treated at a single-center during the period of 2000–2014. We estimated and compared the survival rates with the Kaplan Meier and Long-rank test. Propensity scores were calculated with the generalized boosted regression model and were used in the multivariate Cox regression analysis with the covariate adjustment method.
RESULTS:
We included 288 patients, 111 in the BCS vs. 177 in the TM group. The median follow-up was 102 months. Moreover, the patients in the BCS group had superior OS (85% vs. 81%,
CONCLUSION:
BCS is a safe alternative to TM in Latin-American patients with early-stage TNBC.
Keywords
Background
Breast cancer is the leading neoplastic disease among females, with polyvalent molecular characteristics [1]. The difference in the expression of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER-2) classifies this tumor into four subtypes. One of them, the triple-negative breast cancer (TNBC) – defined as the minimal or non-expression of ER, PR, and HER-2 – is a heterogeneous subtype with an erratic and aggressive behavior [2,3]. Moreover, the overall survival (OS) of Peruvian patients with TNBC at five years is 67% [4,5].
The surgical treatment of early-stage TNBC is with either breast-conservative surgery (BCS) or total mastectomy (TM). Nonetheless, this evidence-based recommendation comes from studies that included the four breast cancer subtypes in their analysis, rather than focusing on the survival outcomes of patients with TNBC [6–9]. Because of the aggressiveness of the TNBC, it would be reasonable to perform a radical surgical approach to reduce the recurrence and mortality risk [10]; however, research reports are contradictory. While some studies show that TM increases the locoregional recurrence risk [11,12], others state that locoregional and distant recurrence, disease-free survival (DFS), and OS of patients that underwent BCS have no difference from TM [13,14]. We aimed to compare the OS and DFS between BCS and TM in early-stage TNBC patients.
Methods
Study design
This is a retrospective cohort study, carried out and approved by the institutional review board of the National Institute of Neoplastic Diseases (INEN, “Instituto Nacional de Enfermedades Neoplásicas”) in Peru. We selected the medical records of patients with the International Code for Disease (ICD)-10: “C50: Malignant neoplasm of breast”. Thereafter, we included: (1) female patients with TNBC treated with either BCS or TM from 2000 to 2014; (2) patients with unilateral tumor stage I-IIa; (3) patients with adjuvant radiotherapy after BCS. We identified 401 medical records; nevertheless, we excluded patients without adjuvant chemotherapy after BCS or TM and loss of follow-up after surgery, which yielded a total of 288 medical records.
Clinicopathological variables
Patients were classified according to the 7th edition of the American Joint Committee on Cancer pathologic staging system [15]. Breast cancer immunophenotype was assessed by conventional immunochemistry, and TNBC was defined as the lack of expression of ER, PR, and HER-2. Patients received paclitaxel (4.5%), doxorubicin and cyclophosphamide (41%), or doxorubicin and cyclophosphamide plus paclitaxel (54.5%) as adjuvant chemotherapy.
Outcomes
Our primary outcome was the difference in mortality risk between BCS and TM at 10 years; we also measured the difference in the recurrence (locoregional or distant) risk. We defined OS from the date of surgery until death or end of the study (December 31, 2016), while DFS from the date of surgery until the diagnosis of locoregional or distant recurrence, death, or end of the study [15]. OS and DFS rates were reported at 5 and 10 years.
Statistical analysis
We used the program R version 3.5.3 with the packages survival, “survminer”, “twang”, and “survey” for the analysis. Demographic, clinicopathological, surgical, and adjuvant therapy characteristics were compared and described between both groups with the Chi-square test for qualitative variables and the Student’s
For the survival analysis, we included 263 patients (162 in the TM group and 101 in the BCS group). The survival curves and differences for both outcomes (OS and DFS) were estimated with the Kaplan–Meier method and the Log-rank test. Furthermore, we calculated the propensity scores with the generalized boosted regression model and employed the covariate adjustment method in the analysis. We used the average treatment effect (ATE) technique and followed the assumption that patients in our cohort had the same probability to underwent BCS or TM. The propensity score model included the following variables: age, menopausal state, histologic grade, tumor size, and lymph node status; because previous literature identified these variables as risk factors for mortality or recurrence risk [6,16–18]. To verify the balance of the propensity score, we employed the standardized effect size (SES). All the variables in the model had SES < 0.20. Multivariate Cox Regression analysis was employed – with the weighted propensity score as covariate – to identify the mortality and recurrence risk between both groups (BCS vs. TM). We calculated the adjusted proportional Hazard Ratio (HR) with a 95% confidence interval (CI) and considered a
Results
Demographic and oncologic characteristics
We reviewed 288 medical records (111 in the BCS group and 177 in the TM group). Table 1 describes the demographic and clinicopathological characteristics of the study population according to both surgical modalities. Patients in the BCS cohort were younger, more likely to be premenopausal, had lower tumor size, and lower pathologic tumor stage. However, the lymph node status and histologic grade had a similar distribution between both cohorts. The most common histological subtype was invasive ductal carcinoma, followed by invasive lobular carcinoma and medullary carcinoma, with no statistical difference (Table 1).
Demographic and Clinicopathological characteristics of patients according to breast surgery management
Demographic and Clinicopathological characteristics of patients according to breast surgery management
Compare to TM, BCS was more frequently performed during the periods of 2006–2010 and 2011–2014; however, TM was more common in the 2000–2005 period (Table 1). A total of 34 (11.8%) patients had positive margins, 31 (27.9%) cases in BCS and 3 (1.7%) cases in TM group (
Impact of surgery type on survival outcomes
We excluded 25 cases with missing values in the histologic grade variable. Of the remaining 263 patients, the locoregional (21 patients, 7.4% in the BCS vs. 8.9% in the TM group,
The median follow-up was 102 months, the overall mortality rate was 14.8% (16% for BCS and 12.9% for TM,
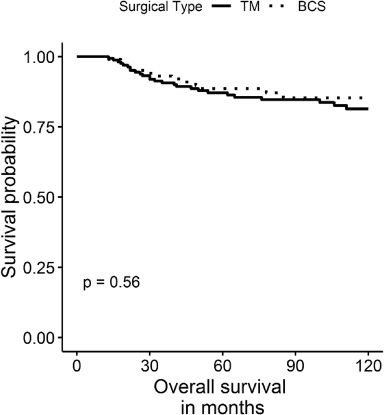
Overall survival of early-stage triple-negative breast cancer patients according to surgical treatment (BCS: breast-conserving surgery and TM: total mastectomy).
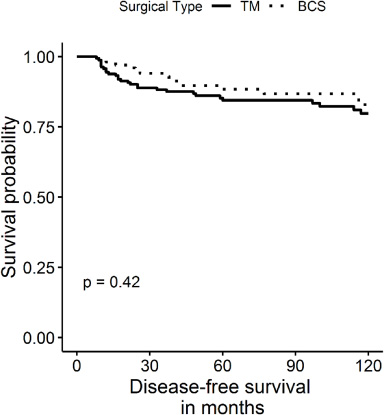
Disease-free survival of early-stage triple-negative breast cancer patients according to surgical treatment (BCS: breast-conserving surgery and TM: total mastectomy).
Showing the balance of covariates before and after weighting by the propensity score
Multivariate Cox regression analysis of mortality and recurrence risk
∗Adjusted for propensity score.
The surgical treatment for patients with early-stage TNBC is with either BCS or TM [6,19]. Our study identified that both surgical approaches had equivalent survival outcomes (OS and DFS); therefore, BCS is a safe alternative to TM in this population. Similar to previous studies, we support the practice of BCS. For example, Chen et al. [17] found that BCS reduced ∼40% the mortality risk, and Adkins et al. [18] identified that TM increased the mortality risk by 22%. Nevertheless, both studies analyzed patients with pathologic tumor stages I–III, which negatively affects the survival outcomes due to the inclusion of patients with higher tumor stages. Moreover, Abdulkarim et al. [12] included breast cancer patients with stages I–II, and reported an OS of 87% and 82% at 5 years in the BCS and the TM groups, respectively; although OS was not their primary objective. In contrast, we included patients with pathologic tumor stages I–IIa, and our principal outcome was mortality risk.
Two factors may explain the non-significant mortality risk between BCS and TM in the multivariate Cox regression analysis. First, the frequency of performance between both surgical approaches differed across time. At the beginning of our observation period, surgeons conducted TM more frequently, while BCS was an uncommon procedure, and its use increased during the following years. Therefore, a learning curve within the surgeons was still developing during the first years of the BCS implementation. Second, the proportion of positive surgical margins were higher in the BCS group, which shows a lack of experience from the surgeons in the first interventions of the BCS. Although both factors could have impacted the OS, our results acknowledge BCS as a safe alternative to TM.
On the other hand, previous studies state that BCS compared to TM has an equivalent locoregional and distant recurrence risk at 5 years [6,18,20]. Parker et al. [20] identified that the recurrence risk (locoregional or distant) between both types of surgery was non-significant, while Adkins et al. [18] reported that BCS did not increase the locoregional recurrence risk. Our results are consistent with these previous experiences; the multivariate analysis did not show a higher locoregional or distant recurrence risk in patients that underwent BCS. However, we reported our results at 10 years compared to the previous investigations, which enhance a long-term favorable outcome for BCS.
Our study has some limitations. First, we excluded medical records due to missing data on the treatment characteristics and loss of follow-up. Second, BCS became more common after 5 years of the beginning of the study, which implies a lack of experience from the surgeons performing this surgery during that period. This is the most likely reason for the high positive margins in the BCS cohort, however, the majority of patients had a re-excision with radiotherapy. Third, this is a single-center investigation, thereby, the generalization of our results must be taken with caution.
Conclusion
In conclusion, we support the use of BCS as a safe alternative to TM in patients with early-stage TNBC. Our results evidence a slight benefit from BCS on OS and DFS (without statistical significance). These results will help physicians and surgeons make evidence-based decisions in their daily practice.
Abbreviations
Breast-conserving surgery
Confidence interval
Disease-free survival
Estrogen receptors
Human epidermal growth factor receptor 2
Hazard ratio
International Code for Disease
Instituto Nacional de Enfermedades Neoplásicas
Overall survival
Progesterone receptors
Total mastectomy
Triple-negative breast cancer
Footnotes
Acknowledgements
We thank to all the healthcare and administrative personnel of the National Institute of Neoplastic Diseases for supporting us in the development of this manuscript. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any financial relationship with a commercial entity that has interest in the subject carried out in this manuscript or any conflict of interest to declare.
