Abstract
BACKGROUND:
The immune system is known to play an important role in tumor cell eradication. Although cancer cells were able to escape from the immune system, many studies showed mononuclear inflammatory cell infiltrates known as tumor-infiltrating lymphocytes (TILs) on breast cancer histopathology specimens showed better prognosis, including in disease-free survival (DFS) and chemotherapy responses.
OBJECTIVE:
This study aimed to reveal the predictive value of tumor-infiltrating lymphocytes (TILs) levels and CD8 expression in invasive breast carcinoma of no special type patients’ samples on response to anthracycline-based neoadjuvant chemotherapy.
METHODS:
75 pre-treatment biopsy samples that were diagnosed as invasive breast carcinoma of no special type were evaluated. TILs level determined following recommendations of International TILs Working Group 2014, CD8 expression assessed semiquantitatively after immunohistochemistry staining. Response to anthracycline-based neoadjuvant chemotherapy evaluated clinically using Response Evaluation Criteria in Solid Tumours (RECIST) criteria and pathologically by evaluating hematoxylin and eosin (H&E)-stained slides from mastectomy specimens after 3 or 4 cycles of neoadjuvant chemotherapy.
RESULTS:
Chi-squared analysis showed a significant relationship between TILs level and CD8 expression with chemotherapy responses clinically (p = 0.011 and p = 0.017 respectively) but not pathologically. Furthermore, the logistic regression test exhibit the predictive value of TILs level was 66.7% and CD8 expression was 64%.
CONCLUSIONS:
This study results suggest that TILs level and CD8 expression may be added as predictive factors to the response of anthracycline-based neoadjuvant chemotherapy, and oncologists may take benefit in breast cancer patient’s management.
Introduction
Breast cancer is still the most common malignancy with the highest mortality in women worldwide. The carcinogenesis of breast cancer as well as other cancer, is caused by fundamental changes in cell physiology known as hallmarks of cancer, including the ability to escape from host immune responses [1].
The cancer immunosurveillance concept was first introduced by Macfarlane Burnet in the 1950s who stated that the physiologic function of the immune system is to recognize and destroy clone of transformed cells before forming tumor cells and eliminating it if it has been formed. This concept was supported by experimental studies on mice and facts of higher incidence of several tumors in a patient with low immune systems [2–5]. However, incomplete tumor elimination may lead to tumor establishment and uncontrolled by the immune system. Moreover, several components of the immune system have a role in tumor establishment. This process is known as immunoediting, which consists of three phases (elimination, equilibrium, and escape), denoting the tumor progression process based on tumor cells and immune system interaction [4].
Although these cancer cells were able to escape from the immune system, many studies showed mononuclear inflammatory cell infiltrates known as tumor-infiltrating lymphocytes (TILs) on breast cancer histopathology specimens showed better prognosis, including in disease-free survival (DFS) and chemotherapy responses [6–12].
One of the TILs subpopulations which are believed to has a significant role in tumor immunoediting is cytotoxic T cells Cluster of Differentiation 8 (CD8). Mahmoud et al.’s investigation on 1.334 breast cancer samples with multivariate analysis showed that higher total CD8 cytotoxic T cells independently related to longer breast cancer-specific survival (BCSS) (HR 0.55; 95% CI 0.9-0.78; p = 0.001) [13]. The result was also parallel in Ali et al.’s investigation but only in ER (estrogen receptor) negative specimens [14].
Neoadjuvant chemotherapy was now recommended for locally advanced breast cancer (Stage IIB & III) and breast cancer with high TILs. The benefits including reduce surgical procedure radicality & enabling in vivo evaluation of chemotherapy responses [15]. Anthracyclines (ANT) is one chemotherapy agent who was largely used and has been shown effective as an anticancer therapy for years [16]. Breast cancer patients with similar staging and chemotherapy agents were not usually present a similar outcome. The oncologist can not always predict the clinical outcome of patients given similar therapy. Hence it is important to identify predictive factors clinically and biologically for better-tailored therapy. This study aimed to investigate the predictive value of TILs level and CD8 expression on the response of anthracycline-based neoadjuvant chemotherapy in invasive breast carcinoma of no special type.
Methods
Setting and subjects
This study involved 75 pre-treatment biopsy samples which were diagnosed as invasive breast carcinoma of No Special Type, treated by Anthracycline-based Neoadjuvant Chemotherapy 3/4 cycles before mastectomy. Clinical data were collected from medical records and data from the Oncology Department of Dr. Wahidin Sudirohusodo Hospital to obtain sex, age, initial clinical T and N stage according to the 7th American Joint Committee on Cancer and to assess clinical chemotherapy responses based on RECIST criteria; the complete and partial response was categorized as chemosensitive samples, while stable and progressive disease were categorized as chemoresistance samples.
Pathological data were obtained from pathology reports, hematoxylin and eosin (H&E)-stained slides and IHC slides for basic biomarkers reviewed by two experienced pathologists to determine histologic tumor type, histologic grade according to Elston & Ellis modification grading system, lymphatic and vascular invasion, estrogen receptor (ER) expression, progesterone receptor (PR) expression, and HER 2 status. The TILs were evaluated in percentage as a semi-quantitative parameter using recommendations of International TILs Working Group 2014, categorized as >10% and ≤10%. Pathological responses were evaluated in mastectomy specimens of the same patient who were undergone surgical procedures after 3 or 4 cycles of neoadjuvant chemotherapy. This study was approved by the Institutional Review Board of Medical Faculty of Hasanuddin University, Makassar, Indonesia.
Immunohistochemistry and CD8 quantification
The paraffin blocks from the pre-treatment biopsy of 75 patients were collected from the Pathology Anatomy Laboratory of Dr. Wahidin Sudirohusodo Hospital (Makassar, Sulawesi Selatan, Indonesia). A 4-μm thick were processed and stained using an IHC method for CD8+ T cells. The IHC staining was performed with the primary antibody, rabbit monoclonal antibody (SP16; CellMarque), and incubated for 24 min at 37 °C. The secondary antibody from the kit was used for the second incubation for ∼20 min at 36 °C. The staining was evaluated using the light microscope (Olympus), and cells expressing membranous staining for CD8+ were interpreted as positive.
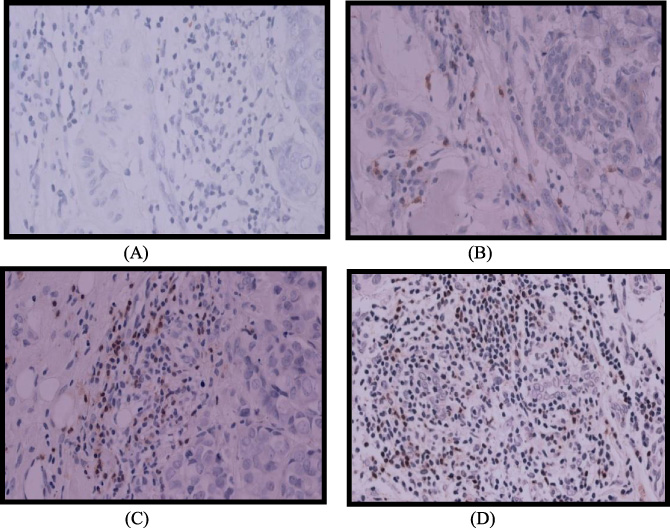
No expression of CD8 (A). Minimal expression of CD8 (B). High expression of CD8 (C, D).
This is a semi-quantitative analysis conducted by two experienced pathologists (U.A.M. & R.M) who were blind to the patients’ clinical data. The calculation was performed in areas of maximum lymphocytic infiltration intratumoral and peritumoral, in three high power fields (magnification ×400), areas of hemorrhage and/or necrosis were excluded. The CD8+ expression levels were classified as high and low based on values ≤ or > of the median value, respectively.
The data collected were analyzed using SPSS version 25 (IBM Company). The associations between TILs, CD8 expression, and other clinicopathological characteristics with chemotherapy responses were analyzed using the chi-squared and logistic regression tests.
Result
The patients’ age was between 32 and 80 years old with a mean age of 49.52 years old. Of the 75 samples, 6 (8%) were low grade, 57 (76%) were moderate grade, and 12 (16%) were high grade. The immunohistochemistry data for ER, PR, and Her 2 were unavailable for 16 samples, so only 59 samples were included for analysis. 16 (27.1%) samples were ER (+), PR (+/−), Her 2 (−); 25 (42.4%) samples were Her 2 (+); and 9 (15.3%) were ER (+), PR (+/−), Her 2 (−) and triple-negative respectively. The lymph node status was positive for 19 (25.4%) samples and negative for 56 (74.6%). The TILs level were ≥10% in 41 (54.5%) samples and <10% in 34 (45.5%) samples. The CD8 expression was high in 35 (46.7%) samples and low in 40 (53.3%) samples (Fig. 1). The clinical chemotherapy responses were chemosensitive for 46 (61.3%) samples and chemoresistant for 29 (38.7%) samples. The data for a pathological response only available for 51 samples, 5 (9.8%) were complete pathological responses, and 46 (90.2%) were incomplete responses.
Analysis result for clinical chemotherapy response
Analysis result for clinical chemotherapy response
Analysis result for pathological chemotherapy response
The chi-squared analysis showed significance only for the association between TILs level with clinical chemotherapy responses and CD8 expression with clinical chemotherapy responses (p = 0.011 (OR 3.93, IC 1.47-10.49) and p = 0.017 (OR 0.73, IC 1.37-10.18) respectively). Other parameters showed no significance, as shown in Tables 1 and 2.
In multivariate logistic regression analysis, TILs level and CD8 expression had predictive significance for clinical chemotherapy responses. Test result between TILs level and CD8 expression with clinical chemotherapy responses obtained p = 0.019, OR = 3.39, 95%CI = 1.22–9.39 and p = 0.030, OR = 3.19, 95%CI 1.12–9.05, respectively. Logistic regression models showed the predictive value of TILs level and CD8 expression was 66.7%.
Immune responses to tumor progression are both carried out by innate and adaptive immunity. The major agent of adaptive immunity against tumor cells is cytotoxic T cells CD8 which requires cross-presentation of host antigen-presenting cells (APCs), specially dendritic cells. Initially, the tumor cells or their antigens ingested and processed inside the APCs, the peptides derived were then displayed bound class I MHC molecules for recognition by CD8 T cells. The APCs at the same time express costimulator needed for differentiation of CD8 T cells into anti-tumor cytotoxic T lymphocytes (CTLs). The effector CTLs are then ready and able to recognize and destroy the target cell/tumor cells by delivering proteins (perforin and granzymes) that induce cell death [2].
This recent study showed a significant relationship between TILs and CD8 expression on pre-treatment samples with anthracycline-based neoadjuvant chemotherapy responses clinically (p = 0.011 and p = 0.017, respectively). This study results signify the role of CD8 T cells and TILs level in breast tumor cell eradication. There were many studies that showed similar results with variable dependent parameters, including PCR, OS, DFS, and BCSS. Al-Saleh K. et al., in their research, showed that a high level of CD8 expression in pre-treatment samples of breast cancer (luminal b/Her2 negative) was significantly correlated with OS (p = 0.023) and was a predictive factor for pCR. Multivariate Cox regression analysis also showed that CD8 expression was an independent prognosis factor for OS but not for DFS [17].
The chemotherapy agents, including an anthracycline, were proven to has an immunomodulatory effect against tumor cells. It stimulates immune responses by inducing immunogenic cell death. Several mechanisms occurred in the process, including translocation of calreticulin from endoplasmic reticulum lumen to the cell surface, which serves as an ‘eat me’ signal and stimulates tumor cell elimination by fagosit and DCs [18]. Anthracycline will also trigger ATP release in the premortem phase of tumor cell death and stimulates tumor cells to produce interferon type 1, which eventually induces the release of CXCL10. The ATP and CXCL10 releases will lead to iDCs recruitment [19,20]. Another mechanism is anthracycline may simulate HMGB1 releases, which trigger DCs maturation. The endpoint of this iDCs maturation is the more expansion and differentiation of T cell CD8 against the tumor cells [21]. Another study by Alizadeh D. et al. also showed that anthracycline might eliminate MDSCs (which have a role in tumor evasion and inhibit innate and adaptive immunity) in the spleen, blood, and tumor area [22]. However, to maximize its function as an anti-tumor agent, anthracycline needs intact immune cells like cytotoxic T cells CD8 and IFN-γ along with IL-1β and IL- 17A as shown by the research of Mattarollo et al. [23].
Logistic regression model for prediction clinical chemotherapy responses
Logistic regression model for prediction clinical chemotherapy responses
In our study, multivariate logistic regression analysis showed the overall predictive value of TILs level and CD8 expression is 66.7% for clinical chemotherapy responses (Table 3). These results indicate that CD8 expression and TILs level may be added as predictive factors to determine the success of anthracycline-based neoadjuvant chemotherapy clinically, and therefore may benefit the clinicians/oncologist to be examined before the treatment given for patients with invasive breast carcinoma of no special type.
Many other factors determine the response of chemotherapy either by the immune system or non-immune systems. As for the immune system, some factors contribute positively to the anti-tumor effect such as T cell CD4 Th1, NK cell, and NKT cell, dendritic cell, and IFN-γ, and factors that contribute negatively such as Treg, TAM2, MDSCs, TGF-β, IL-6, IL-10, PD-1/PDL-1 and CTLA-4 [4]. Besides, an internal factor of T cell CD8 itself may contribute, since CD8 may differentiate as T cell effector (TEFF) and T cell memory central/effector (TCM; TEM) but may also differentiate as T cell exhausted (TEX), T cell anergic (TAN) and T cell senescent/regulatory (TSEN) [24].
This study results showed that TILs level and CD8 expression have a significant relationship with the response of anthracycline-based neoadjuvant chemotherapy clinically in a patient with invasive breast carcinoma of no special type and may be added as predictive factors for better tailor therapy.
Footnotes
Acknowledgements
The authors do not have acknowledgments.
Conflict of interest
None.
Funding
None.
